STATES of MATTER Compare solids liquids and gases







- Slides: 10

STATES of MATTER

Compare solids, liquids and gases in terms of: Distribution of particles Volume Density Compressibility Motion of particles Intermolecular forces Kinetic energy

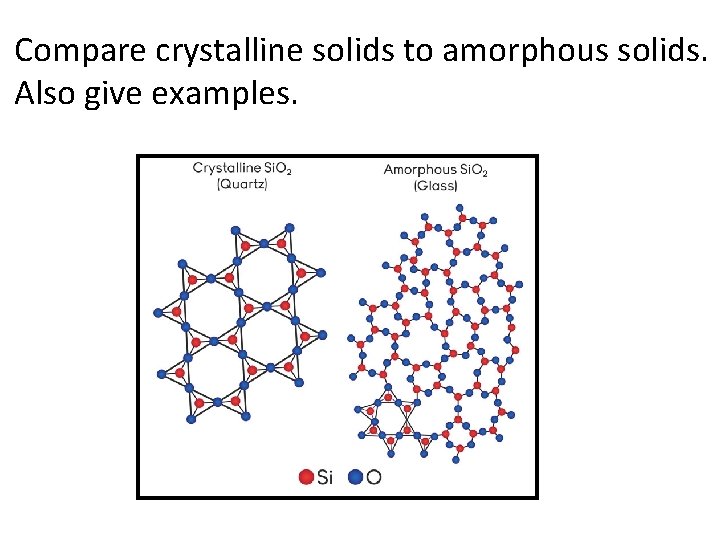
Compare crystalline solids to amorphous solids. Also give examples.

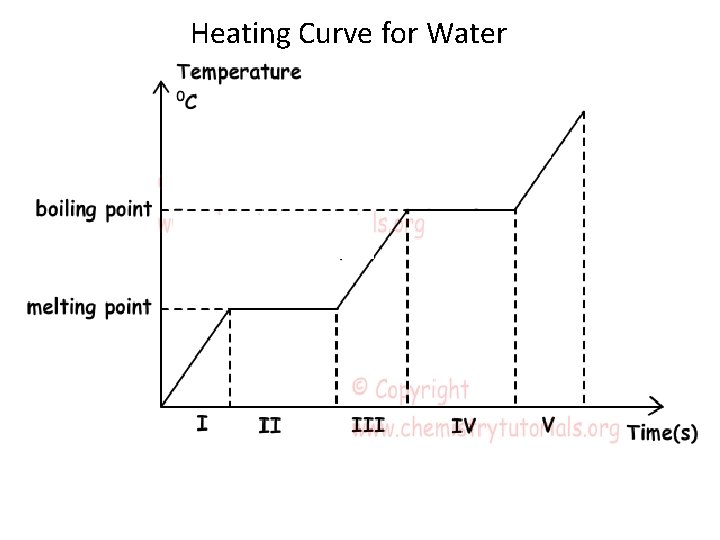
When a substance absorbs heat (energy), what are two things that can happen? 1. The substance can experience a rise in temperature. The heat (energy) causes the particles to move faster thereby increasing the average KE. This increase in average kinetic energy correlates to an increase in temperature. 2. The substance can change state (Melting or boiling). The energy absorbed is used to break the intermolecular forces; that is, the energy absorb is used to change the state of matter. This change does not cause a rise in temperature.

Heating Curve for Water

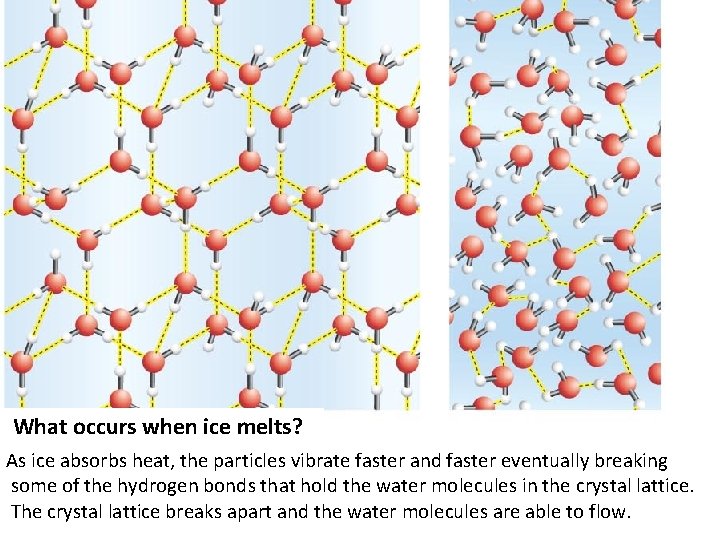
What occurs when ice melts? As ice absorbs heat, the particles vibrate faster and faster eventually breaking some of the hydrogen bonds that hold the water molecules in the crystal lattice. The crystal lattice breaks apart and the water molecules are able to flow.

Liquid vaporization Gas Vaporization is the conversion of a liquid to a gas or vapor. What are two types of vaporization? Explain each.

Evaporation is the process by which particles escape from the surface of a non-boiling liquid and enter the gas state. Evaporation occurs because the particles of a liquid have different kinetic energies. Some surface particles with higher-than-average energies move faster and are able to overcome the intermolecular forces that bind them to the liquid. They can then escape into the gas state. Bromine evaporating

Boiling Heating a liquid increases the average kinetic energy of the particles and the temperature increases. The liquid is heated until all the particles in the liquid have enough kinetic energy to vaporize, the liquid begins to boil. Bubbles of vapor(gas) form throughout the liquid, rise to the surface, and escape into the air. The boiling point is the temperature at which the vapor pressure of the liquid is just equal to the external pressure on the liquid.

Explain why the boiling point of water is different at different altitudes. Water boils when the vapor pressure of water is just equal to the atmospheric pressure. At higher altitudes….