Soil Chemistry Soil Chemistry Mineral salts From weathered




























- Slides: 30

Soil Chemistry

Soil Chemistry • Mineral salts – From weathered rocks – Break down of Organic Matter (OM) – Addition of fertilizer

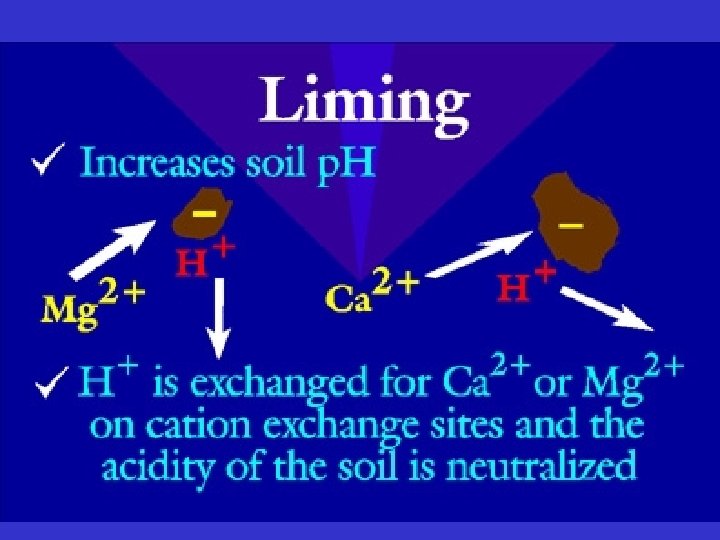
Soil Chemistry • Salt dissolves in soil solution (H 2 O) as ionic forms • Ion: +/- charged particle • Ex: table salt = Na. Cl = Na+ and Cl. Cations H+ Ca 2+ Mg 2+ NH 4+ K+ Anions OHCl. CO 32 SO 42 -

Soil Chemistry • Colloids: particles of clay and O. M. suspended in solution – These attract and exchange cations! • Cation Exchange Capacity (CEC) = the soil fertility • CEC: O. M. > Clay > Silt > Sand

Soil Chemistry • Plants absorb nutrients as ions! • Sandy soils have a low CEC and therefore cannot hold on to ions well • These soils require frequent, light applications of fertilizer

Soil Chemistry • Soil p. H – Relationship of H+ ions to OH- ions Acid Neutral Basic H+ > OHH+ = OHH+ < OH- (also called, “alkaline”)

Soil Chemistry H+ and Al 3+ produce acidic soils Ca 2+ and Mg 2+ produce basic soils High rainfall areas are acidic soils Low rainfall areas are generally basic or neutral soils

Soil Chemistry Salt build-up in low-rainfall location damages crops

• Fungi most active if p. H<5. 5 • Bacteria most active if p. H>5. 5 Properly nodulated legumes add 55 to 300 pounds of nitrogen per acre to soil.



p. H of various liquids household ammonia 11. 9 sea water 8. 5 human blood 7. 4 milk 6. 4 orange juice 3. 5 lemon juice 2. 3 vinegar 2. 8


Plants which like acidic conditions • Ericaceous plants (blueberry, cranberry, azalea, rhododendron)

Plants which like acidic conditions • Ericaceous plants (blueberry, cranberry, azalea, rhododendron)

Plants which like acidic conditions • Ericaceous plants (blueberry, cranberry, azalea, rhododendron)

Plants which like acidic conditions • Ericaceous plants (blueberry, cranberry, azalea, rhododendron)

Plants which like acidic conditions • Ericaceous plants (blueberry, cranberry, azalea, rhododendron)

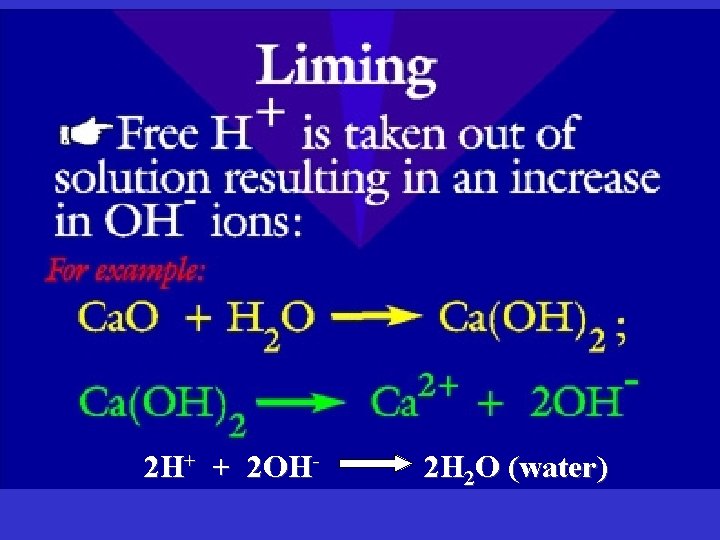
Soil Chemistry • If p. H very low or very high, soil nutrients become unavailable to the plant • Liming - raises the p. H (makes the soil more basic) and is used on acidic soils. – Ca. CO 3 – Mg. CO 3 • Most of Vermont’s soils are acidic • Most tropical soils are acidic due to high rainfall

Liming DON’TS • You should NOT lime your lawn after having applied a fertilizer that contains ammonium nitrate. The ammonium is converted to ammonia gas which can cause burning on the foliage of the grass and surrounding trees and shrubs!

Cliff of limestone


2 H+ + 2 OH- 2 H 2 O (water)

Soil Chemistry • Acidifying the soil or lowering the p. H – Elemental sulfur (effect is temporary) – Ammonium sulfate (effect is temporary) – Aluminum sulfate is also used but not recommended as aluminum is a heavy metal!

Soil Chemistry • Basic soils often cause iron chlorosis in plants • This can be corrected by acidifying the soil or choosing a plant which is tolerant of alkaline soils! • Intervienal chlorosis: often on acid-loving plants like roses, fruit trees, azaleas and rhododendrons




Blue in acidic soils <5. 5 with available aluminum Pink in neutral soils near p. H of 7
