Zumdahl De Coste World of CHEMISTRY Chapter 12





- Slides: 24

Zumdahl De. Coste World of CHEMISTRY

Chapter 12 Chemical Bonding Copyright© by Houghton Mifflin Company. All rights reserved.

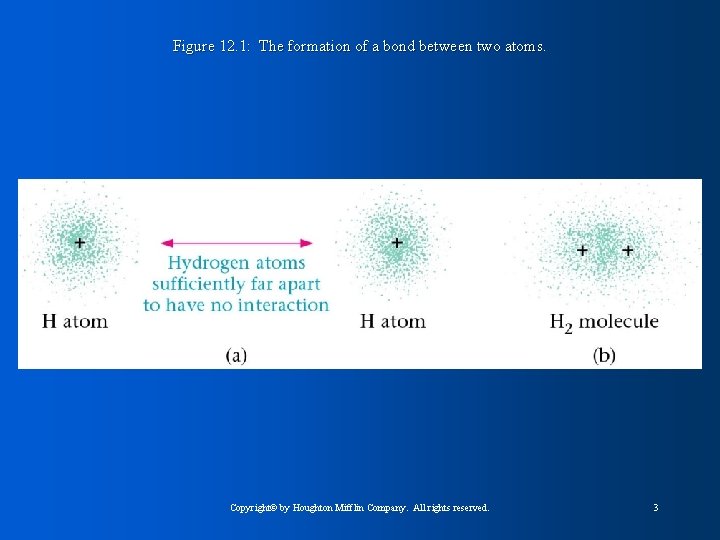
Figure 12. 1: The formation of a bond between two atoms. Copyright© by Houghton Mifflin Company. All rights reserved. 3

Figure 12. 2: Probability representations of the electron sharing in HF. Copyright© by Houghton Mifflin Company. All rights reserved. 4

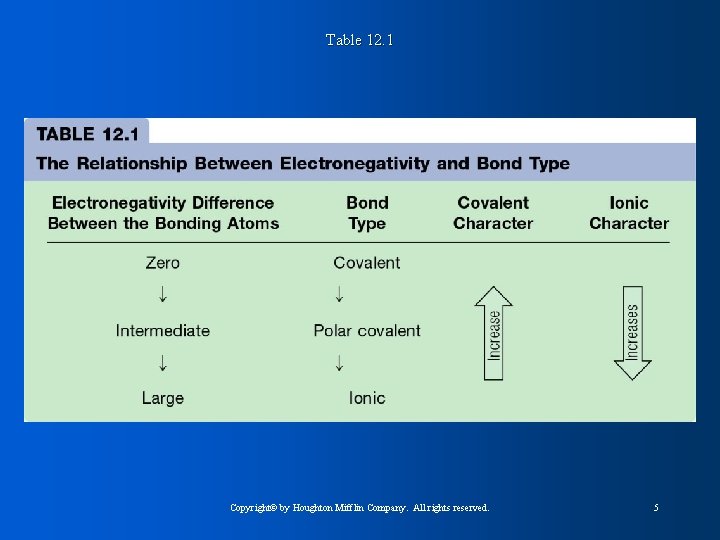
Table 12. 1 Copyright© by Houghton Mifflin Company. All rights reserved. 5

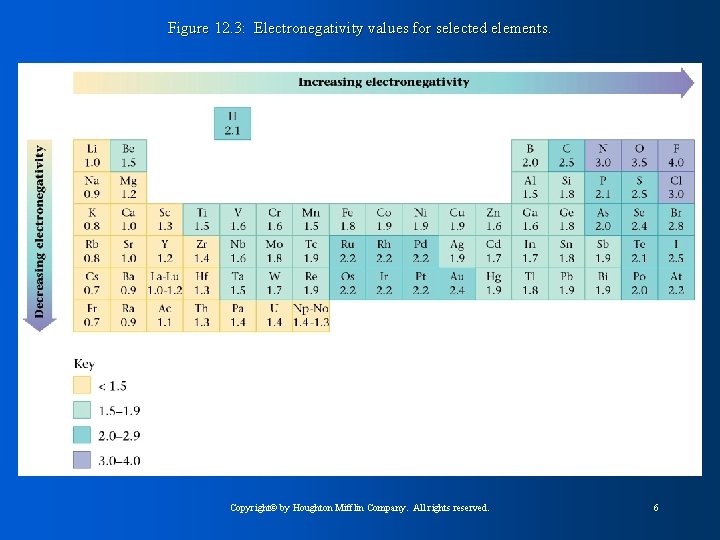
Figure 12. 3: Electronegativity values for selected elements. Copyright© by Houghton Mifflin Company. All rights reserved. 6

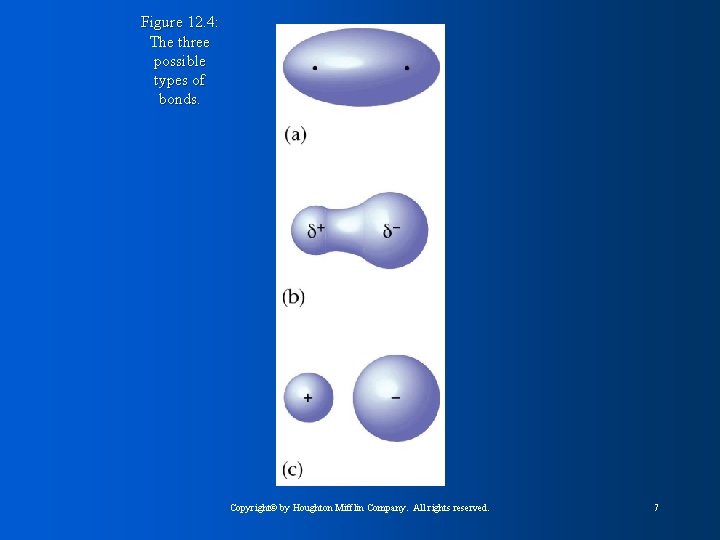
Figure 12. 4: The three possible types of bonds. Copyright© by Houghton Mifflin Company. All rights reserved. 7

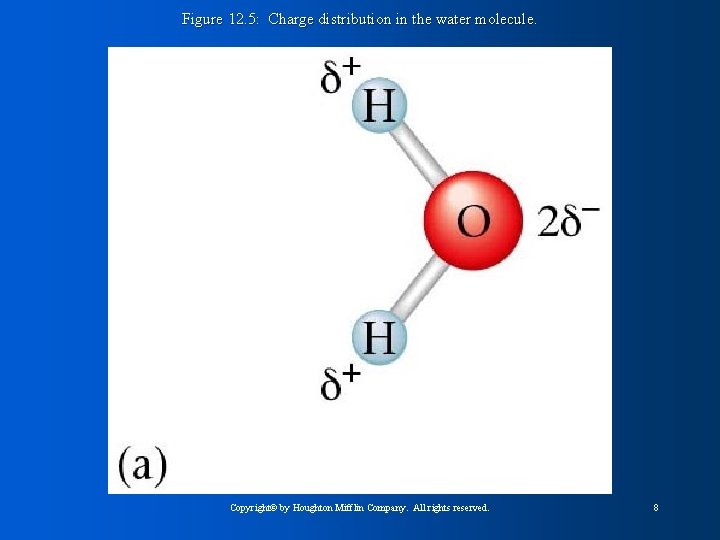
Figure 12. 5: Charge distribution in the water molecule. Copyright© by Houghton Mifflin Company. All rights reserved. 8

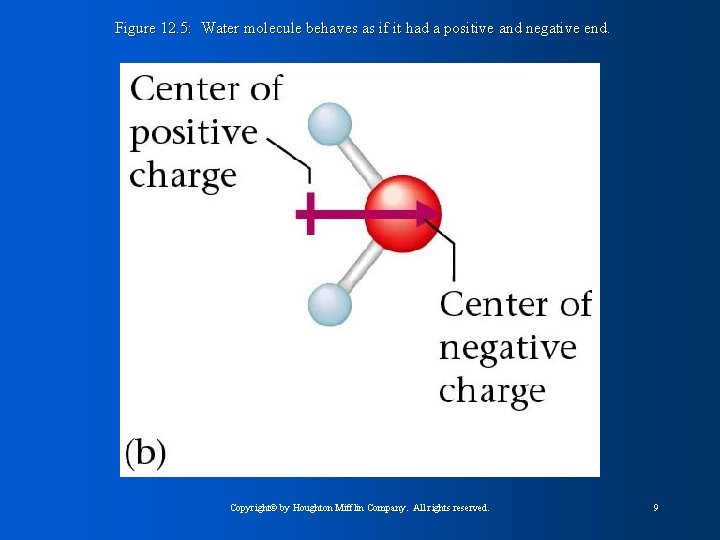
Figure 12. 5: Water molecule behaves as if it had a positive and negative end. Copyright© by Houghton Mifflin Company. All rights reserved. 9

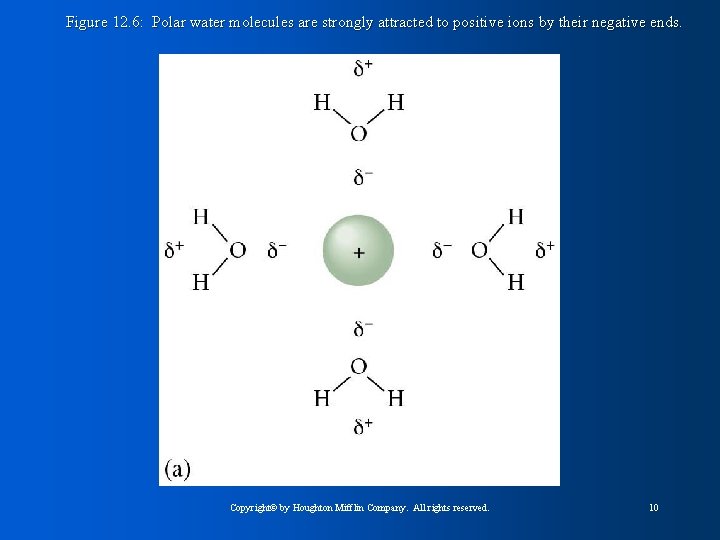
Figure 12. 6: Polar water molecules are strongly attracted to positive ions by their negative ends. Copyright© by Houghton Mifflin Company. All rights reserved. 10

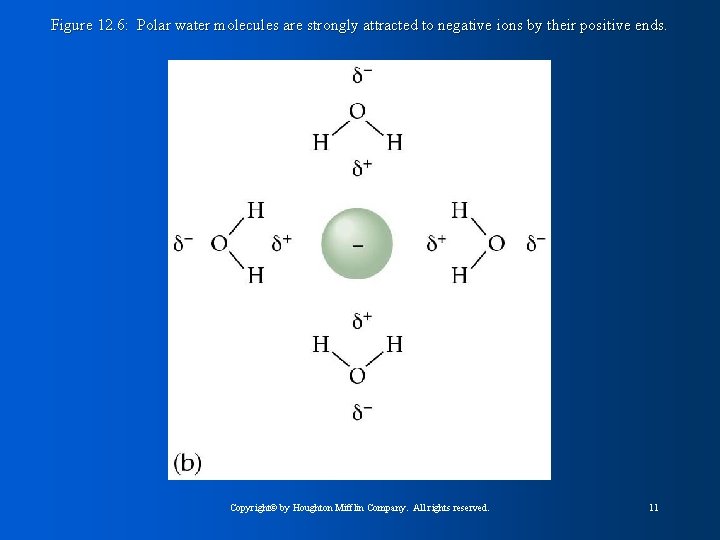
Figure 12. 6: Polar water molecules are strongly attracted to negative ions by their positive ends. Copyright© by Houghton Mifflin Company. All rights reserved. 11

Table 12. 2 Copyright© by Houghton Mifflin Company. All rights reserved. 12

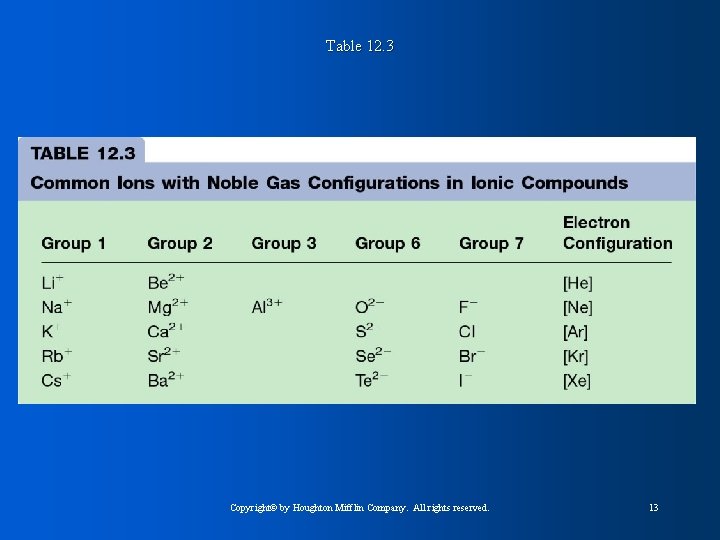
Table 12. 3 Copyright© by Houghton Mifflin Company. All rights reserved. 13

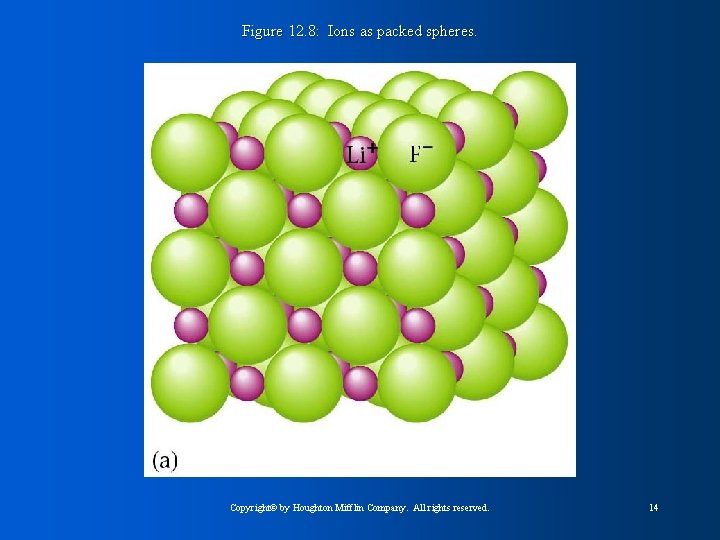
Figure 12. 8: Ions as packed spheres. Copyright© by Houghton Mifflin Company. All rights reserved. 14

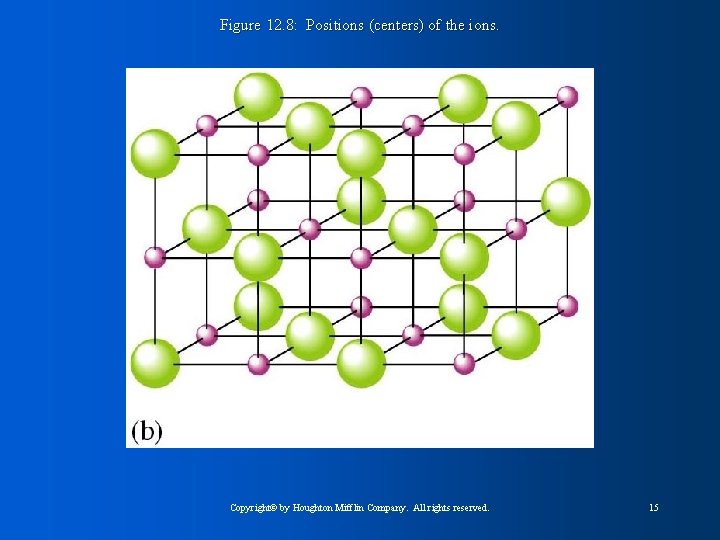
Figure 12. 8: Positions (centers) of the ions. Copyright© by Houghton Mifflin Company. All rights reserved. 15

Figure 12. 9: Relative sizes of some ions and their parent atoms. Copyright© by Houghton Mifflin Company. All rights reserved. 16

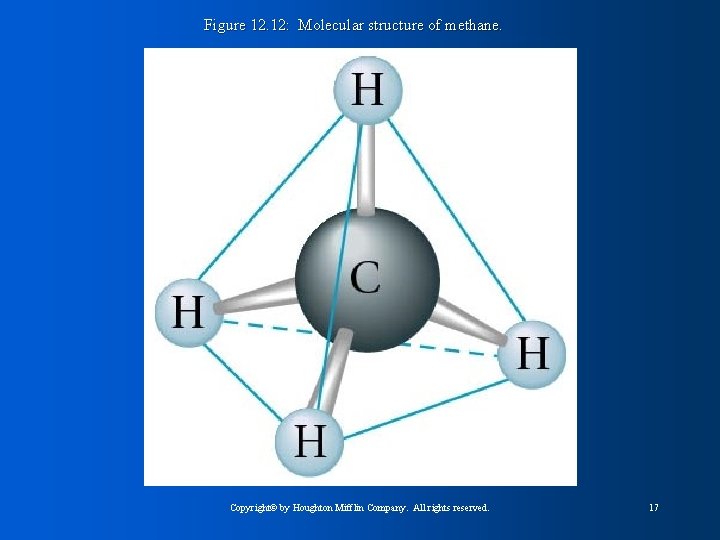
Figure 12. 12: Molecular structure of methane. Copyright© by Houghton Mifflin Company. All rights reserved. 17

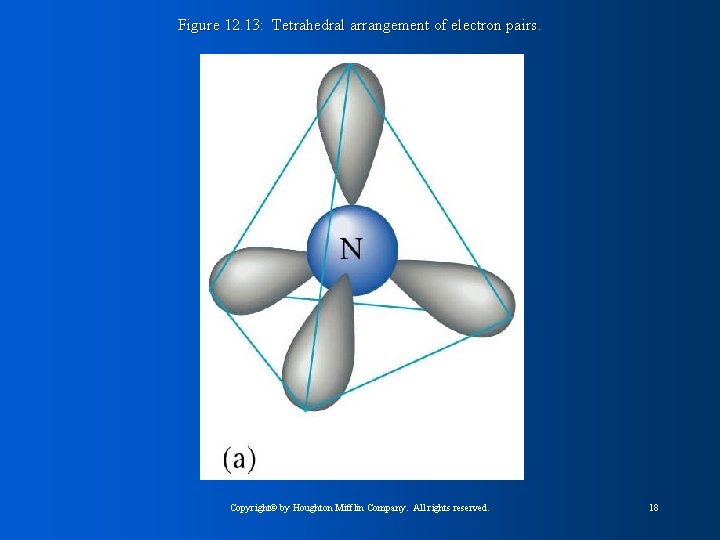
Figure 12. 13: Tetrahedral arrangement of electron pairs. Copyright© by Houghton Mifflin Company. All rights reserved. 18

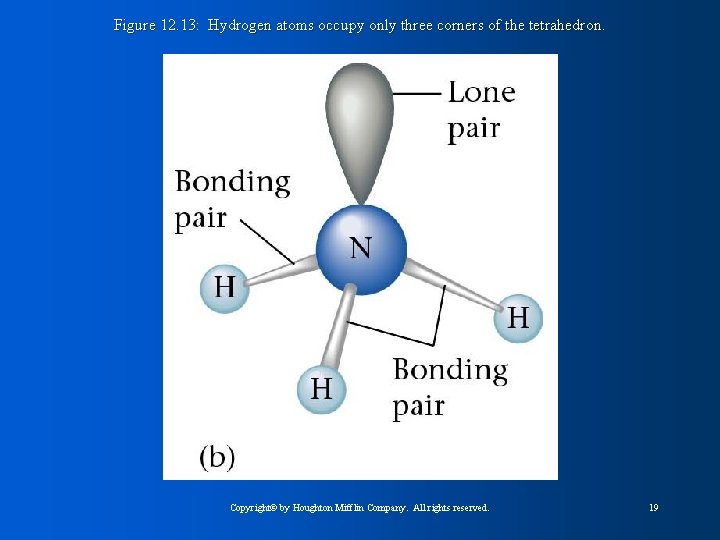
Figure 12. 13: Hydrogen atoms occupy only three corners of the tetrahedron. Copyright© by Houghton Mifflin Company. All rights reserved. 19

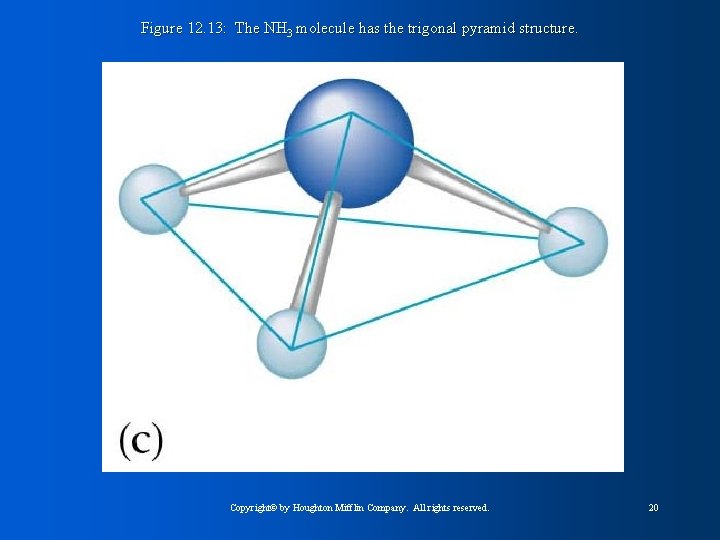
Figure 12. 13: The NH 3 molecule has the trigonal pyramid structure. Copyright© by Houghton Mifflin Company. All rights reserved. 20

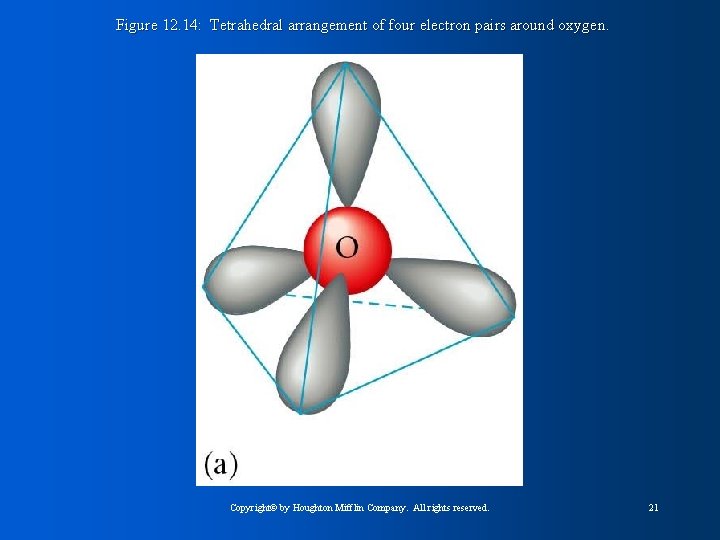
Figure 12. 14: Tetrahedral arrangement of four electron pairs around oxygen. Copyright© by Houghton Mifflin Company. All rights reserved. 21

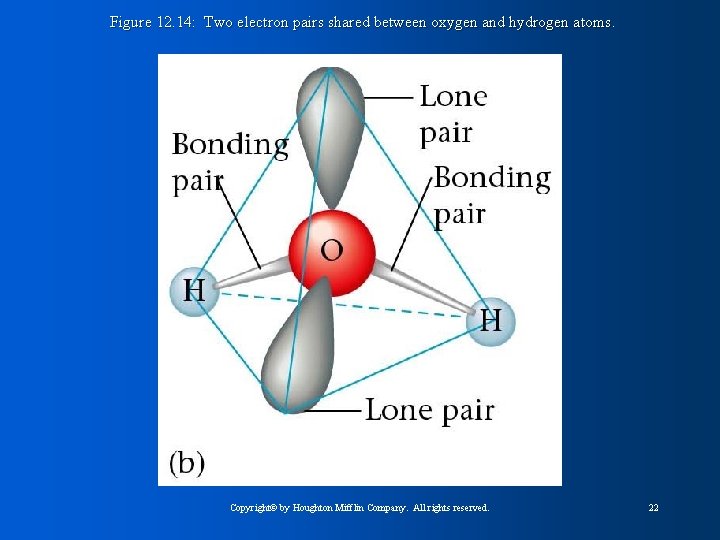
Figure 12. 14: Two electron pairs shared between oxygen and hydrogen atoms. Copyright© by Houghton Mifflin Company. All rights reserved. 22

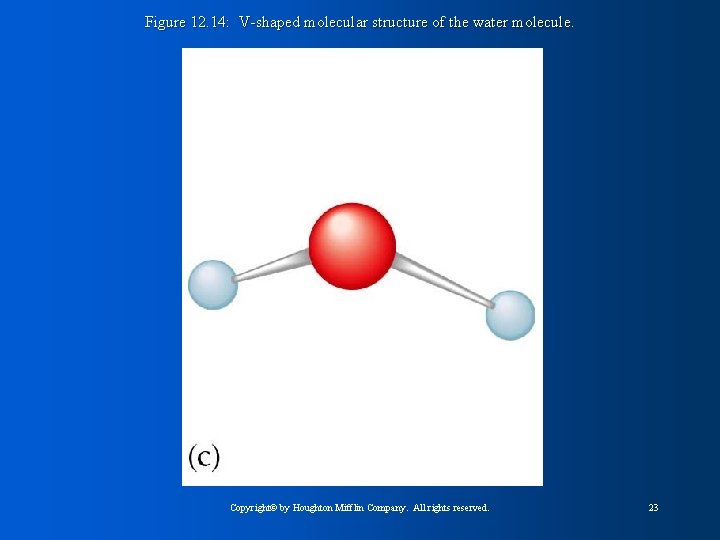
Figure 12. 14: V-shaped molecular structure of the water molecule. Copyright© by Houghton Mifflin Company. All rights reserved. 23

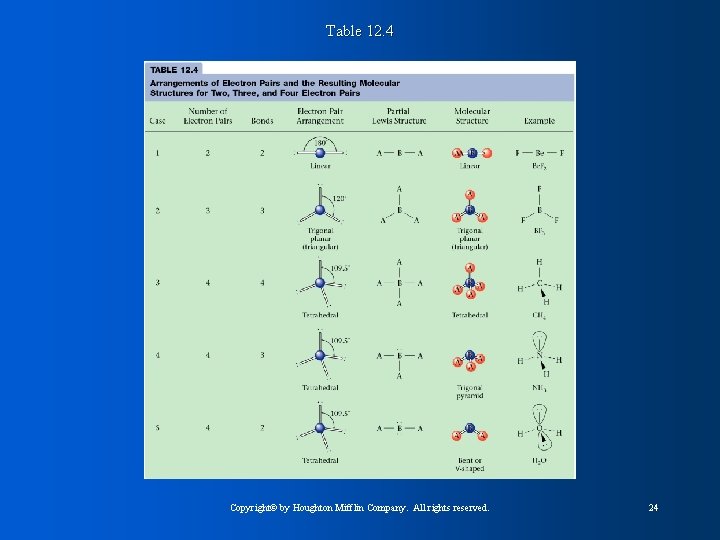
Table 12. 4 Copyright© by Houghton Mifflin Company. All rights reserved. 24