Periods Energy Levels n Each row in the














- Slides: 20

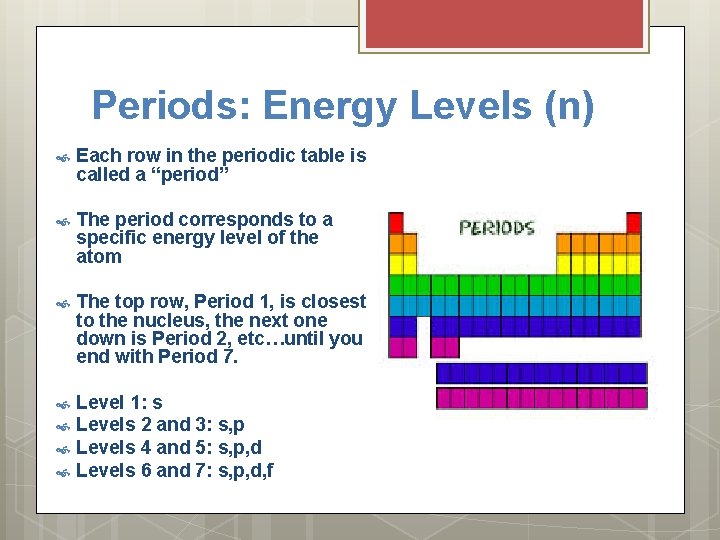
Periods: Energy Levels (n) Each row in the periodic table is called a “period” The period corresponds to a specific energy level of the atom The top row, Period 1, is closest to the nucleus, the next one down is Period 2, etc…until you end with Period 7. Level 1: s Levels 2 and 3: s, p Levels 4 and 5: s, p, d Levels 6 and 7: s, p, d, f

Groups: Valence Electrons Each column in the Periodic Table is called a “group” Each element in a group has the same number of electrons in their outer energy level (the valence level). The electrons in the outer shell are called “Valence Electrons” Red: Group 1 Orange: Group 2 Yellow: Group 13 Green: Group 1 Sky Blue: Group 15 Baby Blue: Group 16 Dark Blue: Group 17 Purple: Group 18

Valence Electrons Valence electrons are the electrons in the highest occupied energy level of the atom. Valence electrons are the only electrons generally involved in bond formation (which we will talk about in the next unit!)

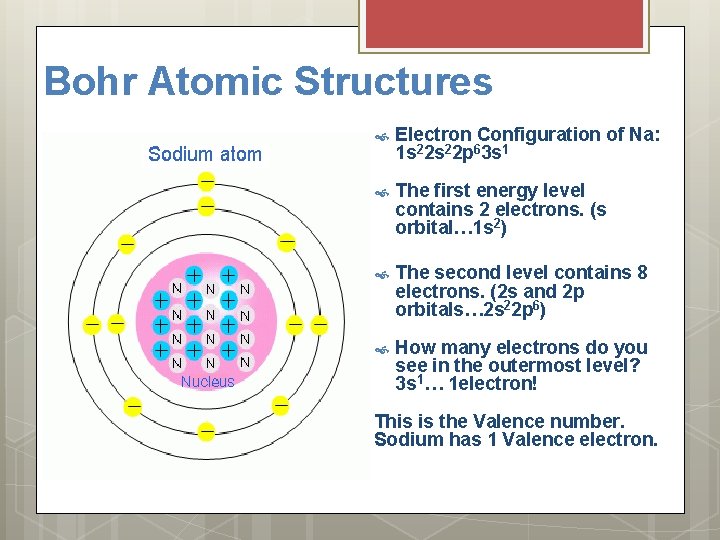
Bohr Atomic Structures Electron Configuration of Na: 1 s 22 p 63 s 1 The first energy level contains 2 electrons. (s orbital… 1 s 2) The second level contains 8 electrons. (2 s and 2 p orbitals… 2 s 22 p 6) How many electrons do you see in the outermost level? 3 s 1… 1 electron! This is the Valence number. Sodium has 1 Valence electron.

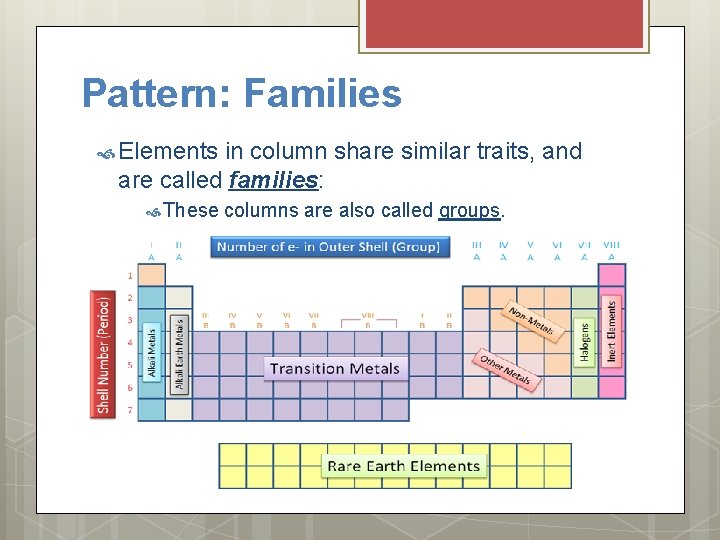
Pattern: Families Elements in column share similar traits, and are called families: These columns are also called groups.

The Alkali Metals 1 valence electron Highly Form Do reactive with water ionic compounds not occur in nature as pure elements (always in compounds)

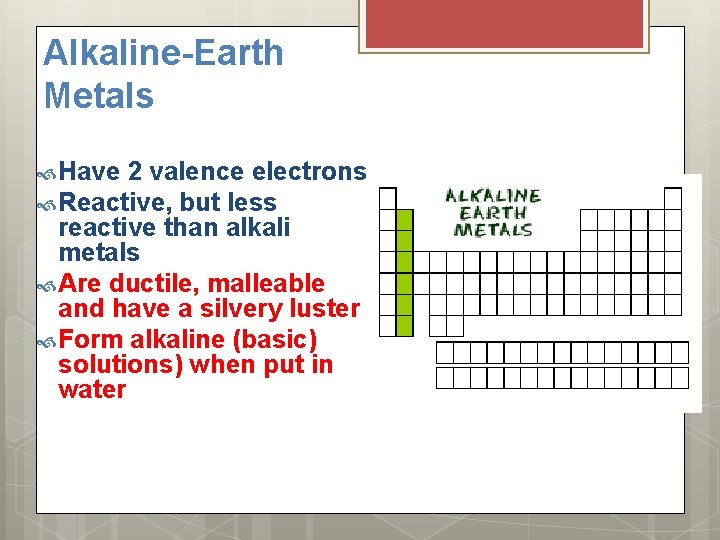
Alkaline-Earth Metals Have 2 valence electrons Reactive, but less reactive than alkali metals Are ductile, malleable and have a silvery luster Form alkaline (basic) solutions) when put in water

Transition metals… and inner transition metals Are less reactive than groups 1 and 2. Tend not to react in water. Are malleable and ductile, but still harder than group 1 & 2. Tend to be solids at room temperature. Are good conductors of **Inner transition electricity and heat. metals tend to be radioactive

Nonmetals Poor conductors of heat and electricity Often are found as gases or liquids, sometimes solids.

Halogens Are nonmetals highly reactive with metalsmost reactive is fluorine, least reactive is astatine Mostly exist as gases or liquids (except At -solid) Have 7 valence electrons

Noble gases At room temperature, exist as gases. Are completely unreactive Have full s and p orbitals Are odorless, colorless, nonflammable

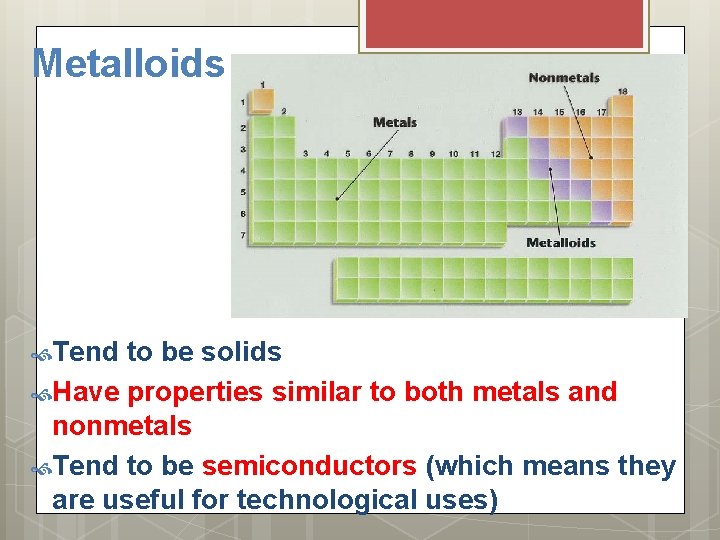
Metalloids Tend to be solids Have properties similar to both metals and nonmetals Tend to be semiconductors (which means they are useful for technological uses)

Valence Electrons: The outermost s & p electrons

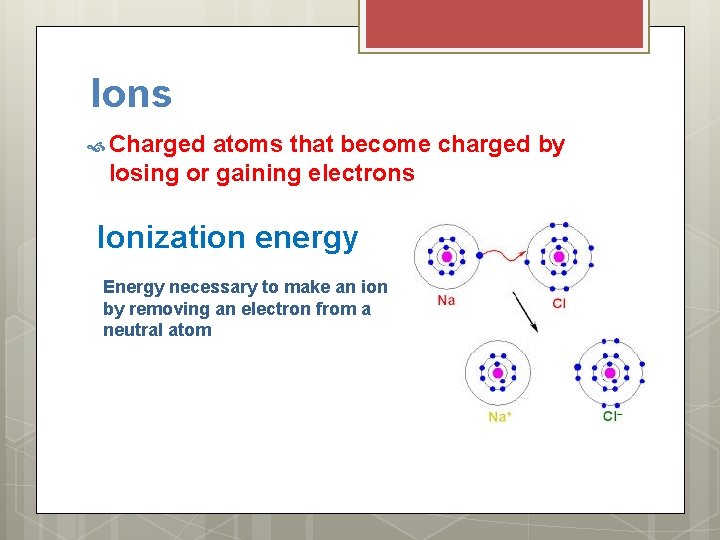
Ions Charged atoms that become charged by losing or gaining electrons Ionization energy Energy necessary to make an ion by removing an electron from a neutral atom

Rule #1 to remember! When an element loses an electron, we can think of it as being given away, which is a good thing or POSITIVE thing to do.

Group 1 Elements H, Li, Na, K, Rb, Cs, Fr Achieve a stable octet (full outer shell) by losing 1 electron, which forms a +1 ion H+ , Li+ , Na+ , etc…

Group 2 Elements Be, Mg, Ca, Sr, Ba, Ra Achieve a stable octet by losing 2 electrons, which forms a +2 ion Be 2+, Mg 2+, Ca 2+, etc…

Rule #2 to remember! When an element gains an electron, we can think of it as it is being stolen from another ion, which is a bad or NEGATIVE thing to do.

Group 7 Elements F, Cl, Br, I, At Achieve a stable octet by stealing (gaining) 1 electron, which forms a -1 ion F -, Cl-, Br- , etc…

Group 6 Elements O, S, Se, Te, Po Achieve a stable octet by stealing (gaining) 2 electrons, which forms a 2 - ion O 2 - , S 2 - , Se 2 -, etc…