Equilibrium and Le Chateliers Principle CA Standards Students

Equilibrium and Le Chatelier’s Principle

CA Standards Students know how to use Le. Chatelier’s principle to predict the effect of changes in concentration, temperature, and pressure. Students know equilibrium is established when forward and reverse reaction rates are equal.

Chemical Equilibrium Reversible Reactions: A chemical reaction in which the products can react to re-form the reactants Chemical Equilibrium: When the rate of the forward reaction equals the rate of the reverse reaction and the concentration of products and reactants remains unchanged 2 Hg. O(s) 2 Hg(l) + O 2(g) Arrows going both directions ( ) indicates equilibrium in a chemical equation

Chemical Equilibrium A. The state where the concentrations of all reactants and products remain constantwith time.
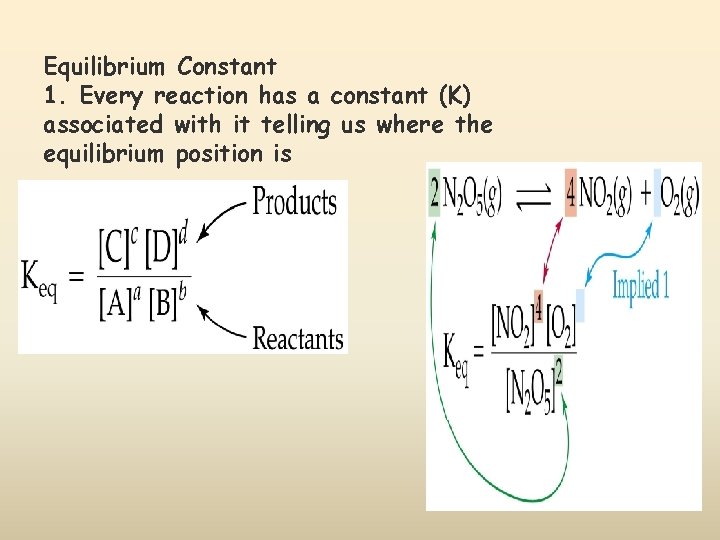
Equilibrium Constant 1. Every reaction has a constant (K) associated with it telling us where the equilibrium position is

2. If we know the concentrations, we can find Keq from its equation 3. K is written without units. 4. K > 1 tells us the equilibrium lies to the right. 5. K < 1 tells us the equilibrium lies to the left. 6. K=1 tells us the equilibrium is exactly in the middle with the concentration of the reactants=concentration of the products.

Le. Chatelier’s Principle When a system at equilibrium is placed under stress, the system will undergo a change in such a way as to relieve that stress. Henry Le Chatelier

Le Chatelier Translated: When you take something away from a system at equilibrium, the system shifts in such a way as to replace what you’ve taken away. When you add something to a system at equilibrium, the system shifts in such a way as to use up what you’ve added.

Le. Chatelier Example #1 A closed container of ice and water at equilibrium. The temperature is raised. Ice + Energy Water The equilibrium of the system shifts to right to use up the added energy. the _______

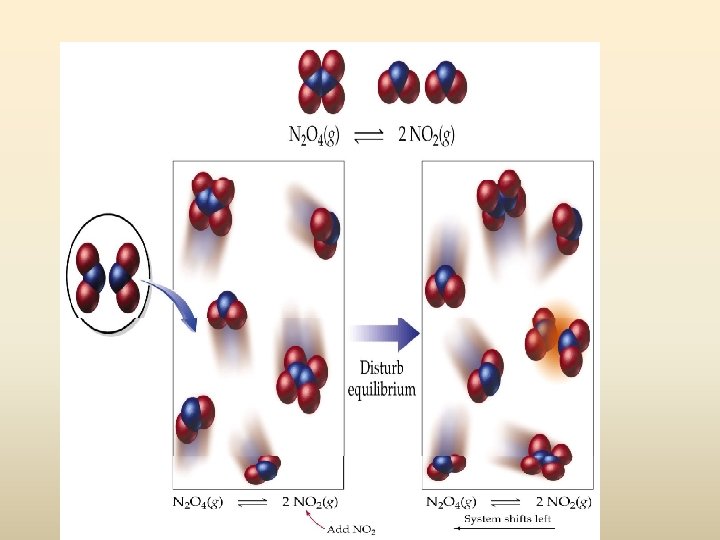
Le. Chatelier Example #2 A closed container of N 2 O 4 and NO 2 at equilibrium. NO 2 is added to the container. N 2 O 4 (g) + Energy 2 NO 2 (g) The equilibrium of the system shifts to left the _______ to use up the added NO 2.

Le. Chatelier Example #3 A closed container of water and its vapor at equilibrium. Vapor is removed from the system. water + Energy vapor The equilibrium of the system shifts to right to replace the vapor. the _______

The Effect of a Change in Pressure 1. Ways to change pressure a. Add or remove a gaseous reactant or product. b. Change the volume of the container. ◊ When the volume of the container holding a gaseous system is reduced, the system responds by reducing its own volume. This is done by decreasing the total number of gaseous molecules in the system ◊ When the container volume is increased, the system will shift so as to increase its volume
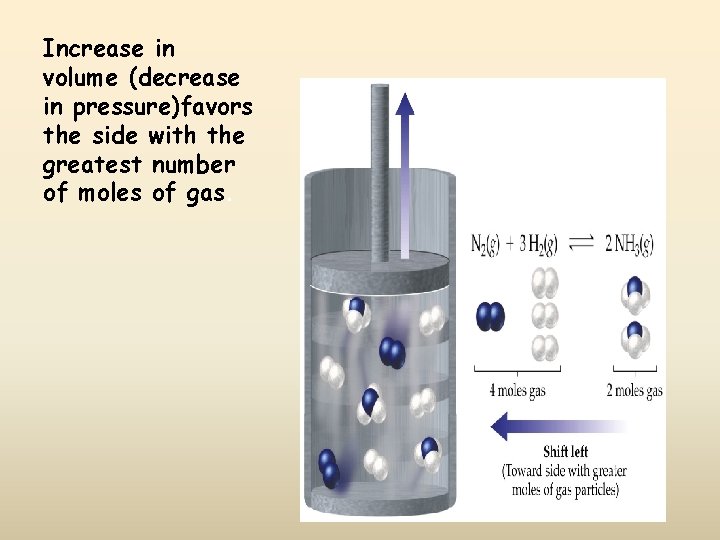
Increase in volume (decrease in pressure)favors the side with the greatest number of moles of gas.

Decrease in volume (increase in pressure) favors the side with the smallest number of moles of gas.

Le. Chatelier Example #4 A closed container of N 2 O 4 and NO 2 at equilibrium. The pressure is increased. N 2 O 4 (g) + Energy 2 NO 2 (g) The equilibrium of the system shifts to left the _______ to lower the pressure, because there are fewer moles of gas on that side of the equation.
- Slides: 16