Properties of Matter n n n Propertiescharacteristics Physical





















- Slides: 23

Properties of Matter n n n Properties=characteristics Physical properties = can be measured/observed w/o changing the identity of the substance Chemical Properties = substance’s ability to undergo change into different substances

Properties of Matter n n Extensive properties –depend on the amount of matter that is present --volume, mass, amount of energy, length Intensive properties – do NOT depend on the amount of matter present -- color, boiling/melting points, density

Changes in Matter --Physical Changes n n Physical Changes – occur without altering the composition of the substance Examples: crumpling, bending, crushing, splitting, changes in state (boiling, melting, condensing, freezing) The temperature at which substances undergo phase changes are important physical properties – can be used for identification purposes. Phase change-change of state

Changes in Matter – Chemical Changes n n n Chemical changes – changes that involve a substance changing into one or more new substances Examples – burning, fermenting, digestion, rusting, rotting, oxidizing Substances that react= reactants Substances that are formed = products Use arrow (yields) to show direction.

Chemical Reaction Example n n Carbon plus oxygen yields (forms) carbon dioxide C + O 2 CO 2 Whenever chemical & physical changes occur, energy is always involved. Energy can take several forms.

Evidences of Chemical Changes n n Always produces a change in properties. The product(s) have different properties from the reactants. Five things to look for: n n n Evolution of a gas Formation of a precipitate (solid falls out of a liquid solution) Color changes – careful! Giving off heat and/or light Change in odor – careful!

Classifications of Matter n Mixture – combination of 2 or more kinds of matter in which each retains its individual identity & properties n Examples: salt & water, sand & water, Cracker Jacks (peanuts & popcorn), Chex Mix, Kool-Aid

Types of Mixtures n n Heterogeneous Mixtures – one where the substances do not blend smoothly throughout, so the individual substances remain distinct Has distinct phases (a phase is a region where properties are different, such as ice in a glass of water. Ice is one phase, the water another phase). n Examples: pizza, chocolate chip cookies, sand & water, nuts & bolts

Types of Mixtures n n Homogeneous Mixtures – constant composition throughout – has a single phase Also called Solutions n n Solutions can be in any form, not just liquids Examples: a solid-solid solution is brass n n Many solid-solid solutions are called ALLOYS (homogeneous mixture of metals or metal & a non-metal where the metal is the largest part) A gas-liquid solution is the carbon dioxide in a soft drink A gas-gas solution is AIR A liquid-gas solution is water droplets in air

Solutions… n n Solvent – the substance present in the greatest quantity (does the dissolving) Solute – the substance(s) present in the least quantity (what is being dissolved)

Separating Mixtures n n n Heterogeneous Mixtures are easily separated. Some can be done by hand, simply by picking out the different phases. Others can be separated by FILTRATION Filtration uses a porous barrier to separate a solid from a liquid, like a coffee filter. In chemistry, we use a filter paper folded into a funnel.

Separating Homogeneous Mixtures n Distillation – separation by a difference in boiling points. Mixture is heated and the substance with the lowest boiling point will vaporize and then condense back into a liquid. n n Crystallization – formation of a solid from a liquid substance n n Example – getting gasoline from crude oil Example – rock candy Chromatography – separates by different travel times of different substances across the surface of another material.

Pure Substances n n n A pure substance has a fixed composition Every sample of a given pure substance has the same exact properties & exactly the same composition. Elements & Compounds are pure substances Element – a pure substance that cannot be separated into simpler substances by either physical or chemical means Examples: carbon, oxygen, iron

Compounds n n Compound – a combination of 2 or more different elements that are combined chemically Approxmately 10 MILLION known compounds, and more added almost daily n Examples: water, table salt, table sugar, aspirin

Compounds n n Compounds can be broken down into simpler forms by chemical means Properties of compounds are different of their component elements.

Elements and Compounds

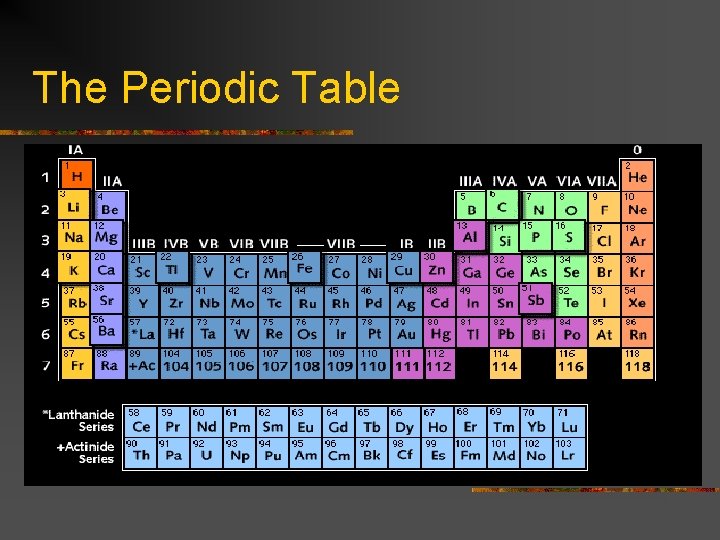
Introduction to the Periodic Table n n Each square shows the element’s: Symbol (C, He, U, Ca) Atomic number (no decimals) Atomic mass (decimals)

The Periodic Table

Periodic Table arrangement n n Vertical columns =“Groups” or “families” Horizontal rows = periods

Types of Elements n n n Metals 1. most are metals 2. on left “side” of table 3. conducts heat, electricity well 4. malleable, ductile, luster (shine) 5. Some soft, some hard

Types of Elements n n Non-metals 1. on right side of table (few elements) 2. solids, liquids, gases 3. do not conduct heat/electricity well

Types of Elements n n Metalloids 1. Found along the “stairstep” of the periodic table 2. Only 8 of them – B, Si, Ge, As, Se, Sb, Te, At 3. Have characteristics of both metals & nonmetals

Types of Elements n n Noble Gases 1. All gases 2. In last group of the table 3. Do not form compounds readily (unreactive)