Reaction Rates and Le Chateliers Principle Rate of











- Slides: 32

Reaction Rates and Le Chatelier’s Principle

Rate of Reaction • The rate of a chemical reaction – Describes how rapidly a chemical change takes place • They are determined by measuring changes in physical properties – Volume, Temperature, Color, Mass, or p. H

Five Factors that Affect Reaction Rate • • • 1. Nature of the reactants 2. Temperature 3. Concentration 4. Surface Area 5. Catalysts

Chemical Equilibrium • At equilibrium the forward reaction and the reverse reaction happen at the same time – There is no change in the amount of any substance in the reaction • Le Chatelier’s principle can be used to predict the effect of a change in conditions on a chemical equilibrium

Le Chatelier’s Principle Summarized • If a chemical system at equilibrium experiences a change (also called a stress) in concentration, temperature, volume, or total pressure; the equilibrium will shift in order to minimize that change.

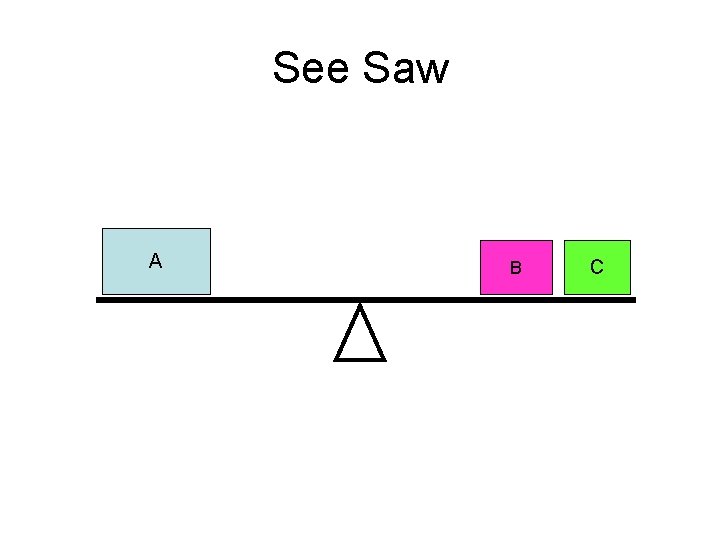
See Saw A B C

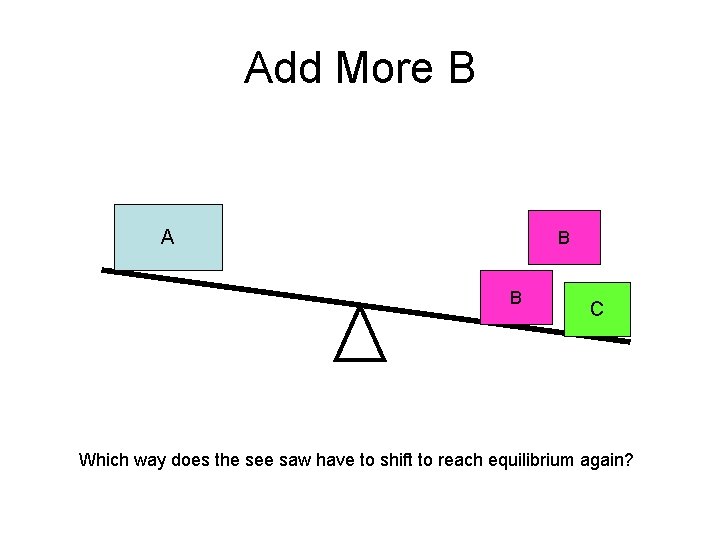
Add More B A B B C Which way does the see saw have to shift to reach equilibrium again?

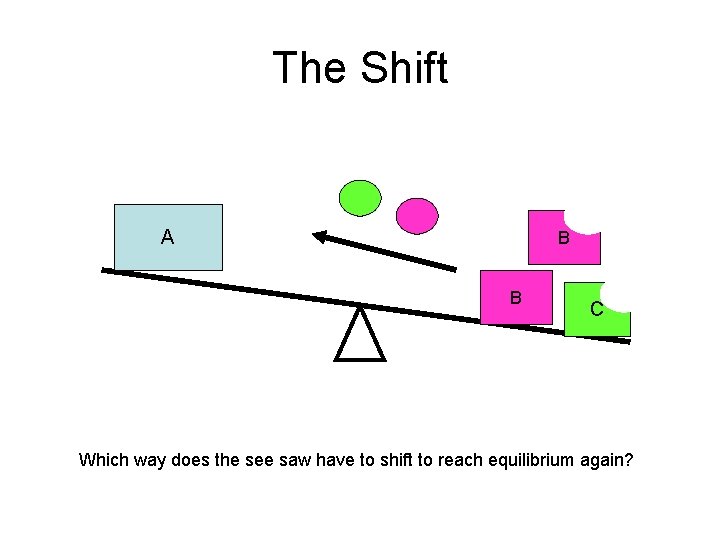
The Shift A B B C Which way does the see saw have to shift to reach equilibrium again?

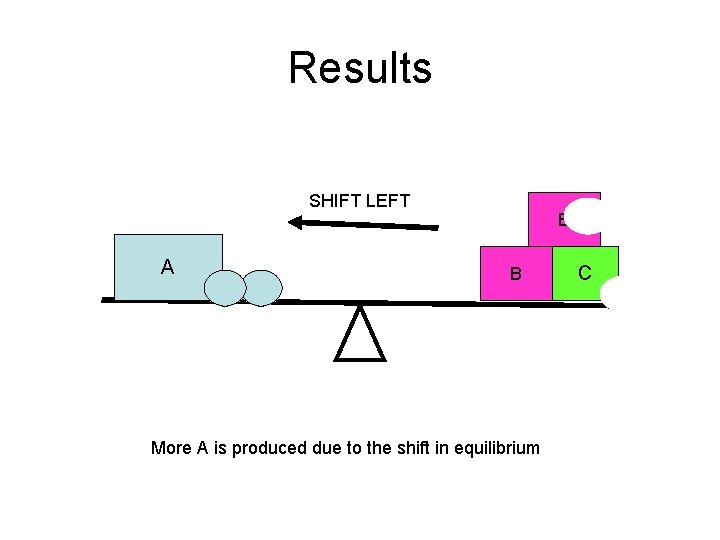
Results SHIFT LEFT A B B More A is produced due to the shift in equilibrium C

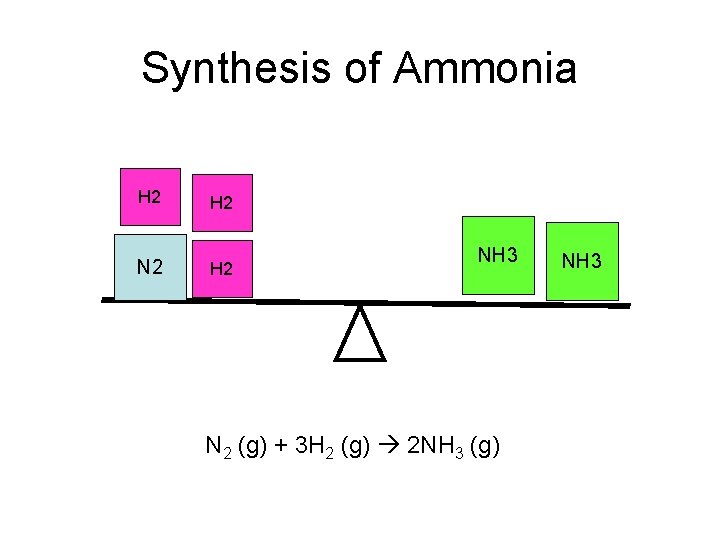
Synthesis of Ammonia H 2 N 2 H 2 NH 3 N 2 (g) + 3 H 2 (g) 2 NH 3 (g) NH 3

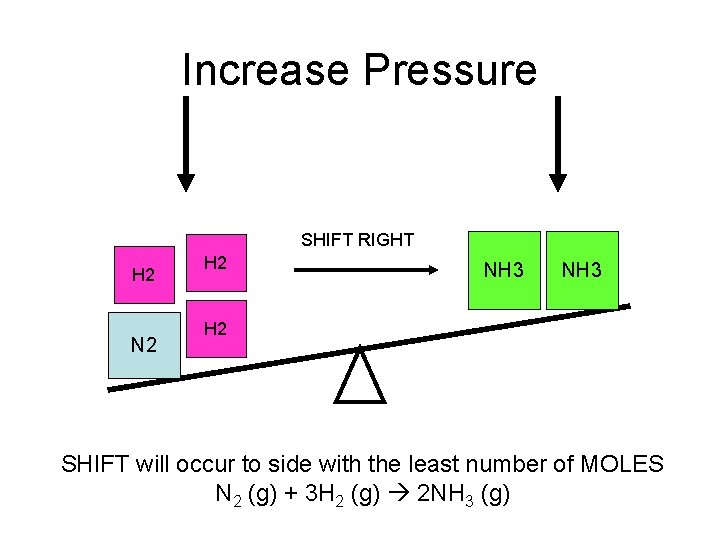
Increase Pressure SHIFT RIGHT H 2 N 2 H 2 NH 3 H 2 SHIFT will occur to side with the least number of MOLES N 2 (g) + 3 H 2 (g) 2 NH 3 (g)

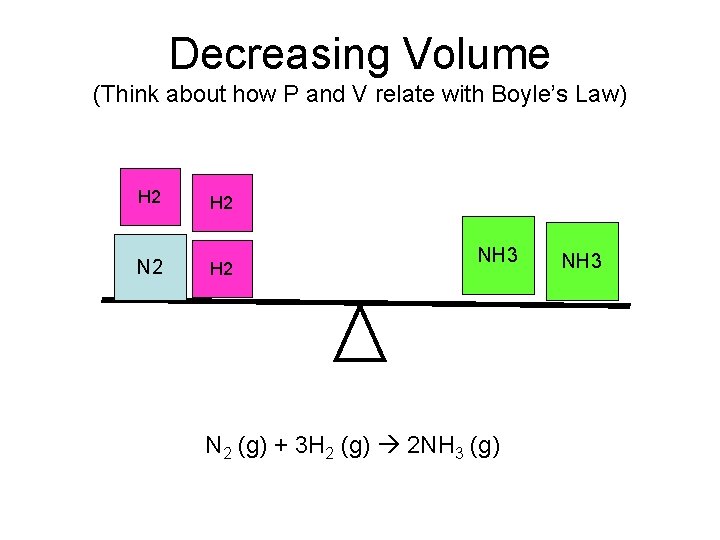
Decreasing Volume (Think about how P and V relate with Boyle’s Law) H 2 N 2 H 2 NH 3 N 2 (g) + 3 H 2 (g) 2 NH 3 (g) NH 3

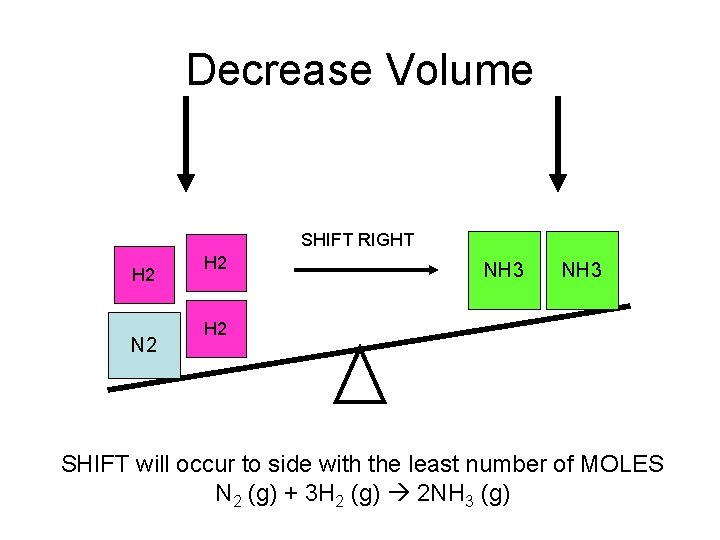
Decrease Volume SHIFT RIGHT H 2 N 2 H 2 NH 3 H 2 SHIFT will occur to side with the least number of MOLES N 2 (g) + 3 H 2 (g) 2 NH 3 (g)

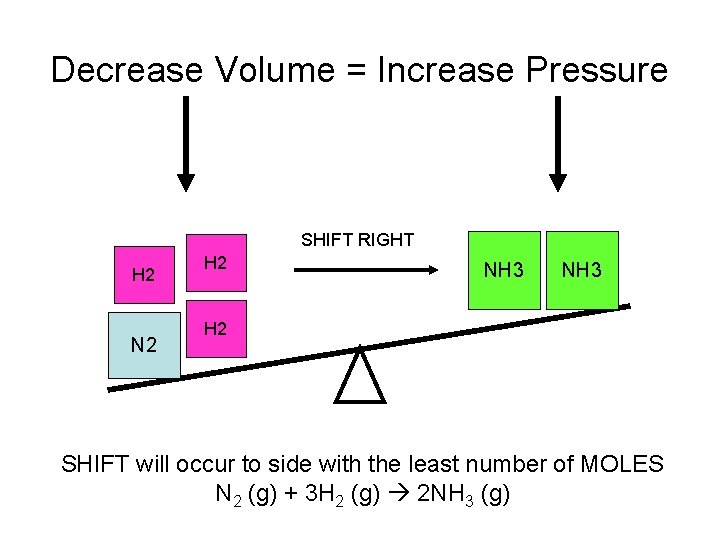
P and V • P and V are inversely related. That means as P increases, then V decreases. • The rule is when P increases or V decreases then the equilibrium will shift to the side with the least number of moles. • This only works if all of the components are gasses.

Decrease Volume = Increase Pressure SHIFT RIGHT H 2 N 2 H 2 NH 3 H 2 SHIFT will occur to side with the least number of MOLES N 2 (g) + 3 H 2 (g) 2 NH 3 (g)

Change in Temperature • Depends if reaction is Endothermic or Exothermic. • Endothermic: Heat on LEFT (+) • Exothermic: Heat on RIGHT (-)

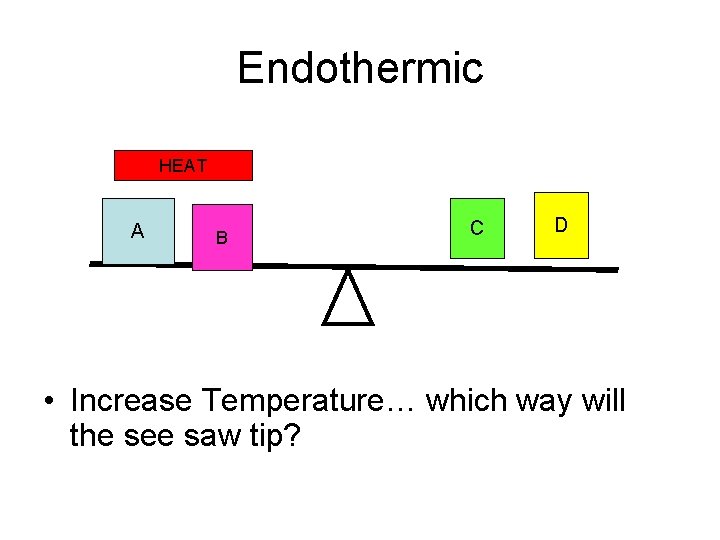
Endothermic HEAT A B C D • Increase Temperature… which way will the see saw tip?

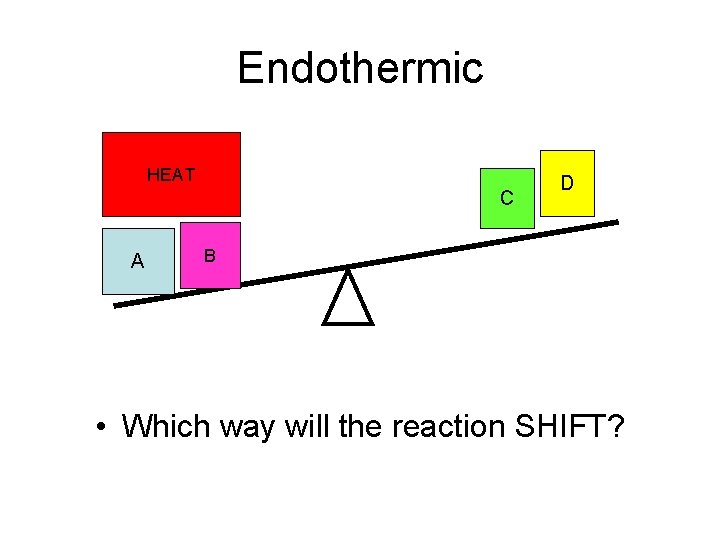
Endothermic HEAT C A D B • Which way will the reaction SHIFT?

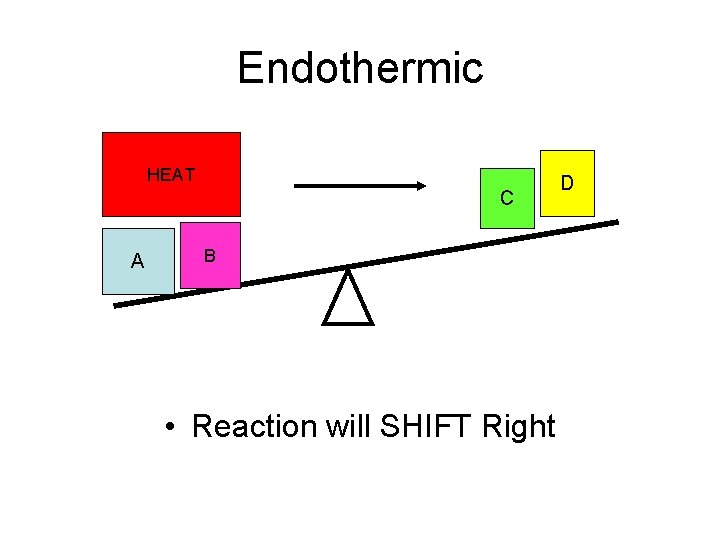
Endothermic HEAT C A B • Reaction will SHIFT Right D

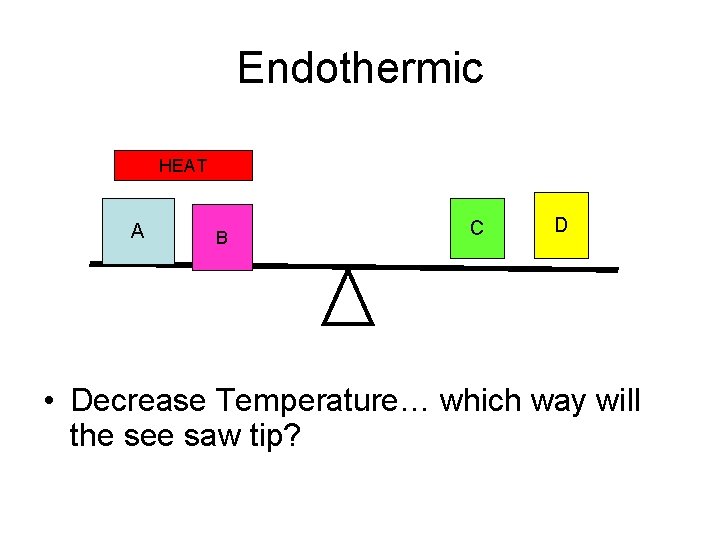
Endothermic HEAT A B C D • Decrease Temperature… which way will the see saw tip?

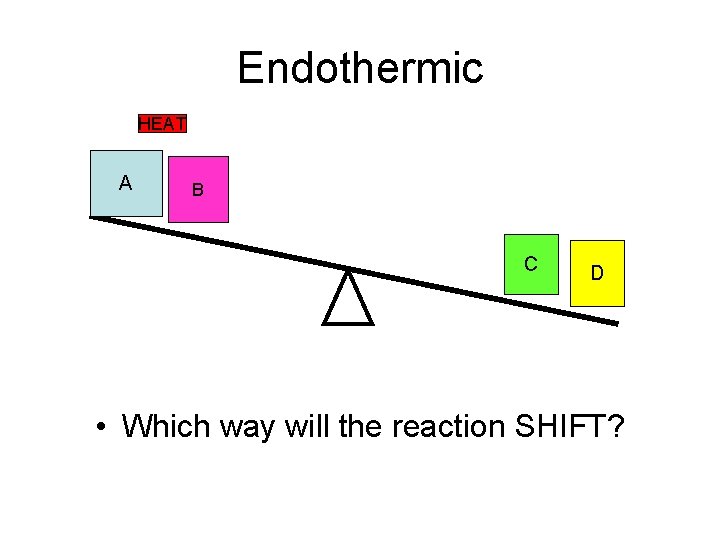
Endothermic HEAT A B C D • Which way will the reaction SHIFT?

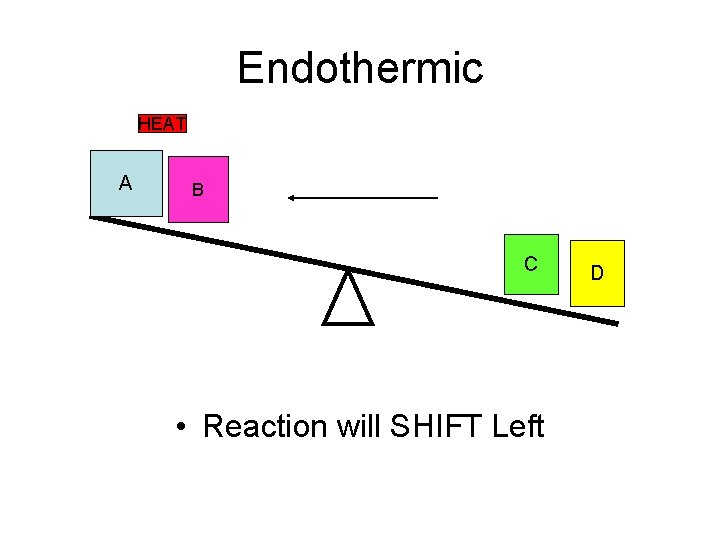
Endothermic HEAT A B C • Reaction will SHIFT Left D

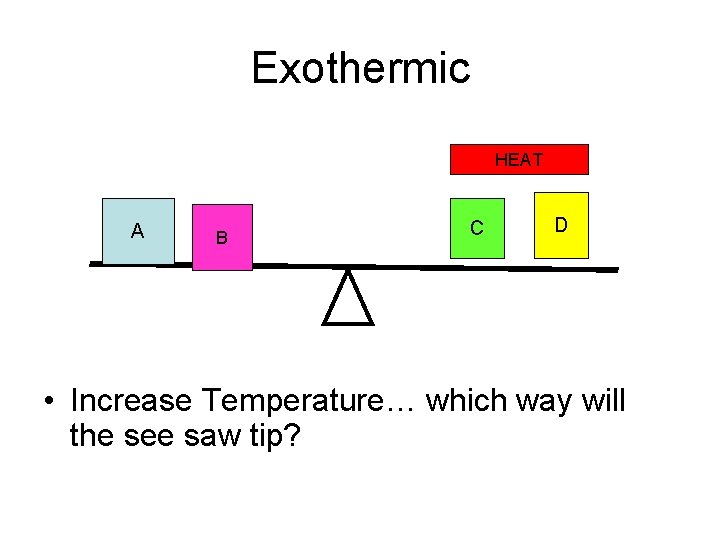
Exothermic HEAT A B C D • Increase Temperature… which way will the see saw tip?

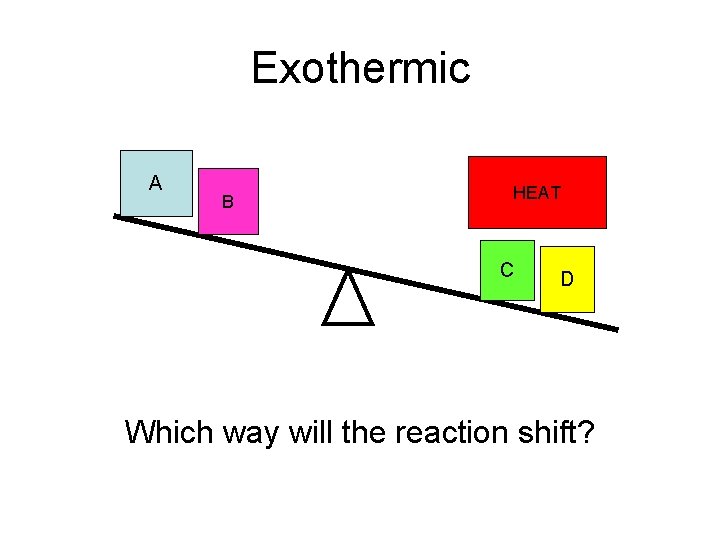
Exothermic A B HEAT C D Which way will the reaction shift?

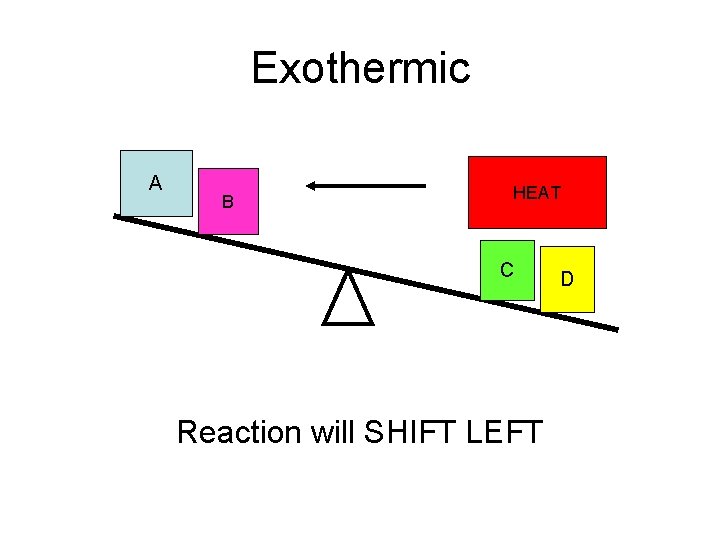
Exothermic A B HEAT C Reaction will SHIFT LEFT D

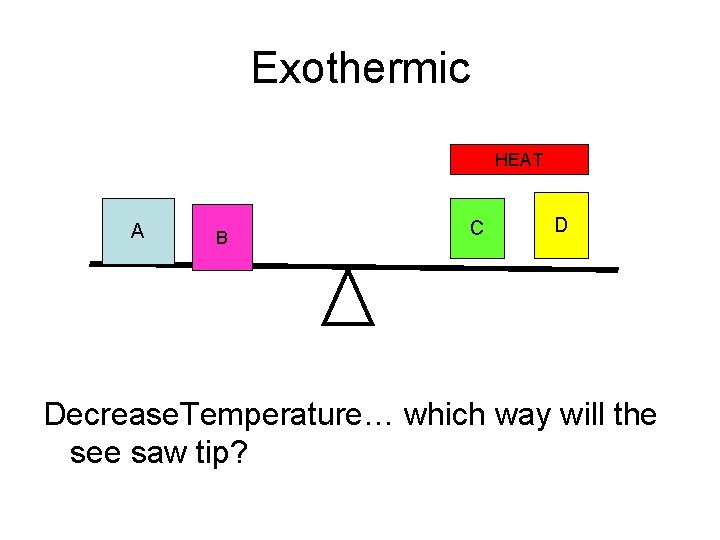
Exothermic HEAT A B C D Decrease. Temperature… which way will the see saw tip?

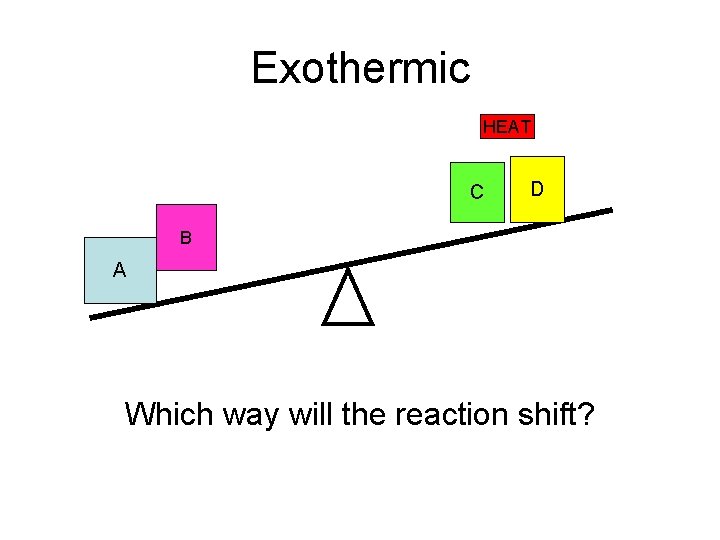
Exothermic HEAT C D B A Which way will the reaction shift?

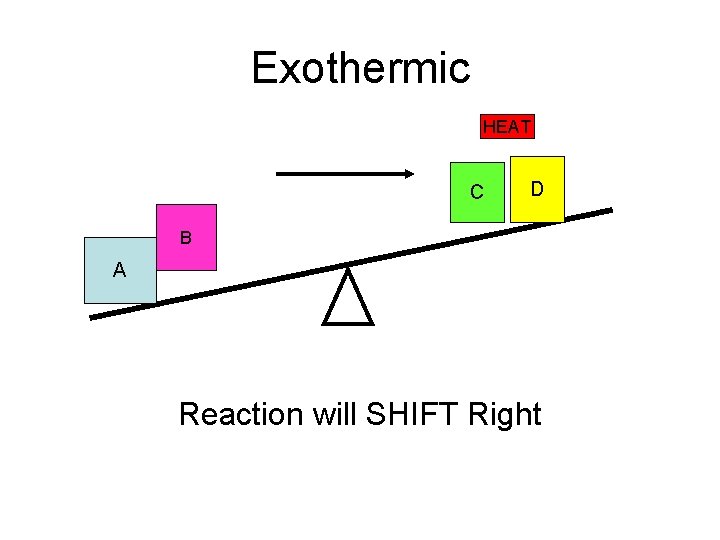
Exothermic HEAT C D B A Reaction will SHIFT Right

How does each situation affect the chemical reaction? 2 H 2 O 2 H 2 + O 2 Cause Effect Increase Reactant Concentration Increase Product Concentration Increase temperature of an endothermic reaction Increase temperature of an exothermic reaction Increase Pressure

Which INCREASE Reaction Rates? • • Increasing Temperature Increasing Concentration Increasing Surface Area Adding a Catalyst. • How are these factors related to Reaction Rates? – DIRECTLY

Nature of Reactants • Reaction rates are affected by the complexity of the bonds that must be broken and formed in the chemical reaction • The state of a reactant can also affect the reaction rate – Gases have the fastest and solids the slowest reaction rats • Because the frequency at which particles collide and the amount of energy they possess increase with increasing motion.

Continued • The faster particles are moving, the more frequent they will collide – Collide with greater kinetic energy. • Because of this TEMPERATURE and REACTION RATE are directly related