Drug Metabolism Learning Outcomes Describe Metabolism Identify the

Drug Metabolism

Learning Outcomes • • • Describe Metabolism Identify the role of Liver in drug metabolism Recognize different metabolic enzymes List the enzymes utilized in different phases of metabolism Sketch out the mechanism of Cytochrome P 450 enzyme. List Non-CYP Drug Metabolizing Enzymes.

Presentation • Introduction • Drug metabolizing enzymes
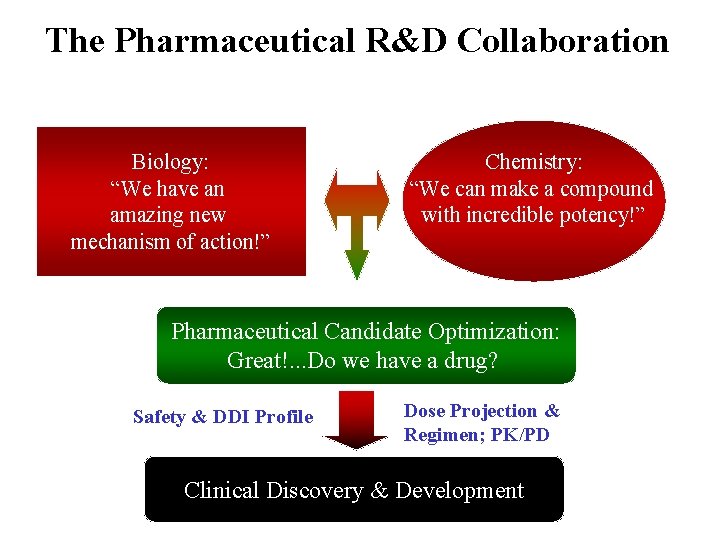
The Pharmaceutical R&D Collaboration Biology: “We have an amazing new mechanism of action!” Chemistry: “We can make a compound with incredible potency!” Pharmaceutical Candidate Optimization: Great!. . . Do we have a drug? Safety & DDI Profile Dose Projection & Regimen; PK/PD Clinical Discovery & Development
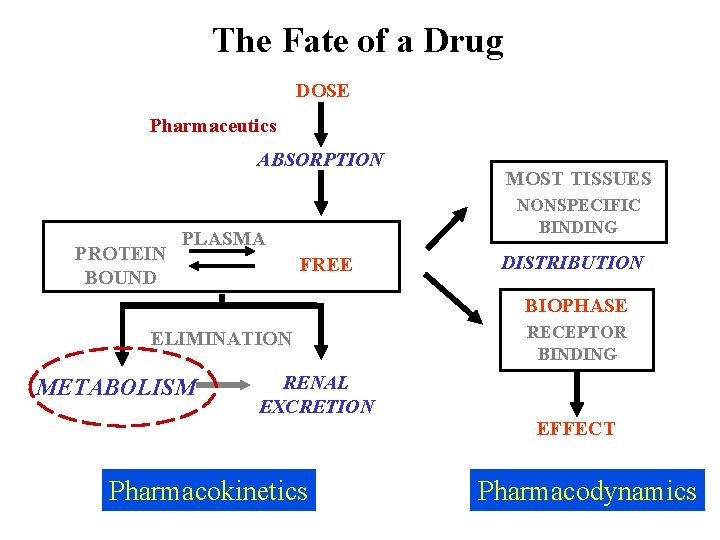
The Fate of a Drug DOSE Pharmaceutics ABSORPTION PROTEIN BOUND MOST TISSUES NONSPECIFIC BINDING PLASMA FREE DISTRIBUTION BIOPHASE ELIMINATION METABOLISM RENAL EXCRETION Pharmacokinetics RECEPTOR BINDING EFFECT Pharmacodynamics
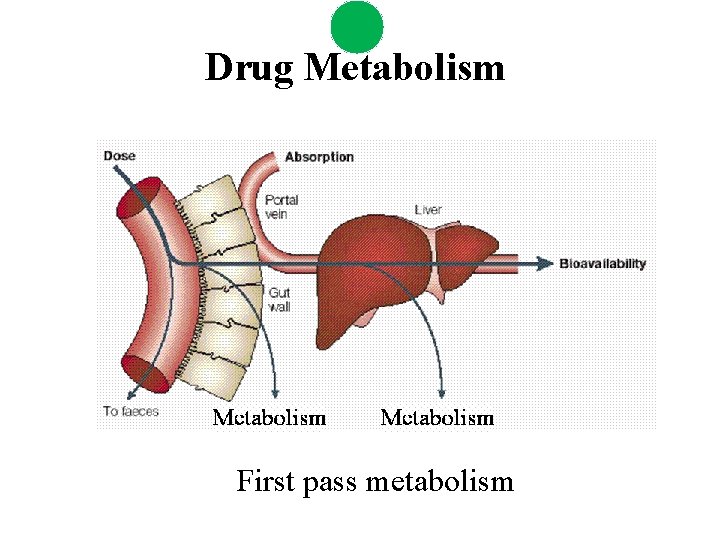
Drug Metabolism First pass metabolism

Drug Metabolism • Drug metabolism can occur in every tissue (e. g. gut, lung and kidney). However, the major drug metabolizing enzymes (DMEs) are expressed at the highest levels in the liver, which thus serves as the major organ of metabolic clearance • Drug metabolism serves to control the exposure of a potentially harmful substance. Usually via oxidation of a lipophilic xenobiotic, DMEs increase the polarity and aqueous solubility thus facilitating its elimination from the body • DMEs also help to regulate endogenous function (e. g. cytochrome P 450 s are involved in steroid and fatty-acid metabolism; and the glucuronosyl-S-transferase, is involved in the clearance of bilirubin)

Drug Metabolism Factors affecting drug metabolism: • Tissue differences • Genetics • Species differences • Co-administered substrates (inhibitors or inducers) • Auto-induction • Diet • Disease (especially hepatic or renal) • Protein-binding • Age • Gender • Route of administration

Drug Metabolism DMEs broadly classified into two types of reactions • PHASE I: typically a functional group (e. g. hydroxyl) is created or exposed in a drug molecule • PHASE II: conjugation of either the parent compound and/or its metabolite(s) involving a polar endogenous substrate that is able to react with the functional groups formed via Phase I reactions
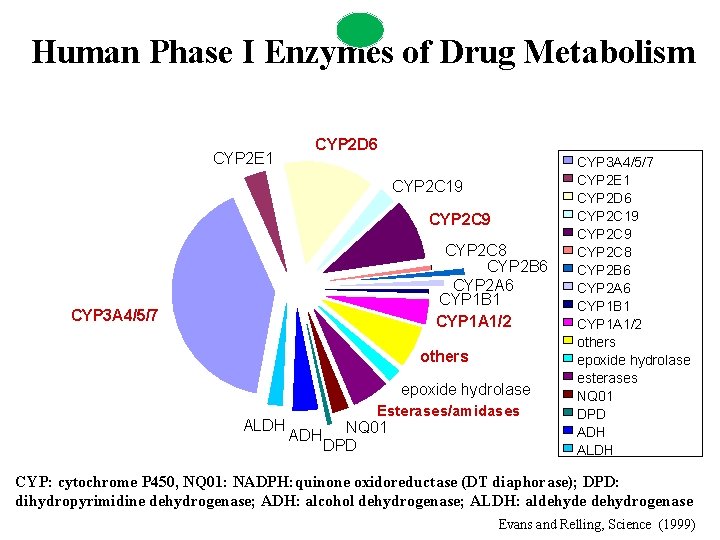
Human Phase I Enzymes of Drug Metabolism CYP 2 E 1 CYP 2 D 6 CYP 2 C 19 CYP 2 C 8 CYP 2 B 6 CYP 2 A 6 CYP 1 B 1 CYP 1 A 1/2 CYP 3 A 4/5/7 others epoxide hydrolase ALDH Esterases/amidases ADH NQ 01 DPD CYP 3 A 4/5/7 CYP 2 E 1 CYP 2 D 6 CYP 2 C 19 CYP 2 C 8 CYP 2 B 6 CYP 2 A 6 CYP 1 B 1 CYP 1 A 1/2 others epoxide hydrolase esterases NQ 01 DPD ADH ALDH CYP: cytochrome P 450, NQ 01: NADPH: quinone oxidoreductase (DT diaphorase); DPD: dihydropyrimidine dehydrogenase; ADH: alcohol dehydrogenase; ALDH: aldehyde dehydrogenase Evans and Relling, Science (1999)
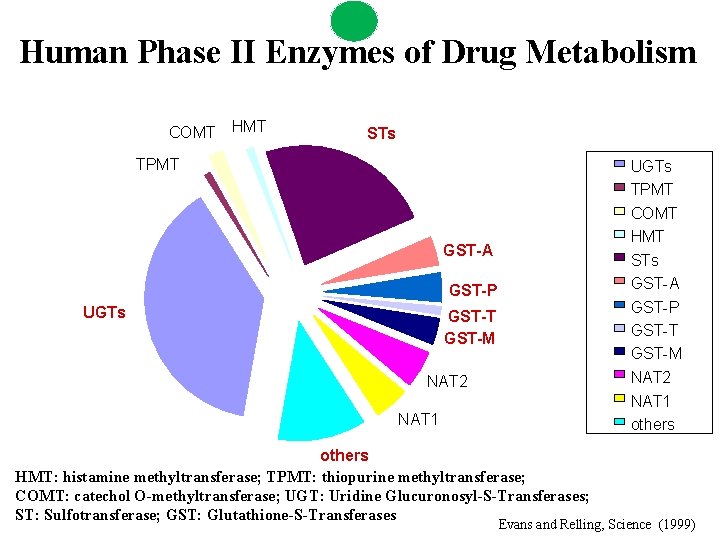
Human Phase II Enzymes of Drug Metabolism COMT HMT STs TPMT UGTs TPMT COMT HMT STs GST-A GST-P UGTs GST-P GST-T GST-M NAT 2 NAT 1 others HMT: histamine methyltransferase; TPMT: thiopurine methyltransferase; COMT: catechol O-methyltransferase; UGT: Uridine Glucuronosyl-S-Transferases; ST: Sulfotransferase; GST: Glutathione-S-Transferases Evans and Relling, Science (1999)

Drug Clearance A typical drug exhibits the following characteristics: • Cytochrome P 450 -mediated clearance – 55 % (90% of Phase I metabolism is CYP mediated) • Unchanged drug (i. e. non-metabolic clearance) – 25 % (urine, bile, expired air, feaces) • Other metabolism – 20 % (UGT, ST, MAO, FMO etc) Clearance is the sum process of all in vivo elimination pathways Any one pathway can dominate (. . . case-by-case analysis)
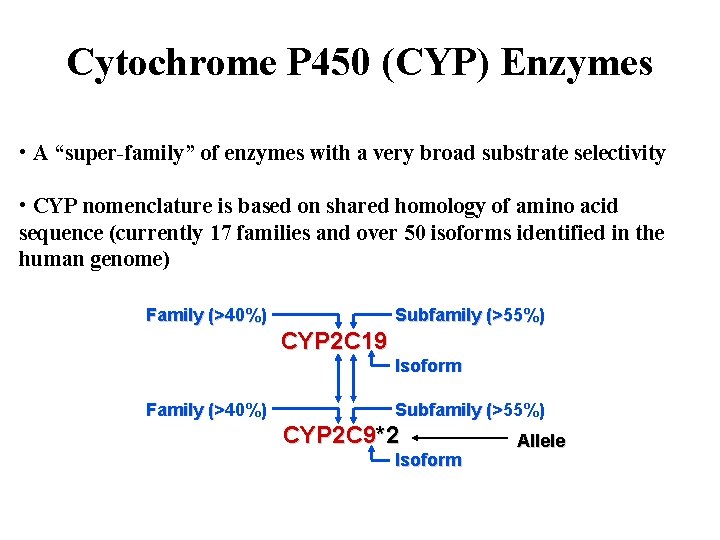
Cytochrome P 450 (CYP) Enzymes • A “super-family” of enzymes with a very broad substrate selectivity • CYP nomenclature is based on shared homology of amino acid sequence (currently 17 families and over 50 isoforms identified in the human genome) Family (>40%) Subfamily (>55%) CYP 2 C 19 Isoform Family (>40%) Subfamily (>55%) CYP 2 C 9*2 Isoform Allele
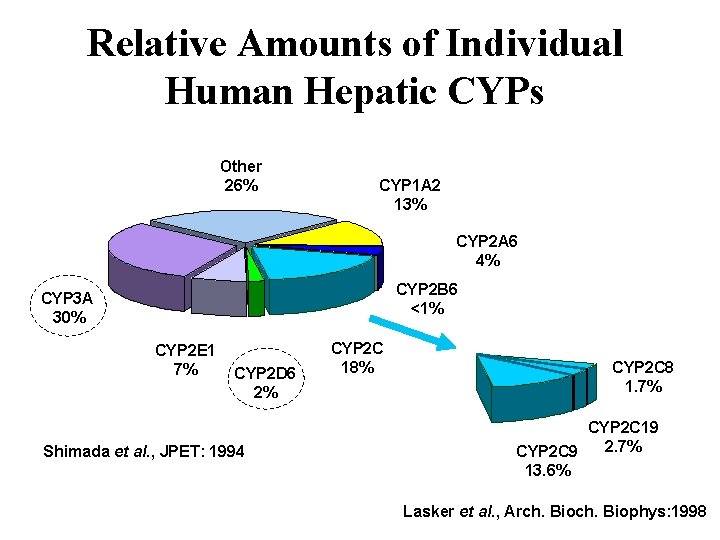
Relative Amounts of Individual Human Hepatic CYPs Other 26% CYP 1 A 2 13% CYP 2 A 6 4% CYP 2 B 6 <1% CYP 3 A 30% CYP 2 E 1 7% CYP 2 D 6 2% Shimada et al. , JPET: 1994 CYP 2 C 18% CYP 2 C 8 1. 7% CYP 2 C 19 2. 7% CYP 2 C 9 13. 6% Lasker et al. , Arch. Biophys: 1998
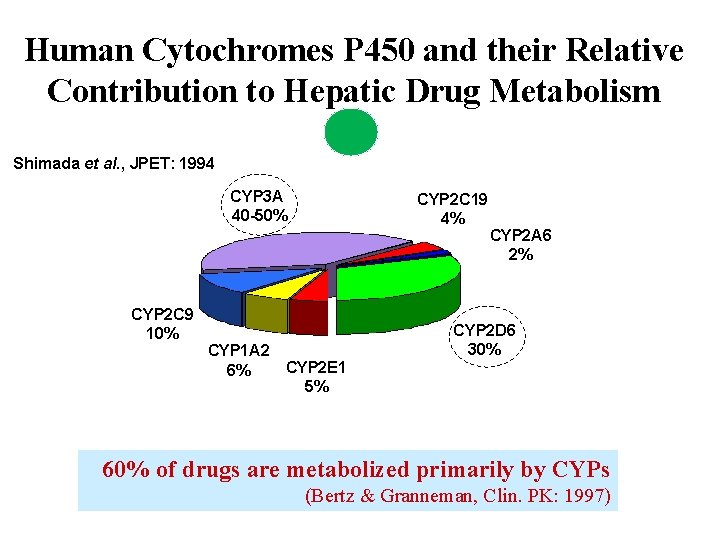
Human Cytochromes P 450 and their Relative Contribution to Hepatic Drug Metabolism Shimada et al. , JPET: 1994 CYP 3 A 40 -50% CYP 2 C 9 10% CYP 2 C 19 4% CYP 1 A 2 CYP 2 E 1 6% 5% CYP 2 A 6 2% CYP 2 D 6 30% 60% of drugs are metabolized primarily by CYPs (Bertz & Granneman, Clin. PK: 1997)
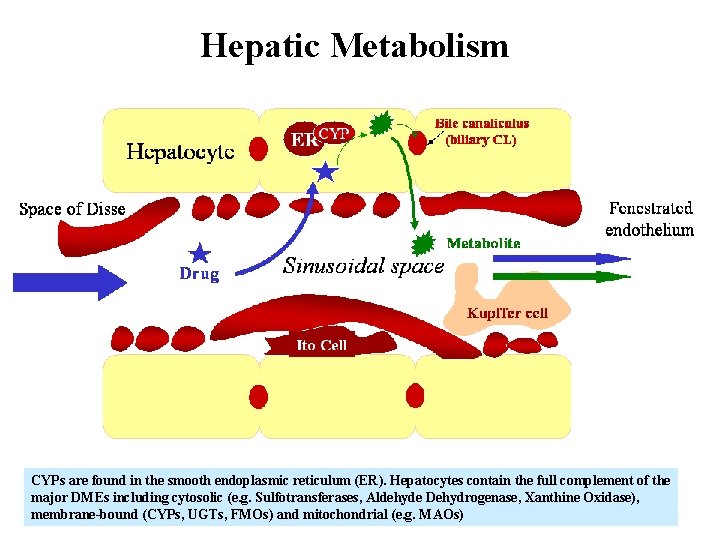
Hepatic Metabolism CYPs are found in the smooth endoplasmic reticulum (ER). Hepatocytes contain the full complement of the major DMEs including cytosolic (e. g. Sulfotransferases, Aldehyde Dehydrogenase, Xanthine Oxidase), membrane-bound (CYPs, UGTs, FMOs) and mitochondrial (e. g. MAOs)
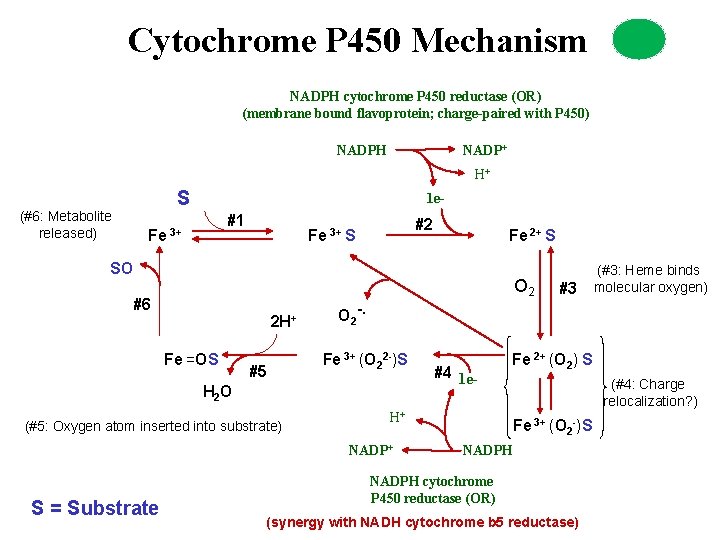
Cytochrome P 450 Mechanism NADPH cytochrome P 450 reductase (OR) (membrane bound flavoprotein; charge-paired with P 450) NADPH NADP+ H+ S (#6: Metabolite released) 1 e- #1 Fe 3+ #2 Fe 3+ S Fe 2+ S SO O 2 #6 2 H+ Fe =OS #5 O 2 -. Fe 3+ (O 22 -)S H 2 O (#5: Oxygen atom inserted into substrate) #4 1 e- H+ NADP+ S = Substrate #3 (#3: Heme binds molecular oxygen) Fe 2+ (O 2) S (#4: Charge relocalization? ) Fe 3+ (O 2 -)S NADPH cytochrome P 450 reductase (OR) (synergy with NADH cytochrome b 5 reductase)
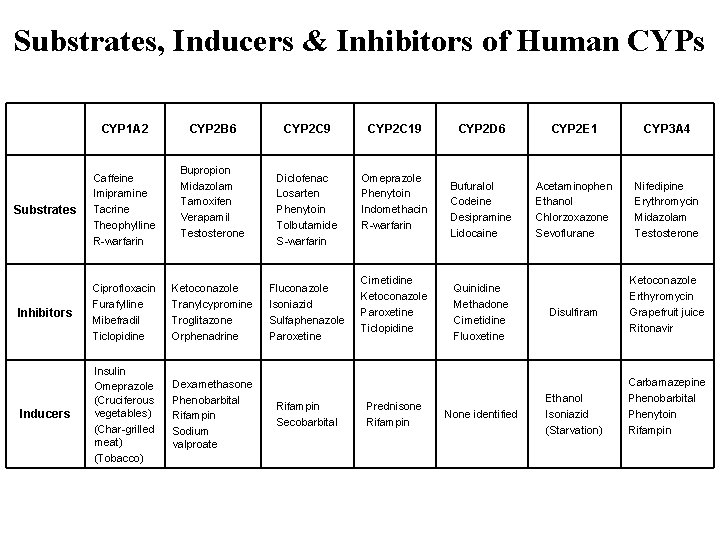
Substrates, Inducers & Inhibitors of Human CYPs CYP 1 A 2 CYP 2 B 6 Bupropion Midazolam Tamoxifen Verapamil Testosterone Substrates Caffeine Imipramine Tacrine Theophylline R-warfarin Inhibitors Ciprofloxacin Furafylline Mibefradil Ticlopidine Ketoconazole Tranylcypromine Troglitazone Orphenadrine Inducers Insulin Omeprazole (Cruciferous vegetables) (Char-grilled meat) (Tobacco) Dexamethasone Phenobarbital Rifampin Sodium valproate CYP 2 C 9 CYP 2 C 19 Diclofenac Losarten Phenytoin Tolbutamide S-warfarin Omeprazole Phenytoin Indomethacin R-warfarin Fluconazole Isoniazid Sulfaphenazole Paroxetine Rifampin Secobarbital Cimetidine Ketoconazole Paroxetine Ticlopidine Prednisone Rifampin CYP 2 D 6 CYP 2 E 1 CYP 3 A 4 Bufuralol Codeine Desipramine Lidocaine Acetaminophen Ethanol Chlorzoxazone Sevoflurane Nifedipine Erythromycin Midazolam Testosterone Quinidine Methadone Cimetidine Fluoxetine None identified Disulfiram Ethanol Isoniazid (Starvation) Ketoconazole Erthyromycin Grapefruit juice Ritonavir Carbamazepine Phenobarbital Phenytoin Rifampin
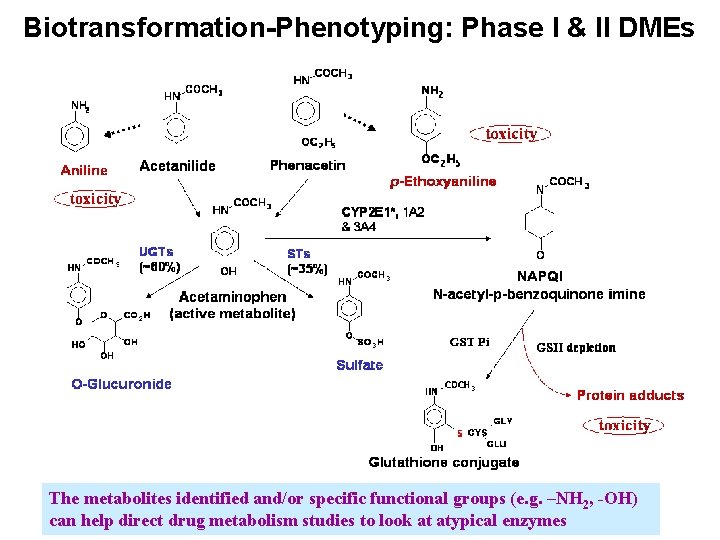
Biotransformation-Phenotyping: Phase I & II DMEs The metabolites identified and/or specific functional groups (e. g. –NH 2, -OH) can help direct drug metabolism studies to look at atypical enzymes

Non-CYP Drug Metabolizing Enzymes (I) Non-CYP Oxidations • Monoamine Oxidase (MAO; mitochondrial) – oxidatively deaminates endogenous substrates including neurotransmitters (dopamine, serotonin, norepinephrine, epinephrine); drugs include triptans • Alcohol & Aldehyde Dehydrogenase (non-specific enzymes; liver cytosol) – ethanol metabolism • Xanthine Oxidase (XO) – converts hypoxanthine to xanthine, and then to uric acid (drugs include theophylline, 6 -mercaptopurine. Allopurinol is a substrate and inhibitor of xanthine oxidase • Flavin Monooxygenases (FMOs; membrane-bound & NADPH-dependent) – catalyze oxygenation of nitrogen, phosphorus, sulfur; particularly facile formation of N-oxides (e. g. cimetidine) • Many others: e. g. O-Methylation, S-Methylation, Amino Acid Conjugation: glycine, taurine, glutathione – metabolites or functional groups offer clues to the likely enzyme involved
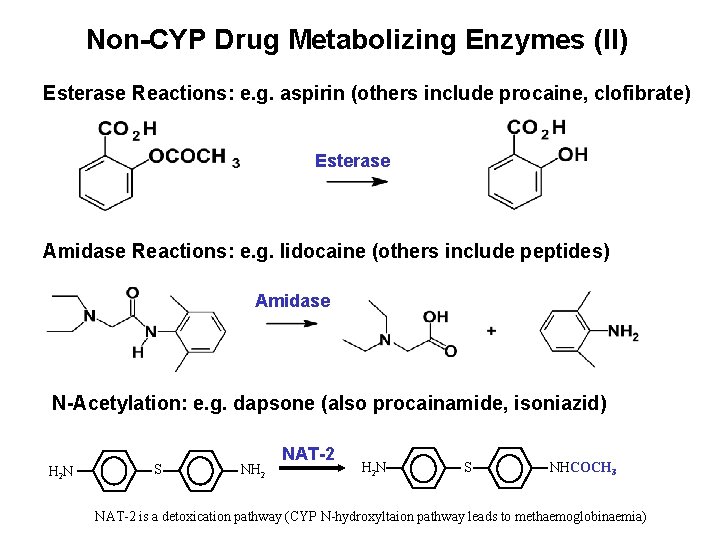
Non-CYP Drug Metabolizing Enzymes (II) Esterase Reactions: e. g. aspirin (others include procaine, clofibrate) Esterase Amidase Reactions: e. g. lidocaine (others include peptides) Amidase O O N-Acetylation: e. g. dapsone (also procainamide, isoniazid) S NH 2 NAT-2 H 2 N S NHCOCH 3 O O H 2 N NAT-2 is a detoxication pathway (CYP N-hydroxyltaion pathway leads to methaemoglobinaemia)

Summary • Metabolism is the major contributor to the systemic exposure and total in vivo clearance of many drugs and thus an important consideration in Drug Discovery and Development • The liver is the major organ of metabolic clearance (however, drug metabolism can occur elsewhere) • The cytochromes P 450 are the major enzymes of drug metabolism, but there are many others to consider on a case-by-case basis
- Slides: 22