The European Association Medical devices Notified Bodies Medical

The European Association Medical devices Notified Bodies Medical Device Survey 2018 Data from all 23 members (end 2018) Team-NB-MD-Survey-2018 1
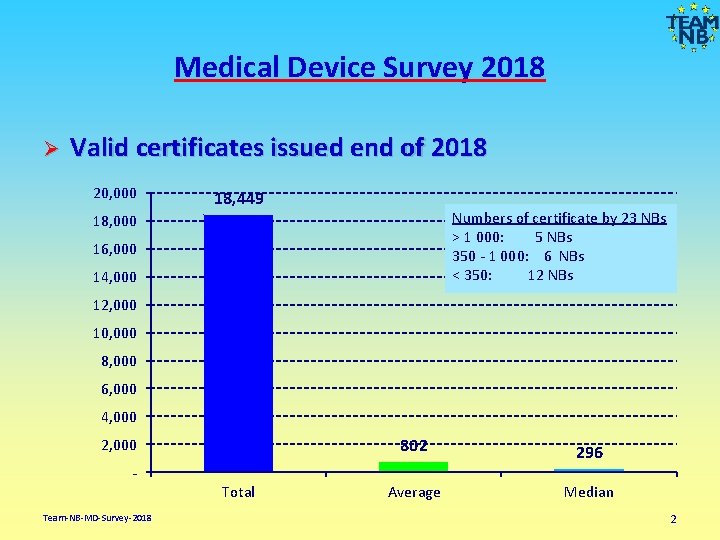
Medical Device Survey 2018 Ø Valid certificates issued end of 2018 20, 000 18, 449 Numbers of certificate by 23 NBs > 1 000: 5 NBs 350 - 1 000: 6 NBs < 350: 12 NBs 18, 000 16, 000 14, 000 12, 000 10, 000 8, 000 6, 000 4, 000 2, 000 802 296 Average Median Total Team-NB-MD-Survey-2018 2
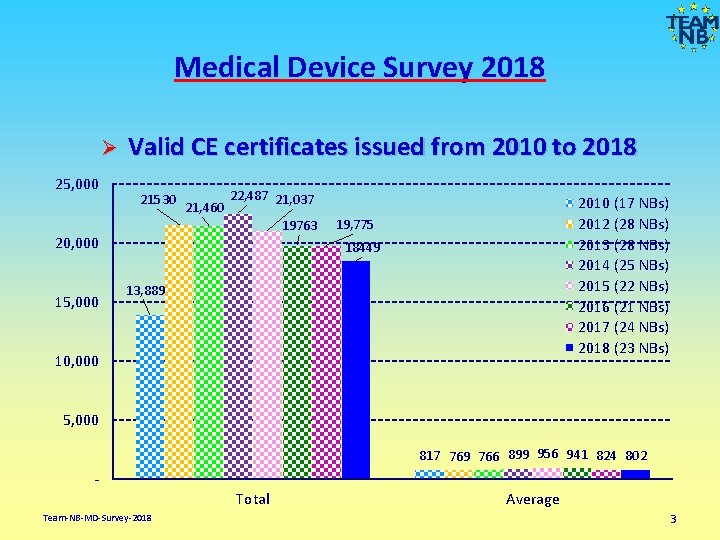
Medical Device Survey 2018 Ø 25, 000 Valid CE certificates issued from 2010 to 2018 21530 21, 460 22, 487 21, 037 19763 20, 000 15, 000 2010 (17 NBs) 2012 (28 NBs) 2013 (28 NBs) 2014 (25 NBs) 2015 (22 NBs) 2016 (21 NBs) 2017 (24 NBs) 2018 (23 NBs) 19, 775 18449 13, 889 10, 000 5, 000 817 769 766 899 956 941 824 802 Total Team-NB-MD-Survey-2018 Average 3
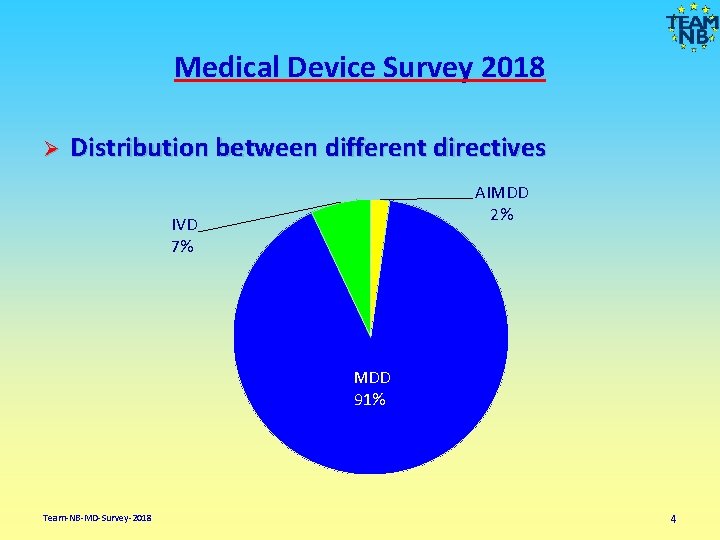
Medical Device Survey 2018 Ø Distribution between different directives AIMDD 2% IVD 7% MDD 91% Team-NB-MD-Survey-2018 4
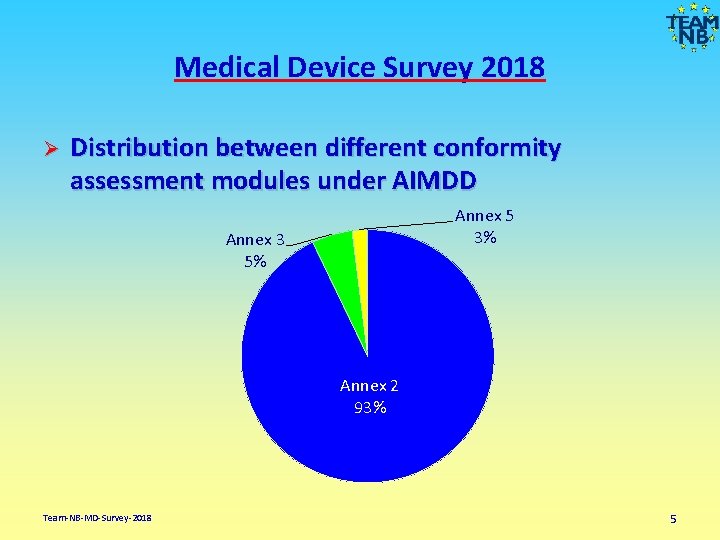
Medical Device Survey 2018 Ø Distribution between different conformity assessment modules under AIMDD Annex 5 3% Annex 3 5% Annex 2 93% Team-NB-MD-Survey-2018 5
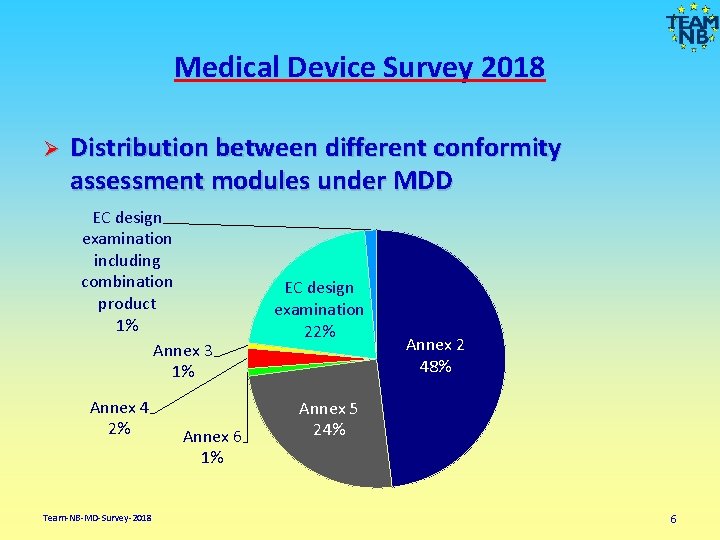
Medical Device Survey 2018 Ø Distribution between different conformity assessment modules under MDD EC design examination including combination product 1% Annex 3 1% Annex 4 2% Team-NB-MD-Survey-2018 Annex 6 1% EC design examination 22% Annex 2 48% Annex 5 24% 6
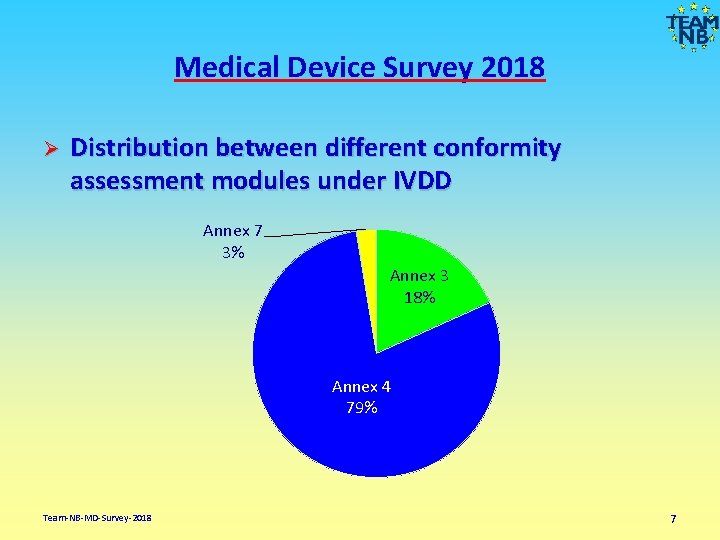
Medical Device Survey 2018 Ø Distribution between different conformity assessment modules under IVDD Annex 7 3% Annex 3 18% Annex 4 79% Team-NB-MD-Survey-2018 7
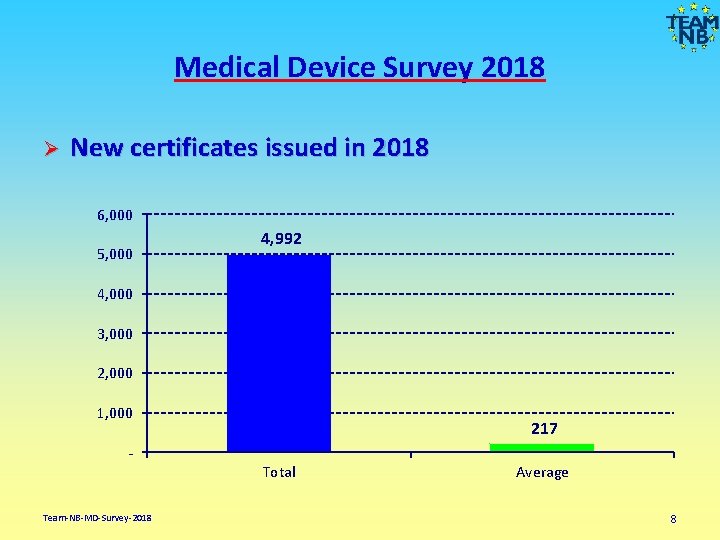
Medical Device Survey 2018 Ø New certificates issued in 2018 6, 000 5, 000 4, 992 4, 000 3, 000 2, 000 1, 000 217 Total Team-NB-MD-Survey-2018 Average 8
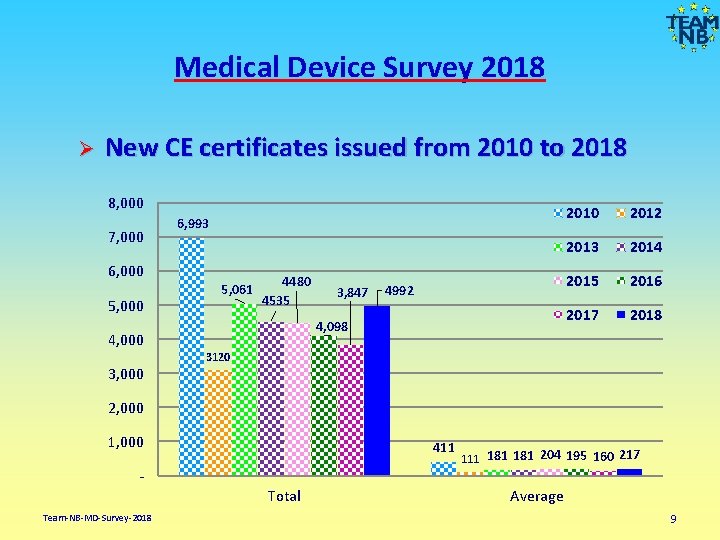
Medical Device Survey 2018 Ø New CE certificates issued from 2010 to 2018 8, 000 7, 000 6, 000 5, 000 6, 993 5, 061 4480 4535 4992 4, 098 4, 000 3, 000 3, 847 2010 2012 2013 2014 2015 2016 2017 2018 3120 2, 000 1, 000 411 Total Team-NB-MD-Survey-2018 111 181 204 195 160 217 Average 9
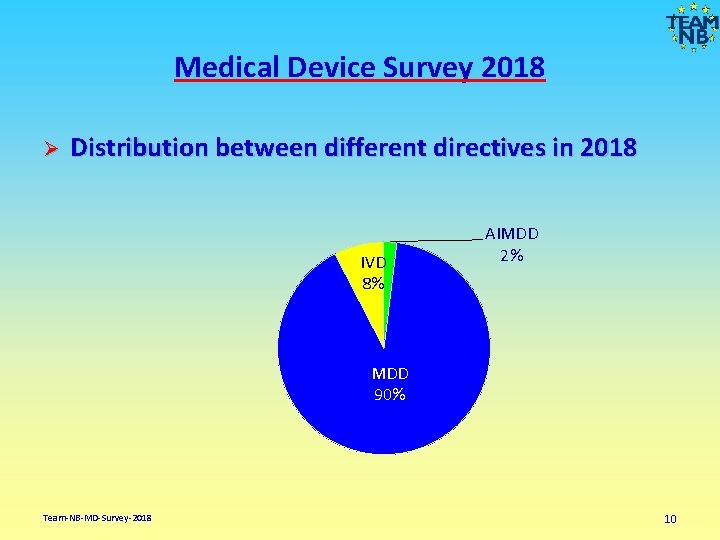
Medical Device Survey 2018 Ø Distribution between different directives in 2018 IVD 8% AIMDD 2% MDD 90% Team-NB-MD-Survey-2018 10
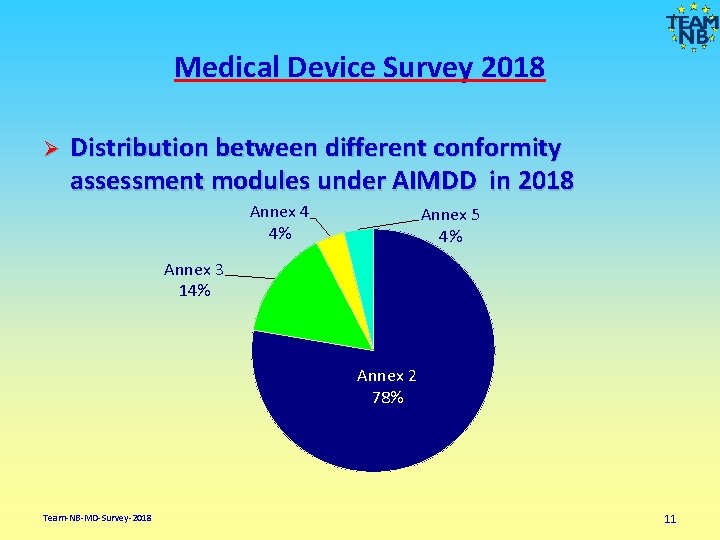
Medical Device Survey 2018 Ø Distribution between different conformity assessment modules under AIMDD in 2018 Annex 4 4% Annex 5 4% Annex 3 14% Annex 2 78% Team-NB-MD-Survey-2018 11
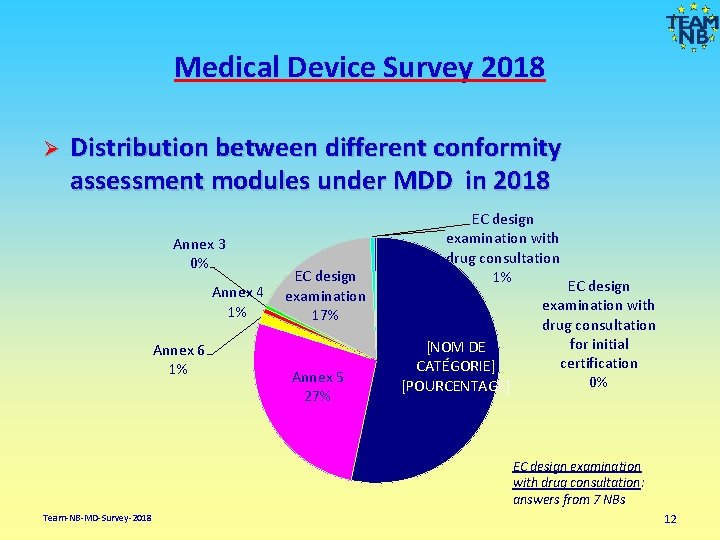
Medical Device Survey 2018 Ø Distribution between different conformity assessment modules under MDD in 2018 Annex 3 0% Annex 4 1% Annex 6 1% EC design examination 17% Annex 5 27% EC design examination with drug consultation 1% [NOM DE CATÉGORIE] [POURCENTAGE] EC design examination with drug consultation for initial certification 0% EC design examination with drug consultation: answers from 7 NBs Team-NB-MD-Survey-2018 12
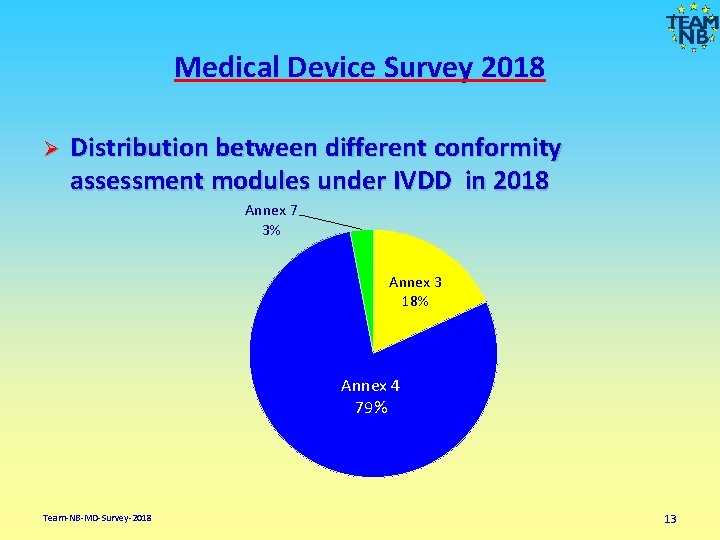
Medical Device Survey 2018 Ø Distribution between different conformity assessment modules under IVDD in 2018 Annex 7 3% Annex 3 18% Annex 4 79% Team-NB-MD-Survey-2018 13
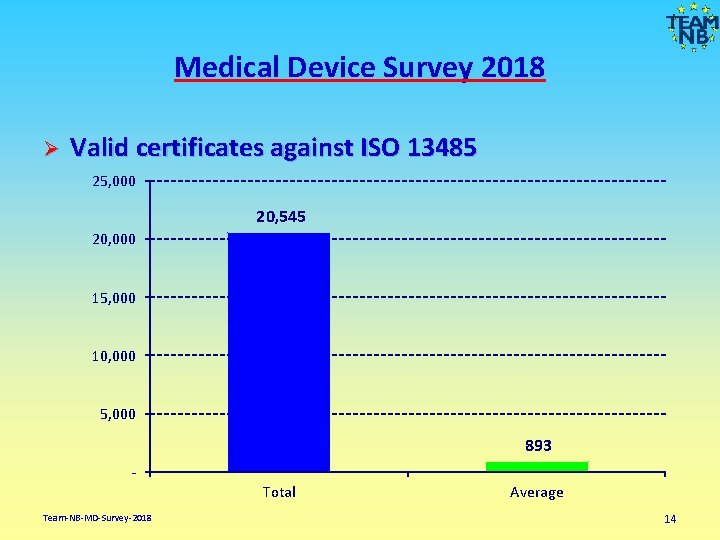
Medical Device Survey 2018 Ø Valid certificates against ISO 13485 25, 000 20, 545 20, 000 15, 000 10, 000 5, 000 893 Total Team-NB-MD-Survey-2018 Average 14
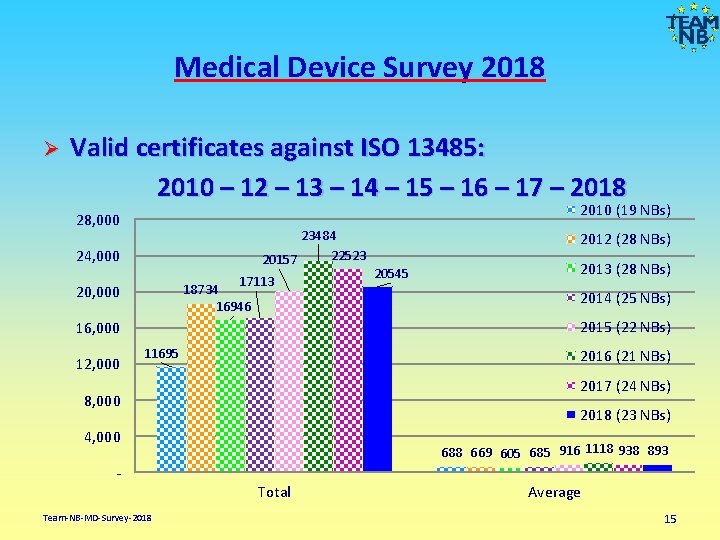
Medical Device Survey 2018 Valid certificates against ISO 13485: 2010 – 12 – 13 – 14 – 15 – 16 – 17 – 2018 Ø 2010 (19 NBs) 28, 000 23484 22523 20157 24, 000 17113 18734 16946 20, 000 20545 2013 (28 NBs) 2014 (25 NBs) 2015 (22 NBs) 16, 000 12, 000 2012 (28 NBs) 11695 2016 (21 NBs) 2017 (24 NBs) 8, 000 2018 (23 NBs) 4, 000 688 669 605 685 916 1118 938 893 Total Team-NB-MD-Survey-2018 Average 15
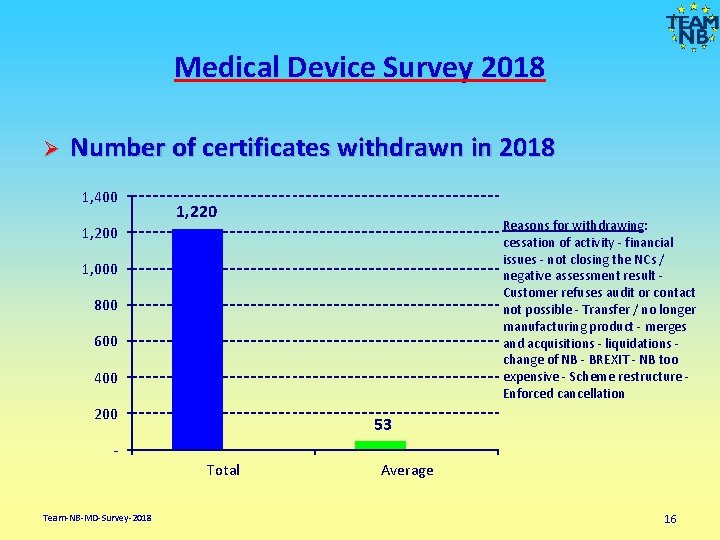
Medical Device Survey 2018 Ø Number of certificates withdrawn in 2018 1, 400 1, 220 Reasons for withdrawing: cessation of activity - financial issues - not closing the NCs / negative assessment result - Customer refuses audit or contact not possible - Transfer / no longer manufacturing product - merges and acquisitions - liquidations - change of NB - BREXIT - NB too expensive - Scheme restructure - Enforced cancellation 1, 200 1, 000 800 600 400 200 53 Total Team-NB-MD-Survey-2018 Average 16
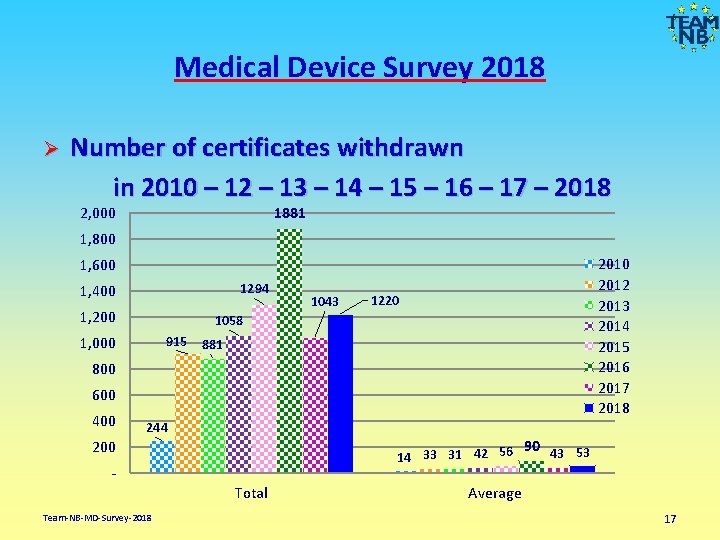
Medical Device Survey 2018 Number of certificates withdrawn in 2010 – 12 – 13 – 14 – 15 – 16 – 17 – 2018 Ø 2, 000 1881 1, 800 2012 2013 2014 2015 2016 2017 2018 1, 600 1294 1, 400 1, 200 1043 1220 1058 915 881 1, 000 800 600 400 244 90 43 53 14 33 31 42 56 200 Total Team-NB-MD-Survey-2018 Average 17
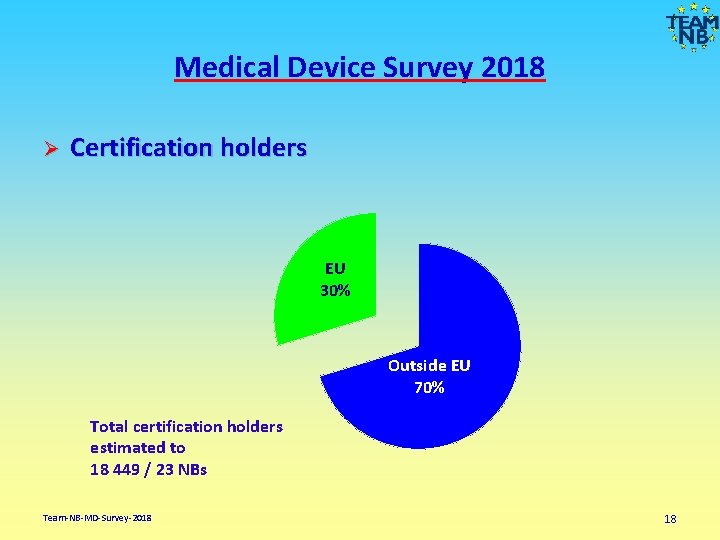
Medical Device Survey 2018 Ø Certification holders EU 30% Outside EU 70% Total certification holders estimated to 18 449 / 23 NBs Team-NB-MD-Survey-2018 18
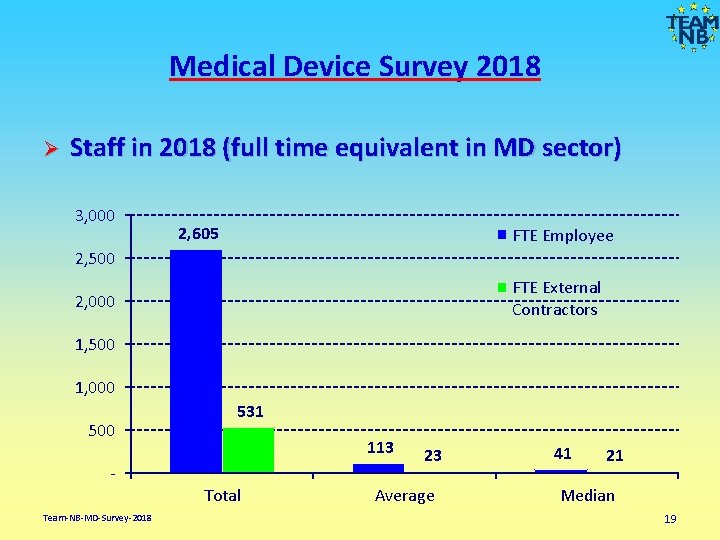
Medical Device Survey 2018 Ø Staff in 2018 (full time equivalent in MD sector) 3, 000 2, 605 FTE Employee 2, 500 FTE External Contractors 2, 000 1, 500 1, 000 500 Team-NB-MD-Survey-2018 531 113 Total 23 Average 41 21 Median 19
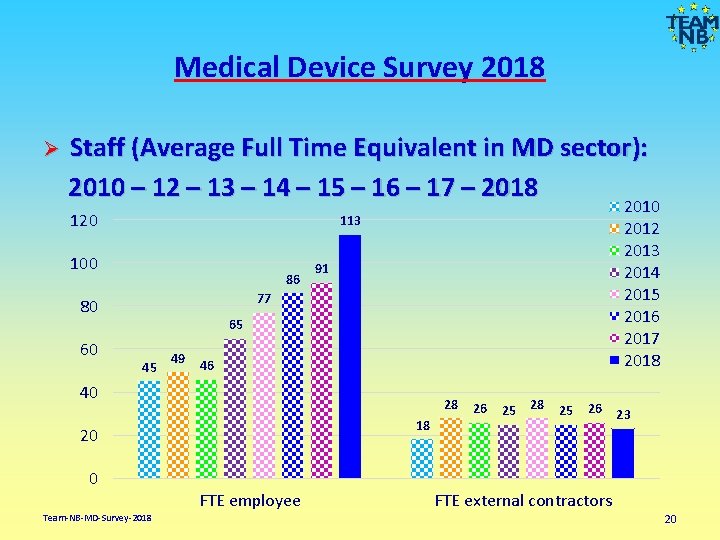
Medical Device Survey 2018 Staff (Average Full Time Equivalent in MD sector): 2010 – 12 – 13 – 14 – 15 – 16 – 17 – 2018 Ø 120 100 86 91 77 80 60 2012 2013 2014 2015 2016 2017 2018 113 65 45 49 46 40 28 18 20 0 Team-NB-MD-Survey-2018 FTE employee 26 25 28 25 26 23 FTE external contractors 20
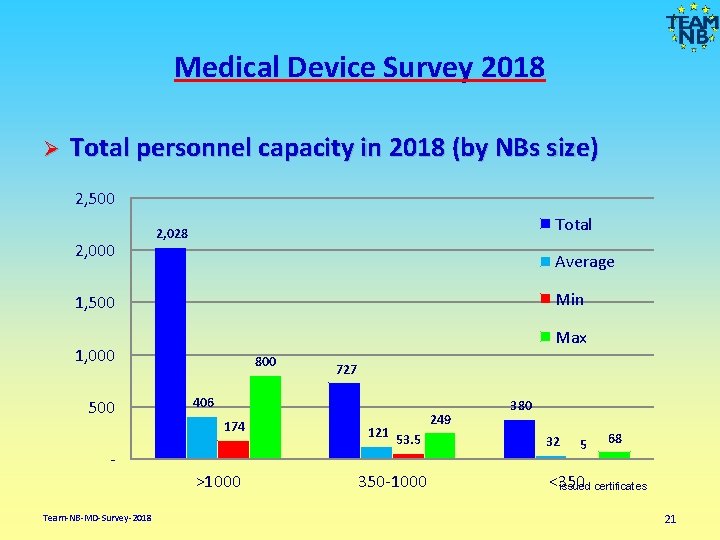
Medical Device Survey 2018 Ø Total personnel capacity in 2018 (by NBs size) 2, 500 2, 000 Total 2, 028 Average Min 1, 500 Max 1, 000 500 800 406 174 Team-NB-MD-Survey-2018 727 >1000 121 249 53. 5 350 -1000 380 32 5 68 <350 issued certificates 21

Members Team-NB-MD-Survey-2018 22
- Slides: 22