Double salts and coordination compounds Ferric alum NH

Double salts and coordination compounds Ferric alum (NH 4)2 SO 4. Fe 2(SO 4)3. 24 H 2 O - Double salt Ø It is an addition compound of two stable compounds ammonium sulphate and ferric sulphate. Ø When crystals of ferric alum are dissolved in water, the solution shows the properties of NH 42+, SO 42 - and Fe 3+ ions. Ø On dissolution , it breaks down to its component simple ions. Ø They exist only in the solid state. 1
![Coordination compound ØPotassium ferrocyanide K 4[Fe(CN)6] does not break down to simple ions Ø Coordination compound ØPotassium ferrocyanide K 4[Fe(CN)6] does not break down to simple ions Ø](http://slidetodoc.com/presentation_image_h/1cba22de1cea21bba50e9ec182e9712b/image-2.jpg)
Coordination compound ØPotassium ferrocyanide K 4[Fe(CN)6] does not break down to simple ions Ø When dissolved in water, its solution consists of K+ and [Fe(CN)6]4 - (latter called as complex ion) Coordination number Ø The total number of monodendate ligands attached to the central metal in a complex Eg. [Pd. F 6]2 - (central metal ion – Pd 4+) [Ag(NH 3)] + (central metal ion – Ag+) 2

Oxidation number ØIt denotes the charge, the central metal atom would have if all the ligands in the complex were removed along with their electron pairs that were shared with the central atom. Ø It is represented by a Roman numeral. [Fe(CO)5] [Oxidation number is O)] [Co(NH 3)6]3+ [Oxidation number is +3 --- (III)] [Zn(CN)4]2 - [Oxidation number is +2 --- (II)] 3
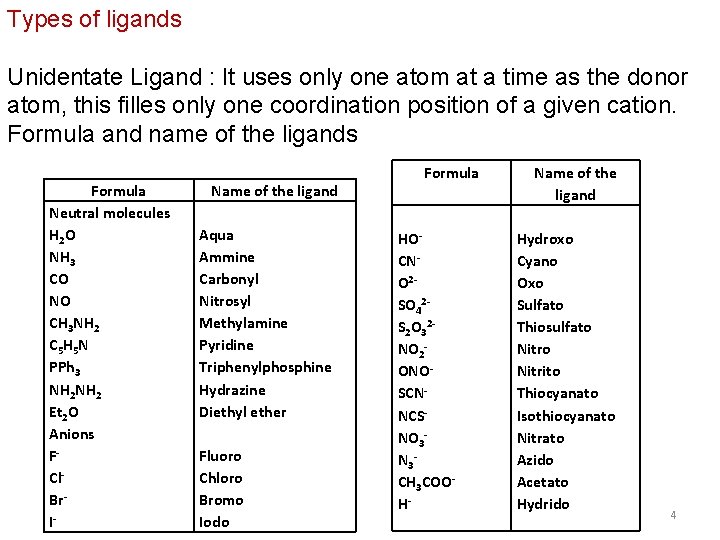
Types of ligands Unidentate Ligand : It uses only one atom at a time as the donor atom, this filles only one coordination position of a given cation. Formula and name of the ligands Formula Neutral molecules H 2 O NH 3 CO NO CH 3 NH 2 C 5 H 5 N PPh 3 NH 2 Et 2 O Anions FCl. Br. I- Name of the ligand Aqua Ammine Carbonyl Nitrosyl Methylamine Pyridine Triphenylphosphine Hydrazine Diethyl ether Fluoro Chloro Bromo Iodo Formula HOCNO 2 SO 42 S 2 O 32 NO 2 ONOSCNNCSNO 3 N 3 CH 3 COOH- Name of the ligand Hydroxo Cyano Oxo Sulfato Thiosulfato Nitrito Thiocyanato Isothiocyanato Nitrato Azido Acetato Hydrido 4

Polydentate Ligands: It uses more than one atom per ligand as a donor atom, it fills more than one coordination position of a given cation simultaneously. Bidentate Ligand : It can form two coordinate bonds with a cation Eg: Oxalate ligand Tridentate ligand: It can form three coordinate bonds with a cation Eg: diethylenetriamine ligand Quadritendate ligand: It can form four coordinate bonds with a cation Eg: triethylenetetraamine ligand 5
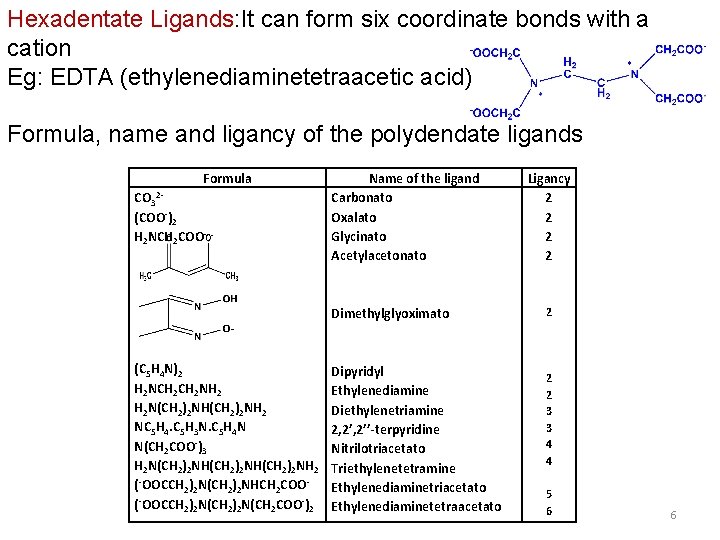
Hexadentate Ligands: It can form six coordinate bonds with a cation Eg: EDTA (ethylenediaminetetraacetic acid) Formula, name and ligancy of the polydendate ligands Formula CO 32(COO-)2 H 2 NCH 2 COO Name of the ligand Carbonato Oxalato Glycinato Acetylacetonato Dimethylglyoximato (C 5 H 4 N)2 H 2 NCH 2 NH 2 H 2 N(CH 2)2 NH 2 NC 5 H 4. C 5 H 3 N. C 5 H 4 N N(CH 2 COO-)3 H 2 N(CH 2)2 NH(CH 2)2 NH 2 (-OOCCH 2)2 N(CH 2)2 NHCH 2 COO(-OOCCH 2)2 N(CH 2 COO-)2 Dipyridyl Ethylenediamine Diethylenetriamine 2, 2’’-terpyridine Nitrilotriacetato Triethylenetetramine Ethylenediaminetriacetato Ethylenediaminetetraacetato Ligancy 2 2 2 2 3 3 4 4 5 6 6

Bridging Ligands: it simultaneously binds two metals and forms a bridge between the two metals, acting as a ligand to both the metals Ambidentate ligand: It is capable of bonding through one type of donor atom in one complex but through a different donor atom in another complex. It has potentially two ligating atoms. Eg. NO 2 - can bond to a metal either through its O or N atom producing two isomeric complexes [(NH 3)5 Co-ONO]Cl 2 - Red complex [(NH 3)5 Co-NO 2]Cl 2 - Yellow complex 7
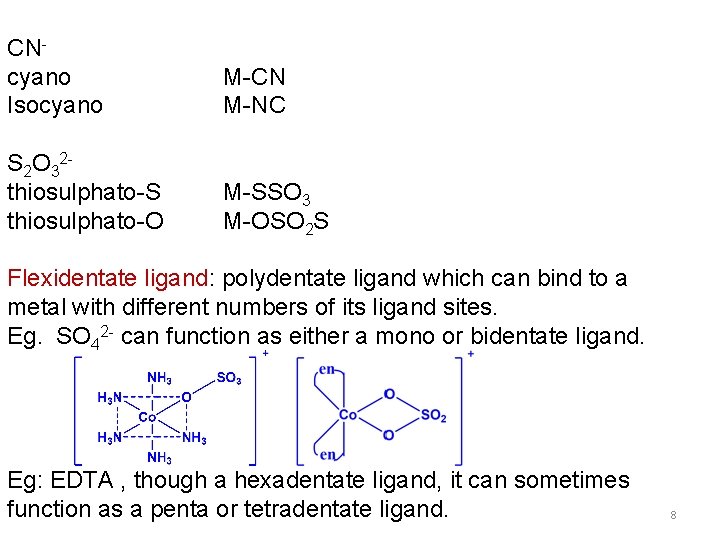
CNcyano Isocyano M-CN M-NC S 2 O 32 thiosulphato-S thiosulphato-O M-SSO 3 M-OSO 2 S Flexidentate ligand: polydentate ligand which can bind to a metal with different numbers of its ligand sites. Eg. SO 42 - can function as either a mono or bidentate ligand. Eg: EDTA , though a hexadentate ligand, it can sometimes function as a penta or tetradentate ligand. 8

WERNER’S THEORY (In 1913, Nobel prize in chemistry-first inorganic chemist) Postulates: 1. Elements exhibit two types of valencies, namely, primary (ionisable) and secondary (nonionisable) valence 2. Anions can satisfy primary valence and anions or neutral molecules can satisfy secondary valence. 3. Primary valence corresponds to the oxidation number and the secondary valence corresponds to the coordination number. 4. All elements tend to satisfy both primary and secondary valences. But the fulfillment of secondary valence is more essential. 5. Secondary valences are directed towards some fixed positions in space. Eg: 4 -coordinated complexes are arranged in a tetrahedral and 6 -coordinated complexes are arranged in a octahedral manner. 9

WERNER’S COBALT COMPLEXES Complex Co. Cl 3. 6 NH 3 (A) Co. Cl 3. 5 NH 3 (B) Co. Cl 3. 4 NH 3 (C) Co. Cl 3. 3 NH 3 (D) No. of Cl- ions precipitated as Ag. Cl Formula 3 2 1 0 [Co(NH 3)6]Cl 3 [Co(NH 3)5 Cl]Cl 2 [Co(NH 3)4 Cl 2]Cl [Co(NH 3)3 Cl 3] No. of ions in solution 4 3 2 0 Molar conductivity (ohm-1) app. 404 app. 229 app. 97 0 Complex A [Co(NH 3)6]Cl 3 • Primary valences are satisfied by three chloride ions (dotted lines) • Six secondary valences (solid lines) are satisfied by six ammonia molecules. • ammonia molecules are tightly bound to cobalt and they do not dissociate in solution 10
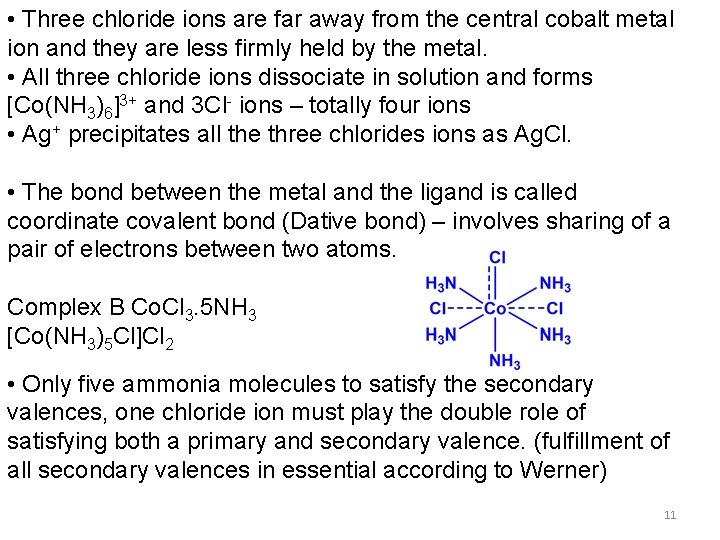
• Three chloride ions are far away from the central cobalt metal ion and they are less firmly held by the metal. • All three chloride ions dissociate in solution and forms [Co(NH 3)6]3+ and 3 Cl- ions – totally four ions • Ag+ precipitates all the three chlorides ions as Ag. Cl. • The bond between the metal and the ligand is called coordinate covalent bond (Dative bond) – involves sharing of a pair of electrons between two atoms. Complex B Co. Cl 3. 5 NH 3 [Co(NH 3)5 Cl]Cl 2 • Only five ammonia molecules to satisfy the secondary valences, one chloride ion must play the double role of satisfying both a primary and secondary valence. (fulfillment of all secondary valences in essential according to Werner) 11
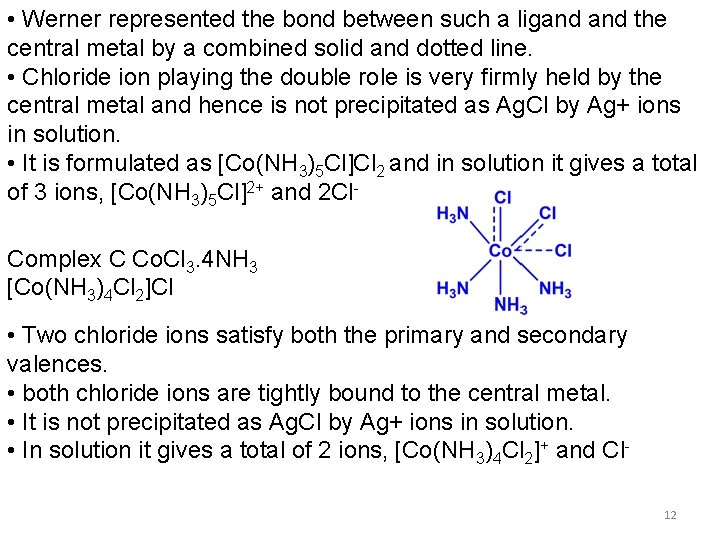
• Werner represented the bond between such a ligand the central metal by a combined solid and dotted line. • Chloride ion playing the double role is very firmly held by the central metal and hence is not precipitated as Ag. Cl by Ag+ ions in solution. • It is formulated as [Co(NH 3)5 Cl]Cl 2 and in solution it gives a total of 3 ions, [Co(NH 3)5 Cl]2+ and 2 Cl. Complex C Co. Cl 3. 4 NH 3 [Co(NH 3)4 Cl 2]Cl • Two chloride ions satisfy both the primary and secondary valences. • both chloride ions are tightly bound to the central metal. • It is not precipitated as Ag. Cl by Ag+ ions in solution. • In solution it gives a total of 2 ions, [Co(NH 3)4 Cl 2]+ and Cl 12
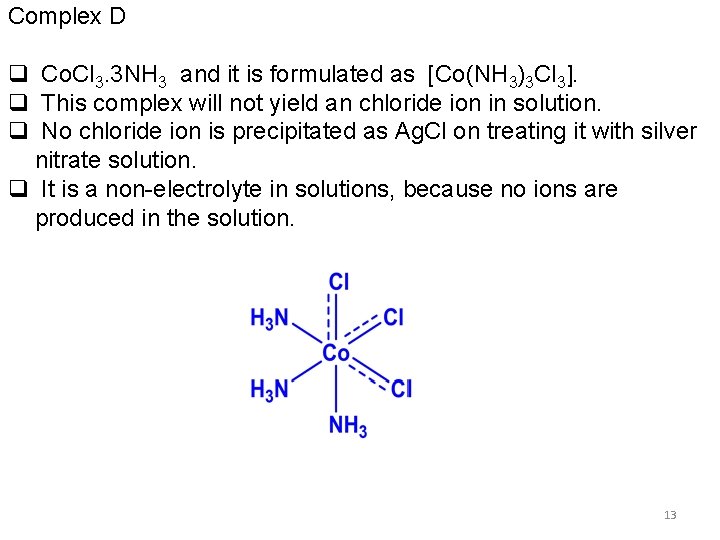
Complex D q Co. Cl 3. 3 NH 3 and it is formulated as [Co(NH 3)3 Cl 3]. q This complex will not yield an chloride ion in solution. q No chloride ion is precipitated as Ag. Cl on treating it with silver nitrate solution. q It is a non-electrolyte in solutions, because no ions are produced in the solution. 13

DEFECTS OF WERNER’S THEORY 1. Werner’s theory describes the structures of many coordination compounds successfully. However, it does not explain the nature of bonding within the coordination sphere. 2. More than 90% of the known complexes were 4 coordinated or 6 -coordinated at Werner’s time. His theory is unable to account for the preference for 4 - and 6 coordination among complexes. 3. Werner’s theory fails to account for the fact certain 4 coordinated complexes are square planar whereas some others are tetrahedral. 14
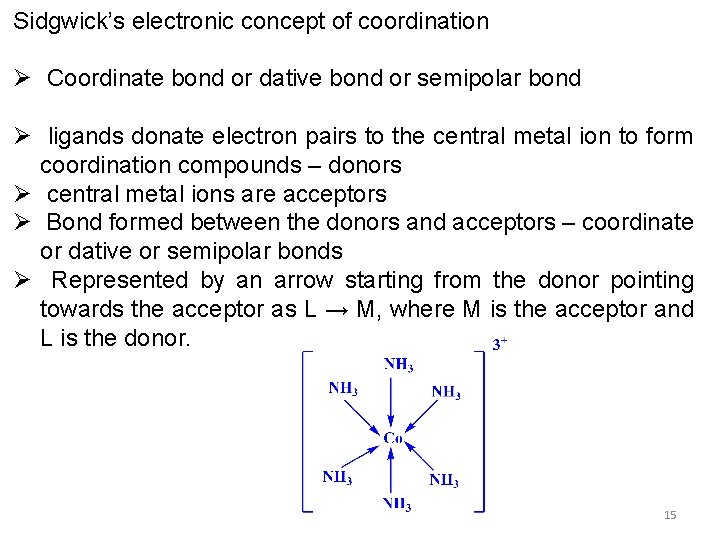
Sidgwick’s electronic concept of coordination Ø Coordinate bond or dative bond or semipolar bond Ø ligands donate electron pairs to the central metal ion to form coordination compounds – donors Ø central metal ions are acceptors Ø Bond formed between the donors and acceptors – coordinate or dative or semipolar bonds Ø Represented by an arrow starting from the donor pointing towards the acceptor as L → M, where M is the acceptor and L is the donor. 15
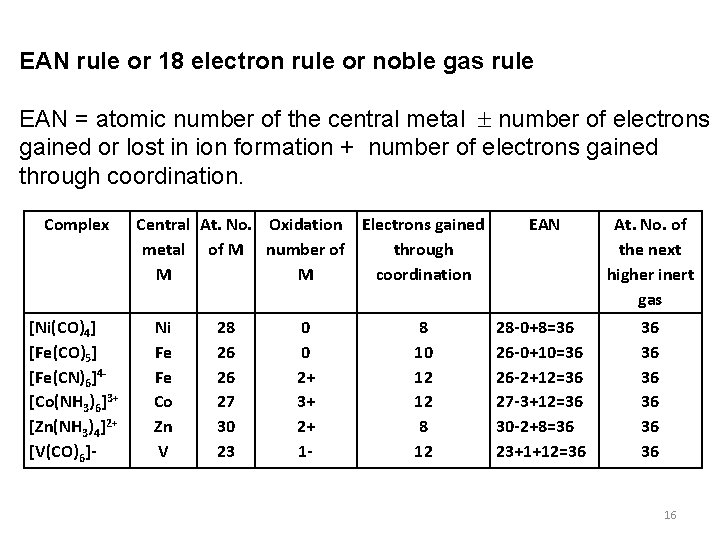
EAN rule or 18 electron rule or noble gas rule EAN = atomic number of the central metal number of electrons gained or lost in ion formation + number of electrons gained through coordination. Complex [Ni(CO)4] [Fe(CO)5] [Fe(CN)6]4[Co(NH 3)6]3+ [Zn(NH 3)4]2+ [V(CO)6]- Central At. No. Oxidation Electrons gained metal of M number of through M M coordination Ni Fe Fe Co Zn V 28 26 26 27 30 23 0 0 2+ 3+ 2+ 1 - 8 10 12 12 8 12 EAN At. No. of the next higher inert gas 28 -0+8=36 26 -0+10=36 26 -2+12=36 27 -3+12=36 30 -2+8=36 23+1+12=36 36 16
![Complexes do not obey EAN rule Complex [V(CO)6] [Mn(CN)4]2[Fe(CN)6]3[Co(NH 3)6]2+ [Ni(NH 3)6]2+ Central At. Complexes do not obey EAN rule Complex [V(CO)6] [Mn(CN)4]2[Fe(CN)6]3[Co(NH 3)6]2+ [Ni(NH 3)6]2+ Central At.](http://slidetodoc.com/presentation_image_h/1cba22de1cea21bba50e9ec182e9712b/image-17.jpg)
Complexes do not obey EAN rule Complex [V(CO)6] [Mn(CN)4]2[Fe(CN)6]3[Co(NH 3)6]2+ [Ni(NH 3)6]2+ Central At. Oxidation metal No. number of M M V Mn Fe Co Ni 23 25 26 27 28 0 2+ 3+ 2+ 2+ Electrons gained through coordination 12 8 12 12 12 EAN 23 -0+12=35 25 -2+8=31 26 -3+12=35 27 -2+12=37 28 -2+12=38 At. No. of the next higher inert gas 36 36 36 17

DEFECTS OF Sidgwick’s THEORY 1. Donation of electrons by the ligands to the central metal ion would cause an accumulation of unfavourable negative charge over the electropositive central metal and it will reduce the stability of the complex. 2. The electron pair donated by H 2 O, NH 3 etc. , to the central metal ion is 2 s 2 pair of electrons. This 2 s 2 pair has no bonding characteristics. In order to make them useful for binding purpose, these electrons should be promoted to high energy level. This requires more energy than what is usually available in the bond formation. 3. According to Sidgwick, coordination compounds must be covalent, But there are complexes which are predominantly ionic in nature, Hence, the forces acting between the central metal ion and the ligands may be regarded as essentially electrostatic. 18
- Slides: 18