Pharmacotherapy of pain Types of pain 1 Acute





































- Slides: 55

Pharmacotherapy of pain

Types of pain 1 Acute pain preventive function localised has vegetative Chronic pain without preventive function sympthomatology and doesn´t lasts 3 -6 months cause severe psychological changes often associated with clearly defined beginning and severe psychological ending changes analgesics are usually effective without clearly defined in treatment beginning malignant (caused by cancer)or non-malignant

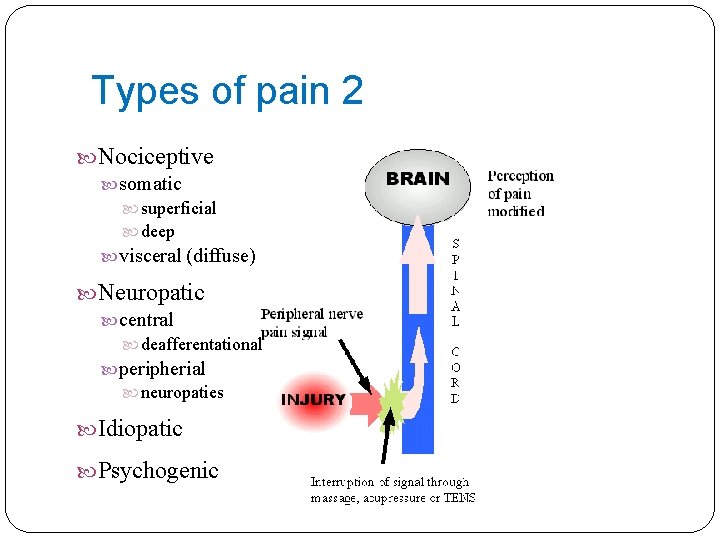
Types of pain 2 Nociceptive somatic superficial deep visceral (diffuse) Neuropatic central deafferentational peripherial neuropaties Idiopatic Psychogenic

Types of pain 2 SOMATIC: often well localised to specific dermal, subcutaneous or musculosceletal tissue VISCERAL: often poorly localized and can irradiate to somatic structures (myocardial infarction), originating in thoracic or abdominal structures NEUROPATHIC: nerve damage (compression, inflammation, diabetes), e. g. trigeminal neuralgia, postherpetic neuralgia, fibromyalgia (antidepressants, anticonvulsives)

(According to: Stahl's Essential Psychopharmacology, 4 th Edition)

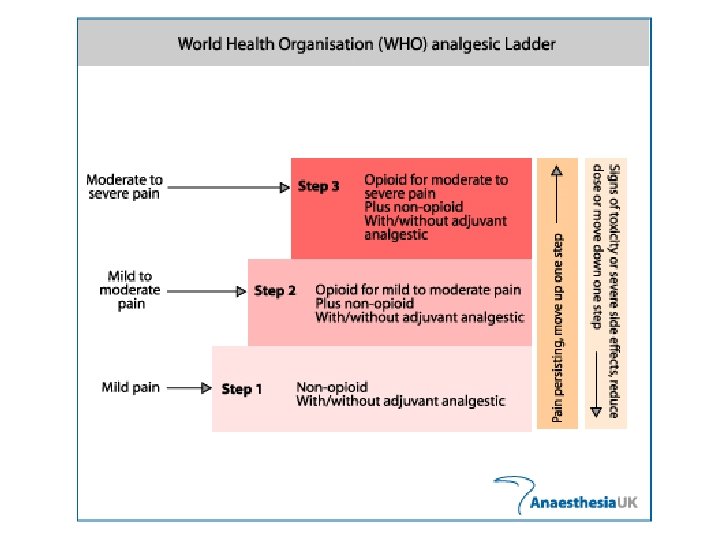
Therapy of pain Analgesics and co-analgesics Non-opioid analgesics Analgesics/antipyretics Non-steroidal anti-inflammatory drugs (NSAIDs) Opioid analgesics Natural (morphine, dihydrocodeine) Synthetic (fentanyl, buprenorphine) Co-analgesics (Antidepressants, Anxiolytics, Myorelaxants, Anticonvulsives, Neuroleptics, Glucocorticoids, Anesthetics, 2 -sympaticomimetics, H 1 -antihistamines)


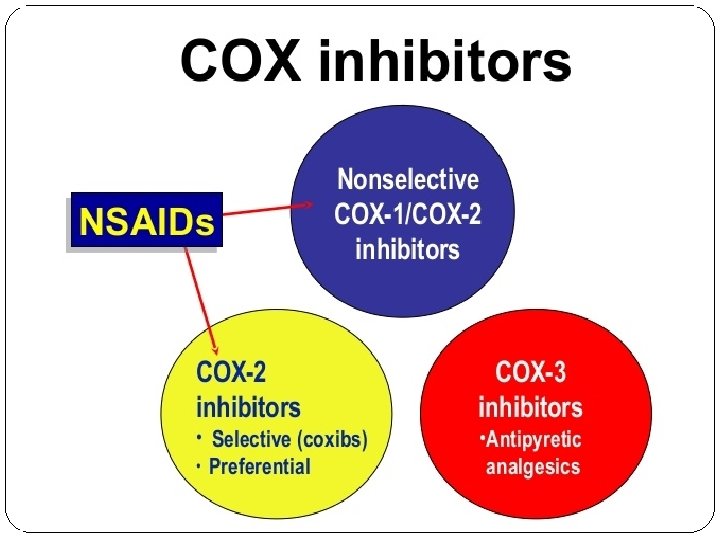
Non-opioid analgesics Analgesics/antipyretics (no anti-inflammatory) aspirin (acetylsalicylic acid- ASA) paracetamol (USA- acetaminophen) metamizole Non-steroidal anti-inflammatory drugs (NSAIDs) (used to treat inflammation in rheumatoid arthritis) Non-selective inhibitors of COX-1 and COX-2 (ibuprofen, diclofenac, ASA) Preferential inhibitors of COX-2 (nimesulide, meloxicam) Selective inhibitors of COX-2/coxibs (celecoxib, rofecoxib (dereg. ))


Analgesics/antipyretics

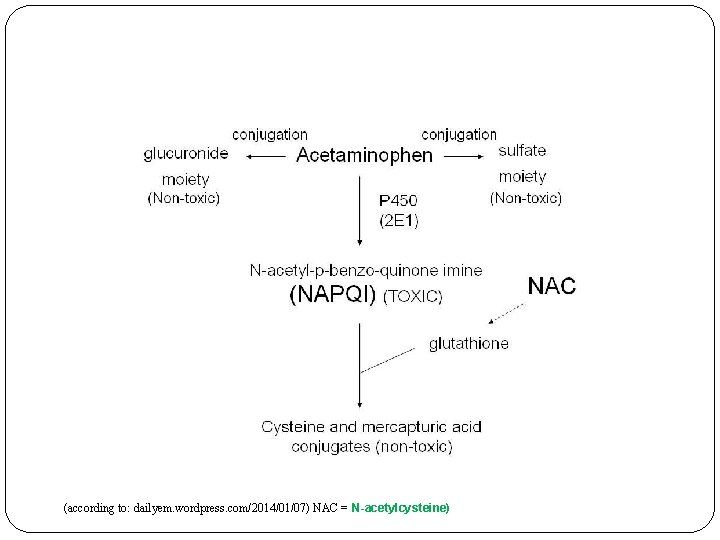
Paracetamol (Acetaminophen) Derivate of anilin Mechanism of action – unknown, possibly inhibition of COX-3 in CNS Good absorption from GIT Biological half life - 1, 5 -3 h Only a small percentage of the drug binds on plasma proteins (10%) Elimination – through the kidnies in the form of glucuronides and sulfates In doses over 5 g/day can cause HEPATAL DAMAGE In the world, approximately 200 different preparates containing paracetamol are being used Antidote: N-acetylcysteine (mucolytic drug – used in therapy of productive cough)

(according to: dailyem. wordpress. com/2014/01/07) NAC = N-acetylcysteine)

Metamizole (Dipyrone) Derivate of pyrazolone Mechanism of action: complex; inhibition of a central COX-3 and activation of the opioidergic system and cannabinoid system Has spasmolytic properties Serious adverse effect: depression of bone marrow leading to AGRANULOCYTOSIS Good absorption in the case of both peroral and rectal administration. More than 50 % binds onto plasmatic proteins; short plasmatic half-life (i. v. metamizole - 14 min); elimination mostly through kidneys

Non-steroidal anti-inflammatory drugs (NSAIDs)


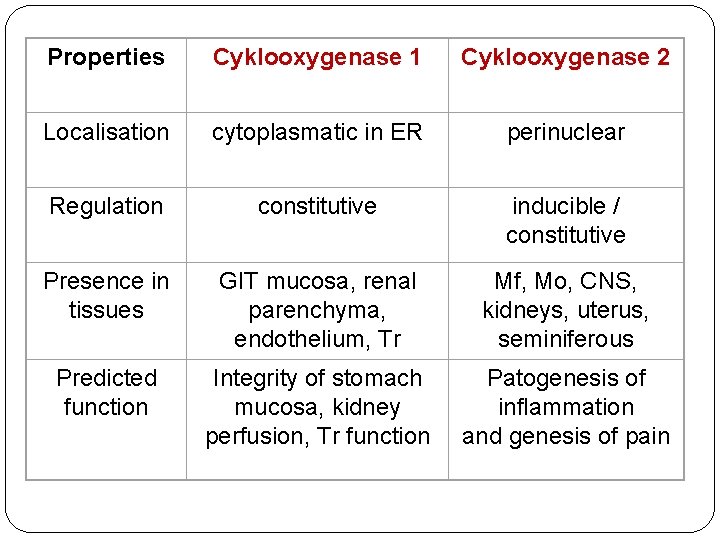
Properties Cyklooxygenase 1 Cyklooxygenase 2 Localisation cytoplasmatic in ER perinuclear Regulation constitutive inducible / constitutive Presence in tissues GIT mucosa, renal parenchyma, endothelium, Tr Mf, Mo, CNS, kidneys, uterus, seminiferous Predicted function Integrity of stomach mucosa, kidney perfusion, Tr function Patogenesis of inflammation and genesis of pain

Non-steroidal anti-inflammatory drugs Division according to COX selectivity: Selective inhibitors of COX-1: Acetylsalicylic acid (ASA)- in a dose of 30 - 300 mg/day (secondary prevention if atherosclerosis) Non-selective inhibitors of COX: Acetylsalicylic acid- in a dose of 500 mg/day or more, ibuprofen, diclofenac, ketoprofen, tiaprofen, indomethacin and many more Preferential inhibitors of COX-2: nimesulide, meloxicam etc. Selective inhibitors of COX-2: celecoxib, rofecoxib, etoricoxib, valdecoxib etc.

Acetylsalicylic acid (Aspirin) in a dose of 30 - 300 mg/day (secondary prevention if atherosclerosis) in a dose of 500 mg (pain, fever) high doses (antiinflammatory effect; high risk of gastrotoxicity) not in children: Reye´s syndrome

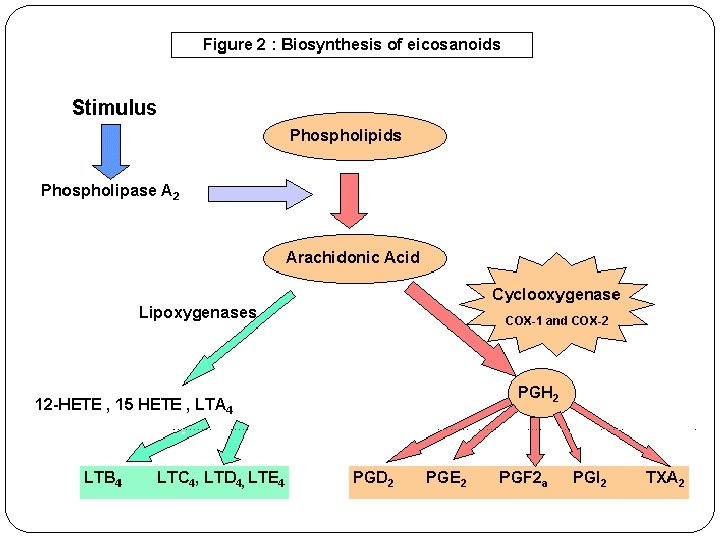
Effects of NSAIDs 1. Analgesic effect 2. Antipyretic effect 3. Antiflogistic effect (mainly higher doses) 4. Antiaggregatory effect (not every NSAID, most important is ASA because of irreversible blocade of COX-1 - be careful with combination of ASA for anti-aggregation and other NSAIDs- mostly ibuprofen) 5. Other effects- for example reduced incidence of some tumors (for example colorectal carcinoma), uricosuric effect. . .

NSAIDs – one of the most widely used drug groups→ their ADRs represent a serious medical / public health / economical problem there are big differences between various NSAIDs in the level risk of particular possible ADRs

Adverse effects of NSAIDs Ø GIT- erosions and ulcers of the gastric mucosa (also in other localisations in the GIT), nausea, vomitus, meteorism, diarrhoea, constipation. . . (mainly inhibition of COX-1) Ø kidney- reduction of glomerular filtration, retention of Na and fluids, edema, hyperkaliemia, kidney failure, interstitial nephrits. . . (inhibition of COX-1 and 2) Ø CVS- thrombotic events, increase of blood pressure, heart failure. . . (mainly COX-2 (mostly in thrombotic events)) Ø CNS- cephalea, weakness, sleep disorders, dizziness, epileptic seizures. . . Ø other- hepatotoxicity, bleeding, provocation of asthmatic attack, Ray´s syndrom, prolonged childbirth, urticaria, decreased number of white blood cells. . .

Gastrointestinal ADRs (inhibition of COX-1) most serious – ↓ production of prostaglandins in the gastric mucosa → peptic ulcer (most often in the stomach and duodenum; the mucosa can get damaged by NSAIDs also in other places in the GIT) roughly 25 % of chronic NSAID users might develop erosions and ulcers, in 2 -4 % perforation or bleeding can occure

Kidney ADRs Decreased production of prostanoids → negative effect on the perfusion of the kidneys, glomerular filtration, excretion of sodium and water and on production of renin → circulation overload, oedemas, hypertension ; hyperkalemia ; in severe cases symptoms of acute kidney failure serious complication– interstitial nephritis (immunological reasons) Incidence of kidney ADRs is approx. 18%, severe cases- roughly 1%

Cardiovascular ADRs (inhibition of COX-2) Ø Increased blood pressure- mostly in hypertensive patients treated with antihypertensives (mainly ACEIs, ARBs, beta blockers), there are big differences between various NSAIDs Ø Development/worsening of heart failure- the risk is highest during the first weeks of treatment, mainly in patients with preexisting congestive heart failure; possibly 19% of all cases of congestive heart failure could be caused (at least partially) by NSAID therapy Ø Thrombotic events- acute myocardial infarction, stroke, thromboembolic disease

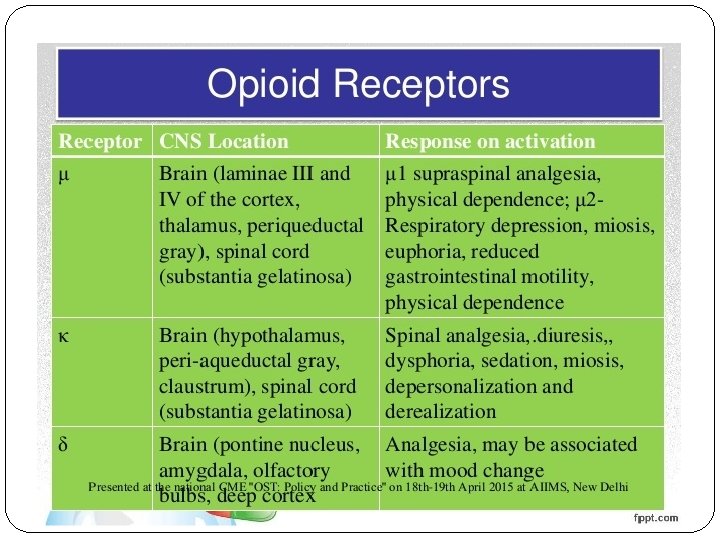
Opioid analgesics Dampen strong somatic and visceral pain – strongest analgesics Dampen algognostic (perception and localisation) and algothymic (psychical and emotional) part of pain Tolerance – results from down-regulation of opioid receptors (not to miosis and constipation) Physical dependence – a person´s continued use of drug is required for his or her well being (in therapy is used methadone, buprenorphine)

HYDROCODONE

Opioid analgesics – strong opioids NO CEILING (´ROOF´) EFFECT The Ceiling effect: when the dosage of a drug is increased, its effect increases proportionally; but after a certain dosage, any amount of increase in the dosage of the drug shows no additional effect

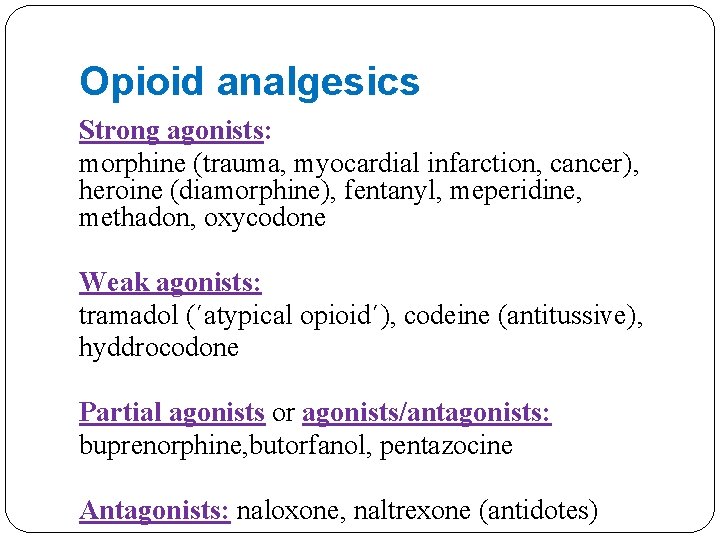
Opioid analgesics Strong agonists: morphine (trauma, myocardial infarction, cancer), heroine (diamorphine), fentanyl, meperidine, methadon, oxycodone Weak agonists: tramadol (´atypical opioid´), codeine (antitussive), hyddrocodone Partial agonists or agonists/antagonists: buprenorphine, butorfanol, pentazocine Antagonists: naloxone, naltrexone (antidotes)

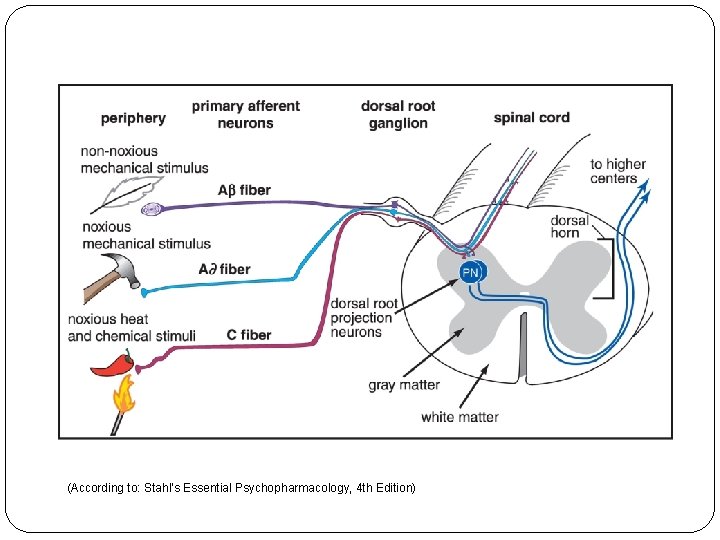
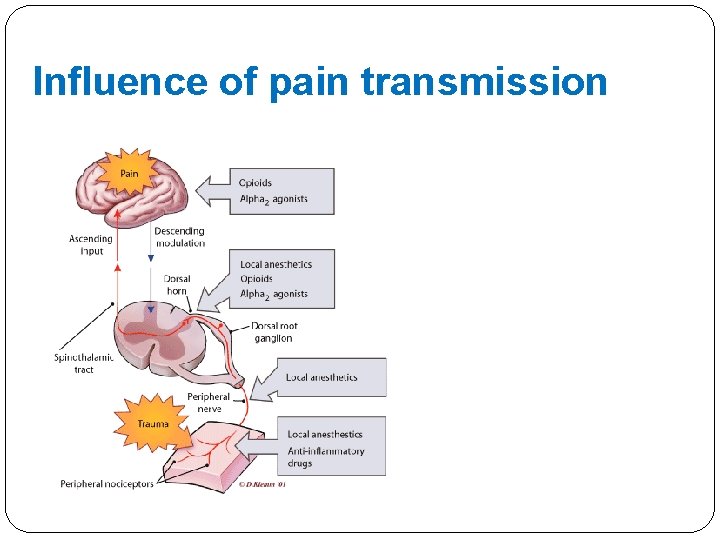
Influence of pain transmission



Pharmacological effects Depressor Stimulatig Analgesia Vomiting Depression of breathing Miosis Antitussive effect Increased tonus of Decreased secretion and smooth muscles, sphincters Induction of histamine release Euphoria motility in the GIT – constipation Sedation

Adverse effects of opioids Neuropsychiatric Sedation, clouded consciousness, euphoria, sleep disorders Cardiopulmonal Depression of breathing centre (the most feared ADR), bronchoconstriction (high doses), orthostatic hypotension, bradycardia (high doses) Gastrointestinal Nausea, vomiting, constipation, gastrointestinal or gallbladder colics Urinary system Retention of urine Endocrine Decreased levels of testosterone, problems with menstrual cycle Allergic and immunologic Pruritus, immunosupression

Morphine Prototypical opioid Isolated from sap from unriped skulls of poppy (1803) Fast resorption after p. o. application, bioavailability is only 30% (significant first pass effect) Most common adverse effects- nausea, vomiting, constipation, gallbladder and uretral spasms Depression of breathing is the most common cause of death in case of intoxication. Most common signs of intoxication – unconstiousness, bradypnoea a miosis

Fentanyl Synthetic derivate of phenylpiperidine Strong agonist of receptors 80 -100 times more potent than morphine Lipophilic character – rapid onset of action, but the effect lasts only for a short time Contraindicated in pregnancy Derivates of fentanyl- sufentanyl and alfentanyl are used in anestesiology Big advantage – availability of fentanyl in the form of TRANSDERMAL PATCHES – „Durogesic“ The second most widely used opioid

Tramadol Atypical opioid (dual-action opioid: agonist at µ receptors + inhibits the neuronal reuptake of serotonin and norepinephrine) For moderate pain High bioavailability is an advantage In therapeutic doses lacks most of the adverse effects of opioids The most common adverse effects are sweating, nausea and dry mouth Suitable for treatment of mild to severe pain in adults and children Available in different drug forms. It is cheap

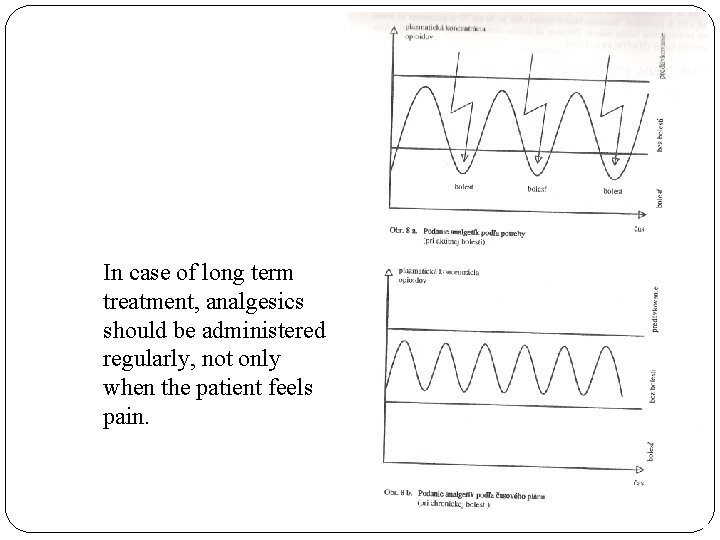
In case of long term treatment, analgesics should be administered regularly, not only when the patient feels pain.

Muscle Relaxants

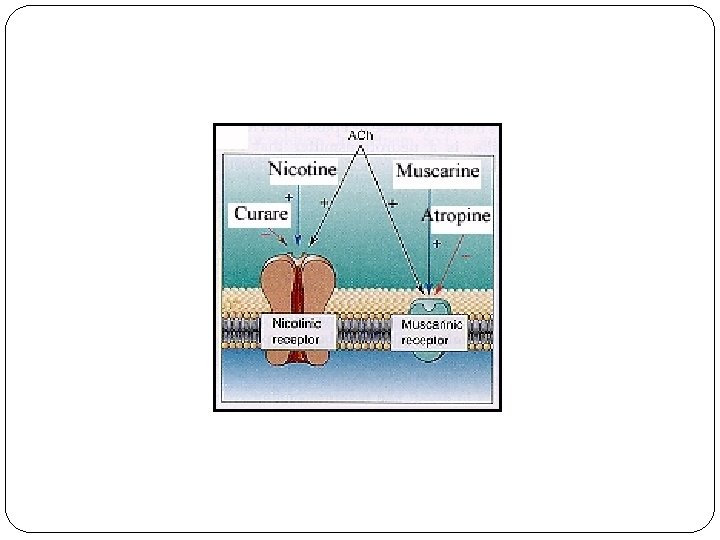
Cholinergic Transmission CNS neuromuscular junction ganglia of the autonomic nervous system (sympathetic and parasympathetic) postganglionic parasympathetic neurons

Receptors for Ach muskarinic: M 1 = CNS, ganglions, stomach M 2 = heart M 3 = glands, smooth muscles M 4, 5 = CNS nicotinic: NM (muscular) = neuromuscular junction NN (neuronal) = veg. ganglions

Degradation of Ach = acetate + choline Ach esterase – synapsis Butyrylcholinesterase – plasma, tissues (atypic form of BCh esterase = ↓ activity !!!)

Muscle Relaxants Peripheral – acting on the neuromuscular plate a. non-depolarising b. depolarising Centrally acting

kurare - tubokurarine


Muscle Relaxants = relaxation of skeletal muscles structure similar to Ach, peripheral + central 1. Peripheral: a. nondepolarising • competitive blockade of nicotinic (N) receptor on neuromuscular junction • fast elimination (kidneys, ↓ liver) • effect starts quickly, lasts about 1 hour after administration (i. v. injection, contin. infusion) • always OT intubation !!!

Muscle Relaxants – peripheral, nondepolarising muscle relaxation: mimic, chewing, oculomotory muscles, than head, neck, limbs, belly, at last diaphragma and intercostal muscles Ind. : anestesiology (abdominal operations) ADR: hypotension, tachycardia, release of histamine interactions: potentiation of myorelaxation after inhalatory anesthetics and aminoglycosides antidote: inhibitors of Ach esterase (neostigmine) + atropine

Muscle Relaxants – peripheral, nondepolarising examples: atracurium, pancuronium, vecuronium, pipecuronium = less ADR as original d-tubocurarine (release of histamine, blockade of N recept. of veget. ganglions) atracurium, cis-atracurium: spontaneous nonenzymat. cleavage through Hoffmann´s elimination (independently from kidney and liver function) pancuronium: action till 60 min.

Muscle Relaxants – peripheral, depolarising b. depolarising sukccinylcholine (suxamethonium) = depolarisation of neuromuscular junction, i. v. administration • effect: fast and short (cca 5 min. ) • on the beginning fasciculations and spasms – always general anesthesia before administration !!! • degradation = butyrylcholinesterase • genet. defect of Bch esterase = long lasting paralysis of muscles and breathing • no antidote – assisted breathing !!!

Muscle Relaxants – peripheral, depolarising Ind. : short lasting manipulations (OT intubation, reposition of fractures and luxations, electroconvulsive therapy in psychiatry) ADR: fasciculations, spasms, hypotension, bradycardia, ↑ intraocular pressure, hyperkalemia

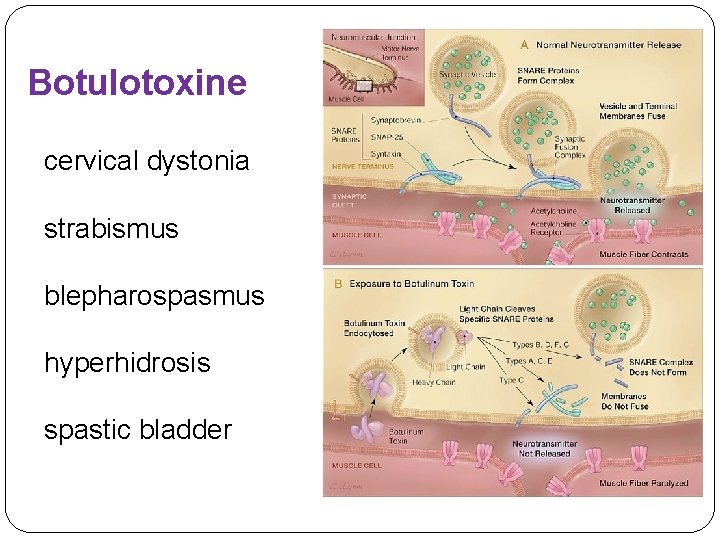
Botulotoxine cervical dystonia strabismus blepharospasmus hyperhidrosis spastic bladder


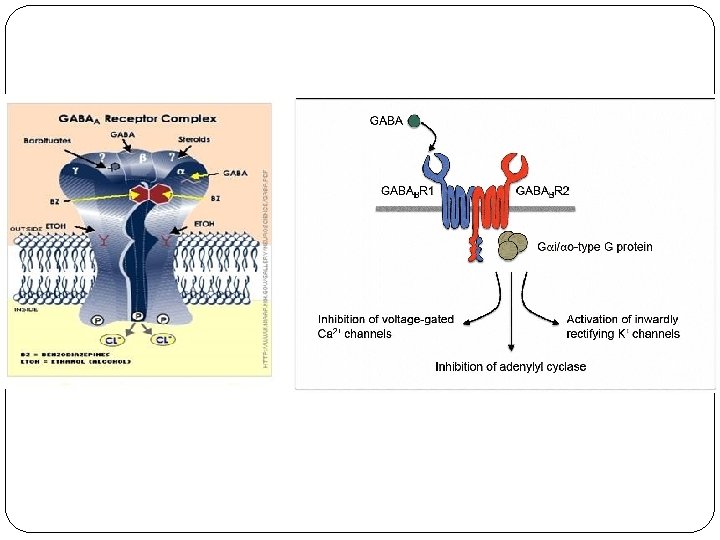
Muscle Relaxants – centrally acting 2. Centrally acting: • act on the level of CNS + inhibition of polysynapt. spinal reflexes, through GABA (baclofen) • Ind. : neurology, rheumatology (painful spasms of skeletal muscles), only symptomatic therapy !!! • ADR: dose-dependent = sedation, fatigue, dizziness – be careful at older pac. !!! • interactions: alcohol, benzodiazepines = ↑↑ ADR

Muscle Relaxants – centrally acting examples: • mefenoxalon, carisoprodol, tolperizon (the smallest suppressing effect) • baclofen (acts through GABA neurotransmission, at abrupt discontinuation of treatment risk of spasms as rebound phenomenon) • guaifenezin (also anxiolytic and expectorans) • benzodiazepines (risk of tollerance and addiction)


Dantrolen inhibits Ca 2+ ion release from sarkoplasmatic reticulum = suppression of muscle contraction, ↓ heat production Ind. : • malignant hyperthermia (rare serious complic. of general anesthesia, more after halothane and suxamethonium) • malignant neurolept. syndrome (adjuvant treatment) • repeated i. v. injection