Absorption of Radiation Zumdahl De Coste World of
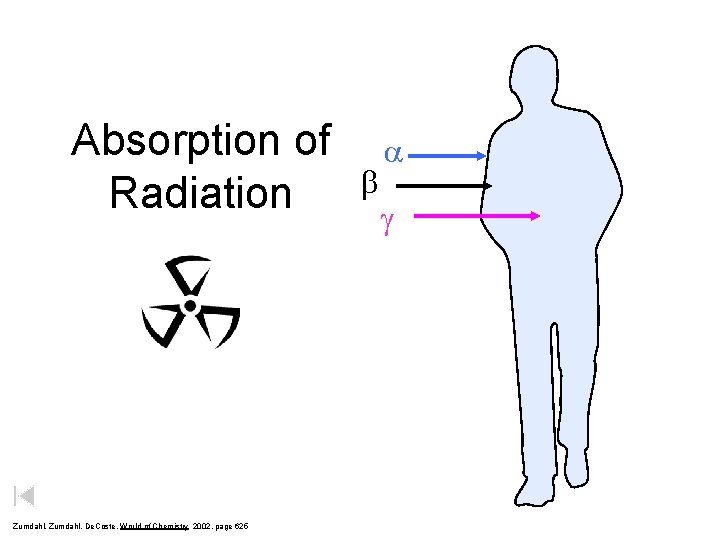
Absorption of Radiation Zumdahl, De. Coste, World of Chemistry 2002, page 625 b a g
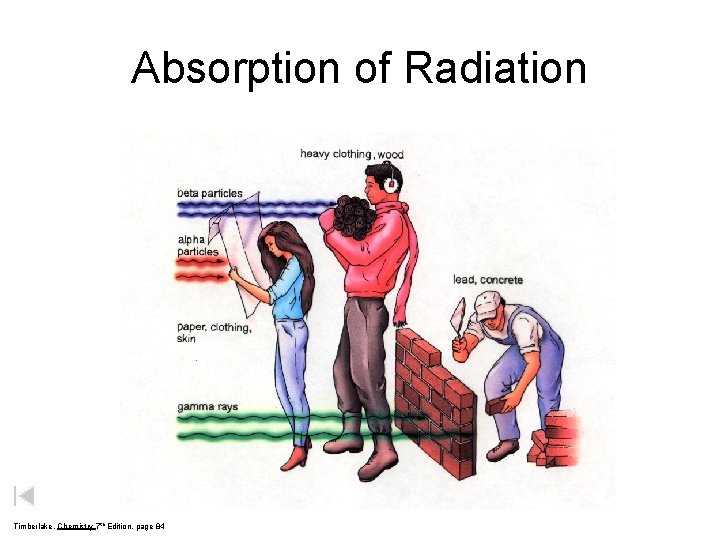
Absorption of Radiation Timberlake, Chemistry 7 th Edition, page 84
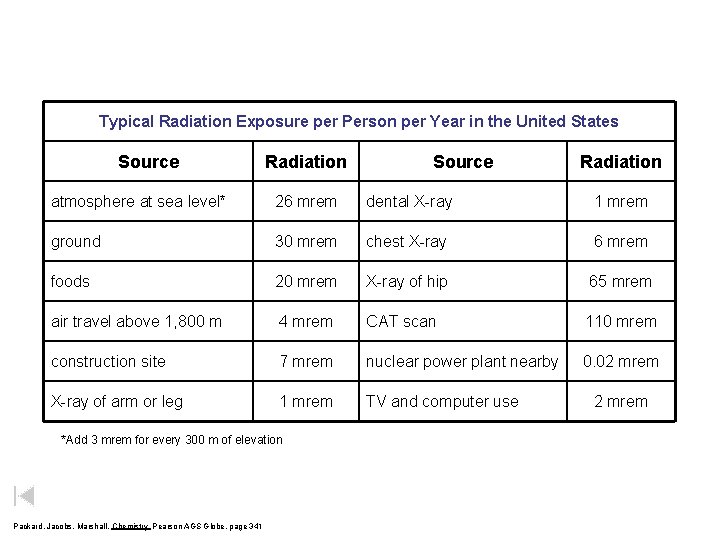
Typical Radiation Exposure per Person per Year in the United States Source Radiation atmosphere at sea level* 26 mrem dental X-ray 1 mrem ground 30 mrem chest X-ray 6 mrem foods 20 mrem X-ray of hip 65 mrem air travel above 1, 800 m 4 mrem CAT scan 110 mrem construction site 7 mrem nuclear power plant nearby 0. 02 mrem X-ray of arm or leg 1 mrem TV and computer use *Add 3 mrem for every 300 m of elevation Packard, Jacobs, Marshall, Chemistry Pearson AGS Globe, page 341 2 mrem
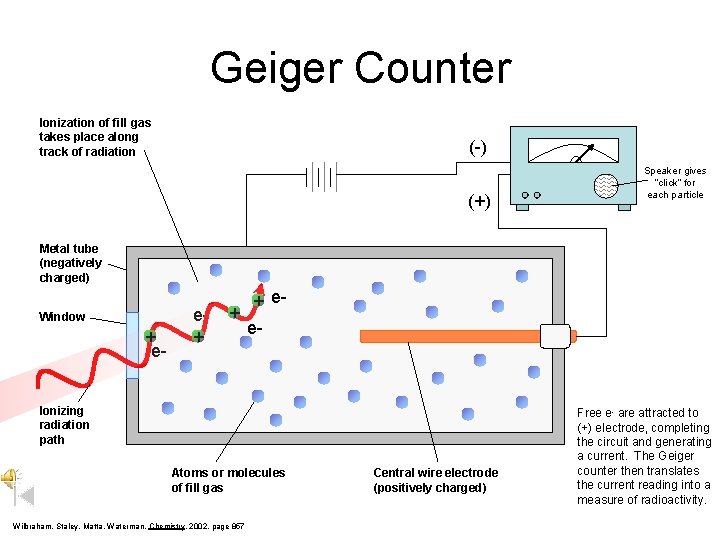
Geiger Counter Ionization of fill gas takes place along track of radiation (-) (+) Speaker gives “click” for each particle Metal tube (negatively charged) Window + e- e+ + + ee- Ionizing radiation path Atoms or molecules of fill gas Wilbraham, Staley, Matta, Waterman, Chemistry, 2002, page 857 Central wire electrode (positively charged) Free e- are attracted to (+) electrode, completing the circuit and generating a current. The Geiger counter then translates the current reading into a measure of radioactivity.
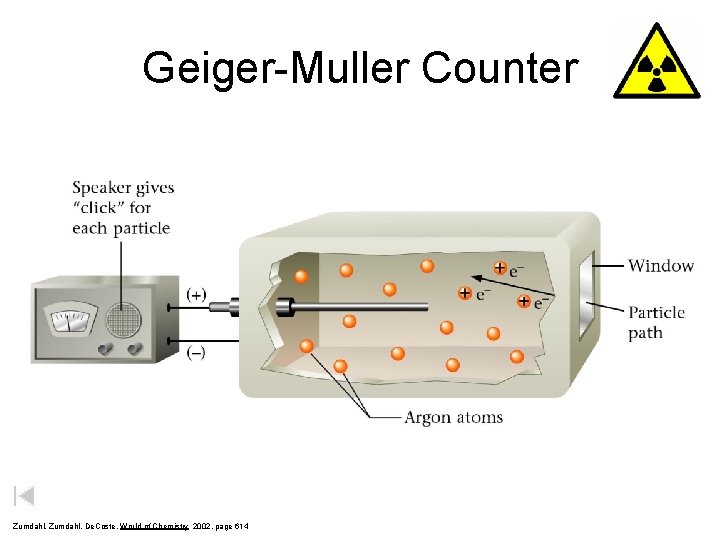
Geiger-Muller Counter Zumdahl, De. Coste, World of Chemistry 2002, page 614
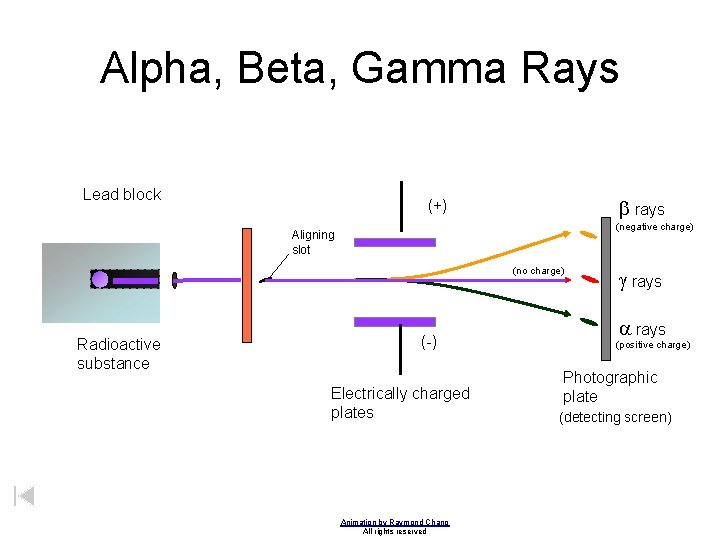
Alpha, Beta, Gamma Rays Lead block b rays (+) (negative charge) Aligning slot (no charge) Radioactive substance (-) Electrically charged plates Animation by Raymond Chang All rights reserved g rays a rays (positive charge) Photographic plate (detecting screen)

Types of Radiation Type Alpha particle Beta particle Positron Gamma ray Symbol Charge Mass (amu) 2+ 4. 015062 1 - 0. 0005486 1+ 0. 0005486 0 0
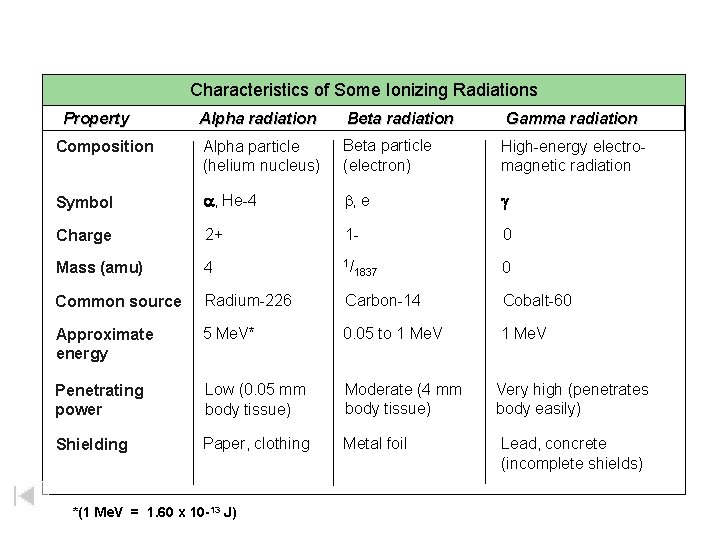
Characteristics of Some Ionizing Radiations Characteristics of Some Ionizing Property Alpha radiation Beta radiation Composition Alpha particle (helium nucleus) Beta particle (electron) High-energy electromagnetic radiation Symbol a, He-4 b, e g Charge 2+ 1 - 0 Mass (amu) 4 1/ 0 Common source Radium-226 Carbon-14 Cobalt-60 Approximate energy 5 Me. V* 0. 05 to 1 Me. V Penetrating power Low (0. 05 mm body tissue) Moderate (4 mm body tissue) Very high (penetrates body easily) Shielding Paper, clothing Metal foil *(1 Me. V = 1. 60 x 10 -13 J) 1837 Gamma radiation Lead, concrete (incomplete shields)

Nuclear reactions Nuclear equations show atoms decay. Similar to chemical equations. - must still balance mass and charge. Differ from chemical equations because - we can change the elements. …transmutation - the type of isotope is important.
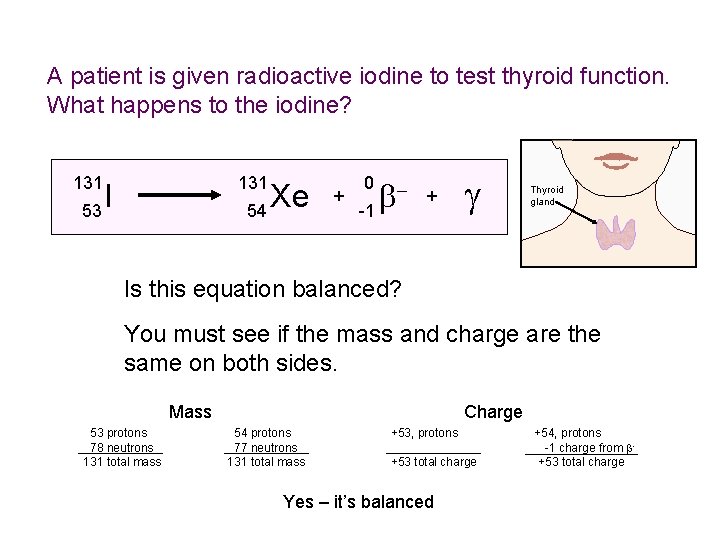
A patient is given radioactive iodine to test thyroid function. What happens to the iodine? 131 I 53 Xe 54 + 0 b -1 + g Thyroid gland Is this equation balanced? You must see if the mass and charge are the same on both sides. Mass 53 protons 78 neutrons 131 total mass Charge 54 protons 77 neutrons 131 total mass +53, protons +53 total charge Yes – it’s balanced +54, protons -1 charge from b+53 total charge
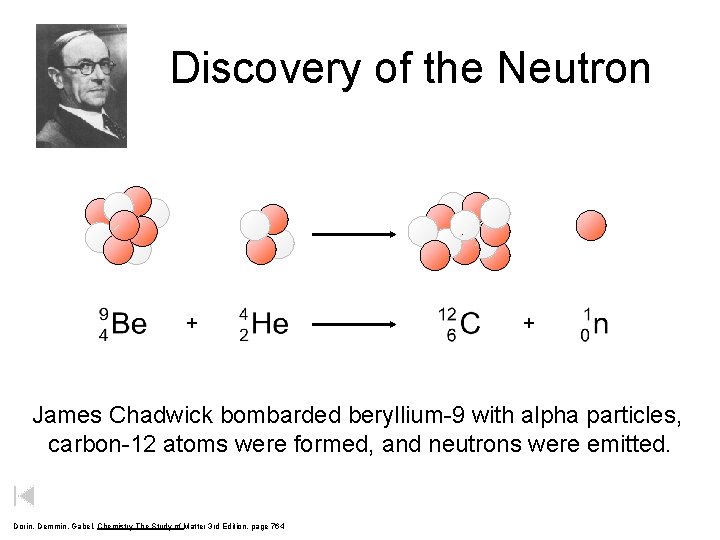
Discovery of the Neutron + + James Chadwick bombarded beryllium-9 with alpha particles, carbon-12 atoms were formed, and neutrons were emitted. Dorin, Demmin, Gabel, Chemistry The Study of Matter 3 rd Edition, page 764
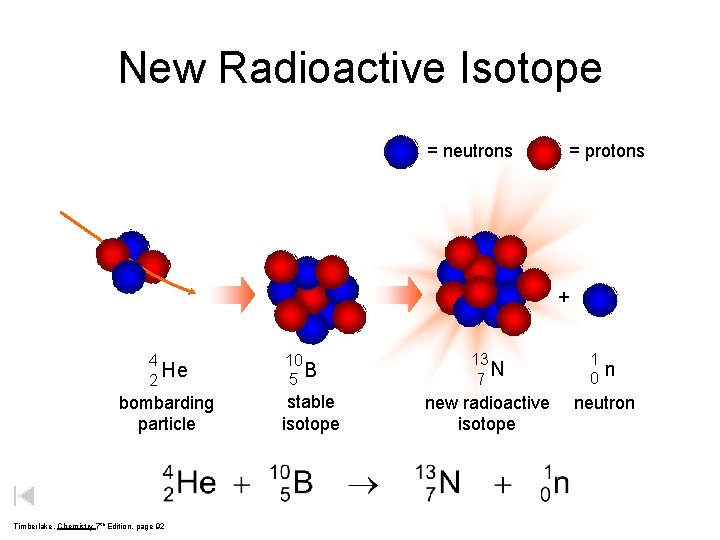
New Radioactive Isotope = neutrons = protons + 4 He 2 bombarding particle Timberlake, Chemistry 7 th Edition, page 92 10 B 5 stable isotope 13 N 7 new radioactive isotope 1 0 n neutron
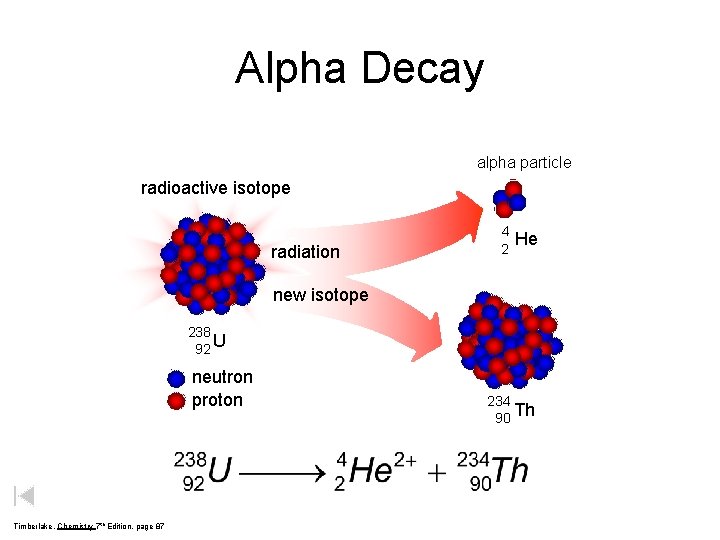
Alpha Decay alpha particle radioactive isotope radiation 4 2 He new isotope 238 92 U neutron proton Timberlake, Chemistry 7 th Edition, page 87 234 90 Th
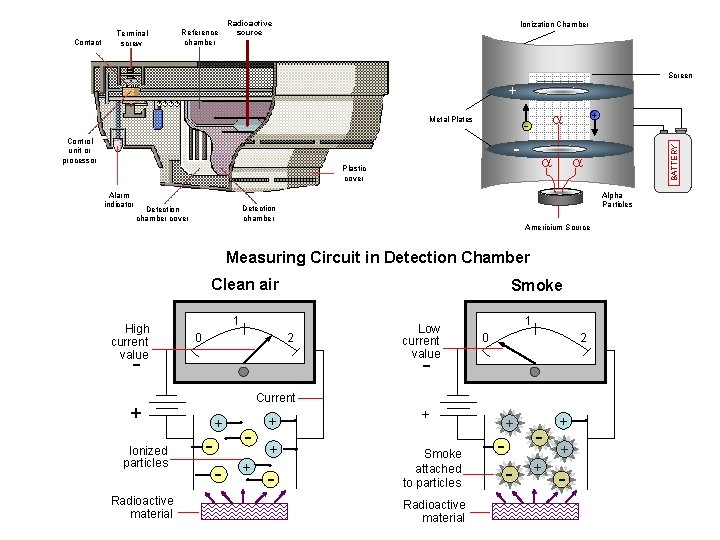
Contact Terminal screw Reference chamber Radioactive source Ionization Chamber Screen + Control unit or processor - Alpha Particles Detection chamber cover + a a Plastic cover Alarm indicator a - Americium Source Measuring Circuit in Detection Chamber Clean air High current value Smoke 1 2 0 - Ionized particles Radioactive material Low current value 1 0 2 - Current + + - + + + - BATTERY Metal Plates + Smoke attached to particles Radioactive material + - + + + -
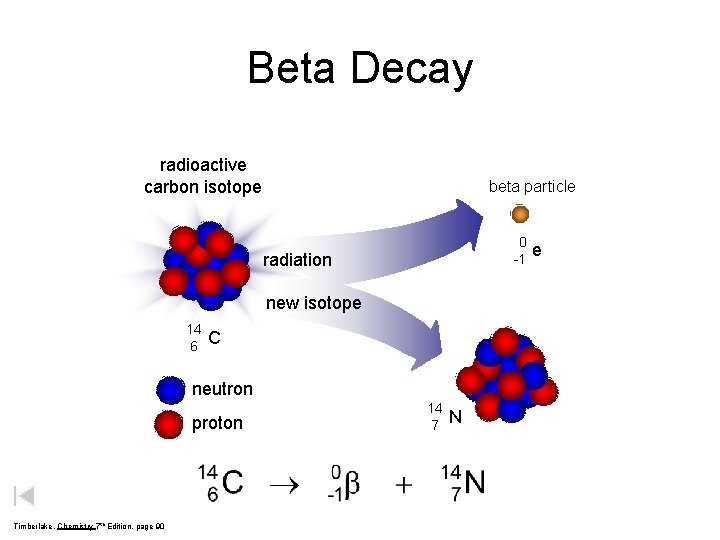
Beta Decay radioactive carbon isotope beta particle 0 -1 radiation new isotope 14 6 C neutron proton Timberlake, Chemistry 7 th Edition, page 90 14 7 N e
- Slides: 15