PROPERTIES OF MATTER Chapter Twelve Properties of Matter





























































- Slides: 70

PROPERTIES OF MATTER

Chapter Twelve: Properties of Matter Ø 12. 1 Properties of Solids Ø 12. 2 Properties of Fluids Ø 12. 3 Buoyancy

Chapter 12. 1 Learning Goals ØDistinguish chemical properties from physical properties of matter. ØIdentify differences between crystalline and amorphous solids. ØExplain how the arrangement of atoms and molecules in solids determines their properties.

Investigation 12 A Mystery Material ØKey Question: How do solids and liquids differ?

12. 1 Properties of Solids Ø Different kinds of matter have different characteristics. Ø Characteristics that can you observe directly are called physical properties. Ø Physical properties include color, texture, density, brittleness, and state (solid, liquid, or gas). Ex. Iron is solid at room temp.

12. 1 Properties of Solids Ø A physical change is any change in the size, shape, or phase of matter in which the identity of a substance does not change. Ø For example, when water is frozen, it changes from a liquid to a solid.

12. 1 Properties of Solids Ø Properties that can only be observed when one substance changes into a different substance are called chemical properties. Ø Any change that transforms one substance into a different substance is called a chemical change. Iron. Ex. reacts with oxygen If you leave a to form iron oxide. nail outside, it rusts.

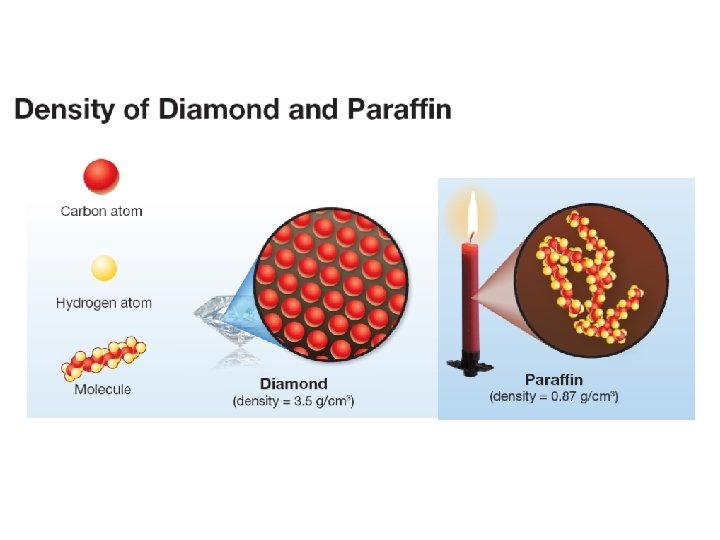
12. 1 Properties of Solids The density of a solid material depends on two things: 1. the individual mass of each atom or molecule, Carbon atoms in diamond are packed very tightly. 2. how closely the atoms or molecules are packed together.

12. 1 Properties of Solids Ø Paraffin wax is also mostly carbon, but its density is only 0. 87 g/cm 3. Ø Paraffin’s carbon atoms are mixed with hydrogen atoms in long molecules that take up more space. The density of paraffin is low compared to diamond.

12. 1 Properties of Solids The atoms or molecules in a solid are arranged in two ways. 1. If the particles are arranged in an orderly, repeating pattern, the solid is crystalline. 2. If the particles are arranged in a random way, the solid is amorphous.


12. 1 Properties of Solids Ø Examples of crystalline solids include salts, minerals, and metals.

12. 1 Properties of Solids Ø Metals don’t look like “crystals” because solid metal is made from very tiny crystals fused together in a jumble of different orientations.

12. 1 Properties of Solids Ø The atoms or molecules in amorphous solids are randomly arranged. Ø Examples of amorphous solids include rubber, wax, and glass.

12. 1 Mechanical properties Ø“Strength” describes the ability of a solid object to maintain its shape even when force is applied.

12. 1 Mechanical properties ØTensile strength is a measure of how much stress a material can withstand before breaking.

12. 1 Mechanical properties ØHardness measures a solid’s resistance to scratching. How might you compare the hardness of these two metals?

12. 1 Mechanical properties ØElasticity describes a solid’s ability to be stretched and then return to its original size. ØBrittleness is defined as the tendency of a solid to crack or break before stretching very much.

12. 1 Mechanical properties ØA ductile material can be bent a relatively large amount without breaking. ØThe ductility of many metals, like copper, allow then to be drawn into wire.

12. 1 Mechanical properties ØMalleability measures a solid’s ability to be pounded into thin sheets. ØAluminum is a highly malleable metal.

12. 1 Mechanical properties ØAlmost all solid materials expand as the temperature increases. ØThe increased vibration makes each particle take up a little more space, causing thermal expansion. Sidewalks and bridges have grooves that allow for thermal expansion.

Chapter Twelve: Properties of Matter Ø 12. 1 Properties of Solids Ø 12. 2 Properties of Fluids Ø 12. 3 Buoyancy

Chapter 12. 2 Learning Goals ØExplain how pressure is created in fluids. ØDiscuss differences between the density of solids and fluids. ØApply Bernoulli’s principle to explain how energy is conserved in fluids.

12. 2 Properties of Fluids Ø A fluid is defined as any matter that flows when force is applied. Ø Liquids like water or silver are kinds of fluid.

12. 2 Pressure ØA force applied to a fluid creates pressure. ØPressure acts in all directions, not just the direction of the applied force.

12. 2 Forces in fluids ØForces in fluids are more complicated than forces in solids because fluids can change shape.

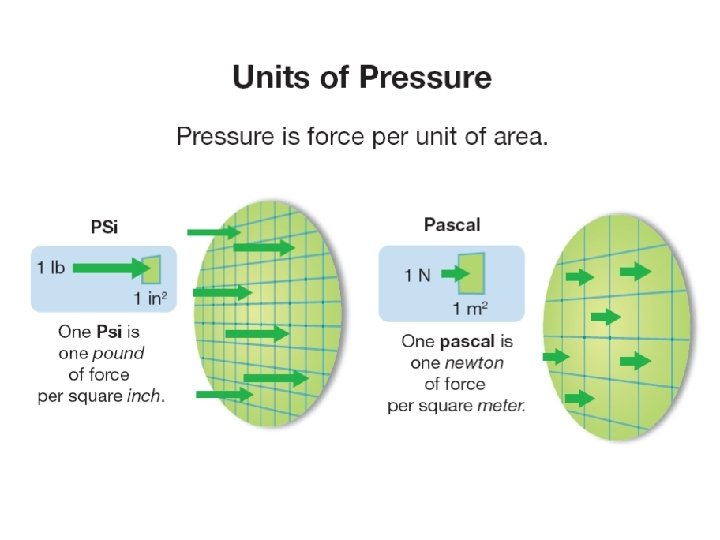
12. 2 Units of pressure ØThe units of pressure are force divided by area. ØOne psi is one pound per square inch.

12. 2 Units of pressure ØThe S. I. unit of force is the pascal. ØOne pascal (unit of force) is one newton of force per square meter of area (N/m 2).


12. 2 Pressure ØIf your car tires are inflated to 35 pounds per square inch (35 psi), then a force of 35 pounds acts on every square inch of area inside the tire. What might happen if you over-inflate a tire?

12. 2 Pressure ØOn the microscopic level, pressure comes from collisions between atoms. ØEvery surface can experience a force from the constant impact of trillions of atoms. ØThis force is what we measure as pressure.

12. 2 Pressure ØIn a car engine high pressure is created by an exploding gasoline-air mixture.

12. 2 Energy conservation and Bernoulli’s Principle ØStreamlines are imaginary lines drawn to show the flow of fluid. ØBernoulli’s principle tells us that the energy of any sample of fluid moving along a streamline is constant.

12. 2 Bernoulli’s Principle ØBernoulli’s principle says the three variables of height, pressure, and speed are related by energy conservation.

12. 2 Three Variables and Bernoulli’s Principle ØIf one variable increases along a streamline, at least one of the other two must decrease. ØFor example, if speed goes up, pressure goes down.

12. 2 The air foil ØOne of the most important Ø The difference in applications of Bernoulli’s principle is is thewhat airfoilcreates shape pressure of wings on a plane. the lift force that ØWhen a plane is plane moving, supports the inthe pressure on the top surface air. of the wings is lower than the pressure beneath the wings.

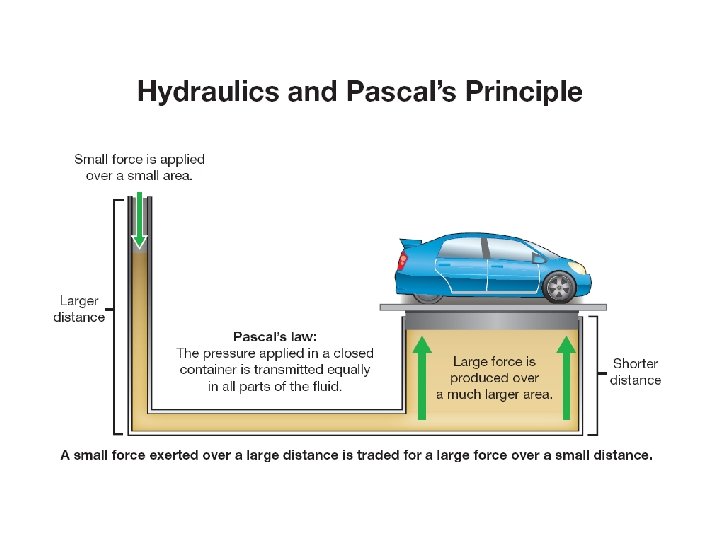
12. 2 Hydraulics and Pascal’s Principle ØHydraulic lifts and other hydraulic devices use pressure to multiply forces and do work. ØThe word hydraulic refers to anything that is operated by a fluid under pressure. ØHydraulic devices operate on the basis of Pascal’s principle, named after Blaise Pascal.

12. 2 Hydraulics and Pascal’s Principle ØPascal’s principle states that the pressure applied to an incompressible fluid in a closed container is transmitted equally in all parts of the fluid. ØAn incompressible fluid does not decrease in volume when pressure is increased.

12. 2 Hydraulics and Pascal’s Principle ØA small force exerted over a large distance is traded for a large force over a small distance.


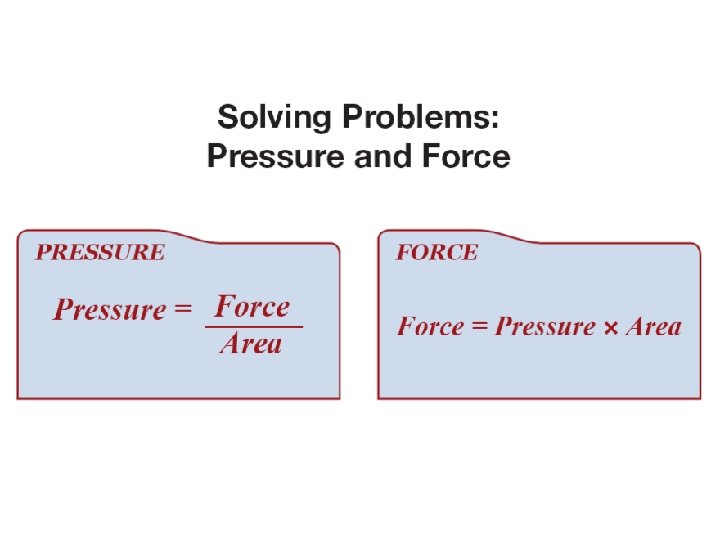
12. 2 Pressure ØPressure is force divided by area.

12. 2 Force ØYou can calculate the force exerted if you know the pressure and area.


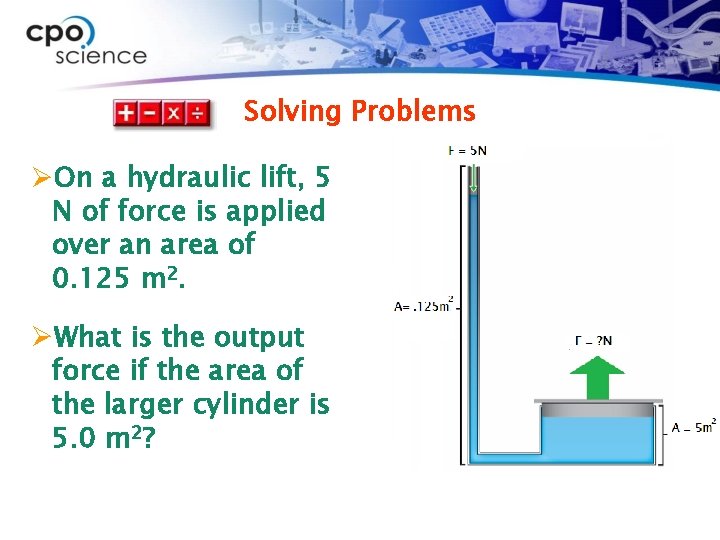
Solving Problems ØOn a hydraulic lift, 5 N of force is applied over an area of 0. 125 m 2. ØWhat is the output force if the area of the larger cylinder is 5. 0 m 2?

Solving Problems 1. Looking for: Ø …output force 2. Given Ø …input force = 5 N; input area =. 125 m 2 ; output area = 5 m 2 3. Relationships: Ø Pressure = Force = P x A Area

Solving Problems 4. Solution Ø Solve for pressure using input force. Ø Pressure = 5 N. 125 m 2 = 40 N/m 2 Ø Use Pascal’s law principle and use equivalent pressure to solve for output force. Ø Force = 40 N x 5 m 2 = m 2 200 N

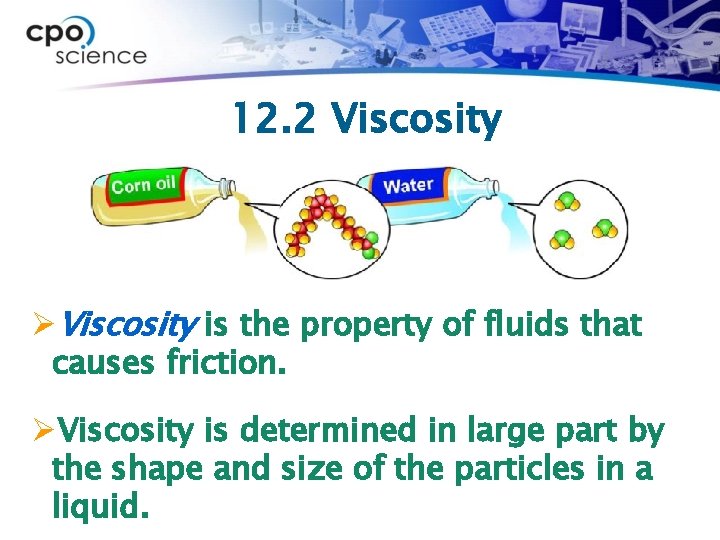
12. 2 Viscosity ØViscosity is the property of fluids that causes friction. ØViscosity is determined in large part by the shape and size of the particles in a liquid.

12. 2 Viscosity and temperature ØAs the temperature of a liquid increases, the viscosity of a liquid decreases. ØIncreasing the kinetic energy of the substance allows the particles to slide past one another more easily.

Investigation 12 C Density of Fluids ØKey Question: What is the maximum load a boat can hold before sinking? How is the maximum load affected by the density of the water in which the boat floats?

Chapter Twelve: Properties of Matter Ø 12. 1 Properties of Solids Ø 12. 2 Properties of Fluids Ø 12. 3 Buoyancy

Chapter 12. 3 Learning Goals ØDefine buoyancy. ØExplain the relationship between density and buoyancy. ØDiscuss applications of Archimedes’ principle.

Investigation 12 B Buoyancy of Fluids ØKey Question: Can you make a clay boat?

12. 3 Buoyancy is a force ØBuoyancy is a measure of the upward force a fluid exerts on an object that is submerged. The water in the pool exerts an upward force that acts in a direction opposite to the boy’s weight.

12. 3 Volume and buoyancy ØThe strength of the buoyant force on an object in water depends on the volume of the object that is underwater. As you keep pushing downward on the ball, the buoyant force gets stronger and stronger. Which ball has more volume underwater?

12. 3 Weight and buoyancy ØWeight is a force, like any other pushing or pulling force, and is caused by Earth’s gravity. ØIt is easy to confuse mass and weight, but they are not the same. What is the rock’s weight? ØWeight is the downward force of gravity acting on mass. What is the rock’s mass?

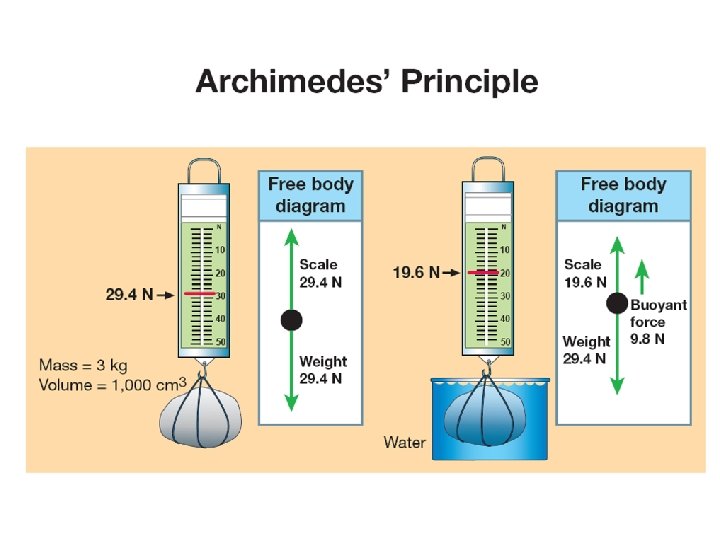
12. 3 Weight and buoyancy ØIn the third century BC, a Greek mathematician named Archimedes realized that buoyant force is equal to the weight of fluid displaced by an object. ØA simple experiment can be done to measure the buoyant force on a rock with a spring scale when it is immersed in water.


12. 3 Weight and buoyancy ØIn air the buoyant force on the rock is 29. 4 N. ØWhen the rock was submerged, the scale read 19. 6 N. ØThe difference is a force of 9. 8 N, exactly the amount of force the displaced water exerts.

12. 3 Weight and buoyancy These blocks are the same total volume. Which block has more buoyant force acting on it? Which block weighs more in air?

12. 3 Weight and buoyancy ØBuoyancy explains why some objects sink and others float. ØWhether an object sinks or floats depends on how the buoyant force compares with the weight.

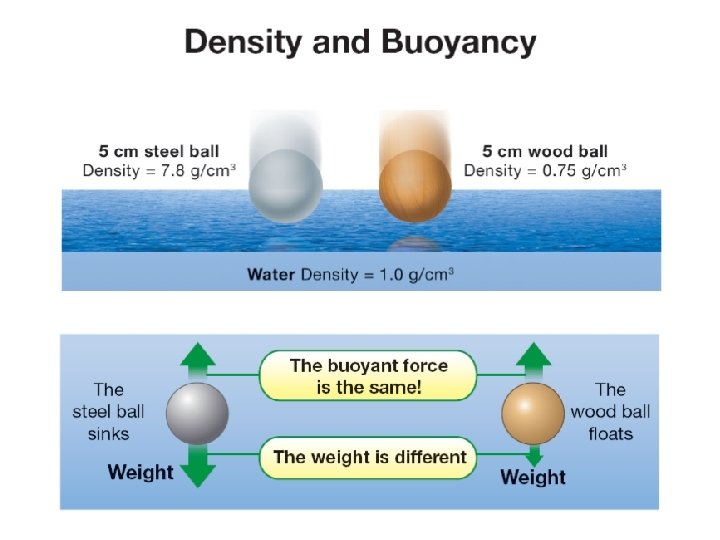
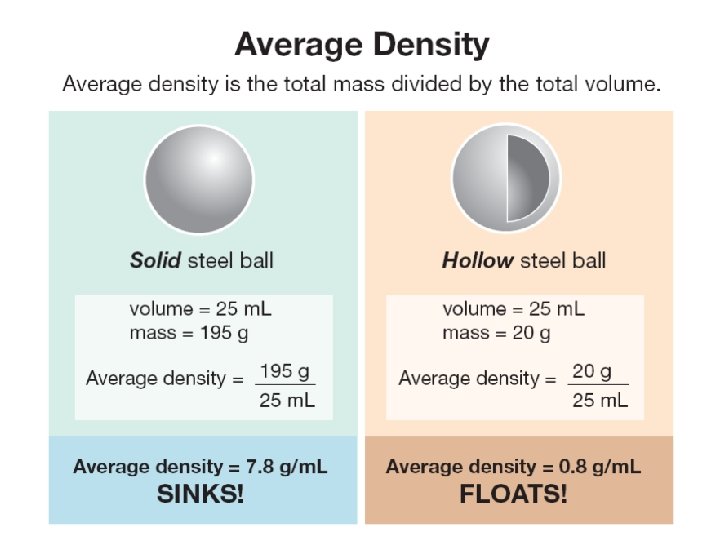
12. 3 Density and buoyancy ØIf you know an object’s density you can quickly predict whether it will sink or float. Which ball will sink in water? Which ball will float in water?

12. 3 Density and buoyancy Average density helps determine whether objects sink or float. Ø An object with an average density GREATER than the density of water will sink. Ø An object with an average density LESS than the density of water will float.

12. 3 Density and buoyancy What can you say about the average density of these blocks?

12. 3 Density and buoyancy ØØWhen they the are steel completely underwater, However, ball has more weight both have the same buoyant force sinceballs it has a higher density. because they displace the same volume of water.


12. 3 Boats and average density ØUse your understanding of average density to explain how steel boats can be made to float.

12. 3 Boats and average density ØA If you full ship havehas seen more a loaded mass cargo than an ship, empty you ship. might have noticed that it sat lower in the Øwater This means a full ship must than an unloaded shipdisplace nearby. more water (sink deeper) to make the buoyant force large enough to balance the ship’s weight.


Investigation 12 C Density of Fluids ØKey Question: What is the maximum load a boat can hold before sinking? How is the maximum load affected by the density of the water in which the boat floats?

The Hull ØThere are many different types of boats, but all have one thing in common—the hull. ØThe hull is the main body of the boat. It displaces the water that provides the upward buoyant force. It also provides stability.