TEAM NB The European Association Medical devices Notified

TEAM NB The European Association Medical devices - Notified Bodies 1

TEAM-NB • To improve communications with – – the EC Commission, Industry, Competent Authorities and User Groups • To promote high technical and ethical standards • Members are Notified Bodies under any or all of the 3 medical devices directives: – 90/385/EEC – 93/42/EEC – 98/79/EC 2

List of Team-NB members • • • • BSI (0086 – UK)* • CERMET (0476 – I) • DGM (0543 – DK) • DNV (0434 – N) • DQS (0297 – D) • ECM (0481 – D) • EUROCAT (0535 – D) • ITC (1023 – CZ) • INTERTEK AMTAC CERT (0473 – UK) • INTERTEK ETL SEMKO (0359 – UK) • • INTERTEK SEMKO AB (0413 – S) • KEMA (0344 – NL) • LNE - G-MED (0459 – F) *(Identification number - Country) LRQA (0088 – UK) MDC (0483 – D) MEDCERT (0482 – D) NSAI (0050 – IRL) ORKI (1011 – HUN) SGS (0120 - UK) SNCH (0499 – L) TÜV AUSTRIA (0408 – A) TÜV NORD CERT(0044 – D) TÜV Rheinland (0197 – D) TÜV Süd Product Service (0123 – D) UL International (0843 – UK) VTT AUTOMATION (0537 -FI) 3
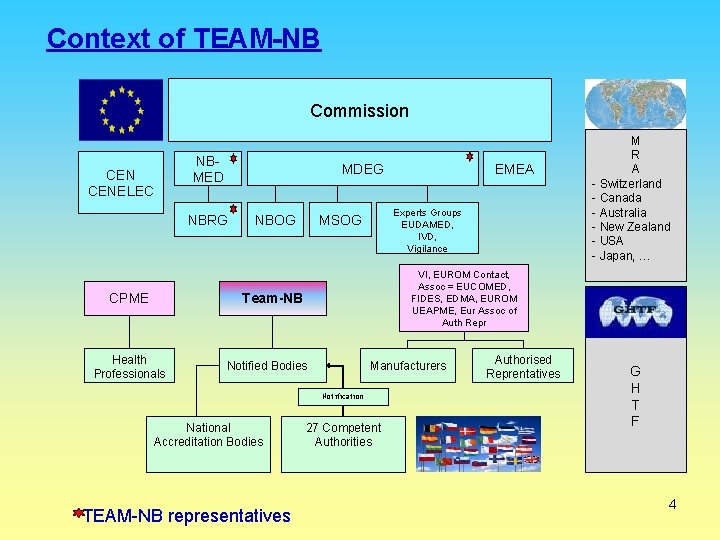
Context of TEAM-NB Commission CENELEC NBMED MDEG NBRG CPME NBOG Experts Groups EUDAMED, IVD, Vigilance MSOG VI, EUROM Contact, Assoc = EUCOMED, FIDES, EDMA, EUROM UEAPME, Eur Assoc of Auth Repr Team-NB Health Professionals Notified Bodies Manufacturers Notification National Accreditation Bodies TEAM-NB representatives EMEA 27 Competent Authorities Authorised Reprentatives M R A - Switzerland - Canada - Australia - New Zealand - USA - Japan, … G G H T F 4

Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance NB-MED (Notified Bodies Medical Devices) Aim : Recommendations to Notified Bodies active in the medical devices field Documents provided: Guidelines, Recommendations and preparation of MEDDEV’s Stakeholders : - Experts - NB’s - Members states representatives - Industry representatives - Commission representatives 5

Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance NB-RG (Notified Bodies Recommendation Group) Aim : Preparing in smaller expert group Recommendations paper Documents provided: Guidelines, Recommendations and preparation of MEDDEV’s Stakeholders : Experts 6
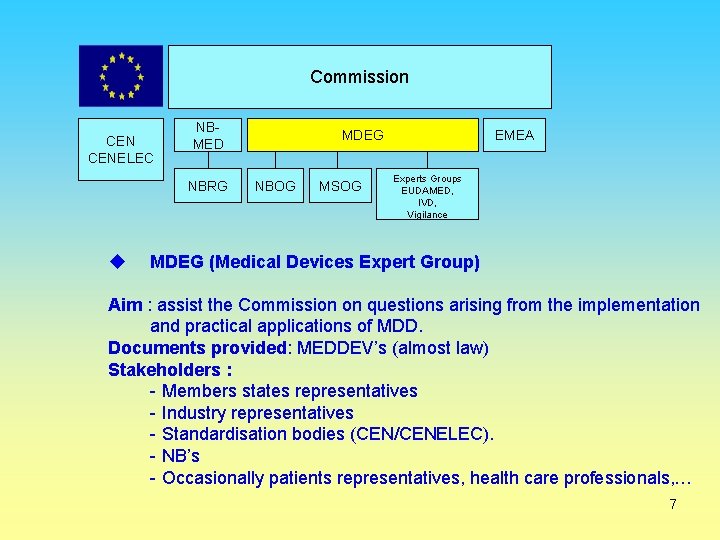
Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance MDEG (Medical Devices Expert Group) Aim : assist the Commission on questions arising from the implementation and practical applications of MDD. Documents provided: MEDDEV’s (almost law) Stakeholders : - Members states representatives - Industry representatives - Standardisation bodies (CEN/CENELEC). - NB’s - Occasionally patients representatives, health care professionals, … 7

Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance NBOG (Notified Bodies Operational group) Aim : to improve the overall performance of the Notified Bodies in the MD sector by identifying and promulgating examples of best practices to be adopted by both Notified Bodies and those organisations responsible for their designation and control. Documents provided: Guidelines Stakeholders : - Members states representatives - NB-Med representative accepted as observer for some subjects 8
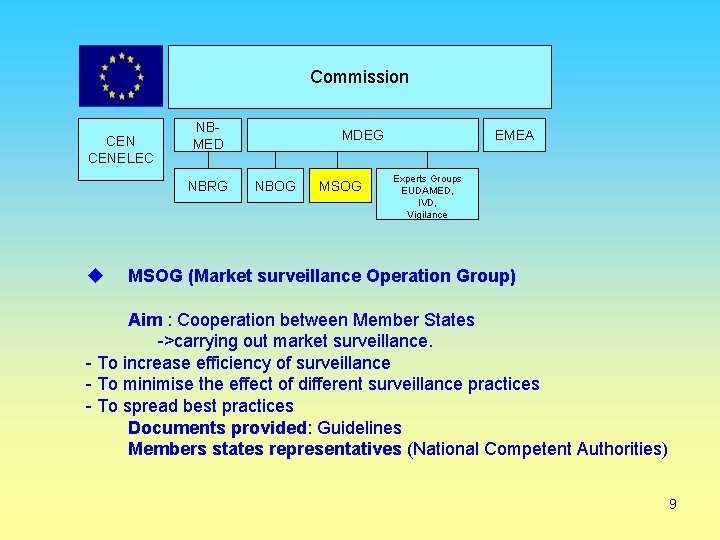
Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance MSOG (Market surveillance Operation Group) Aim : Cooperation between Member States ->carrying out market surveillance. - To increase efficiency of surveillance - To minimise the effect of different surveillance practices - To spread best practices Documents provided: Guidelines Members states representatives (National Competent Authorities) 9
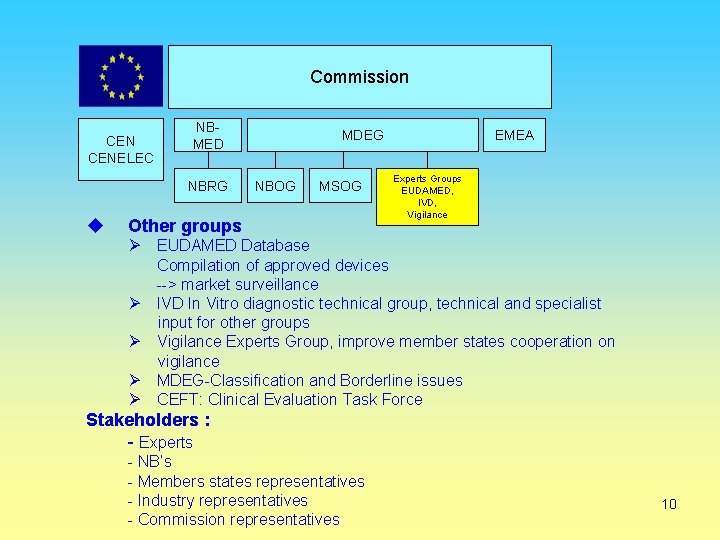
Commission CENELEC NBMED NBRG MDEG NBOG MSOG Other groups EMEA Experts Groups EUDAMED, IVD, Vigilance Ø EUDAMED Database Compilation of approved devices --> market surveillance Ø IVD In Vitro diagnostic technical group, technical and specialist input for other groups Ø Vigilance Experts Group, improve member states cooperation on vigilance Ø MDEG-Classification and Borderline issues Ø CEFT: Clinical Evaluation Task Force Stakeholders : - Experts - NB’s - Members states representatives - Industry representatives - Commission representatives 10
Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance CENELEC : CEN : “Centre Européen de Normalisation” - Integrated system for European standardization to deliver formal standards. CENELEC : « Centre Européen de Normalisation Electrique » 11

Commission CENELEC NBMED NBRG MDEG NBOG MSOG EMEA Experts Groups EUDAMED, IVD, Vigilance EMEA (European Medicines Agency) Decentralised body in London Protection and promotion of public and animal health - evaluation and supervision of medicines Responsible for scientific evaluation -> approval Pharmacovigilance monitoring 12

TEAM-NB’s website TEAM-NB stated for The European Association Medical devices - Notified Bodies. We are a non-profit association. Our members are Notified Bodies (see list) under any or all of the three medical device new approach directives: 90/385/EEC; 93/42/EEC; 98/79/EC. Our aims are : " To improve communications with the EC Commission, Industry, Competent Authorities and User Groups by acting as a focal point and the single voice of Notified Bodies". " To promote high technical and ethical standards in the functioning of Notified Bodies " " To protect the legal and commercial interests of Notified Bodies in their vital role in the functioning of the three medical device directives ". 13

Future developments - Best Practice Guide Relation with national association Development of closer contact with European Commission Development of closer contact with European Parliament representatives Development of closer contact with industry associations Web site with availability of MEDDEV & NB-Med Recommendations 14

Advantages • to protect the legal and commercial interests of Notified Bodies in their vital role in the functioning of the three medical device directives. • to join an association which is seen as the authentic and representative voice of Notified Bodies in the field of medical devices 15

Contacts Management: • President : Chris Jepson at Chris_Jepson@sgs. com • Treasurer : Aud Loken Eiklid at aud. loken. eiklid@dnv. com • Vice-president : Werner Kexel at Kexel@eurocat. de Secretary: Gero Viola at gero. Viola@de. tuv. com Director and Secretariat: • Françoise Schlemmer at Schlemmer@quasys. com 16
- Slides: 16