REACTION RATES FACTORS AFFECTING REACTION RATES I can





































































![�Equation: rate = k[A]x[B]y �[A] and [B] are molar concentrations. �K is a rate �Equation: rate = k[A]x[B]y �[A] and [B] are molar concentrations. �K is a rate](https://slidetodoc.com/presentation_image_h/10c07f8e694b11d4cba4202dd272a26a/image-73.jpg)


![�Example: NO 2(g) + CO(g) NO(g) + CO 2(g) Rate = [NO 2]2 �Example: NO 2(g) + CO(g) NO(g) + CO 2(g) Rate = [NO 2]2](https://slidetodoc.com/presentation_image_h/10c07f8e694b11d4cba4202dd272a26a/image-76.jpg)


- Slides: 78

REACTION RATES

FACTORS AFFECTING REACTION RATES

�I can identify and describe the five factors that affect reaction rates.

�There are five general factors that affect the rate of reaction.

NATURE OF REACTANTS �The rate of reaction depends on the particular reactants and the complexity of the bonds that have to be broken and formed. �Reactions where the rearrangement of atoms is simple occur faster. �More complex reactions are much slower.

NATURE OF REACTANTS �Reactions that involve covalent bonds are usually slower because they are more complex. �The states of a reactant in a chemical reaction can also affect reaction rate.

NATURE OF REACTANTS �Reactions that involve gases will be the fastest, followed by those that involve liquids, and reactions that involve solids are the slowest.

TEMPERATURE �Most reactions occur more rapidly with increased temperatures. �The higher the temperature, the faster the molecules move.

TEMPERATURE �The faster molecules move, the more energy they have, and the more often they collide, which helps them react. �Therefore, increasing temperature increases the rate of reaction.

CONCENTRATION �The rate of reaction depends on concentrations of the reactants. �The higher the concentrations, the faster the reaction rate. �The reason for the increase is due to the fact that higher concentrations result in more collisions.

SURFACE AREA �The more surface area a reactant has, the greater the number of particles that are expose for reaction. �A larger surface area increases the frequency at which particle collide. �To increase the surface area of a solid, scientists use powders.

CATALYSTS �A catalyst is a substance that increases the rate of a reaction without being used up. �Catalysts can be used over and over again. �Catalysts in the human body are called enzymes.

CATALYSTS �Catalysts are not a reactant or a product. �They can be re-used after a chemical reaction occurs. �Catalysts increase reaction rates because they lower the activation energy for a reaction to occur.

CATALYSTS �The lower the activation energy, the faster the reaction rate. �It’s not a good idea to speed up all chemical reactions. �Substances that slow a chemical reaction are inhibitors.

THE COLLISION THEORY

�I can describe the collision theory. � I can define an effective vs. ineffective collision. � I can define both kinetic and potential energy. � I can interpret an energy diagram.

�In order for a chemical reaction to occur, particles must come together. �Combining two substances means forcing their particles to hit or collide. �The collision theory states that molecules must collide to react.

� In order for a molecule, atom, or ion to react, it must collide with another molecule, atom, or ion or with the wall of a container. � If there were no collisions among reactants, there would be no reactions. � Not all collisions are effective in leading to the formation of products.

�Effective collisions lead to the formation of products. �Ineffective collisions do not lead to the formation of products. �If every collision lead to a reaction, the rates of reaction would be much faster.

� For a reaction to occur, particles must be in the correct position that allows the bonds to break and atoms to rearrange. � If the orientation is not right, the molecules bounce off each other. � Colliding particles must also have enough energy so that bonds can be broken and new bonds formed.

ENERGY IN REACTIONS �Energy is required to break bonds that hold reactants together. �There are two types of energy: Kinetic and potential. �Potential energy is stored and kinetic energy is the energy of motion.

�Remember, energy can not be created or destroyed, only transformed. �Particles of matter have both potential and kinetic energy. �The energy to break bonds comes form the energy of the reacting particles.

�During a collision, kinetic energy is converted to potential energy as particles are formed, bonds are broken, and atoms rearranged. �Kinetic energy depends on the mass and velocity of a particle. �Particles must have a minimum amount of energy to react.

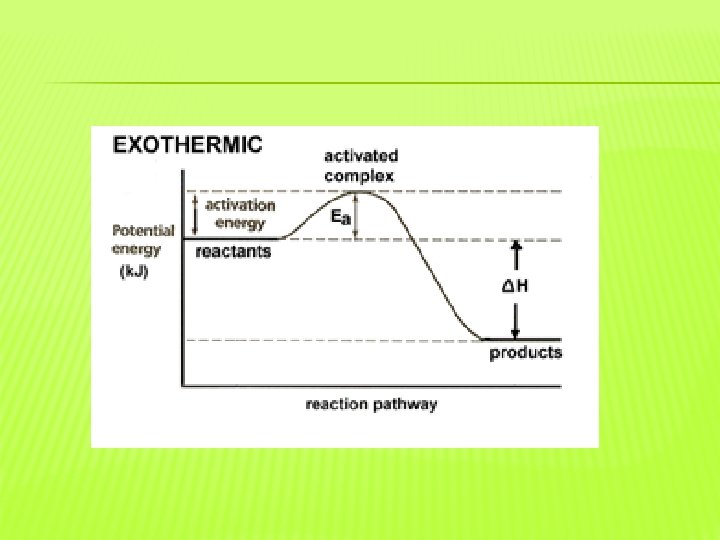
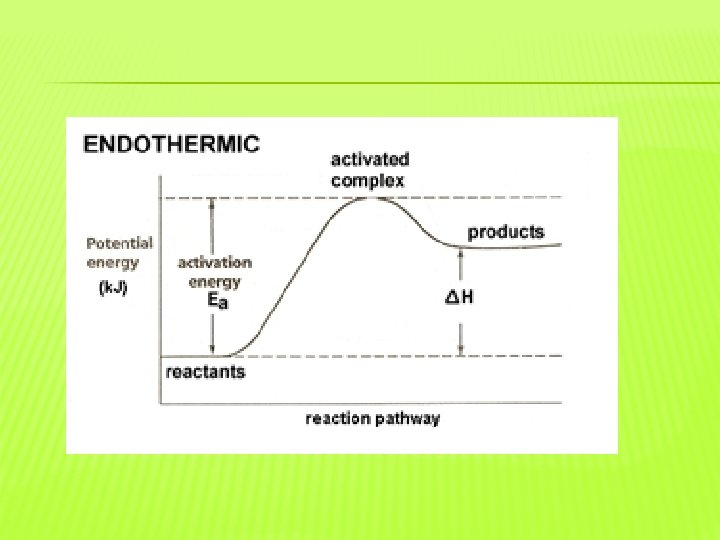
�Activation energy is the energy needed to start a reaction. �Energy diagrams show the changes in energy that occur during a chemical reaction. �The difference between the peak and reactants is the activation energy.



�An exothermic reaction releases heat. �Therefore, the products have less energy than the reactants. �An endothermic reaction absorbs heat. �Therefore the products have more energy than the reactants.

�The activated complex is a structure that forms in a reaction that is not a reactant or a product. �The activated complex exists where the energy is greatest. �Reactions occur at different rates because of energy.

� Particles must collide at the correct orientation to react. � Particles must also have enough energy to overcome the activation energy to react. � Reaction rates are directly related to activation energy because every reaction has a different activation energy.

FREE ENERGY

�I can define free energy. � I can describe a spontaneous and nonspontaneous reaction. � I can define both entropy and enthalpy. � I can describe the conditions under which a spontaneous and nonspontaneous reaction occur.

�Many chemical and physical processes release energy that can be used to bring about other changes. �Free energy is the energy that is available to do work.

�Even though free energy is available, that does not mean it can be used efficiently. �Car engines are only 30% efficient. �That means only 30% of the free energy released from burning gasoline is used to move the car.

�The other 70% is lost to friction and heat. �Efforts are constantly underway to increase efficiency. �However, no mechanical process can be made 100% efficient.

� Energy can only be obtained from a reaction if the reaction actually occurs. � Not all balanced chemical reactions actually occur in nature. � Spontaneous reactions are reactions that occur naturally and that favor the formation of products at the specified conditions.

� All spontaneous reactions release free energy. � Nonspontaneous reactions are reactions that do not favor the formation of products at the specified conditions. � In most reversible reactions, one reaction is spontaneous and one is nonspontaneous.

�The terms spontaneous and nonspontaneous do not refer to how fast reactants go to products. �Some spontaneous reactions are extremely slow, and they look like they are nonspontaneous.

�Some reactions that are nonspontaneous at one set of conditions may be spontaneous at other conditions. �Changing the temperature or pressure can change a reaction to be spontaneous.

�Heat change accompanies most chemical and physical processes. �Both exothermic and endothermic reactions can be spontaneous. �Heat is also called enthalpy. �Enthalpy change is symbolized as H.

�To determine whether or not a reaction is spontaneous, we must consider heat and disorder. �The disorder of a system is called entropy. �The more disorder, the more entropy.

�The law of disorder states that processes move in the direction of maximum disorder or randomness. �The entropy of gasses is higher than the entropy of liquids. �The entropy of liquids is higher than the entropy of solids.

�Entropy increases when solid reactants form liquids. �Entropy also increases when liquid reactants form gases. �Entropy increases when a substance is divided into parts.

�Therefore, entropy increases when a solid is dissolved in water because the solute particles are more separated than the solid. �Entropy tends to increase in chemical reactions when the total number of product molecules is greater than the total number of

�Entropy tends to increase when temperature increases. �As the temperature increases, the molecules move faster and faster, which increases disorder.

�The size and direction of heat changes and entropy changes determine whether a reaction is spontaneous. �In other words, whether it favors products and releases free energy.

� Releasing heat is a favorable condition for a reaction to be spontaneous. � Absorbing heat is an unfavorable condition. � Increasing entropy is a favorable condition for a reaction to be spontaneous. � Decreasing entropy is an unfavorable condition.

�Spontaneous reactions occur under the following conditions: �Heat is released and Entropy increases �The entropy increase is larger than the heat absorbed. �The heat released is larger than the decrease in entropy.

� Reactions will be nonspontaneous under the following conditions: � The heat absorbed is greater than the increase in entropy. � The heat released is less than the decrease in entropy. � Heat is absorbed and entropy decreases.

ENTROPY CALCULATIONS

�Entropy is the measure of disorder of a system. �The symbol for entropy is S. �The units for S is J/K �The standard entropy of a liquid or solid substance at 25 C is designated So.

�The pressure at So for gases is 101. 3 k. Pa. �Standard entropy changes can be calculated using the following equation: So = So(products) – So (reactants)

EXAMPLE What is the standard change in entropy for the reaction below? 2 NO(g) + O 2(g) 2 NO 2(g) So for NO 2 = 240. 5 x So for NO = 210. 6 x So for O 2 = 205. 0 x

GIBBS FREE ENERGY

�In every spontaneous process, some energy becomes available to do work. �This energy is called the Gibbs-free energy change ( G) �It is the maximum amount of energy that can be coupled to another process to do useful work.

�The change in Gibbs free energy is related to the change in entropy and the change in enthalpy. �The change in enthalpy is the change in heat.

�The equation for Gibbs Free Energy: G= H–T S H= Change in enthalpy S = Change in entropy T = Temperature in Kelvin

�All spontaneous reactions release free energy. � G for all spontaneous processes is negative. �All nonspontaneous reactions require work to make them happen. �Therefore, the G for nonspontaneous reactions is

�You can calculate G if you know H and S. �However, if H and S are not known, you can still calculate G.

Equation: G= o f G (products) - o f G (reactants)

�Using G to calculate whether a reaction is spontaneous or not only works if the reactants and products are in their standard states. �A reaction that is nonspontaneous under one set of conditions may be spontaneous under another set.

CHEMICAL KINETICS

�I can explain the term rate. � I can define reaction rates. � I can explain a reaction mechanism. � I can use a rate law to determine what would happen to the reaction rate when concentrations are changed.

�The speed at which chemical reactions occur depends on external conditions. �It’s important to know how fast chemical reactions occur and to understand factors that control speed.

�Knowing the speed at which food spoils, metal rusts, and dental fillings set is important. �Chemical kinetics is the chemistry concerned with the speed of reactions. �Rates measure change over time.

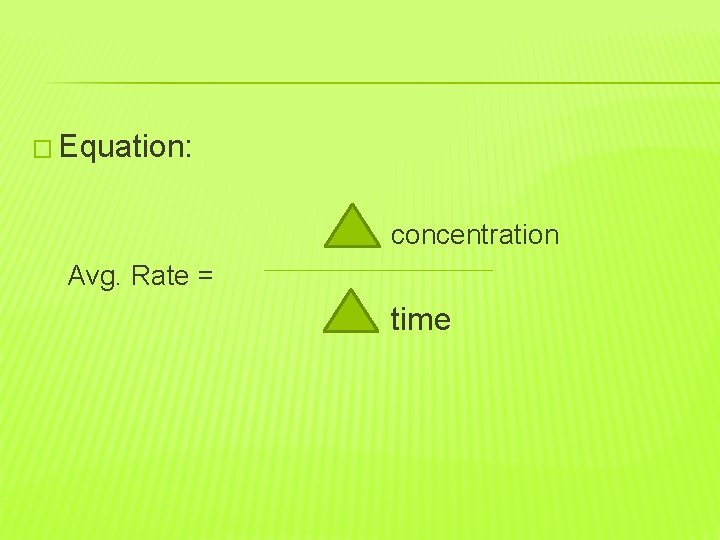
�Chemical changes in a chemical reaction do not occur all at once. �The reaction rate is the rate at which reactants disappear and products appear. �It is the change in concentration of reactants and products in a certain amount of time.

� Equation: concentration Avg. Rate = time

�The Greek letter delta means change in. �Reaction rates change throughout a chemical reaction so it is important to get an average.

REACTION MECHANISMS �During chemical reactions, the rearrangement of atoms is often complicated. �Chemical reactions do not necessarily occur in one step. �Usually chemical reactions occur in several steps made of simple reactions.

�A reaction mechanism is a series of steps that lead from reactants to products. �Detailed reaction mechanisms describe the order in which bonds break and atoms rearrange during a reaction.

�The simple steps in a reaction mechanism are called elementary steps. �The product of the first step becomes the reactant of the second step.

�An intermediate product is a substance that is produces in one step, but used in a later step. �Determining reaction mechanisms is difficult and time consuming and they are determined by experimentation.

RATE LAWS �A rate law is an equation that can be used to calculate the reaction rate for any given concentration of reactants.
![Equation rate kAxBy A and B are molar concentrations K is a rate �Equation: rate = k[A]x[B]y �[A] and [B] are molar concentrations. �K is a rate](https://slidetodoc.com/presentation_image_h/10c07f8e694b11d4cba4202dd272a26a/image-73.jpg)
�Equation: rate = k[A]x[B]y �[A] and [B] are molar concentrations. �K is a rate constant.

�K has a fixed value for a reaction at a particular temperature and is determined by experiment. �X and y are also determined by experiment. �Not all reactants appear in the

�If changing the concentration of a particular reactant does not change the rate, the reactant does not appear in the law.
![Example NO 2g COg NOg CO 2g Rate NO 22 �Example: NO 2(g) + CO(g) NO(g) + CO 2(g) Rate = [NO 2]2](https://slidetodoc.com/presentation_image_h/10c07f8e694b11d4cba4202dd272a26a/image-76.jpg)
�Example: NO 2(g) + CO(g) NO(g) + CO 2(g) Rate = [NO 2]2

�CO is not included in the rate law because it does not change the rate. �What would happen to the reaction rate if the concentration increased 5 times?

�What would happen if the concentration went from 4. 0 M to 3. 0 M? Hint: Take the final concentration and divide by the starting concentration, and then put that number in the rate law.