Zumdahl De Coste World of CHEMISTRY Topic 9













- Slides: 23

Zumdahl • De. Coste World of CHEMISTRY

Topic 9 -2008 Properties of Gases

Figure 13. 2: A glass tube is filled with mercury and inverted in a dish of mercury at sea level. Copyright © Houghton Mifflin Company 3

When water at 25ºC is heated and changes to a gas at 110ºC, the water molecules A. become more organized. B. move farther apart. C. stop moving. D. move closer together. E. move more slowly. Copyright © Houghton Mifflin Company 4

6. A sample of liquid ammonia (NH 3) is completely evaporated (changed to a gas) in a closed container as shown: Which of the following diagrams best represents what you would “see” in the same area of the magnified view of the vapor? Copyright © Houghton Mifflin Company 5

7. A diagram representing carbon dioxide molecules in the solid phase, also known as dry ice, is shown below. Which of these molecular diagrams best shows what dry ice would look like after it sublimates (solid into a gas)? Copyright © Houghton Mifflin Company 6

Comparing Gases Helium Gas Argon Gas 10 m. L 1. How do the radii of these types of atoms compare? 2. How will the mass of these two gases compare? 3. How will the number of atoms compare? Copyright © Houghton Mifflin Company 7

Figure 13. 9: The relationship between volume V and number of moles n. Copyright © Houghton Mifflin Company 8

Postulates of KMT. Copyright © Houghton Mifflin Company 9

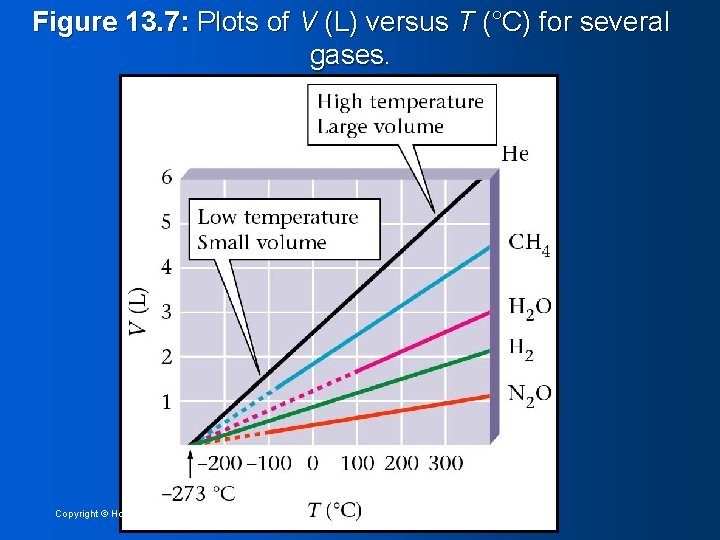
Figure 13. 7: Plots of V (L) versus T (°C) for several gases. Copyright © Houghton Mifflin Company 10

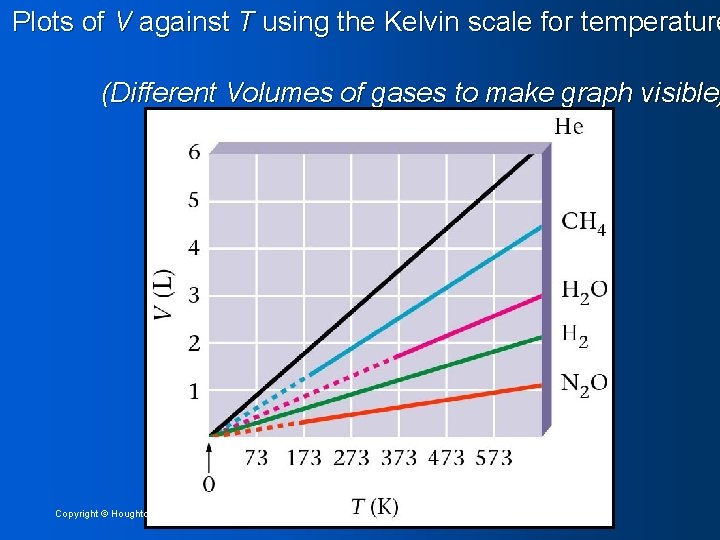
Plots of V against T using the Kelvin scale for temperature (Different Volumes of gases to make graph visible) Copyright © Houghton Mifflin Company 11

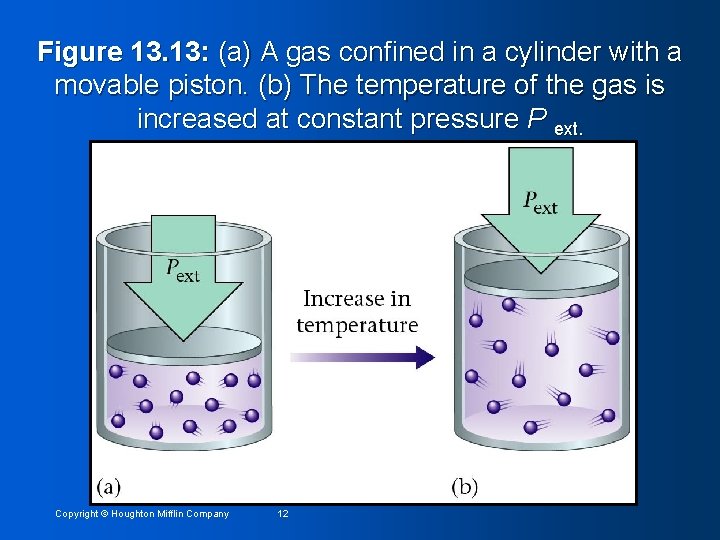
Figure 13. 13: (a) A gas confined in a cylinder with a movable piston. (b) The temperature of the gas is increased at constant pressure P ext. Copyright © Houghton Mifflin Company 12

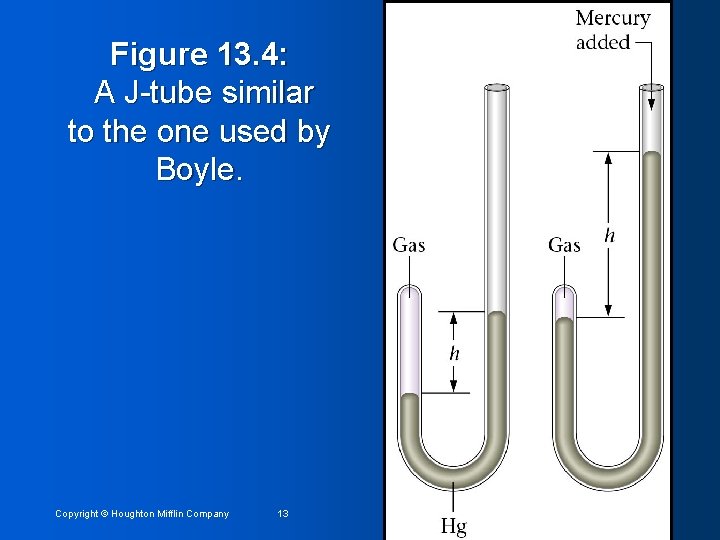
Figure 13. 4: A J-tube similar to the one used by Boyle. Copyright © Houghton Mifflin Company 13

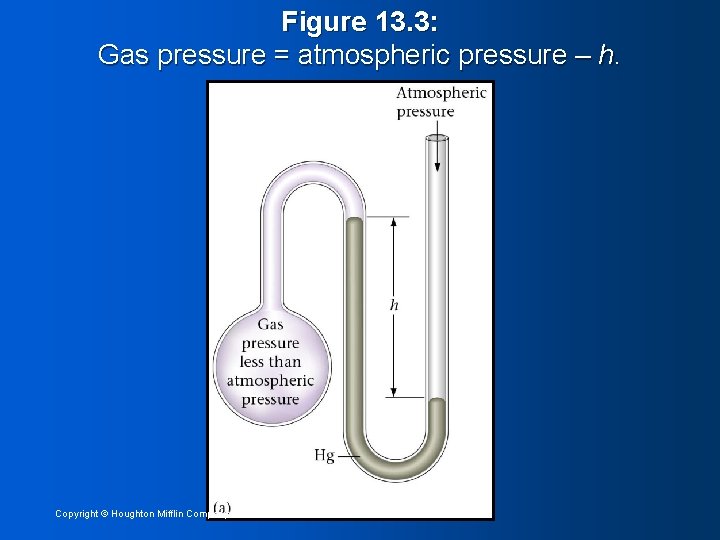
Figure 13. 3: Gas pressure = atmospheric pressure – h. Copyright © Houghton Mifflin Company 14

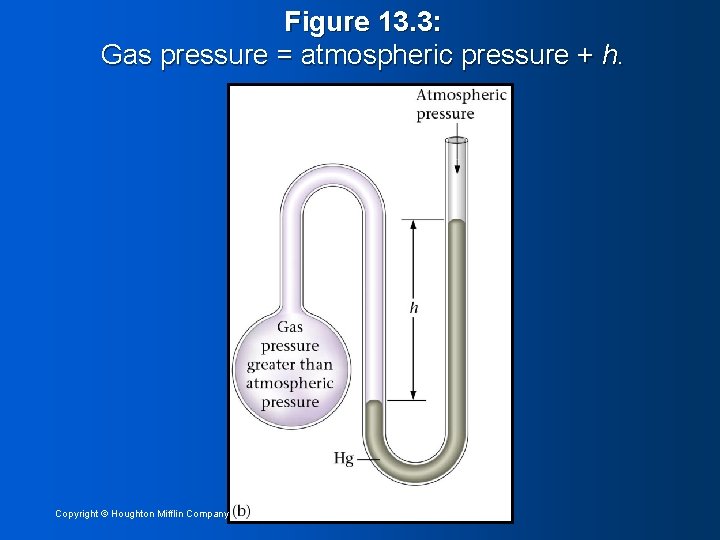
Figure 13. 3: Gas pressure = atmospheric pressure + h. Copyright © Houghton Mifflin Company 15

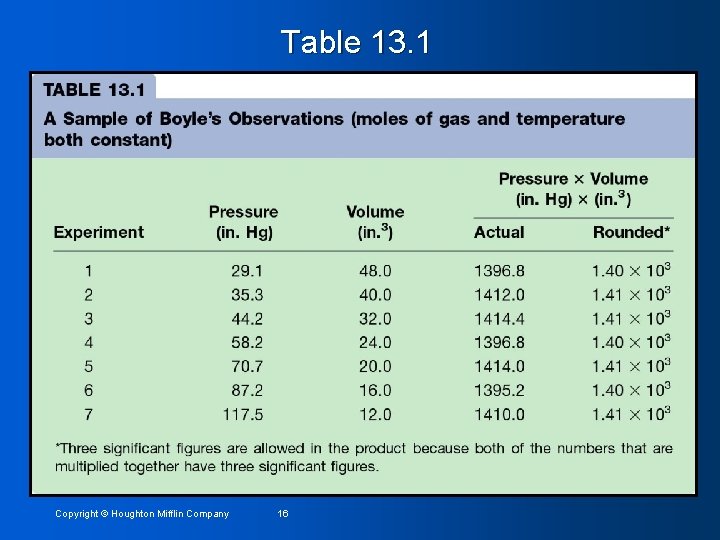
Table 13. 1 Copyright © Houghton Mifflin Company 16

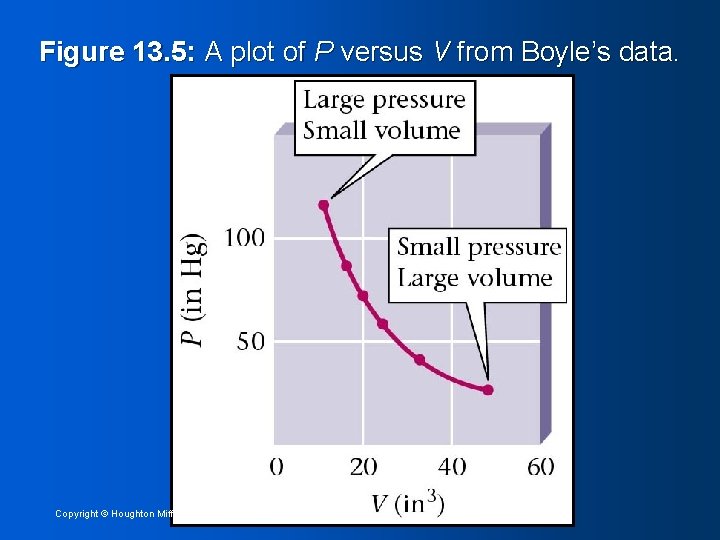
Figure 13. 5: A plot of P versus V from Boyle’s data. Copyright © Houghton Mifflin Company 17

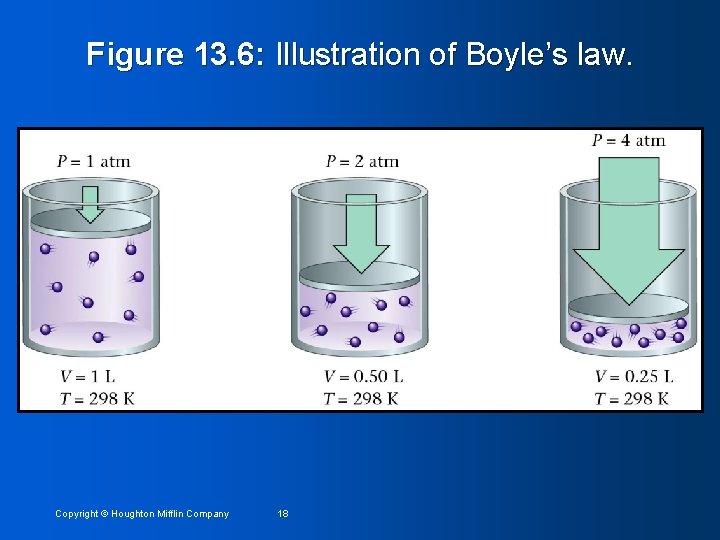
Figure 13. 6: Illustration of Boyle’s law. Copyright © Houghton Mifflin Company 18

“Vacuum-packed Students” Increasing the external pressure reduces the volume. Copyright © Houghton Mifflin Company 19

Figure 13. 10: When two gases are present, the total pressure is the sum of the partial pressures of the gases. Copyright © Houghton Mifflin Company 20

Collecting Gas over Water Pressure. Total = PO 2 + PH 2 O

Figure 13. 12: The production of oxygen by thermal decomposition. Copyright © Houghton Mifflin Company 22

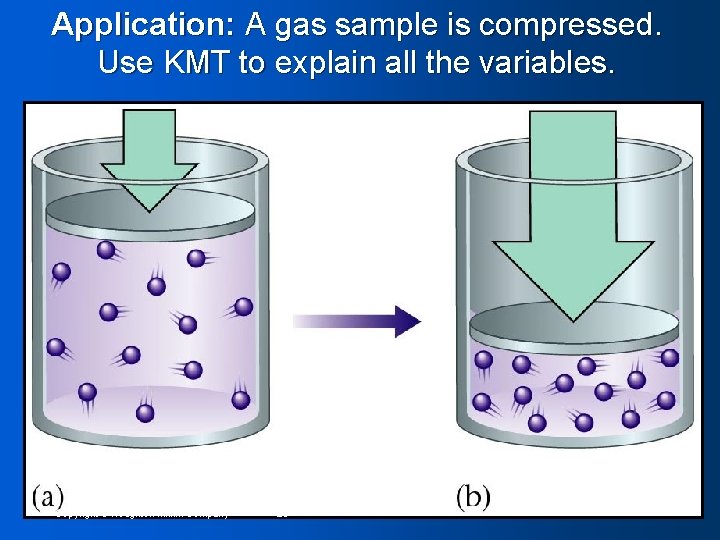
Application: A gas sample is compressed. Use KMT to explain all the variables. Copyright © Houghton Mifflin Company 23