Acids and Bases Properties of Acids and Bases

Acids and Bases

Properties of Acids and Bases BASES ACIDS Tart or sour taste (think of lemons) Bitter taste (think of baking soda) DO NOT TASTE CHEMICALS! Corrosive to metals Corrosive to organic materials React with bases to form a salt and water On the p. H scale: acids have a p. H less than 7 Litmus paper = RED React with acids to form a salt and water On the p. H scale: bases have a p. H greater than 7 Litmus paper = BLUE Feel slippery
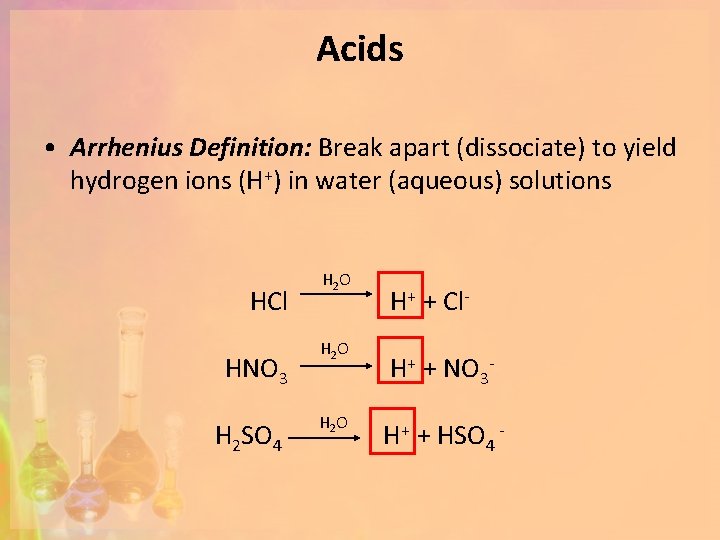
Acids • Arrhenius Definition: Break apart (dissociate) to yield hydrogen ions (H+) in water (aqueous) solutions HCl HNO 3 H 2 SO 4 H 2 O H+ + Cl. H+ + NO 3 H+ + HSO 4 -
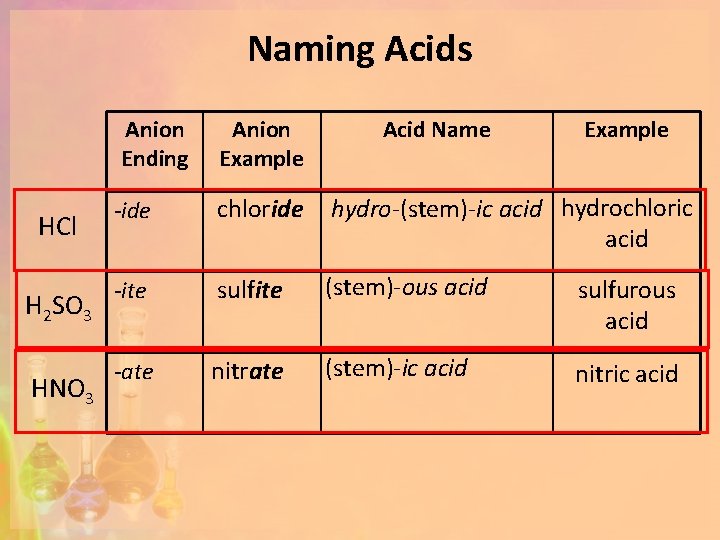
Naming Acids HCl H 2 SO 3 HNO 3 Anion Ending Anion Example Acid Name Example -ide chloride hydro-(stem)-ic acid hydrochloric acid -ite sulfite (stem)-ous acid sulfurous acid -ate nitrate (stem)-ic acid nitric acid

Naming Acids Name the following acids: 1. H 2 Cr. O 4 2. HF 3. H 2 CO 3 4. HCl. O 2 5. CH 3 COOH Anion Ending Acid Name -ide hydro-(stem)-ic acid -ite (stem)-ous acid -ate (stem)-ic acid

Writing Formulas of Acids Write the formulas for the following acids: 1. Phosphorous acid Anion Ending Acid Name -ide hydro-(stem)-ic acid -ite (stem)-ous acid -ate (stem)-ic acid
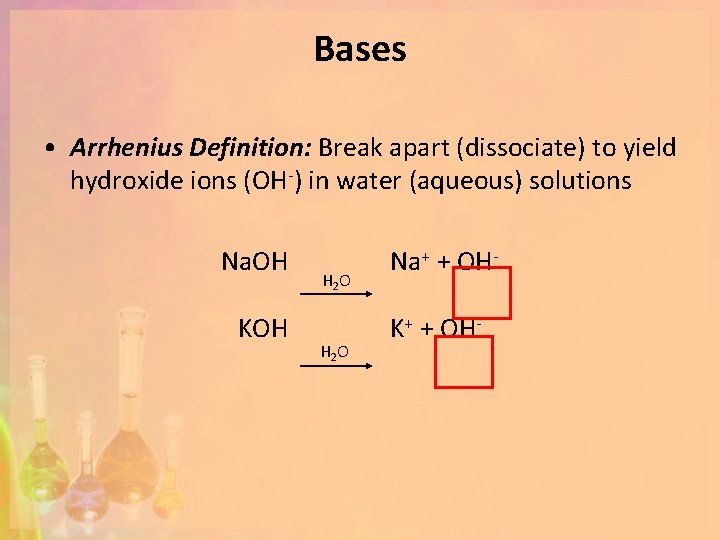
Bases • Arrhenius Definition: Break apart (dissociate) to yield hydroxide ions (OH-) in water (aqueous) solutions Na. OH KOH H 2 O Na+ + OHK+ + OH-
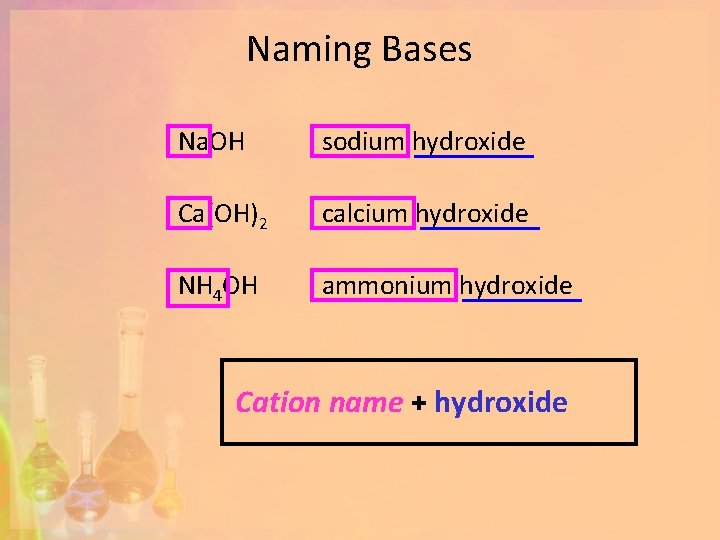
Naming Bases Na. OH sodium hydroxide Ca(OH)2 calcium hydroxide NH 4 OH ammonium hydroxide Cation name + hydroxide

Naming Bases Name the following bases: 1. Ba(OH)2 2. KOH 3. Al(OH)3 Cation name + hydroxide

More Naming Practice Name the following acids/bases or write their formula: 1. H 3 PO 4 2. Nitrous acid 3. Mg(OH)2 Anion Ending Acid Name 4. Hydrobromic acid -ide hydro-(stem)-ic acid 5. Lithium hydroxide -ite (stem)-ous acid 6. H 2 S -ate (stem)-ic acid

Even More Naming Practice! Identify whether the following are acids or bases. Then name them! 1. Ba(OH)2 2. H 2 S 3. CH 3 COOH 4. Fe(OH)2 5. HCl. O 2 Anion Ending Acid Name -ide hydro-(stem)-ic acid -ite (stem)-ous acid -ate (stem)-ic acid

The p. H Scale
Remember the household products lab? • You tested household chemicals with litmus paper and p. H paper. Product 1. 2. … … 9. Color p. H Formula Acid/Base?

What happened to the p. H paper? p. H paper (aka Universal Indicator Paper) • What color(s) do/does the paper turn for acids? Reds and oranges • What color(s) do/does the paper turn for bases? Greens and Blues
What is the p. H scale? • p. H: – stands for “potential of hydrogen” – represents the hydrogen ion (H+) concentration of a solution
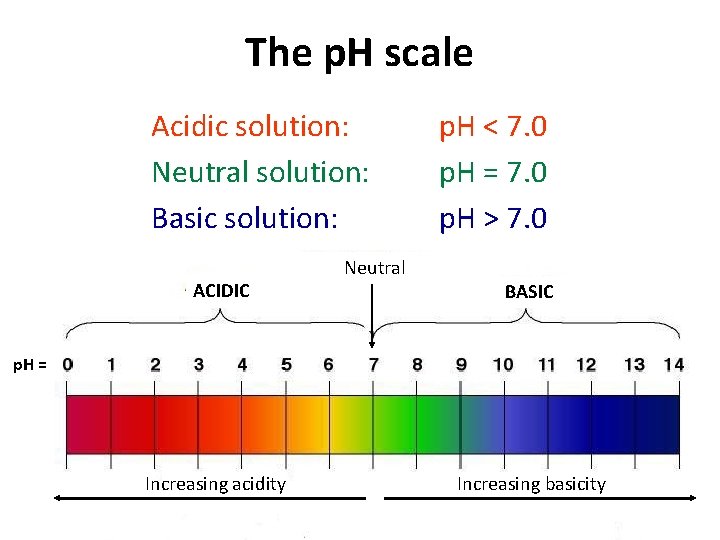
The p. H scale Acidic solution: Neutral solution: Basic solution: ACIDIC Neutral p. H < 7. 0 p. H = 7. 0 p. H > 7. 0 BASIC p. H = Increasing acidity Increasing basicity
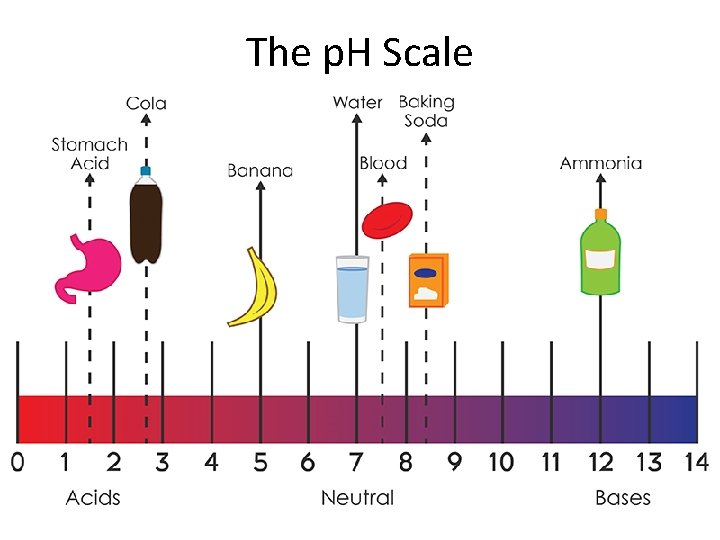
The p. H Scale

The p. H Scale

How can you tell the p. H of a chemical? Use an indicator! 1. Indicator paper (like p. H paper) 2. Liquid indicators (like phenolphthalein) 3. p. H meters
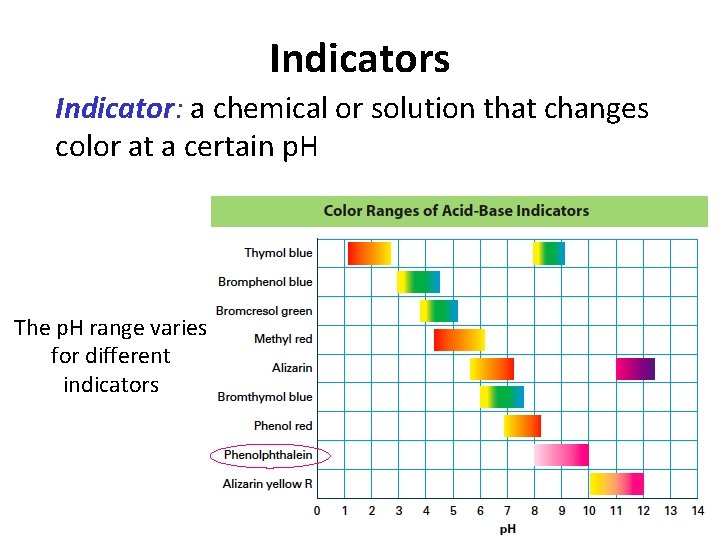
Indicators Indicator: a chemical or solution that changes color at a certain p. H The p. H range varies for different indicators

LAB: Fruit and Vegetable Indicators Purpose: Many naturally colored compounds can behave as acid-base indicators. In this lab, you will be given a choice of several different fruits and vegetables. Your goal is to make indicators from these fruits and vegetables. Your goal today: - With your group, write a DETAILED procedure for how you will prepare your three fruit and vegetable indicator solutions. - Get approved by me before the end of the hour.

Indicator Lab Instructions: Many naturally colored compounds can behave as acid-base indicators. In this lab, you will be given a choice of several different fruits and vegetables. Your goal is to make indicators from these fruits and vegetables. On lined notebook paper, you must do the following: 1. Write a purpose. 2. Provide a complete set of instructions for making the indicator solution from the raw materials and testing the solution. (The indicator is usually in the skin of the fruit or veggie. You will have to boil the fruit or veggie in a small amount of water to extract the indicator). 3. Create a data table listing your results for 3 indicator solutions (see below). 4. Answer the analysis questions. Available Materials: solutions with various p. H, carrots, blueberries, red cabbage, red grapes, red onions, tea leaves, 100 m. L beaker, deionized water, pipettes, well plates, scoopula

DEMO • Why do chemistry teachers like to teach about hydroxides?

Calculating p. H and p. OH
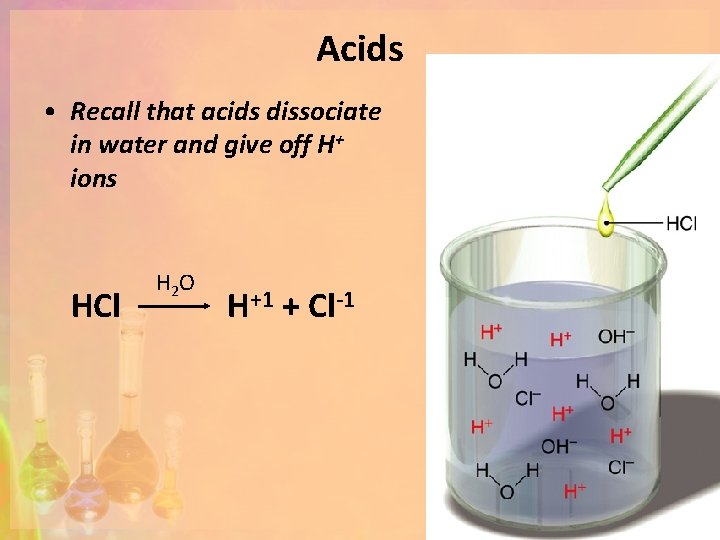
Acids • Recall that acids dissociate in water and give off H+ ions HCl H 2 O H+1 + Cl-1

p. H What is an easy way of representing the concentration of hydrogen ions in a solution? p. H (“potential of hydrogen”): a way of expressing the hydrogen ion concentration p. H = - log [H+] = the hydrogen ion concentration IN MOLARITY

Calculating p. H Calculate the p. H values using the equation p. H = - log [H+]. Do you notice a pattern? [H+] p. H 1 1 x 10 -3 M 3. 0 2 1 x 10 -5 M 5. 0 3 1 x 10 -7 M 7. 0 4 1 x 10 -9 M 9. 0 5 1 x 10 -12 M 12. 0 As the p. H value increases, the H+ concentration decreases
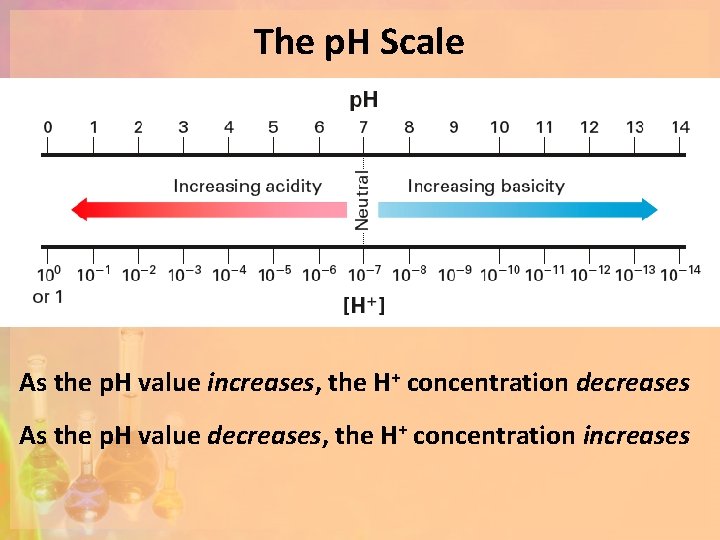
The p. H Scale As the p. H value increases, the H+ concentration decreases As the p. H value decreases, the H+ concentration increases
![Calculating p. H Example p. H = - log [H+] What is the p. Calculating p. H Example p. H = - log [H+] What is the p.](http://slidetodoc.com/presentation_image_h/c36ebc38224c1cc45e2c4bdcdd377143/image-30.jpg)
Calculating p. H Example p. H = - log [H+] What is the p. H of a HCl solution in which [H+] = 3. 9 x 10 -4 M?
![Calculating [H+] How do we calculate the hydrogen ion concentration, [H+], if we know Calculating [H+] How do we calculate the hydrogen ion concentration, [H+], if we know](http://slidetodoc.com/presentation_image_h/c36ebc38224c1cc45e2c4bdcdd377143/image-31.jpg)
Calculating [H+] How do we calculate the hydrogen ion concentration, [H+], if we know the p. H? p. H = - log [H+] = the hydrogen ion concentration IN MOLARITY [H+] = 10 -p. H
![Calculating [H+] Example [H+] = 10 -p. H What is the [H+] of a Calculating [H+] Example [H+] = 10 -p. H What is the [H+] of a](http://slidetodoc.com/presentation_image_h/c36ebc38224c1cc45e2c4bdcdd377143/image-32.jpg)
Calculating [H+] Example [H+] = 10 -p. H What is the [H+] of a solution if the p. H = 10. 8?
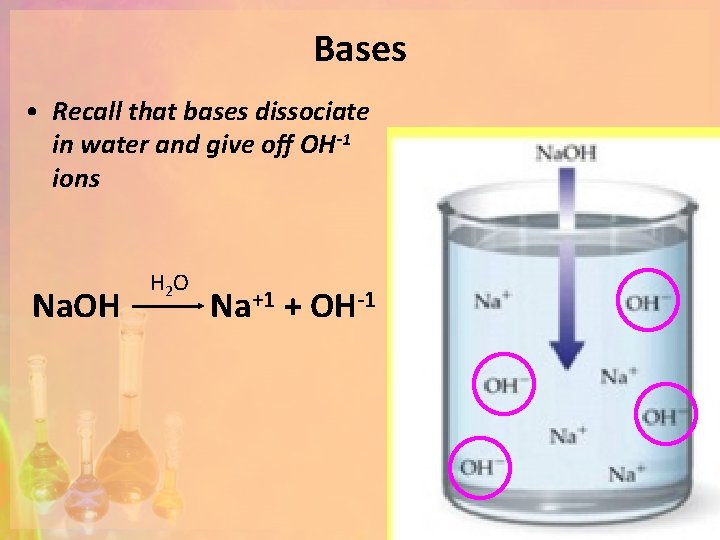
Bases • Recall that bases dissociate in water and give off OH-1 ions Na. OH H 2 O Na+1 + OH-1

Just like with p. H… p. OH (“potential of hydroxide”): a way of expressing the hydroxide ion concentration (the OH-) p. OH = - log [OH-] = the hydroxide ion concentration IN MOLARITY Just like with [H+]: [OH-] = 10 -p. OH The only way to go between H and OH: p. H + p. OH = 14
![Example #1 What is the p. OH of a solution if the [OH-] = Example #1 What is the p. OH of a solution if the [OH-] =](http://slidetodoc.com/presentation_image_h/c36ebc38224c1cc45e2c4bdcdd377143/image-35.jpg)
Example #1 What is the p. OH of a solution if the [OH-] = 3. 5 x 10 -9 M?
![Example #2 What is the p. H of a solution if the [OH-] = Example #2 What is the p. H of a solution if the [OH-] =](http://slidetodoc.com/presentation_image_h/c36ebc38224c1cc45e2c4bdcdd377143/image-36.jpg)
Example #2 What is the p. H of a solution if the [OH-] = 0. 00059 M?
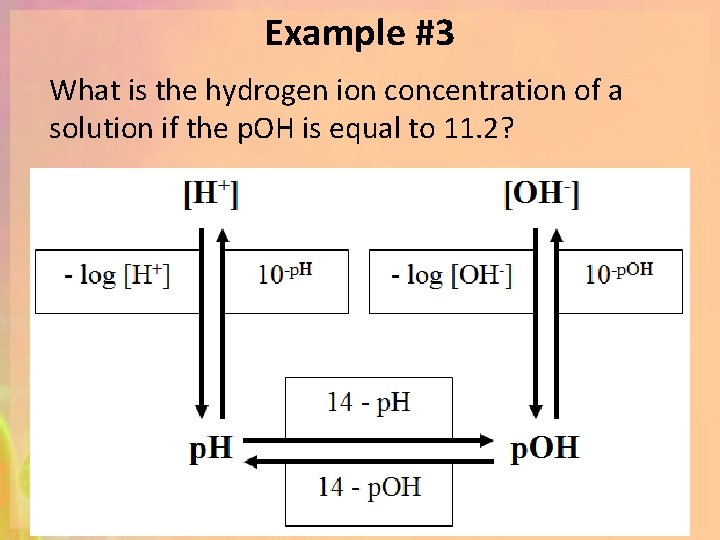
Example #3 What is the hydrogen ion concentration of a solution if the p. OH is equal to 11. 2?

Acid-Base Neutralization Reactions

Neutralization Reactions Remember double replacement reactions? HCl + Na. OH → ____ + ____ To write balanced reaction: 1. Circle any polyatomic ions 2. Label charges 3. Write products – make charges cancel! 4. Balance using coefficients
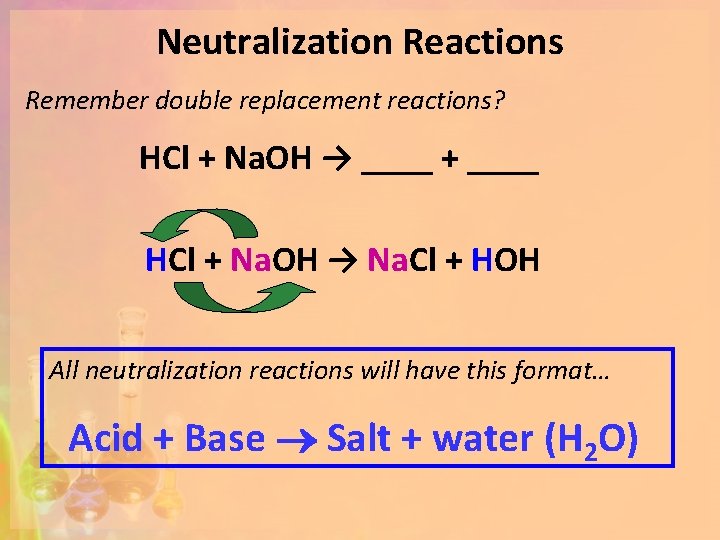
Neutralization Reactions Remember double replacement reactions? HCl + Na. OH → ____ + ____ HCl + Na. OH → Na. Cl + HOH All neutralization reactions will have this format… Acid + Base Salt + water (H 2 O)
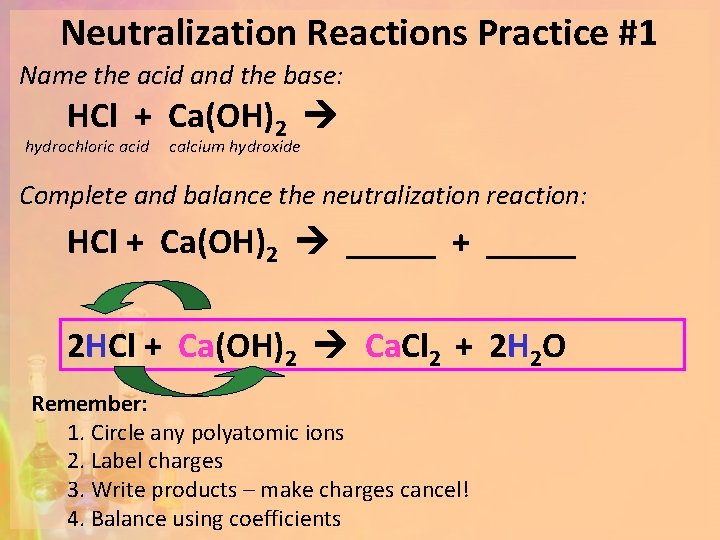
Neutralization Reactions Practice #1 Name the acid and the base: HCl + Ca(OH)2 hydrochloric acid calcium hydroxide Complete and balance the neutralization reaction: HCl + Ca(OH)2 _____ + _____ 2 HCl + Ca(OH)2 Ca. Cl 2 + 2 H 2 O Remember: 1. Circle any polyatomic ions 2. Label charges 3. Write products – make charges cancel! 4. Balance using coefficients
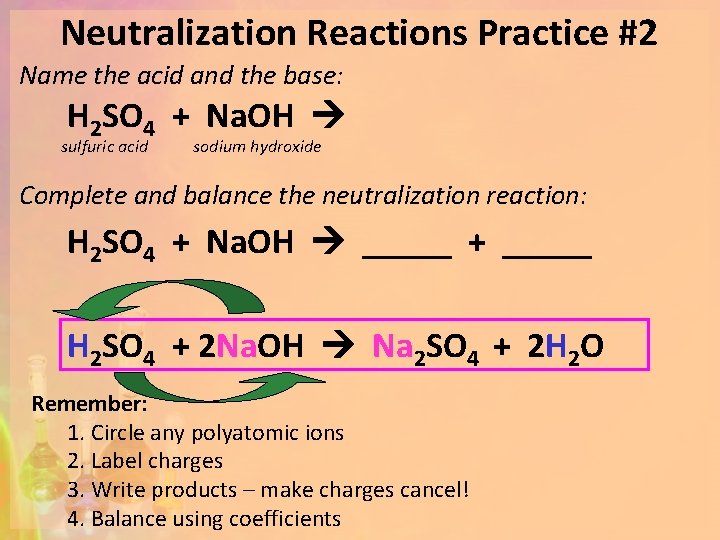
Neutralization Reactions Practice #2 Name the acid and the base: H 2 SO 4 + Na. OH sulfuric acid sodium hydroxide Complete and balance the neutralization reaction: H 2 SO 4 + Na. OH _____ + _____ H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O Remember: 1. Circle any polyatomic ions 2. Label charges 3. Write products – make charges cancel! 4. Balance using coefficients
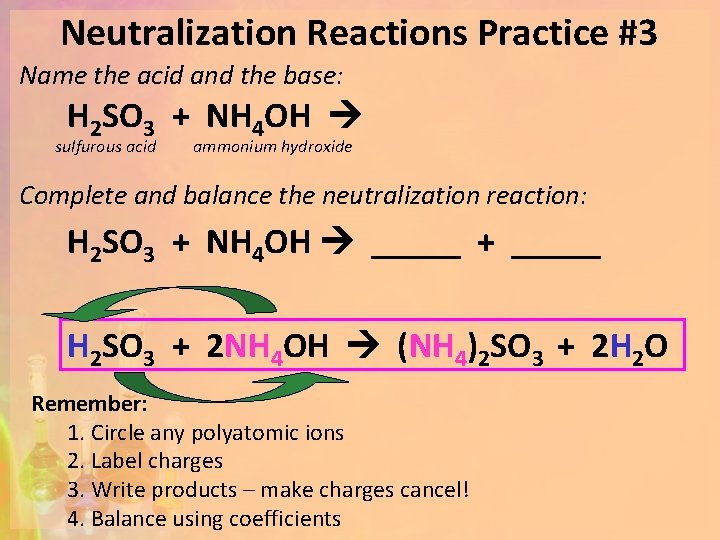
Neutralization Reactions Practice #3 Name the acid and the base: H 2 SO 3 + NH 4 OH sulfurous acid ammonium hydroxide Complete and balance the neutralization reaction: H 2 SO 3 + NH 4 OH _____ + _____ H 2 SO 3 + 2 NH 4 OH (NH 4)2 SO 3 + 2 H 2 O Remember: 1. Circle any polyatomic ions 2. Label charges 3. Write products – make charges cancel! 4. Balance using coefficients

Neutralization Practice
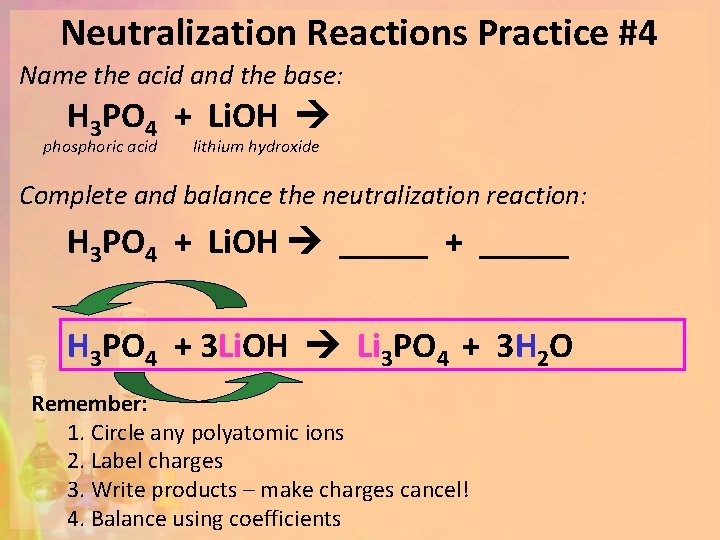
Neutralization Reactions Practice #4 Name the acid and the base: H 3 PO 4 + Li. OH phosphoric acid lithium hydroxide Complete and balance the neutralization reaction: H 3 PO 4 + Li. OH _____ + _____ H 3 PO 4 + 3 Li. OH Li 3 PO 4 + 3 H 2 O Remember: 1. Circle any polyatomic ions 2. Label charges 3. Write products – make charges cancel! 4. Balance using coefficients

Acid-Base Titrations

Titrations Titration: adding a known amount of solution of known concentration to a solution with an unknown concentration Goal: To determine the unknown concentration of a solution

Titrations Endpoint: the point of neutralization in a titration How do we know we reached the endpoint in a titration? We use an indicator and look for a color change!

Titration Example A 2. 5 L solution of H 2 SO 4 is completely neutralized by 0. 5 L of 1. 0 M Na. OH. What is the concentration of the H 2 SO 4 solution? In buret: 1. 0 M Na. OH To solve this problem, we need to use the box method! In Erlenmeyer flask: ? M H 2 SO 4

A 2. 5 L solution of H 2 SO 4 is completely neutralized by 0. 5 L of 1. 0 M Na. OH. What is the concentration of the H 2 SO 4 solution? H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O Step 1: Write the balanced neutralization equation.

A 2. 5 L solution of H 2 SO 4 is completely neutralized by 0. 5 L of 1. 0 M Na. OH. What is the concentration of the H 2 SO 4 solution? H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O x 1/2 Moles (mol) 0. 25 mol 0. 10 M 1. 0 M 2. 5 L 0. 5 L Molarity (M)down = divide Moving Liters (L) Multiply by molar ratio Moving up = multiply Step 2: Draw the box, fill in known information, and calculate missing information.

Titration Practice - #1 on WS 30 m. L of 0. 10 M Na. OH is used to completely neutralize 25. 0 m. L of HCl. Determine the concentration of the acid. Moles (mol) Molarity (M) Liters (L)

HCl Titration Lab – Analysis Questions Work with your partner to complete questions 3 -5 on your lab…

Titration Practice #1 A 1. 5 L solution of HCl is completely neutralized by 2. 0 L of 0. 5 M Al(OH)3. What is the concentration of the HCl solution? Moles (mol) Molarity (M) Liters (L)

Titration Practice #2 A 25 m. L solution of H 2 SO 4 is completely neutralized by 6. 5 m. L of 1. 0 M Na. OH. What is the concentration of the H 2 SO 4 solution? Moles (mol) Molarity (M) Liters (L)
- Slides: 55