MATTER Ch 8 Solids Liquids Gases I States



















- Slides: 23

MATTER Ch. 8 - Solids, Liquids, & Gases I. States of Matter (p. 214 -220) w Kinetic Theory of Matter w Four States of Matter w Thermal Expansion

A. Kinetic Theory of Matter ö KTM w Tiny, constantly moving particles make up all matter. w The kinetic energy (motion) of these particles increases as temperature increases.

States of Matter ö Can by classified by their properties w Ability to flow w Ability to change shape w Ability to change volume w Movement of molecules

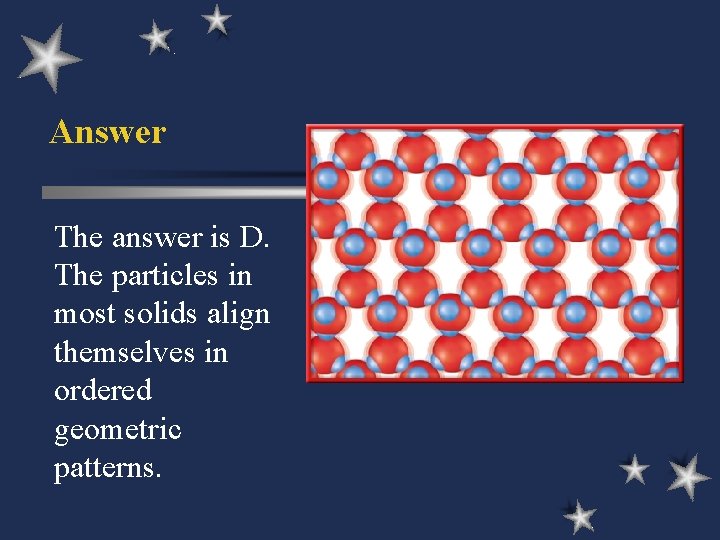
B. Four States of Matter ö Solids w low KE - particles vibrate but can’t move around w definite shape & volume w crystalline - repeating geometric pattern w amorphous - no pattern (e. g. glass, wax)

Water as a crystaline solid Ice

B. Four States of Matter ö Liquids w higher KE - particles can move around but are still close together w indefinite shape Ø Takes the shape of container w definite volume

B. Four States of Matter ö Gases w high KE - particles can separate and move throughout container w indefinite shape & volume

B. Four States of Matter ö Plasma w very high KE - particles collide with enough energy to break into charged particles (+/-) w gas-like, indefinite shape & volume w stars, fluorescent light bulbs, TV tubes

Example of Plasma


States of Matter Clip Show States of Matter. flv

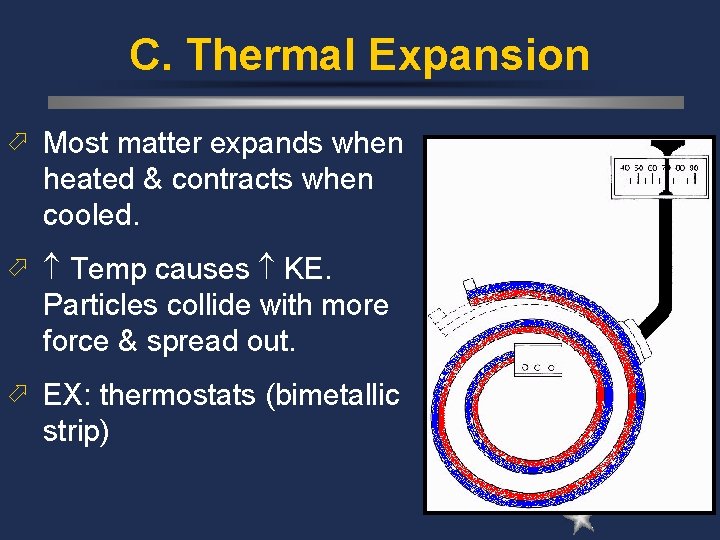
C. Thermal Expansion ö Most matter expands when heated & contracts when cooled. ö Temp causes KE. Particles collide with more force & spread out. ö EX: thermostats (bimetallic strip)

Question 1 Most _____ materials have a specific type of geometric arrangement. A. B. C. D. gaseous inert liquid solid

Answer The answer is D. The particles in most solids align themselves in ordered geometric patterns.

Question 1 Which is represented in the diagram? A. B. C. D. solid liquid gas plasma

Answer The answer is A. The particles in a solid are packed together tightly.


Solids, Liquids, and Gases The matter that surrounds you is either a solid, liquid, or gas. Make the following Foldable to help you organize information about solids, liquids, and gases.

Fold a sheet of paper in half lengthwise. Make the back edge about 5 cm longer than the front edge.

Turn the paper so the fold is on the bottom. Then fold it into thirds.

Unfold and cut only the top layer along both folds to make three tabs.

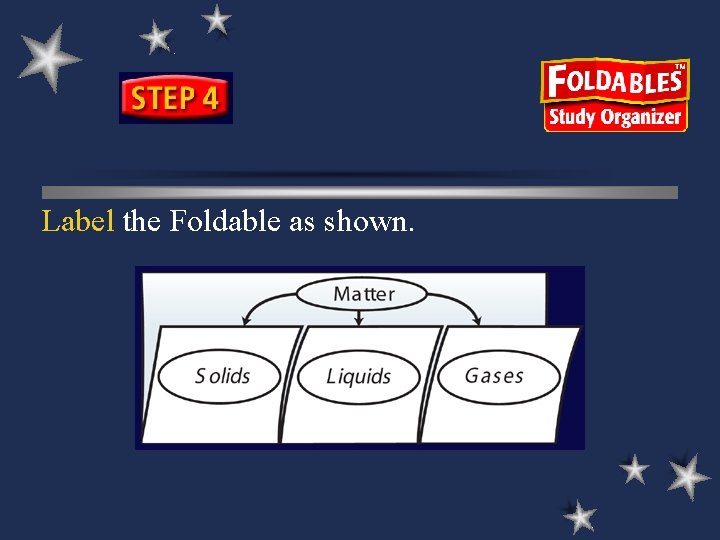
Label the Foldable as shown.

Read for Main Ideas As you read the chapter, list the characteristics of solids, liquids, and gases under the appropriate tab. List some examples of each under the tab also.