5 2 SALTS 1 What are salts Salts

















- Slides: 30

5. 2 SALTS 1

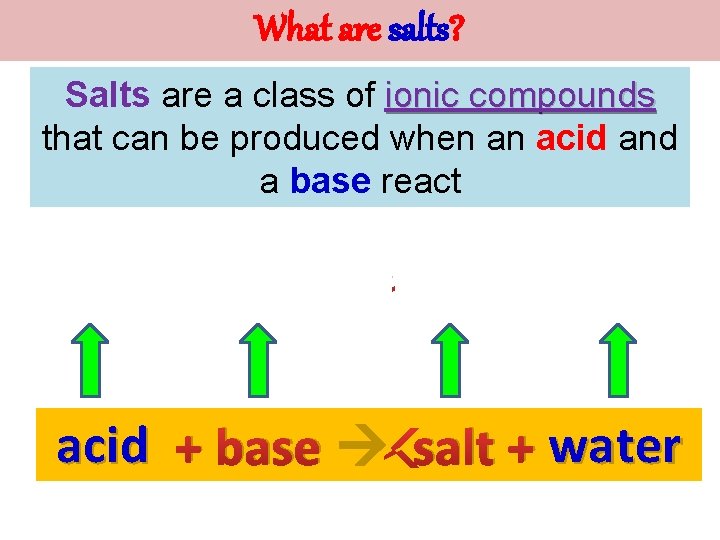
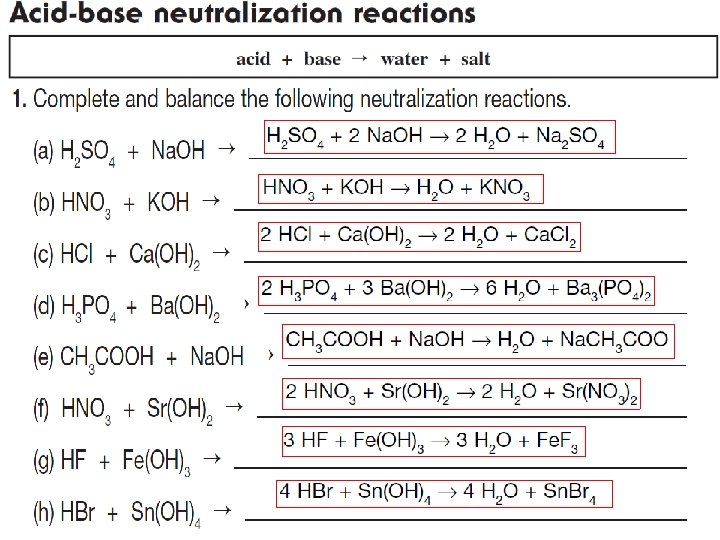
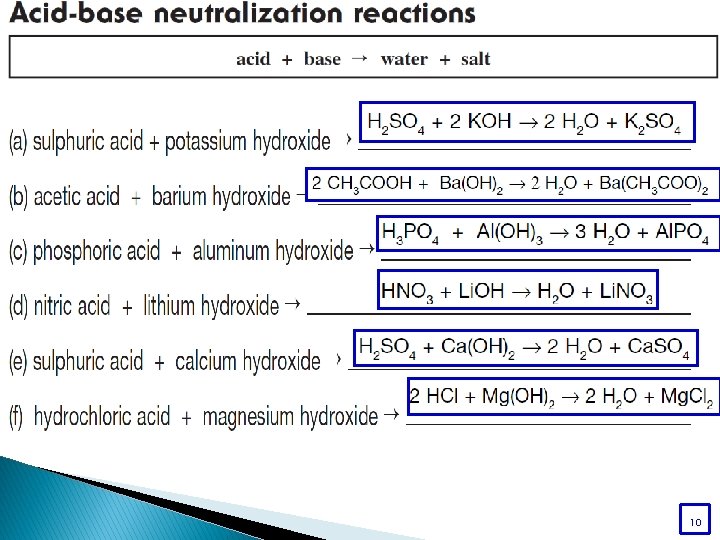
What are salts? Salts are a class of ionic compounds that can be produced when an acid and a base react H A + BOH OH BA + H 2 O acid + base salt + water

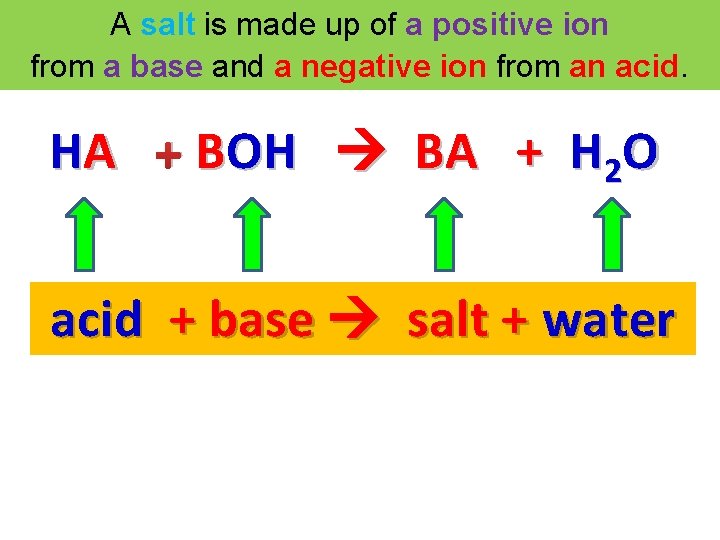
A salt is made up of a positive ion from a base and a negative ion from an acid. HA + BOH BA + H 2 O acid + base salt + water HCl + KOH KCl + H 2 O

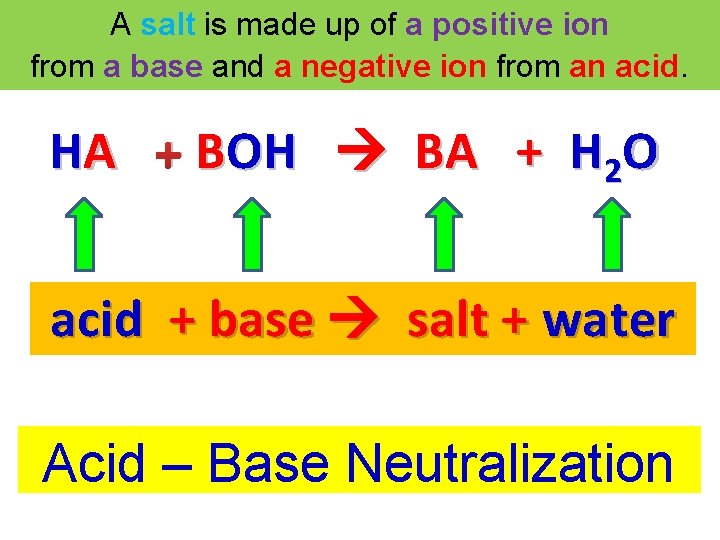
A salt is made up of a positive ion from a base and a negative ion from an acid. HA + BOH BA + H 2 O acid + base salt + water Acid – Base Neutralization

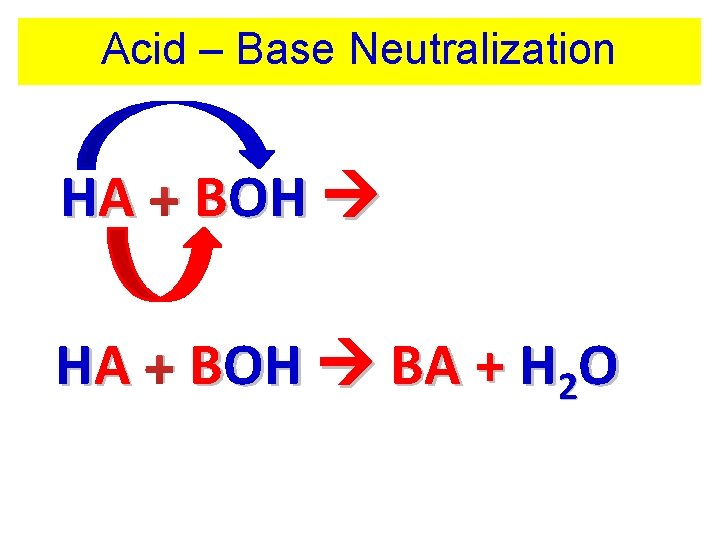
Acid – Base Neutralization HA + BOH BA + HOH HA + BOH BA + H 2 O

Acid – Base Neutralization Read page 234 – 236 and do:

Acid – Base Neutralization WORKBOO K page 91

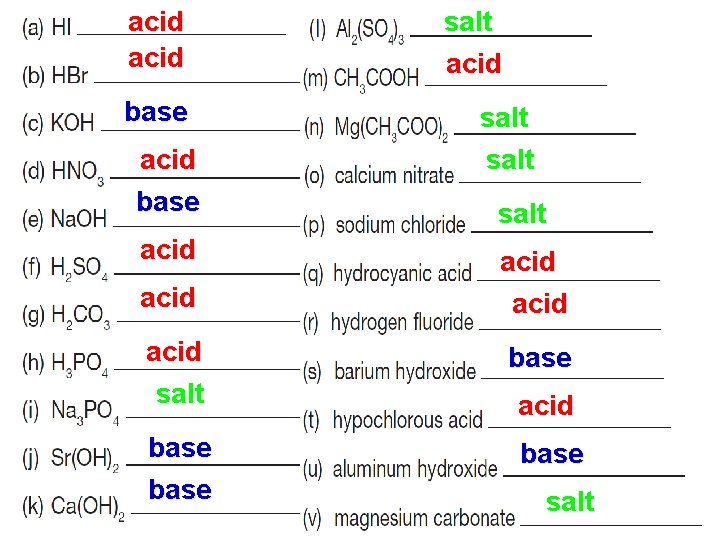
acid base acid salt base salt acid salt acid base salt


10

Metal Oxides • Metals react with oxygen to form metal oxides Metal Oxides Na 2 O K 2 O Ca. O Mg. O

Non - Metal Oxides Non - Metals react with oxygen to form non metal oxides Non - Metal Oxides CO 2 SO 2 NO CO

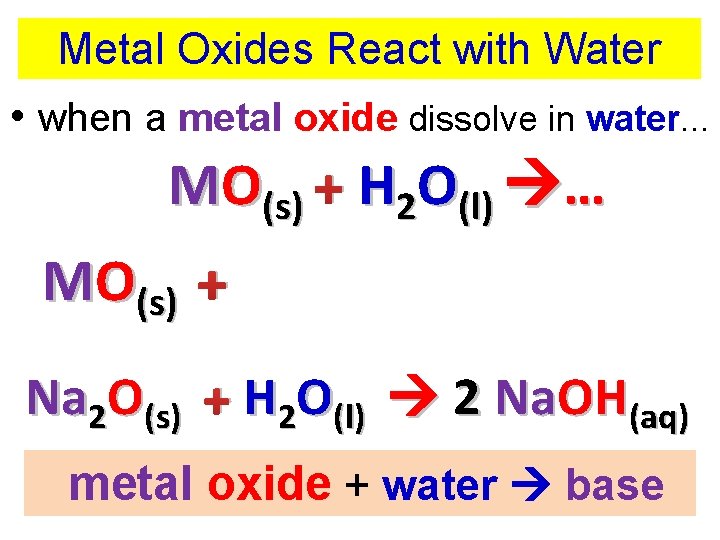
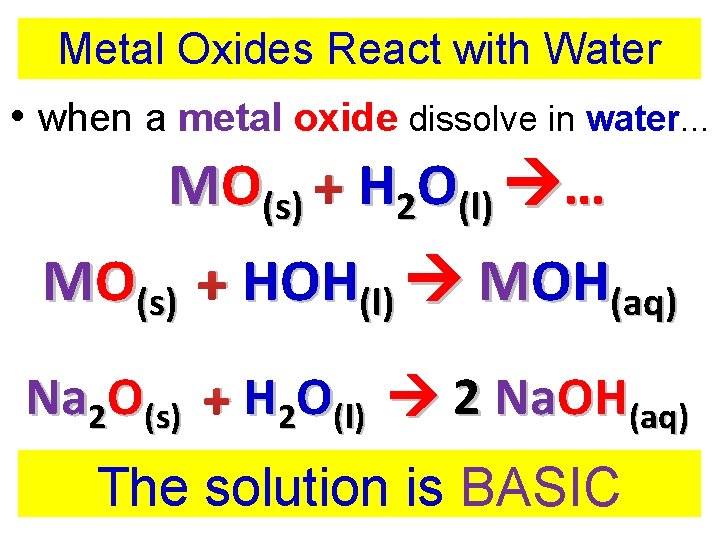
Metal Oxides React with Water • when a metal oxide dissolve in water. . . MO(s) + H 2 O(l) … MO(s) + HOH(l) MOH(aq) Na 2 O(s) + H 2 O(l) 2 Na. OH(aq) metal oxide + water base

Metal Oxides React with Water • when a metal oxide dissolve in water. . . MO(s) + H 2 O(l) … MO(s) + HOH(l) MOH(aq) Na 2 O(s) + H 2 O(l) 2 Na. OH(aq) The solution is BASIC

Non - Metal Oxides React with Water • when a non - metal oxide dissolve in water. . . NO(s) + H 2 O(l) … non - metal oxide + water acid SO 2(s) + H 2 O(l) 2 H 2 SO 3(aq) The solution is ACIDIC

Metal Oxides and Non – Metal Oxides Read page 237 and do:

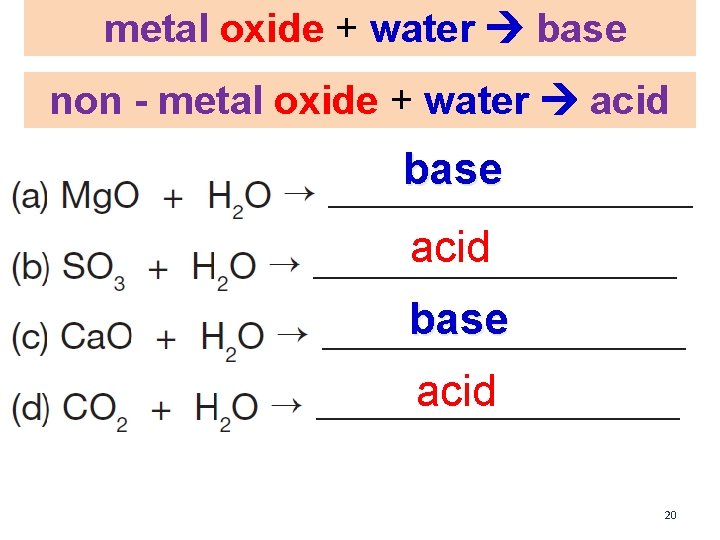
Metal Oxides and Non – Metal Oxides WORKBOO K page 93 metal oxide + water base non - metal oxide + water acid

metal oxide + water base non - metal oxide + water acid 18

metal oxide + water base non - metal oxide + water acid

metal oxide + water base non - metal oxide + water acid base acid 20

Metals and Acids • Metals of Groups 1 and 2 (Alkali Metals and Alkaline Earth Metals)

Metals and Acids • Metals of Groups 1 and 2 (Alkali Metals and Alkaline Earth Metals) react vigorously with water and acids • Other metals are not that reactive with acids

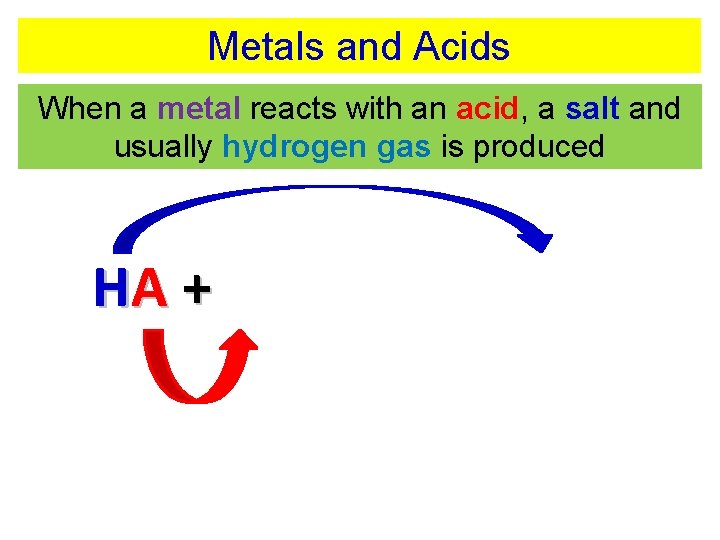
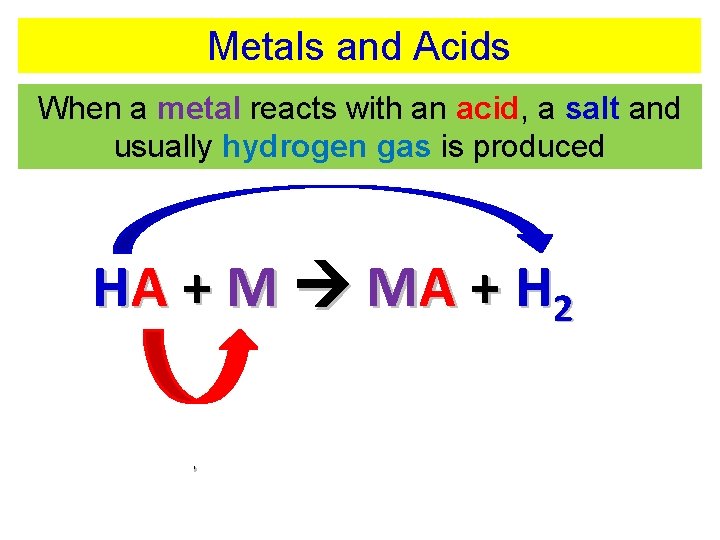
Metals and Acids When a metal reacts with an acid, a salt and usually hydrogen gas is produced HA + M MA + H 2 acid + metal salt + hydrogen

Metals and Acids When a metal reacts with an acid, a salt and usually hydrogen gas is produced HA + M MA + H 2 HCl(aq) + Mg(s) Mg. Cl 2(aq) + H 2(g)

Metals and Acids Read page 238 and do:

Carbonates and Acids CO 3 2 - Metal Carbonates Ca. CO 3 Mg. CO 3 Na 2 CO 3 K 2 CO 3

Carbonates and Acids Carbonate rocks react with acids to produce. . . CO 3 2 - metal carbonate + acid salt + water + carbon dioxide

Carbonates and Acids Carbonate rocks react with acids to produce. . . CO 3 2 - Ca. CO 3 + H 2 SO 4 Ca. SO 4 + H 2 O + CO 2

5. 2 Check Your Understanding TEXTBOOK page 93

5. 2 SALTS QUIZ • Date: Wednesday, Dec 18