Neutron Powder Diffraction a powerful technique for studying













































- Slides: 93

Neutron Powder Diffraction: a powerful technique for studying structural and magnetic phase transitions A-site ordered perovskite YBa. Mn 2 O 6: atomic versus larger scales charge ordering Juan Rodríguez-Carvajal Laboratoire Léon Brillouin (CEA-CNRS), Saclay, France Barcelona, September 2005, MULTIMAT

Content of the talk • Generalities about the properties and use of neutrons for condensed matter research • Neutron powder diffraction. Data treatment. Introduction to the programs of the Full. Prof Suite • Study of the phase transitions in YBa. Mn 2 O 6. Charge/orbital/spin ordering. Barcelona, September 2005, MULTIMAT

Neutrons as constituents of matter Chadwick 1932 Barcelona, September 2005, MULTIMAT

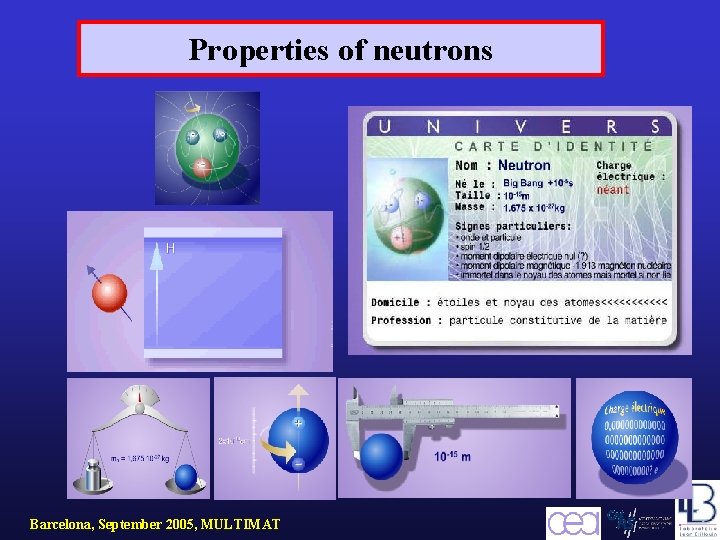
Properties of neutrons Barcelona, September 2005, MULTIMAT

Neutron production: nuclear reactors • The reactor geometry is optimized to produce the maximum number of neutrons. • The fission of a 235 U nucleus produce in average 2. 5 neutrons. 1. 5 are used to keep the chain reaction, only 1 is used for making neutron beams. l. Main research reactors in Europe: n France, l. ILL (57 MW) in Grenoble l. Orpheé (14 MW) in Saclay n Germany l. FRJ-2 (23 MW) in Julich (closed) l. BER-2 (10 MW) in Berlin l. FRM-II (24 MW) in Munich (new) Barcelona, September 2005, MULTIMAT

Neutron production: spallation sources • • H+ accelerated to 1 Ge. V Targets of W, Pb, Hg, U 20 to 25 neutrons by H+ Pulses of 50 Hz Flux of 3. 7 1016 ns-1 (ISIS) Pulse length of µs Relatively small average flux l l l Barcelona, September 2005, MULTIMAT Switzerland, SINQ quasi-continuous ( 10 MW) United Kingdom, ISIS (1. 5 MW) SNS, Oak Ridge (USA) – in construction

Neutron reactors: ILL Barcelona, September 2005, MULTIMAT

Neutron reactors: ILL Barcelona, September 2005, MULTIMAT

Particle-wave properties kinetic energy (E) velocity (v) temperature (T). E= mnv 2/2= k. B T = p 2/2 mn= (ħk)2/2 mn=(h/l)2/2 mn momentum (p) p= mnv = ħ k ħ=h/2 p wavevector (k) Barcelona, September 2005, MULTIMAT wavelength (l) k= 2 p/l = mnv/ħ

Particle-wave properties (moderators) E = mn v 2 /2 = k. B T = (ħk)2/2 mn ; l. Neutrons moderated by heavy water at 300 K (Thermal neutrons) l. Also cold moderating source, liquid deuterium at 25 K (Cold neutrons) l. And hot moderating source, graphite at ~2000 K Barcelona, September 2005, MULTIMAT k = 2 p/l = mnv/ħ

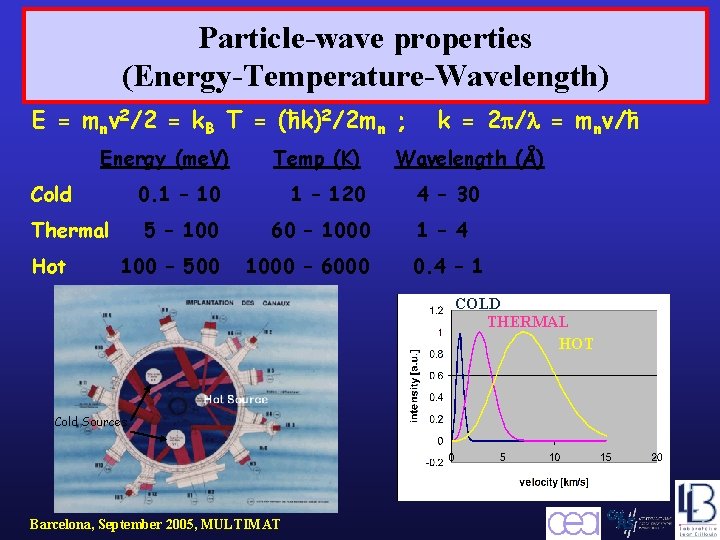
Particle-wave properties (Energy-Temperature-Wavelength) E = mnv 2/2 = k. B T = (ħk)2/2 mn ; Energy (me. V) Cold 0. 1 – 10 1 – 120 5 – 100 60 – 1000 100 – 500 1000 – 6000 Thermal Hot Temp (K) k = 2 p/l = mnv/ħ Wavelength (Å) 4 – 30 1 – 4 0. 4 – 1 COLD THERMAL HOT Cold Sources Barcelona, September 2005, MULTIMAT

Interaction neutron-nucleus k’ k f r Direction q, f d. S q d. W Target q Barcelona, September 2005, MULTIMAT z = number of incident neutrons /cm 2 / second = total number of neutrons scattered/second/ Fermi’s golden rule gives the neutron-scattering Cross-section number of neutrons of a given energy scattered per second in a given solid angle (the effective area presented by a nucleus to an incident neutron)

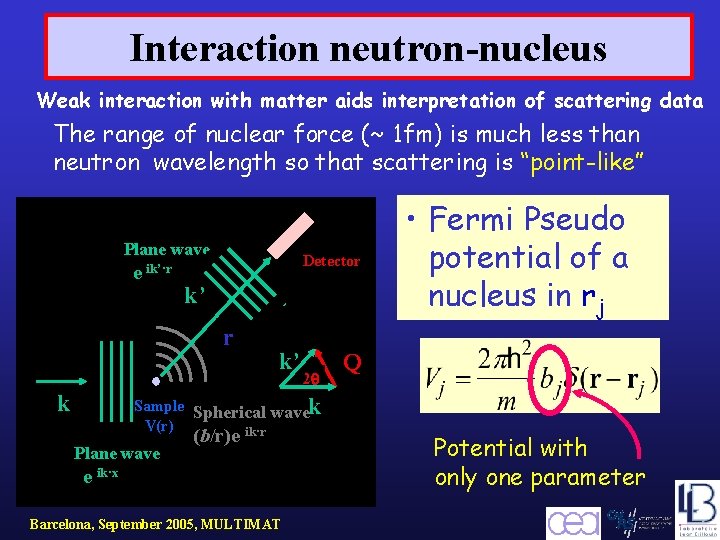
Interaction neutron-nucleus Weak interaction with matter aids interpretation of scattering data The range of nuclear force (~ 1 fm) is much less than neutron wavelength so that scattering is “point-like” Plane wave e ik’·r Detector k’ r k k’ 2 Sample Spherical wavek V(r) ik·r Plane wave (b/r)e e ik·x Barcelona, September 2005, MULTIMAT • Fermi Pseudo potential of a nucleus in rj Q Potential with only one parameter

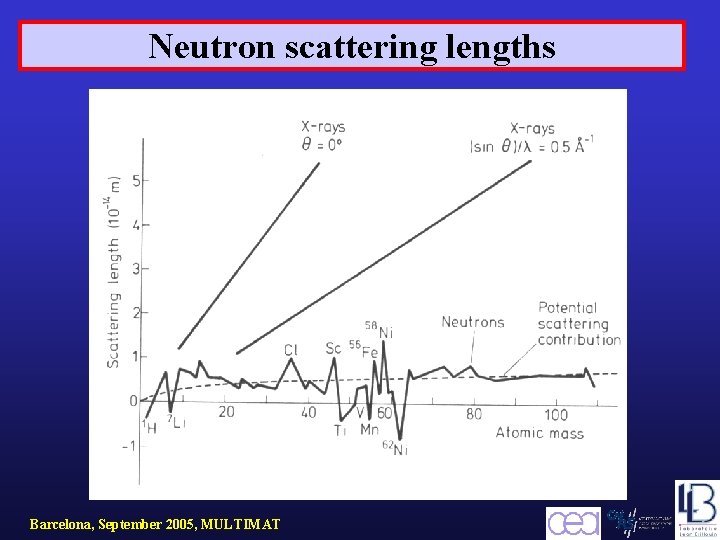
Neutron scattering lengths Barcelona, September 2005, MULTIMAT

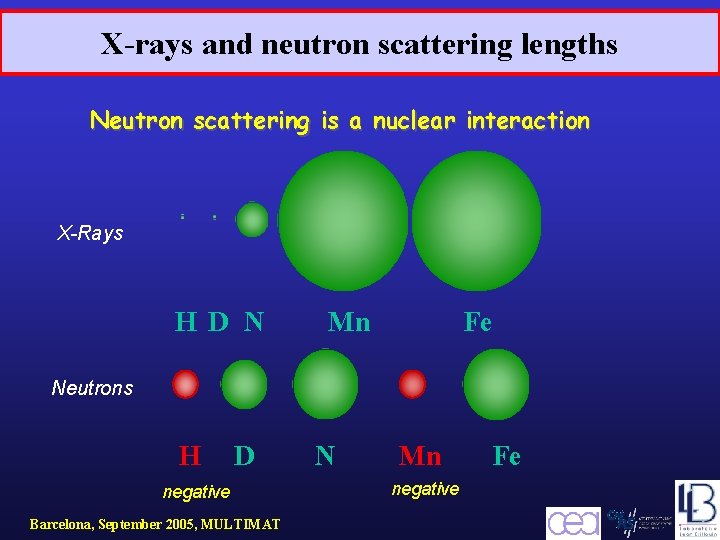
X-rays and neutron scattering lengths Neutron scattering is a nuclear interaction X-Rays H D N Mn Fe Neutrons H D negative Barcelona, September 2005, MULTIMAT N Mn negative Fe

Elastic Scattering: Diffraction For elastic scattering: Where v 0 is the unit cell volume and t a reciprocal lattice vector The coherent elastic scattering takes place only for Bragg Law Barcelona, September 2005, MULTIMAT

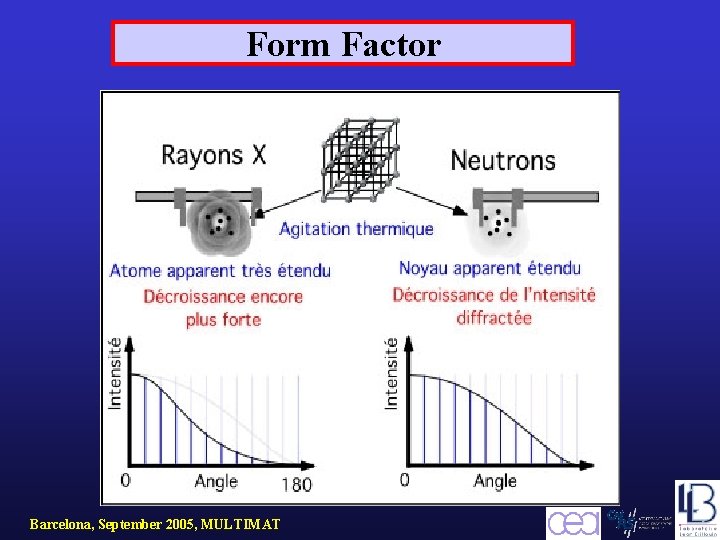
Form Factor Barcelona, September 2005, MULTIMAT

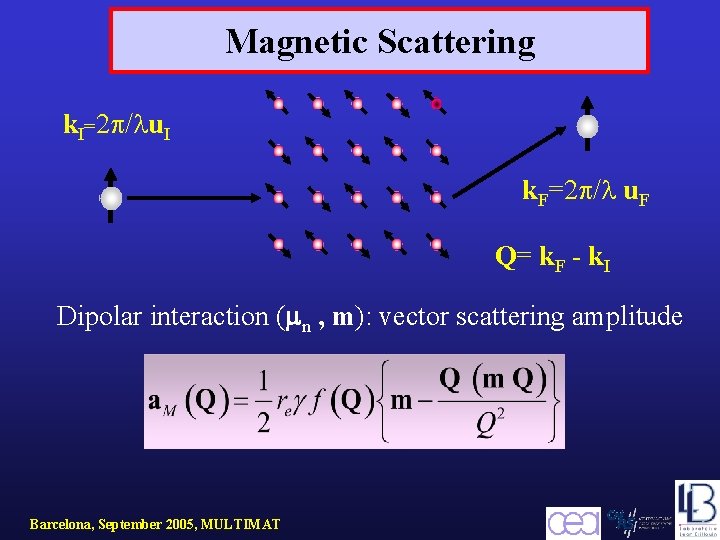
Magnetic Scattering k. I=2 / u. I k. F=2 / u. F Q= k. F - k. I Dipolar interaction ( n , m): vector scattering amplitude Barcelona, September 2005, MULTIMAT

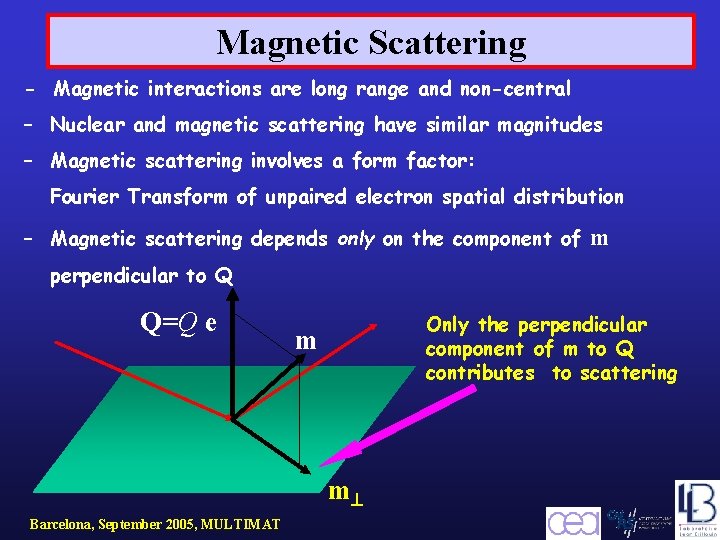
Magnetic Scattering - Magnetic interactions are long range and non-central – Nuclear and magnetic scattering have similar magnitudes – Magnetic scattering involves a form factor: Fourier Transform of unpaired electron spatial distribution – Magnetic scattering depends only on the component of m perpendicular to Q Q=Q e Only the perpendicular component of m to Q contributes to scattering m m Barcelona, September 2005, MULTIMAT

Content of the talk • Generalities about the properties and use of neutrons for condensed matter research • Neutron powder diffraction. Data treatment. Introduction to the programs of the Full. Prof Suite • Study of the phase transitions in YBa. Mn 2 O 6. Charge/orbital/spin ordering. Barcelona, September 2005, MULTIMAT

Diffractometers: Powder high flux D 1 B(ILL) or G 41(LLB) Barcelona, September 2005, MULTIMAT

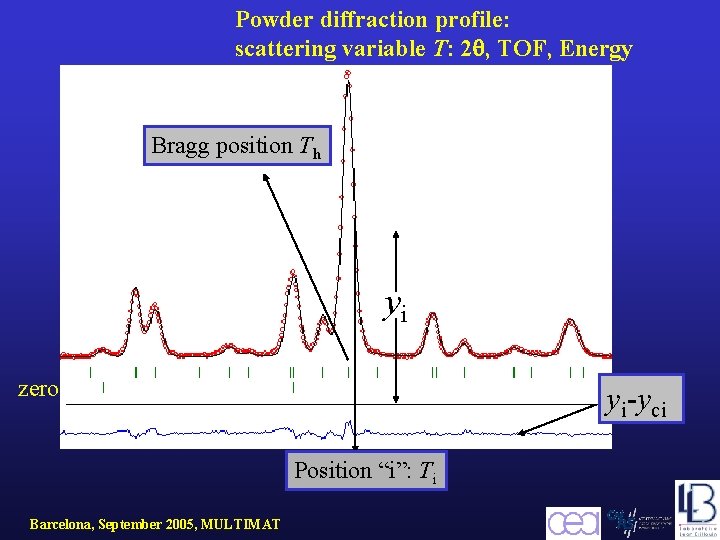
Experimental powder pattern A powder diffraction pattern can be recorded in numerical form for a discrete set of scattering angles, times of flight or energies. We will refer to this scattering variable as : T. The experimental powder diffraction pattern is usually given as three arrays : The profile can be modelled using the calculated counts: at the ith step by summing the contribution from neighbouring Bragg reflections plus the background. Barcelona, September 2005, MULTIMAT yci

Powder diffraction profile: scattering variable T: 2 , TOF, Energy Bragg position Th yi zero yi-yci Position “i”: Ti Barcelona, September 2005, MULTIMAT

The calculated profile of powder diffraction patterns Contains structural information: atom positions, magnetic moments, etc Contains micro-structural information: instr. resolution, defects, crystallite size… Background: noise, incoherent scattering diffuse scattering, . . . Barcelona, September 2005, MULTIMAT

The calculated profile of powder diffraction patterns The symbol {h} means that the sum is extended only to those reflections contributing to the channel “i”. This should be taken into account (resolution function of the diffractometer and sample broadening) before doing the actual calculation of the profile intensity. This is the reason why some Rietveld programs are run in two steps Barcelona, September 2005, MULTIMAT

Several phases ( = 1, n ) contributing to the diffraction pattern Several phases ( = 1, n ) contributing to several (p=1, np) diffraction patterns Barcelona, September 2005, MULTIMAT

Integrated intensities are proportional to the square of the structure factor F. The factors are: Scale Factor (S), Lorentz-polarization (Lp), preferred orientation (O), absorption (A), other “corrections” (C). . . Barcelona, September 2005, MULTIMAT

The Structure Factor contains the structural parameters (isotropic case) Barcelona, September 2005, MULTIMAT

Structural Parameters (simplest case) Atom positions (up to 3 n parameters) Occupation factors (up to n -1 parameters) Isotropic displacement (temperature) factors (up to n parameters) Barcelona, September 2005, MULTIMAT

Structural Parameters (complex cases) As in the simplest case plus additional (or alternative) parameters: • Anisotropic temperature (displacement) factors • Anharmonic temperature factors • Special form-factors (Symmetry adapted spherical harmonics ), TLS for rigid molecules, etc. • Magnetic moments, coefficients of Fourier components of magnetic moments , basis functions, etc. Barcelona, September 2005, MULTIMAT

The Structure Factor in complex cases Complex form factor of object j Anisotropic DPs Anharmonic DPs Barcelona, September 2005, MULTIMAT

The approximation of the peak shape profile function and microstructural effects Precise refinements can be done with confidence only if the intrinsic and instrumental peak shapes are properly approximated. At present The approximation of the intrinsic profile is mostly based in the Voigt (or pseudo-Voigt) function The approximation of the instrumental profile is also based in the Voigt function for constant wavelength instruments For TOF the instrumental+intrinsic profile is approximated by the convolution of a Voigt function with back-to-back exponentials or with the Ikeda. Carpenter function. Barcelona, September 2005, MULTIMAT

The peak shape function of powder diffraction patterns contains the Profile Parameters In most cases the observed peak shape is approximated by a linear combination of Voigt (or pseudo-Voigt) functions Barcelona, September 2005, MULTIMAT

The Voigt function The pseudo-Voigt function Barcelona, September 2005, MULTIMAT

The Rietveld Method consist of refining a crystal (and/or magnetic) structure by minimising the weighted squared difference between the observed and the calculated pattern against the parameter vector: : is the variance of the "observation" yi Barcelona, September 2005, MULTIMAT

Least squares: Gauss-Newton (1) Minimum necessary condition: A Taylor expansion of around allows the application of an iterative process. The shifts to be applied to the parameters at each cycle for improving 2 are obtained by solving a linear system of equations (normal equations) Barcelona, September 2005, MULTIMAT

Least squares: Gauss-Newton (2) The shifts of the parameters obtained by solving the normal equations are added to the starting parameters giving rise to a new set The new parameters are considered as the starting ones in the next cycle and the process is repeated until a convergence criterion is satisfied. The variances of the adjusted parameters are calculated by the expression: Barcelona, September 2005, MULTIMAT

Neutron and synchrotron powder diffraction Neutrons • Constant scattering length. Contrast. • Low absorption: easy sample environment • Magnetic structures • High precision in structure refinement • Moderate resolution Barcelona, September 2005, MULTIMAT Synchrotron X-rays • Extremely high resolution • Subtle distortions • Indexing and Structure determination • Anomalous scattering • Texture effects

Barcelona, September 2005, MULTIMAT

Barcelona, September 2005, MULTIMAT

Barcelona, September 2005, MULTIMAT

Directory structure of the Full. Prof Suite Barcelona, September 2005, MULTIMAT

Win. PLOTR: program to access the whole Full. Prof Suite Barcelona, September 2005, MULTIMAT

Configuration of Win. PLOTR File: winplotr. set Barcelona, September 2005, MULTIMAT

Indexing demo with Win. PLOTR New facility: DICVOL 04 Two other indexing programs: TREOR, ITO Barcelona, September 2005, MULTIMAT

Other features of Win. PLOTR Two other indexing programs: TREOR, ITO Access to other programs: Ed. PCR, Fp_Studio, DICVOL 04, Bas. Ireps Barcelona, September 2005, MULTIMAT

A program for analysis of diffraction patterns: Full. Prof • A program for : Simulation of powder diffraction patterns Pattern decomposition integrated intensities Structure refinement Powder and single crystal data • Crystal and magnetic structures • Multiple data sets: simultaneous treatment of several powder diffraction patterns (CW X-rays & neutrons, Energy dispersive X-rays, TOF neutron diffraction) • Combined treatment of single crystal and powder data • Crystal and magnetic Structure determination capabilities: simulated annealing on integrated intensity data Barcelona, September 2005, MULTIMAT

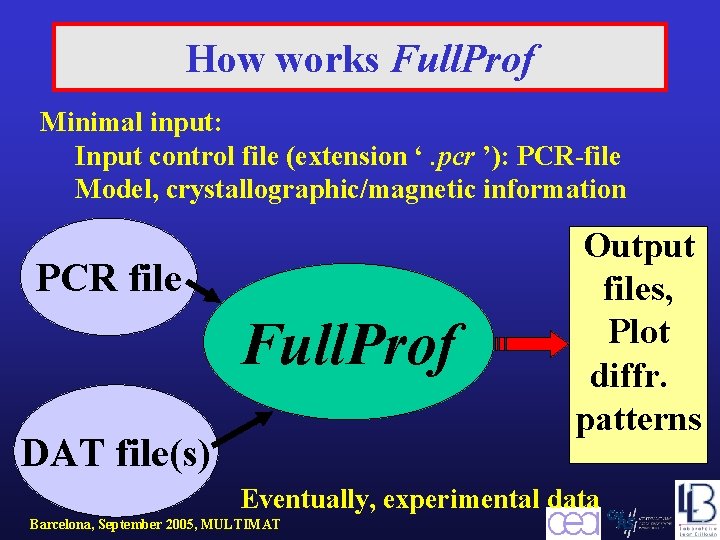
How works Full. Prof Minimal input: Input control file (extension ‘. pcr ’): PCR-file Model, crystallographic/magnetic information PCR file Full. Prof DAT file(s) Output files, Plot diffr. patterns Eventually, experimental data Barcelona, September 2005, MULTIMAT

The PCR file: steep learning curve DAT file(s) PCR file Format depending on the instrument, usually simple Many variables and options Complex to handle Hint: copy an existing (working) PCR-file and modify it for the user case, or. . . USE the new GUI: Ed. PCR Barcelona, September 2005, MULTIMAT

A new GUI for Full. Prof: Ed. PCR GUI using Winteracter: http: //www. winteracter. com Barcelona, September 2005, MULTIMAT

Content of the talk • Generalities about the properties and use of neutrons for condensed matter research • Neutron powder diffraction. Data treatment. Introduction to the programs of the Full. Prof Suite • Study of the phase transitions in YBa. Mn 2 O 6. Charge/orbital/spin ordering. Barcelona, September 2005, MULTIMAT

Example: Study of A-site ordered manganite YBa. Mn 2 O 6 • Introduction to the half-doped (Mn 3+/Mn 4+) manganites • Phase transitions, crystal and magnetic structures of mixed valence YBa. Mn 2 O 6 Barcelona, September 2005, MULTIMAT

Colossal Magnetoresistance Regain of interest in electronic properties of (semi)-conducting magnetic oxides Our team started few years ago to answer some important questions! Charge ordering: In the charge ordered state, is the charge localization at atomic level? What does the conventional ionic states Mn 3+/Mn 4+ mean? … CDW, charge disproportionation, polarons, … Orbital ordering: Is that concept really relevant for explaining the electronic/structural transitions in manganites? Is there any difference with respect to a MO 6 distorting structural transition? Barcelona, September 2005, MULTIMAT

Electronic transitions and phase separation: Role of the chemistry and the disorder. Homogeneous/inhomogeneous states: what is a phase? Role of the defects and microstrains … Magnetic exchange interactions: Are the Goodenough-Kanamori-Anderson rules still applicable? How to explain the canted magnetic structures? Are there other magnetic structures compatible with the observed magnetic powder diffraction patterns? Is the CE structure really collinear? Barcelona, September 2005, MULTIMAT

Nature of the structural transition at TCO in half-doped manganites R 1/2 D 1/2[Mn 3. 5+]O 3 Our previous work : A new interpretation of the electronic localization in charge ordered manganites The single Mn-site in the HT phase breaks up into two sublattices in the COlow temperature phase, having very similar characteristics concerning the average Mn-O distances. The observed distortions are different of what is expected from the generally accepted picture of a concomitant charge (Mn 3+-Mn 4+) and orbital ordering. O 1 Formation of “molecular” ferromagnetic Mn-O-Mn pairs stabilised by local DE and a suitable structural distortion: Zener polaron. A. Daoud-Aladine et al. , Phys. Rev. Lett. 89, 097205 (2002). Barcelona, September 2005, MULTIMAT Mn 2 O 4 q e O 3 O 1 Mn 1 O 4 O 2

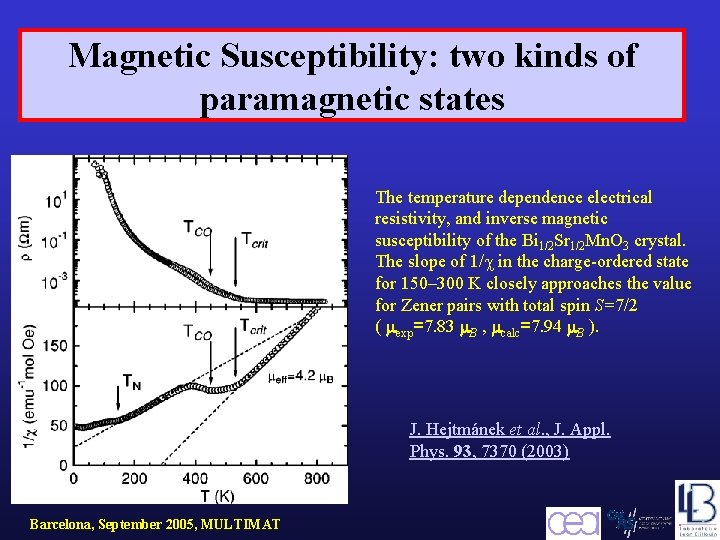
Magnetic Susceptibility: two kinds of paramagnetic states The temperature dependence electrical resistivity, and inverse magnetic susceptibility of the Bi 1/2 Sr 1/2 Mn. O 3 crystal. The slope of 1/ in the charge-ordered state for 150– 300 K closely approaches the value for Zener pairs with total spin S=7/2 ( exp=7. 83 B , calc=7. 94 B ). J. Hejtmánek et al. , J. Appl. Phys. 93, 7370 (2003) Barcelona, September 2005, MULTIMAT

Our previous strategy to study the structural distortions in half-doped manganites: Minimise the size mismatch between atoms in A sites of perovskites AMn. O 3 (Pr, Ca) In common manganites the A-site doping induces disorder and local inhomogeneities (fluctuations of chemical composition) Role of the A-site disorder Using Ba and a heavy rare earth, or Y, a new ordered family of half-doped manganites is obtained Y 1/2 Ba 1/2 Mn. O 3 YBa. Mn 2 O 6 A first report on this compound was published by: T. Nakajima et al. J. Phys. Chem. Solids 63 (2002) 913 Barcelona, September 2005, MULTIMAT

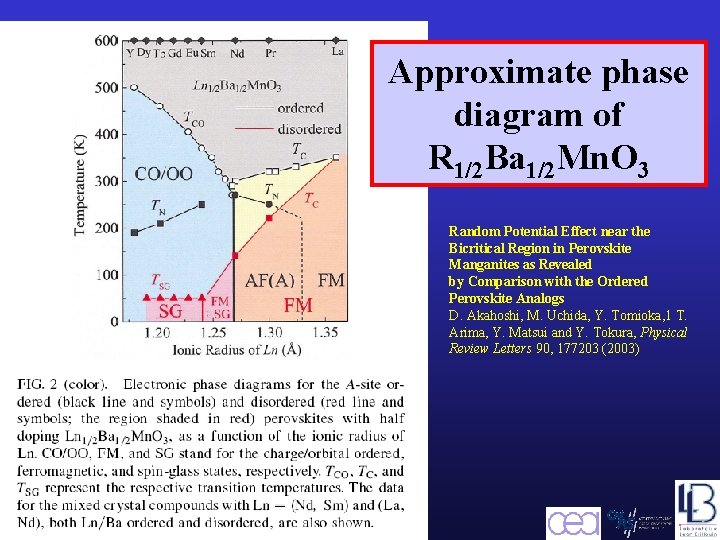
Approximate phase diagram of R 1/2 Ba 1/2 Mn. O 3 Random Potential Effect near the Bicritical Region in Perovskite Manganites as Revealed by Comparison with the Ordered Perovskite Analogs D. Akahoshi, M. Uchida, Y. Tomioka, 1 T. Arima, Y. Matsui and Y. Tokura, Physical Review Letters 90, 177203 (2003) Barcelona, September 2005, MULTIMAT

Phase transitions, crystal and magnetic structures of mixed valence YBa. Mn 2 O 6 Barcelona, September 2005, MULTIMAT

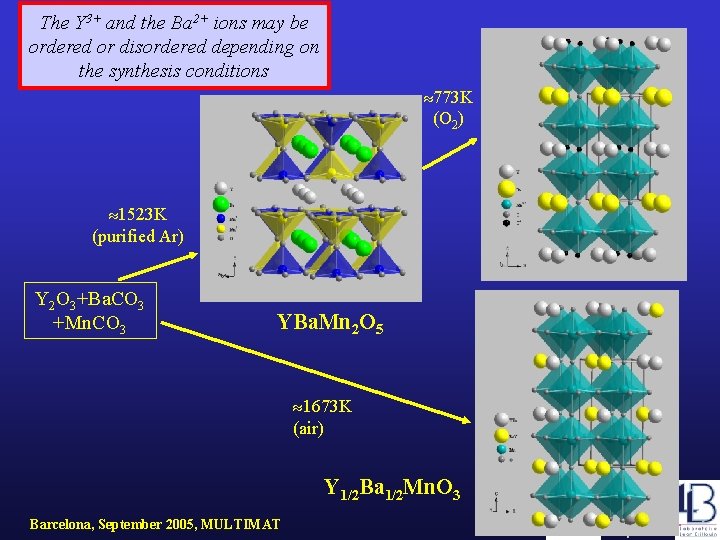
YBa. Mn 2 O 6 The Y 3+ and the Ba 2+ ions may be ordered or disordered depending on the synthesis conditions 773 K (O 2) 1523 K (purified Ar) Y 2 O 3+Ba. CO 3 +Mn. CO 3 YBa. Mn 2 O 5 1673 K (air) Y 1/2 Ba 1/2 Mn. O 3 Barcelona, September 2005, MULTIMAT

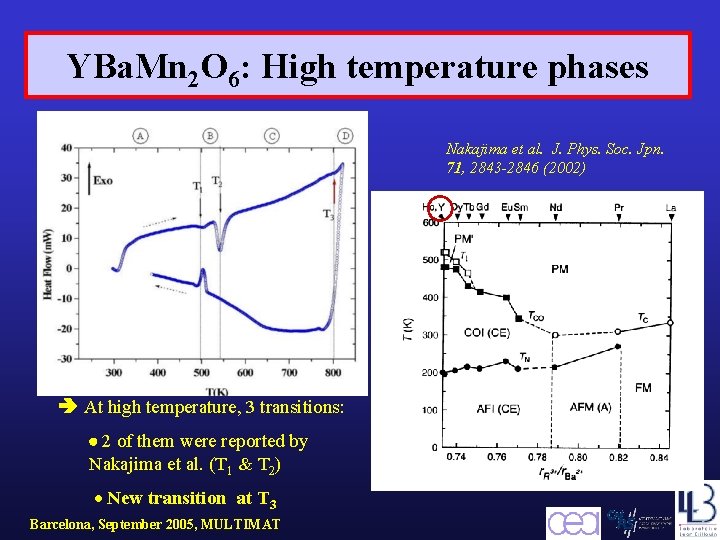
YBa. Mn 2 O 6: High temperature phases Nakajima et al. J. Phys. Soc. Jpn. 71, 2843 -2846 (2002) At high temperature, 3 transitions: 2 of them were reported by Nakajima et al. (T 1 & T 2) New transition at T 3 Barcelona, September 2005, MULTIMAT

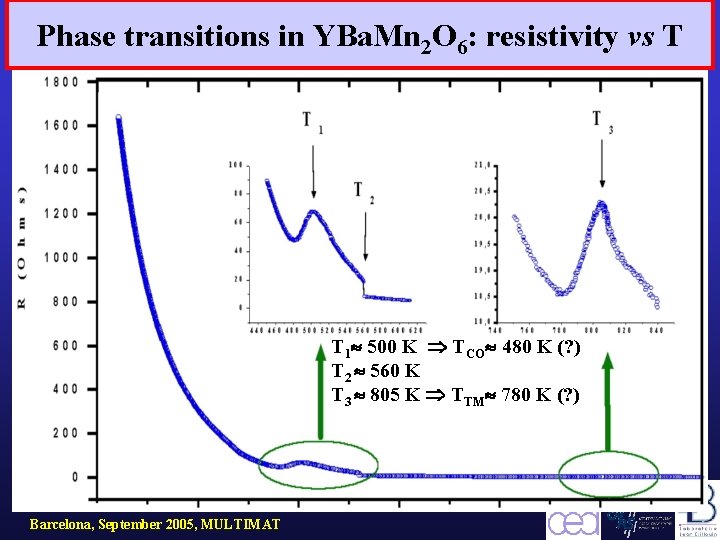
Phase transitions in YBa. Mn 2 O 6: resistivity vs T T 1 500 K TCO 480 K (? ) T 2 560 K T 3 805 K TTM 780 K (? ) Barcelona, September 2005, MULTIMAT

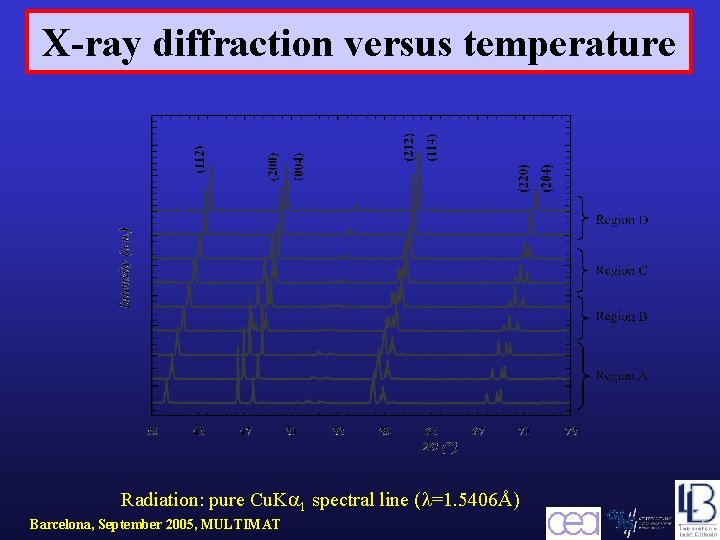
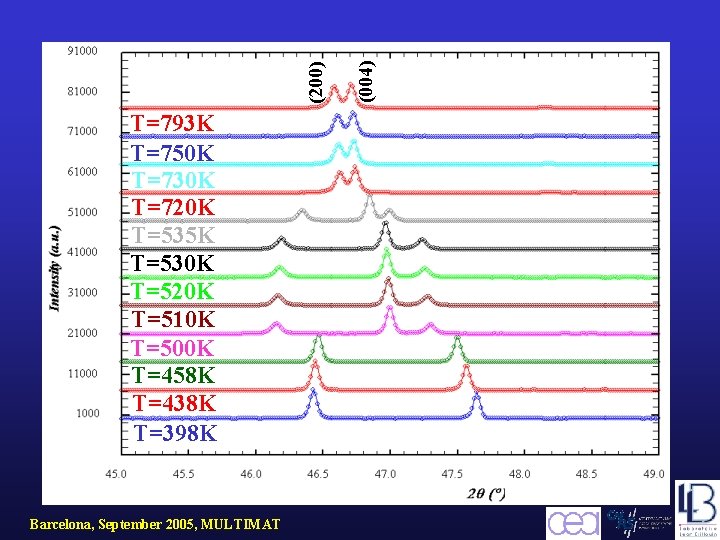
X-ray diffraction versus temperature Radiation: pure Cu. K 1 spectral line ( =1. 5406Å) Barcelona, September 2005, MULTIMAT

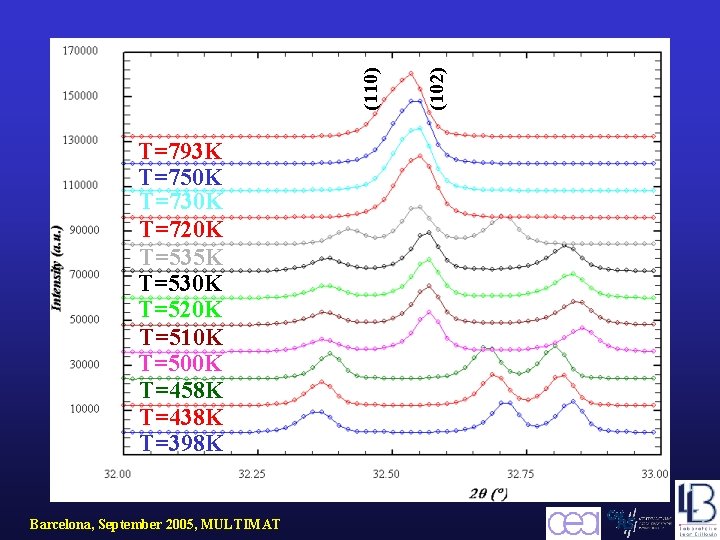
Barcelona, September 2005, MULTIMAT (102) (110) T=793 K T=750 K T=730 K T=720 K T=535 K T=530 K T=520 K T=510 K T=500 K T=458 K T=438 K T=398 K

Barcelona, September 2005, MULTIMAT (004) (200) T=793 K T=750 K T=730 K T=720 K T=535 K T=530 K T=520 K T=510 K T=500 K T=458 K T=438 K T=398 K

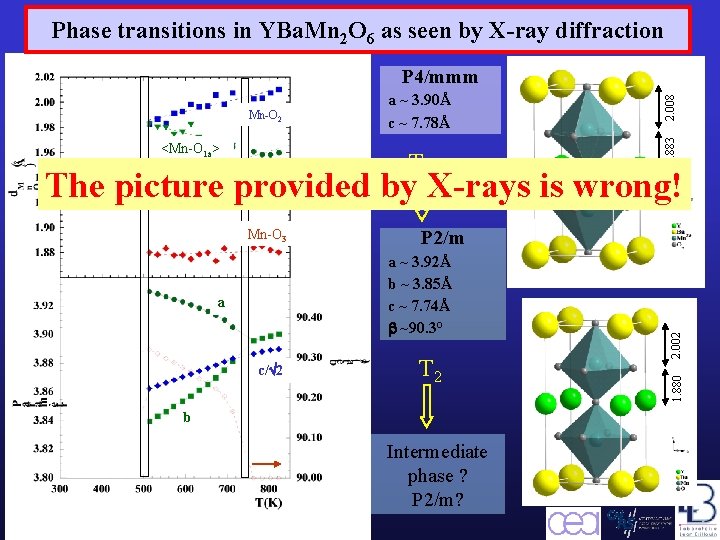
Phase transitions in YBa. Mn 2 O 6 as seen by X-ray diffraction <Mn-O 1 a> a ~ 3. 90Å c ~ 7. 78Å T 3 1. 883 Mn-O 2 2. 008 P 4/mmm The picture provided by X-rays is wrong! Mn-O 1 b b/2 2 c/ 2 T 2 b c/ 2 Barcelona, September 2005, MULTIMAT Intermediate phase ? P 2/m? 2. 002 a ~ 3. 92Å b ~ 3. 85Å c ~ 7. 74Å ~90. 3° a a/ 2 P 2/m 1. 880 Mn-O 3

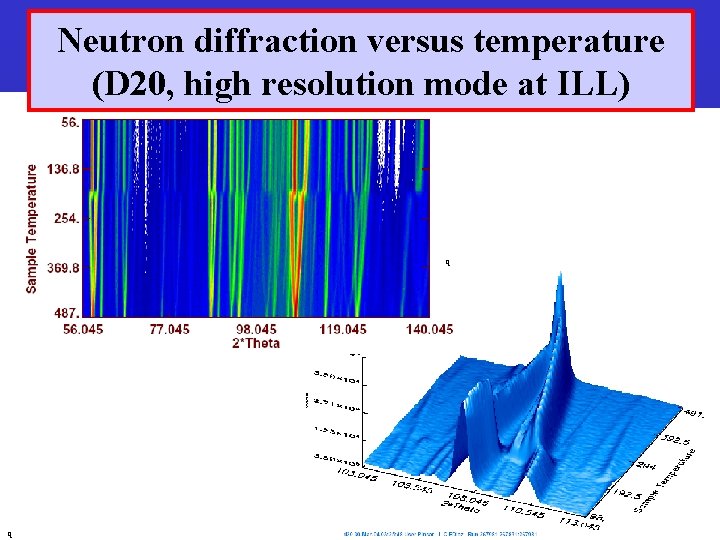
Neutron diffraction versus temperature (D 20, high resolution mode at ILL) q q Barcelona, September 2005, MULTIMAT

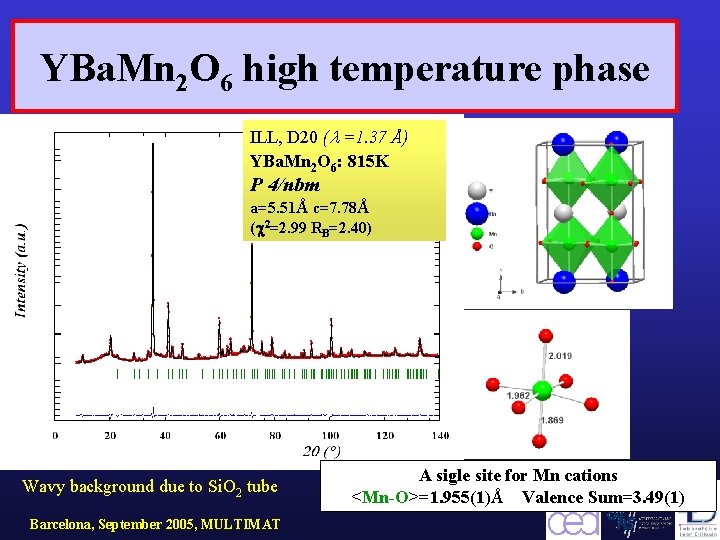
YBa. Mn 2 O 6 high temperature phase ILL, D 20 ( =1. 37 Å) YBa. Mn 2 O 6: 815 K P 4/nbm a=5. 51Å c=7. 78Å ( 2=2. 99 RB=2. 40) Wavy background due to Si. O 2 tube Barcelona, September 2005, MULTIMAT A sigle site for Mn cations <Mn-O>=1. 955(1)Å Valence Sum=3. 49(1)

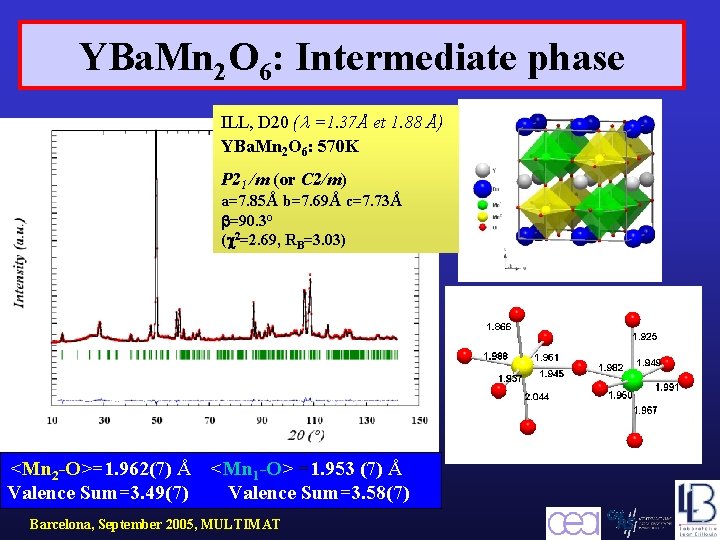
YBa. Mn 2 O 6: Intermediate phase ILL, D 20 ( =1. 37Å et 1. 88 Å) YBa. Mn 2 O 6: 570 K P 21 /m (or C 2/m) a=7. 85Å b=7. 69Å c=7. 73Å =90. 3° ( 2=2. 69, RB=3. 03) <Mn 2 -O>=1. 962(7) Å Valence Sum=3. 49(7) <Mn 1 -O> =1. 953 (7) Å Valence Sum=3. 58(7) Barcelona, September 2005, MULTIMAT

YBa. Mn 2 O 6: Room temperature phase Nakajima et al. : Monoclinic space group P 2 (ap 2 2 ap) with two non-equivalent Mn. O 6 octahedra showing a marked volume difference Conclusion of the authors: This phase is characterised by a CO with a checkerboard pattern in the ab-plane. Williams et al. for Tb. Ba. Mn 2 O 6 (neutron diffraction pattern) The space group P 21/m initially used (ap 2 2 ap), does not take into account few superstructure reflections, with a propagation vector q=(0, 1/2, 0). A good fit to the profiles was obtained by transforming the structure to a bdoubled cell with triclinic P-1 symmetry. Scarce number of observed superstructure peaks : constrained parameter refinements. • strain-relieving displacements in the Tb. O layer • orbital ordering in the Mn. O 2 layers Conclusion of the authors: The obtained structure is characterised by a ‘‘rocksalt’’ three-dimensional Mn 3+/Mn 4+ charge ordering. Barcelona, September 2005, MULTIMAT

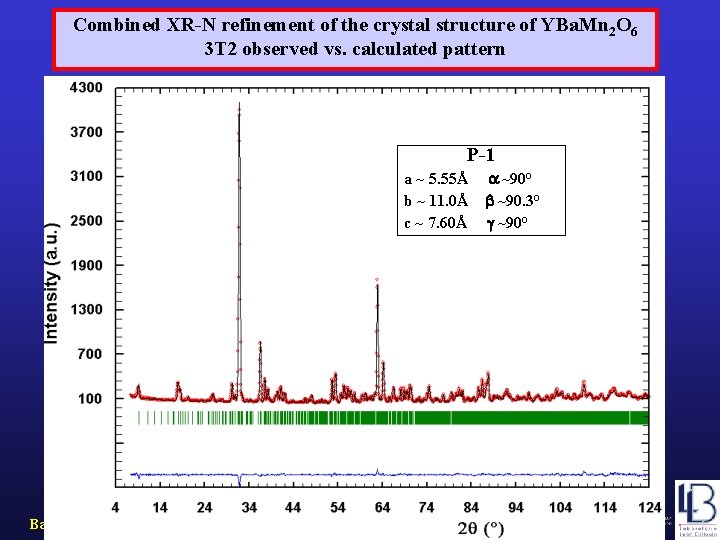
Combined XR-N refinement of the crystal structure of YBa. Mn 2 O 6 3 T 2 observed vs. calculated pattern P-1 a ~ 5. 55Å b ~ 11. 0Å c ~ 7. 60Å Barcelona, September 2005, MULTIMAT ~90° ~90. 3° ~90°

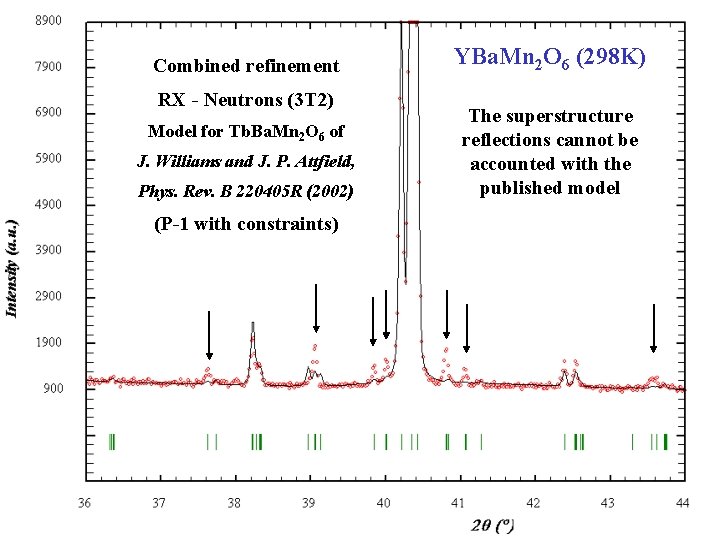
Combined refinement RX - Neutrons (3 T 2) Model for Tb. Ba. Mn 2 O 6 of J. Williams and J. P. Attfield, Phys. Rev. B 220405 R (2002) (P-1 with constraints) Barcelona, September 2005, MULTIMAT YBa. Mn 2 O 6 (298 K) The superstructure reflections cannot be accounted with the published model

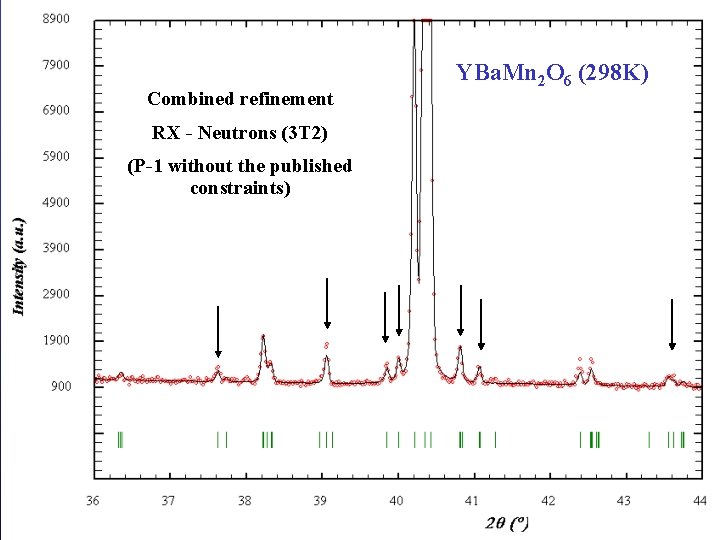
Combined refinement RX - Neutrons (3 T 2) (P-1 without the published constraints) Barcelona, September 2005, MULTIMAT YBa. Mn 2 O 6 (298 K)

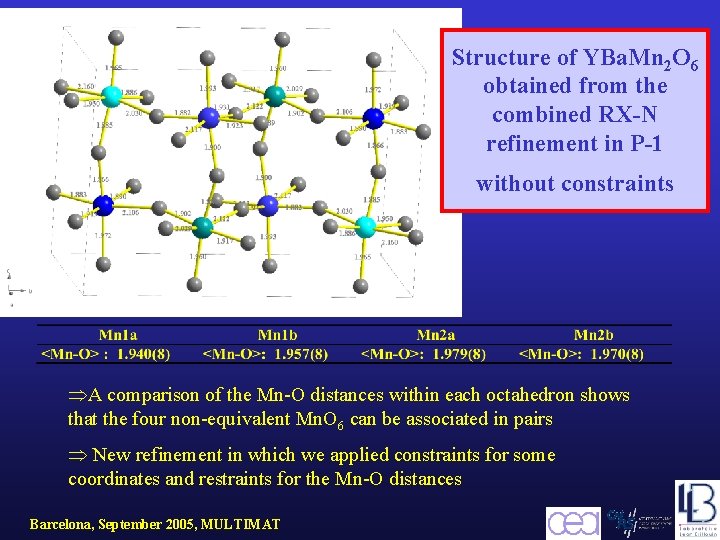
Structure of YBa. Mn 2 O 6 obtained from the combined RX-N refinement in P-1 without constraints A comparison of the Mn-O distances within each octahedron shows that the four non-equivalent Mn. O 6 can be associated in pairs New refinement in which we applied constraints for some coordinates and restraints for the Mn-O distances Barcelona, September 2005, MULTIMAT

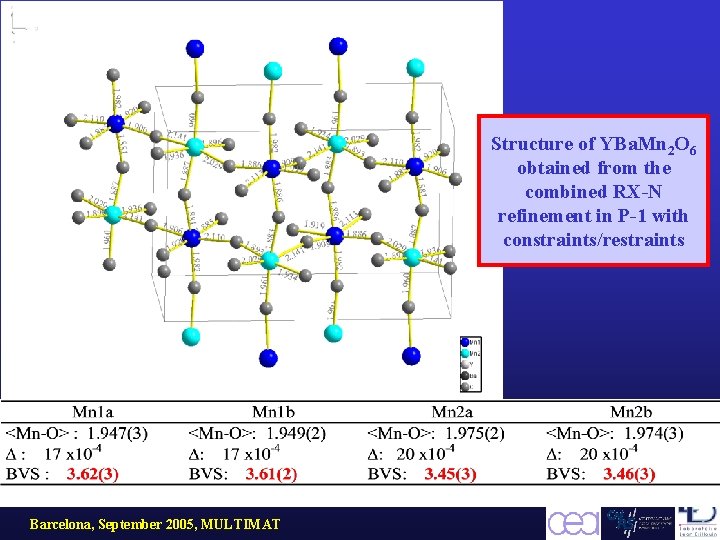
Structure of YBa. Mn 2 O 6 obtained from the combined RX-N refinement in P-1 with constraints/restraints Barcelona, September 2005, MULTIMAT

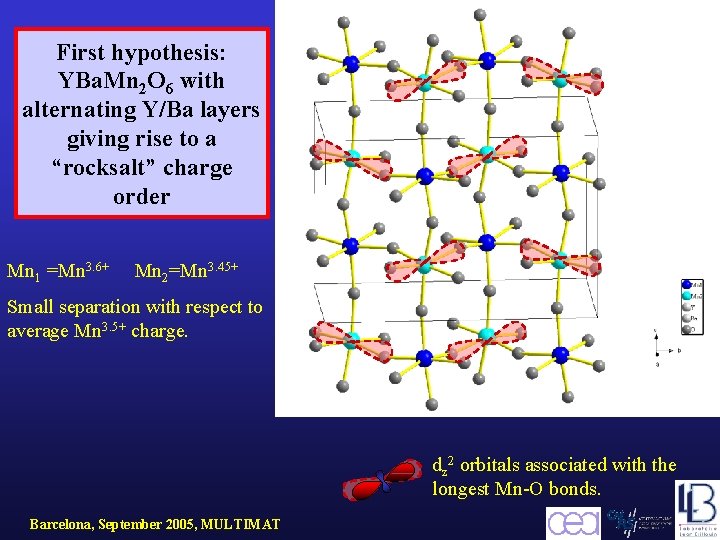
First hypothesis: YBa. Mn 2 O 6 with alternating Y/Ba layers giving rise to a “rocksalt” charge order Mn 1 =Mn 3. 6+ Mn 2=Mn 3. 45+ Small separation with respect to average Mn 3. 5+ charge. dz 2 orbitals associated with the longest Mn-O bonds. Barcelona, September 2005, MULTIMAT

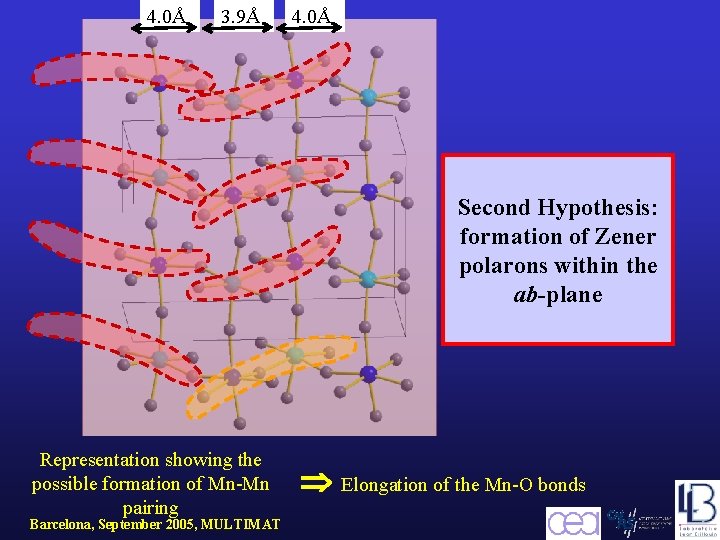
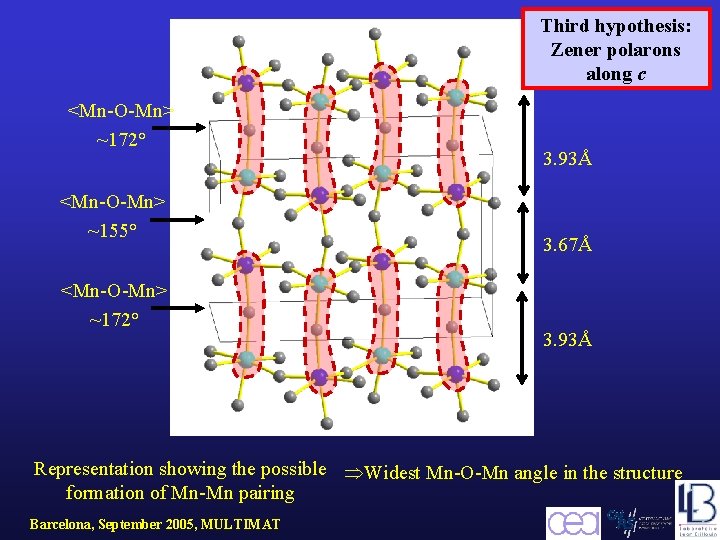
4. 0Å 3. 9Å 4. 0Å Second Hypothesis: formation of Zener polarons within the ab-plane Representation showing the possible formation of Mn-Mn pairing Barcelona, September 2005, MULTIMAT Elongation of the Mn-O bonds

Third hypothesis: Zener polarons along c <Mn-O-Mn> ~172° <Mn-O-Mn> ~155° <Mn-O-Mn> ~172° 3. 93Å 3. 67Å 3. 93Å Representation showing the possible Widest Mn-O-Mn angle in the structure formation of Mn-Mn pairing Barcelona, September 2005, MULTIMAT

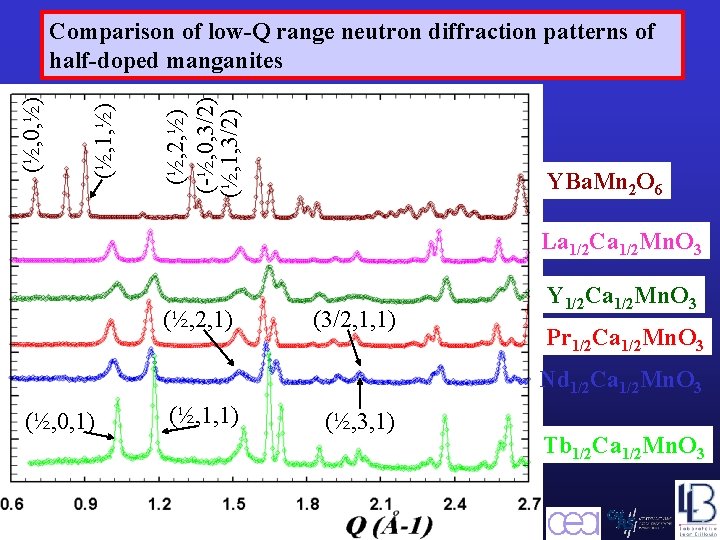
(½, 2, ½) (-½, 0, 3/2) (½, 1, ½) (½, 0, ½) Comparison of low-Q range neutron diffraction patterns of half-doped manganites YBa. Mn 2 O 6 La 1/2 Ca 1/2 Mn. O 3 (½, 2, 1) (3/2, 1, 1) Y 1/2 Ca 1/2 Mn. O 3 Pr 1/2 Ca 1/2 Mn. O 3 Nd 1/2 Ca 1/2 Mn. O 3 (½, 0, 1) (½, 1, 1) Barcelona, September 2005, MULTIMAT (½, 3, 1) Tb 1/2 Ca 1/2 Mn. O 3

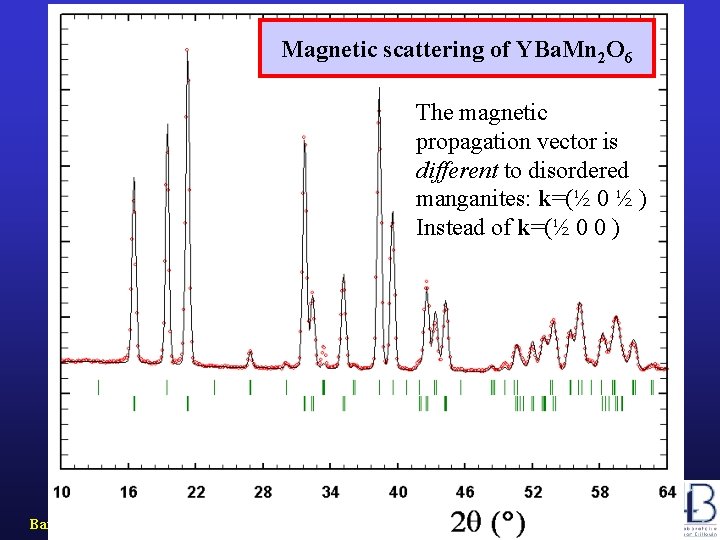
Magnetic scattering of YBa. Mn 2 O 6 The magnetic propagation vector is different to disordered manganites: k=(½ 0 ½ ) Instead of k=(½ 0 0 ) Barcelona, September 2005, MULTIMAT

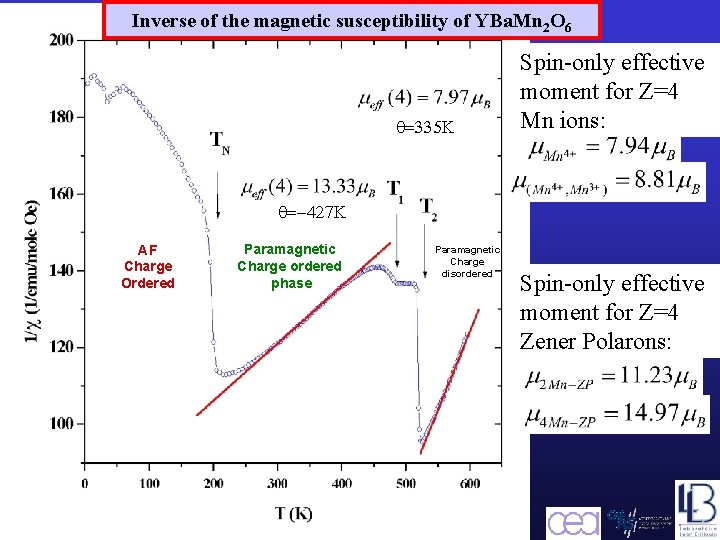
Inverse of the magnetic susceptibility of YBa. Mn 2 O 6 q=335 K Spin-only effective moment for Z=4 Mn ions: q=-427 K AF Charge Ordered Paramagnetic Charge ordered phase Barcelona, September 2005, MULTIMAT Paramagnetic Charge disordered Spin-only effective moment for Z=4 Zener Polarons:

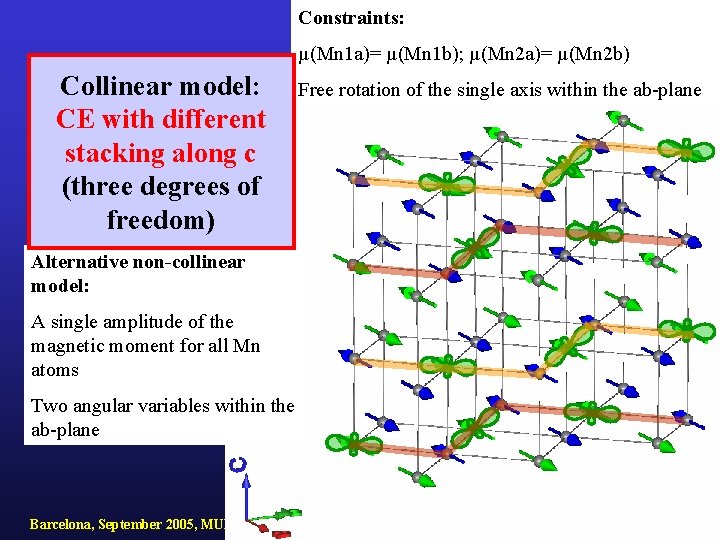
Constraints: µ(Mn 1 a)= µ(Mn 1 b); µ(Mn 2 a)= µ(Mn 2 b) Collinear model: CE with different stacking along c (three degrees of freedom) Alternative non-collinear model: A single amplitude of the magnetic moment for all Mn atoms Two angular variables within the ab-plane Barcelona, September 2005, MULTIMAT Free rotation of the single axis within the ab-plane

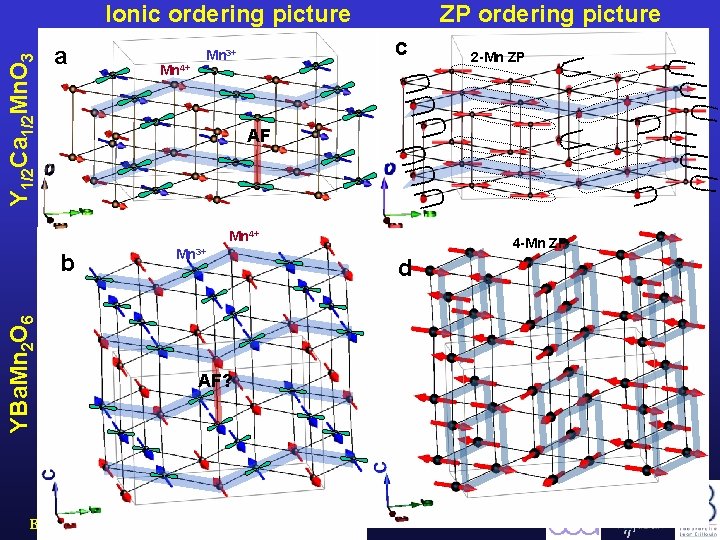
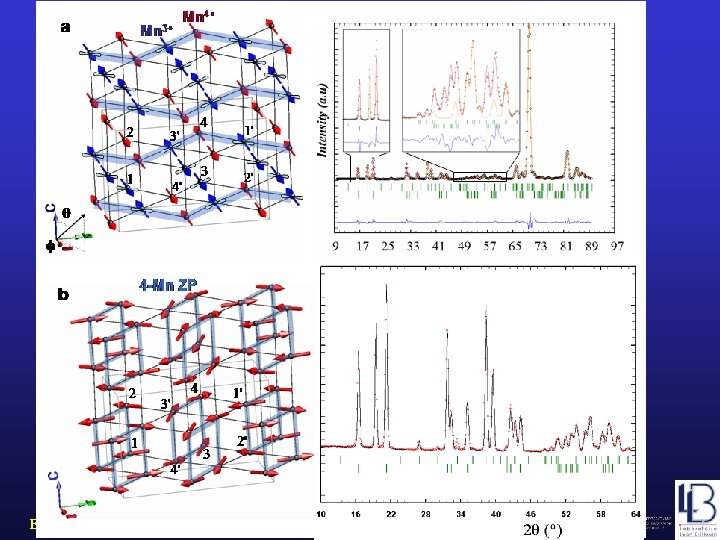
Y 1/2 Ca 1/2 Mn. O 3 Ionic ordering picture a Mn 4+ ZP ordering picture c Mn 3+ AF Mn 4+ b YBa. Mn 2 O 6 2 -Mn ZP Mn 3+ AF? Barcelona, September 2005, MULTIMAT 4 -Mn ZP d

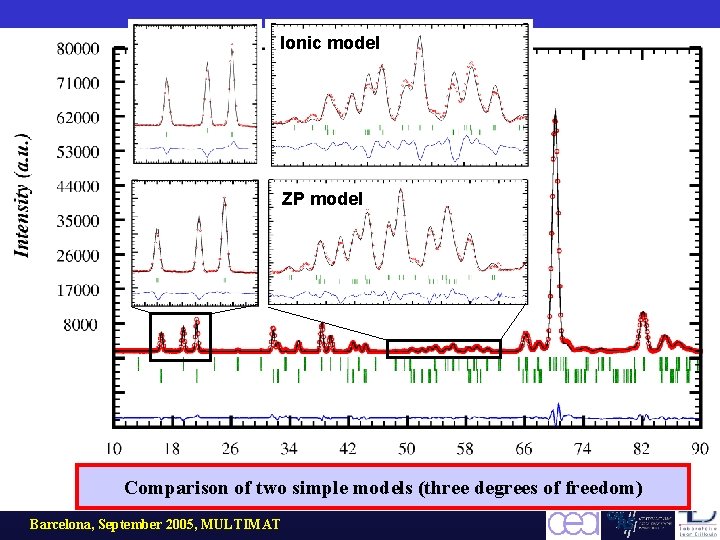
Ionic model ZP model Comparison of two simple models (three degrees of freedom) Barcelona, September 2005, MULTIMAT

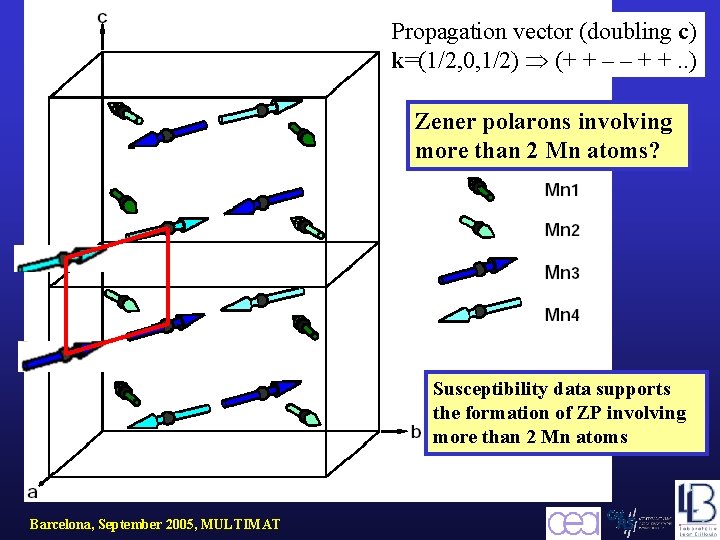
Propagation vector (doubling c) k=(1/2, 0, 1/2) (+ + – – + +. . ) Zener polarons involving more than 2 Mn atoms? Susceptibility data supports the formation of ZP involving more than 2 Mn atoms Barcelona, September 2005, MULTIMAT

Magnetic Structure of YBa. Mn 2 O 6 Barcelona, September 2005, MULTIMAT

Perspectives of research in CO transitions An image of “non-atomic” localisation, conserving the intermediate valence of Mn-ions, is more appropriate and explain most of the observed experimental facts formally Mn 3+/Mn 4+ perovskites. Extension: confinement of de-localised electron in ferromagnetic supraatomic units, …. CO in manganites Order of Zener polarons? Implication for orbital ordering pseudo-molecular orbitals Implication for super-exchange theory extension of GKA rules Single crystal x-rays and neutron diffraction to obtain accurate structures Polarized neutron scattering (3 D-polarimetry): magnetic structures Electron density studies Electronic structure calculations Barcelona, September 2005, MULTIMAT

Access to the LLB neutron beams Barcelona, September 2005, MULTIMAT

Access to the LLB neutron beams Barcelona, September 2005, MULTIMAT

Barcelona, September 2005, MULTIMAT

Barcelona, September 2005, MULTIMAT

Barcelona, September 2005, MULTIMAT

The end Full. Prof Suite and related programs: ftp: //ftp. cea. fr/pub/llb/divers/ Set of directories with different programs, documents, tutorials and examples of powder diffraction data analysis Downloading of Software http: //www. ccp 14. ac. uk Graphical tutorial run-through of most of this software is located via (“look before you try”): http: //www. ccp 14. ac. uk/tutorial/ Barcelona, September 2005, MULTIMAT