Acids Bases Equilibria Part IV Analysis of Salts

Acids – Bases Equilibria Part IV: Analysis of Salts Jespersen Chap. 17 Sec 6 Dr. C. Yau Fall 2014 1
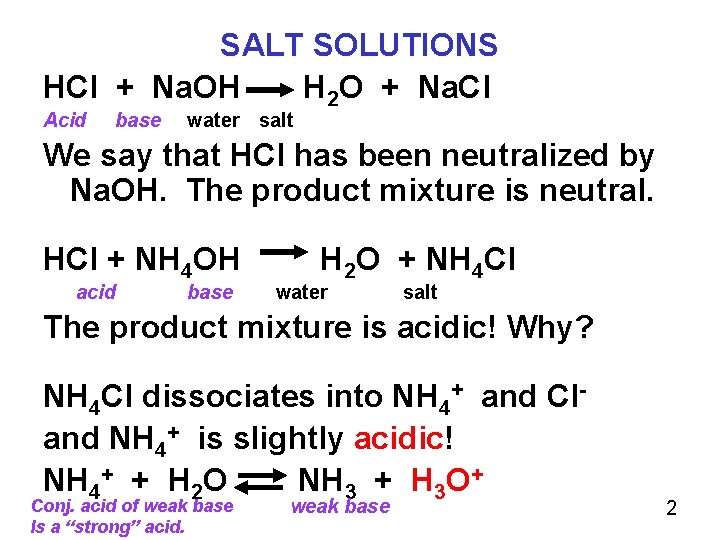
SALT SOLUTIONS HCl + Na. OH H 2 O + Na. Cl Acid base water salt We say that HCl has been neutralized by Na. OH. The product mixture is neutral. HCl + NH 4 OH acid base H 2 O + NH 4 Cl water salt The product mixture is acidic! Why? NH 4 Cl dissociates into NH 4+ and Cland NH 4+ is slightly acidic! NH 4+ + H 2 O NH 3 + H 3 O+ Conj. acid of weak base Is a “strong” acid. weak base 2

In the same way, HC 2 H 3 O 2 + Na. OH H 2 O + Na. C 2 H 3 O 2 The product mixture is not neutral. Na. C 2 H 3 O 2 dissociates into Na+ and C 2 H 3 O 2 - and acetate is the conjugate base of a weak acid (acetic acid). C 2 H 3 O 2 - is a relatively strong base. It will pull a proton off water: C 2 H 3 O 2 - + HOH HC 2 H 3 O 2 + OHproducing OH - ions. THUS SODIUM ACETATE PRODUCES A SLIGHTLY BASIC SOLUTION! 3

Predicting Acid–Base Properties Of A Salt Cation may be acidic. Anion may be basic. 1. If neither the cation nor the anion can affect the p. H, the solution should be neutral. 2. If only the cation of the salt is acidic, the solution will be acidic. 3. If only the anion of the salt is basic, the solution will be basic. 4. If a salt has a cation that is acidic and an anion that is basic, the p. H of the solution is determined by the relative strengths of the acid and base. – Compare Ka and Kb for the ions 4

Predicting Effects Of Ions On p. H • Most metal cations are neutral or extremely weak acids (e. g. Na+, Ca 2+) • Conjugate acids of weak bases are acidic (e. g. NH 3, N 2 H 4 NH 4+, N 2 H 5+) • Conjugate bases of weak acids are basic ) (e. g. HC 2 H 3 O 2, HNO 2 C 2 H 3 O 2¯, NO 2¯ • Conjugate bases of the strong acids are neutral (except HSO 4 - which is still fairly acidic) (e. g. HCl, HNO 3 Cl¯, NO 3¯ ) ¯ acidic! but not H 2 SO 4 Still HSO 4 5

• • 0. 1 M solutions of the following are acidic basic/ neutral or amphoteric? … Na. Cl • neutral • basic Na. CN • acidic NH 4 Cl • acidic HCN Na. Cl. O 4 • neutral • acidic HCl Na 3 PO 4 • basic Do p. 795: Pract Exer 25 & 26. 6

At this point you are expected to recognize whether a salt will produce an acidic or basic solution from looking at its formula. That is a qualitative analysis. We now examine how to analyze the p. H of salt solutions quantitatively. 1. Decide whether cation or anion will affect p. H. 2. Write eqn for the Ka or Kb of ion. 3. Calc Ka or Kb from Kb or Ka of its conjugate acid/base 4. Set up ICE table. Calc p. H. 7

Example 17. 5 p. 796 What is the p. H of a 0. 10 M solution of Na. OCl? What do we look up? Need Kb of OCl-. So find Ka of HOCl = 3. 0 x 10 -8 Then what? Set up ICE table for what? Example What is the p. H of a 0. 20 M solution of hydrazinium chloride N 2 H 5 Cl? What do we look up? Hydrazine N 2 H 4 is a weak base with Kb = 9. 6 x 10 -7 Do Pract Exer 27, 28 & 29 p. 797 8

Example Will an aqueous solution that is 0. 20 M NH 4 F be acidic, basic or neutral? Kb of NH 3 = 1. 8 x 10 -5 Ka of HF = 3. 5 x 10 -4. . . and so? Do Pract Exer 30 & 31 p. 798 9

Summary of what we did in the previous examples Example 17. 5: Calculation for p. H of salt made from strong base and weak acid (Na. OCl). Example with N 2 H 5 Cl. : Calculation for p. H of salt made from weak base and strong acid Example NH 4 F: Analysis of salt made from weak base and weak acid. 10
- Slides: 10