Fig 14 29 Nucleotide degradation PurineAMPGMPuric acid Pyrimidine












- Slides: 34


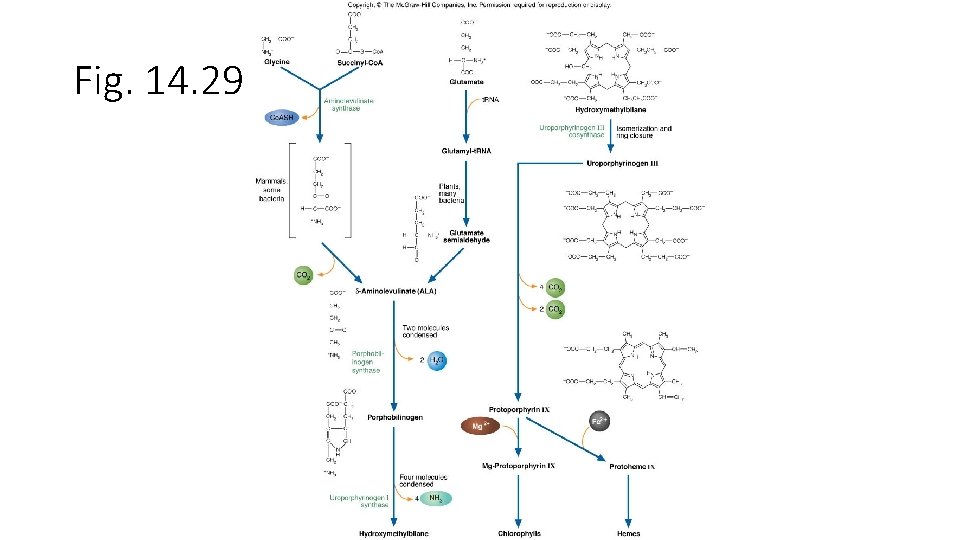
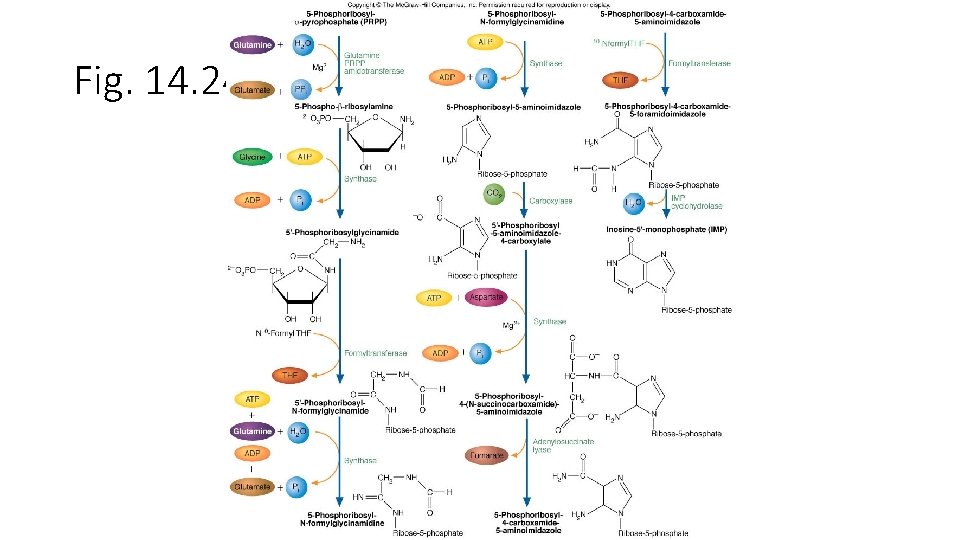
Fig. 14. 29



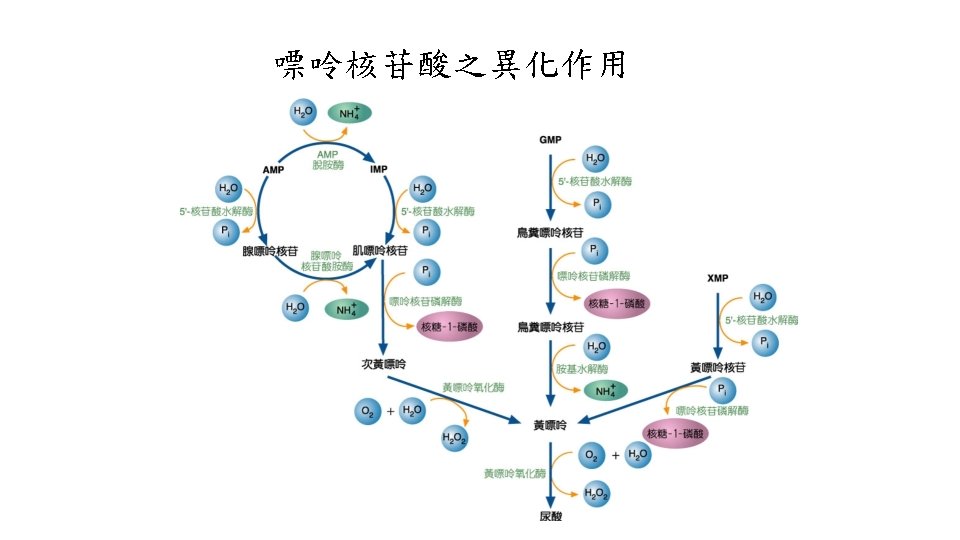
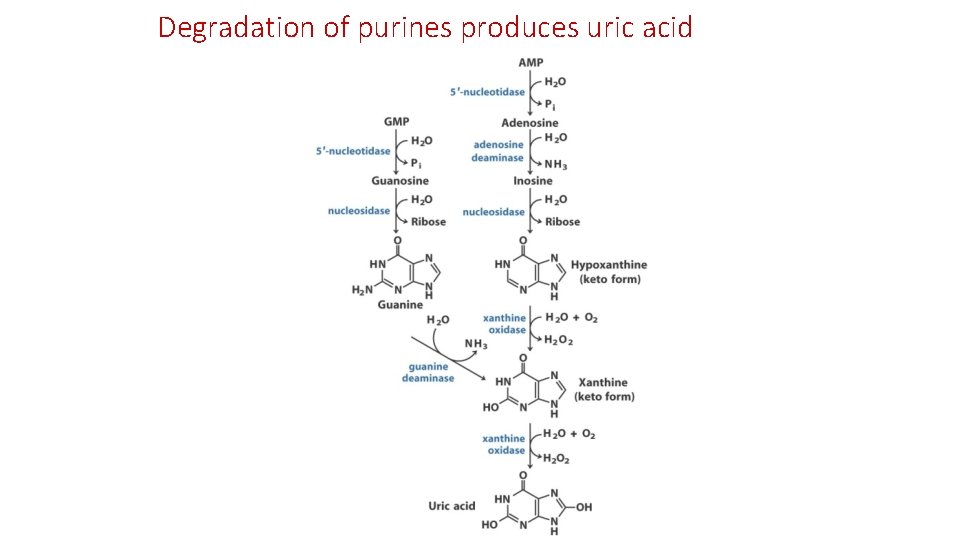
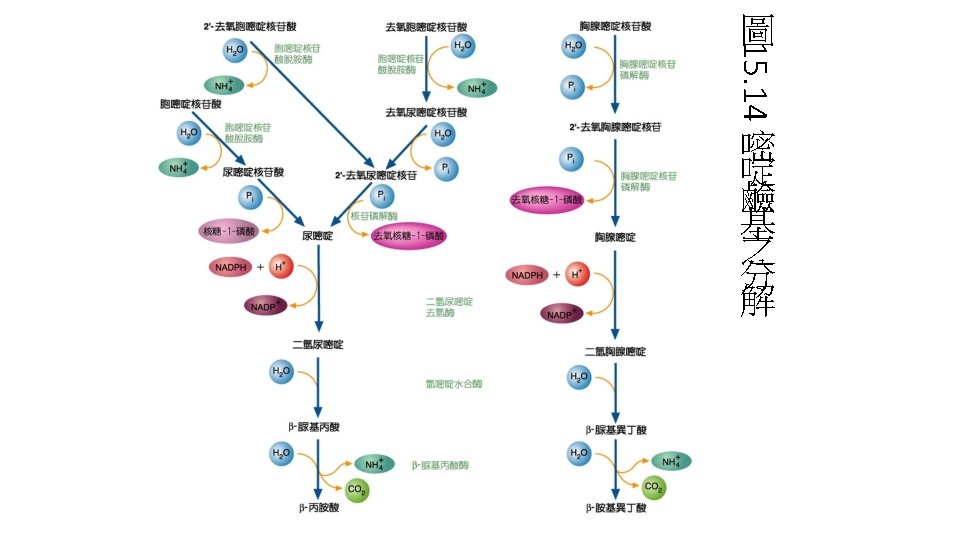
核苷酸分解(Nucleotide degradation) • 在人類與鳥類 • Purine(AMP、GMP)分解的終產物是uric acid • Pyrimidine被分解成 -alanine或 -aminoisobutyric acid • cytidine、uridine -alanine • thymine -aminoisobutyric acid


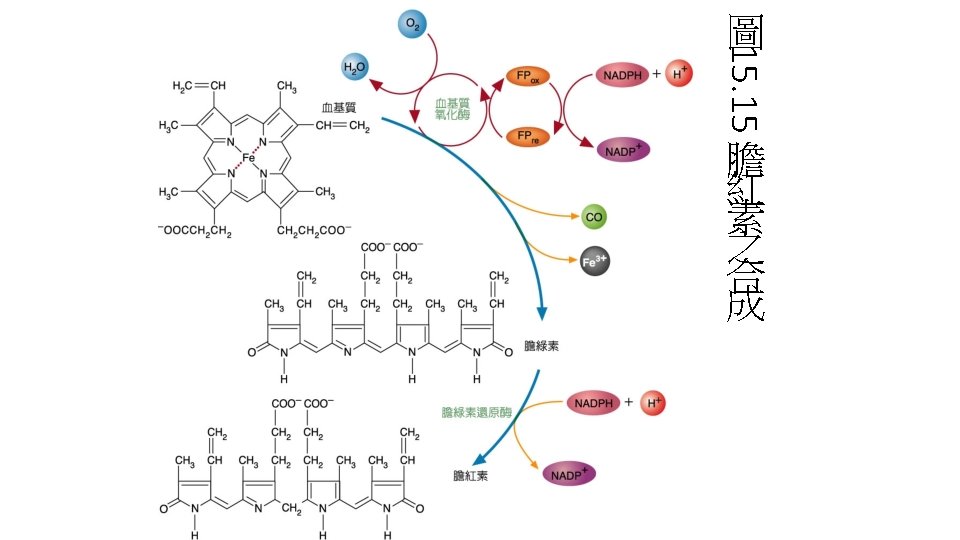
Degradation of purines produces uric acid



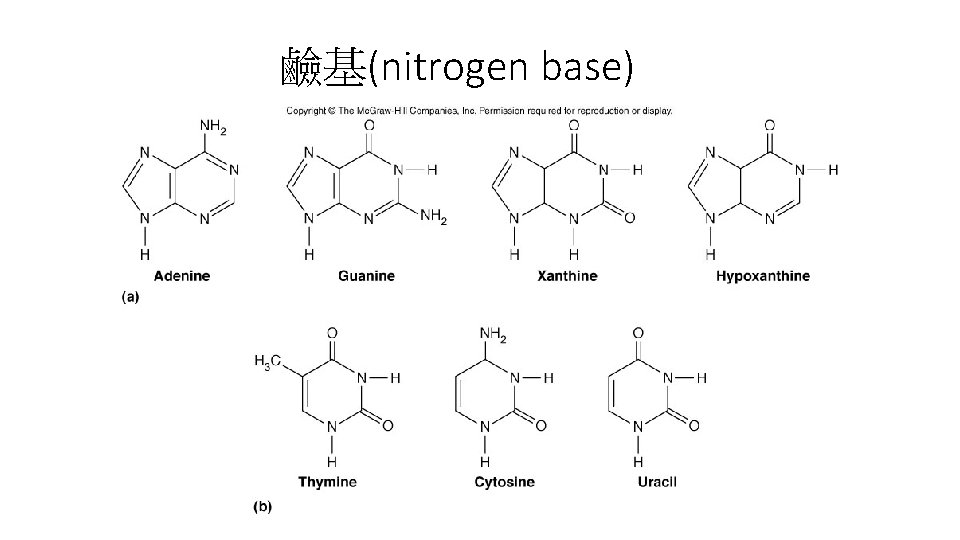
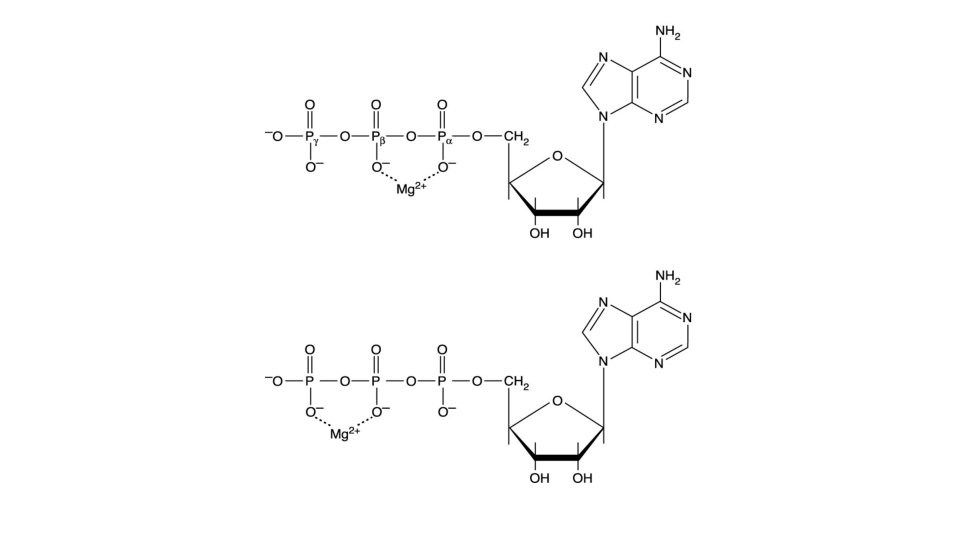
核苷酸(Nucleotide)合成 • Nucleotide: 含三個部分: 一個氮鹼基、一個五碳糖與一 個、二個或三個磷酸。 • 氮鹼基: 含嘌呤(purine)或嘧啶(pyrimidine),由具有類 苯環(aromatic ring)的結構,因此在p. H 7時對260 nm的 UV光具有吸收性。 • Purine: 含adenine、guanine、xanthine與hypoxanthine。 • Pyrimidine: 含thymine、cytocine與uracil

鹼基(nitrogen base)



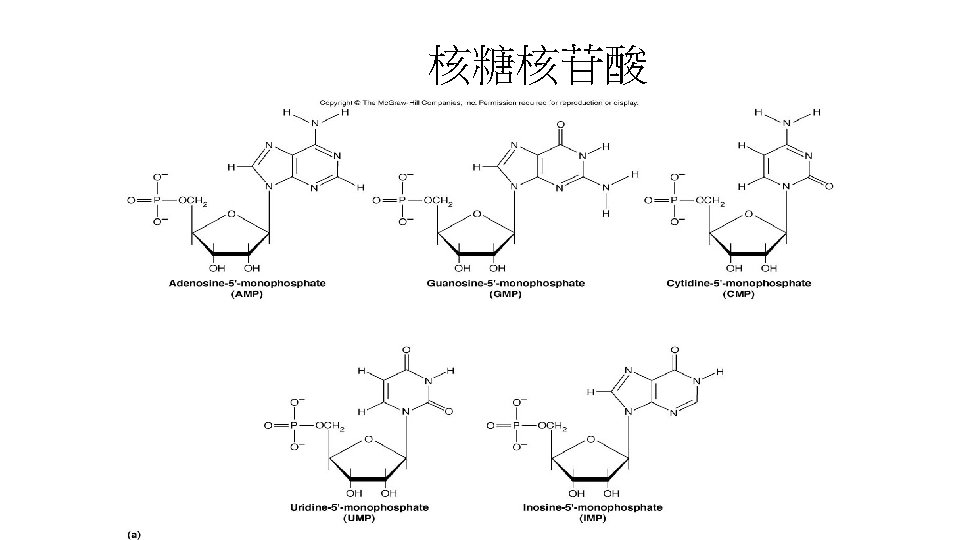
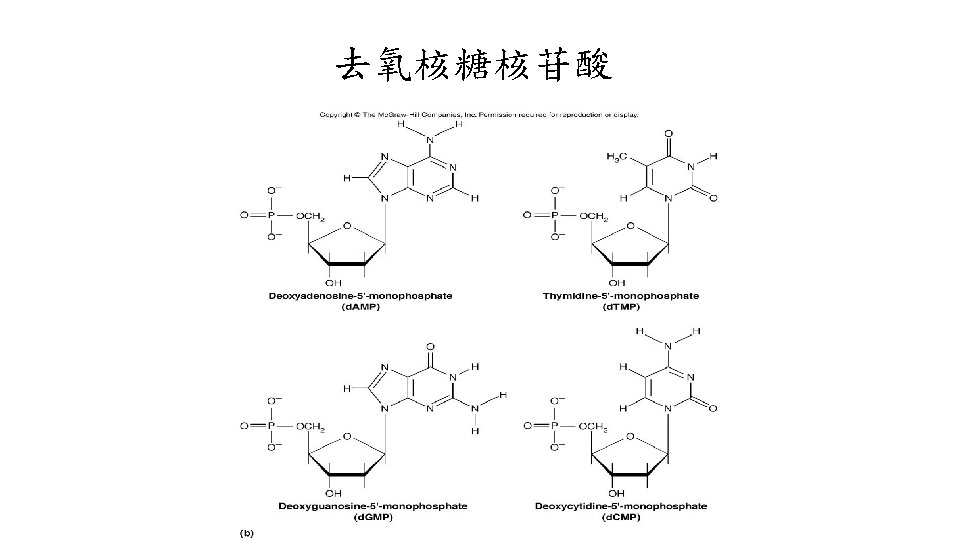
核苷(Nucleosine) • 五碳糖分成兩類: 核糖(ribose)與去氧核糖 (deoxyribose) • Nucleosine: 不接磷酸的核苷。 • purine或pyrimidine接在五碳糖的C-1的部位,以 -NGlycosidic形式接合。 含ribose的nucleiside: adenosine、guanosine、 cytosine與uridine。 • 含deoxyribose的nucleoside: deoxyadenosine、 deoxyguanosine、deoxycytosine與deoxythyminosine。



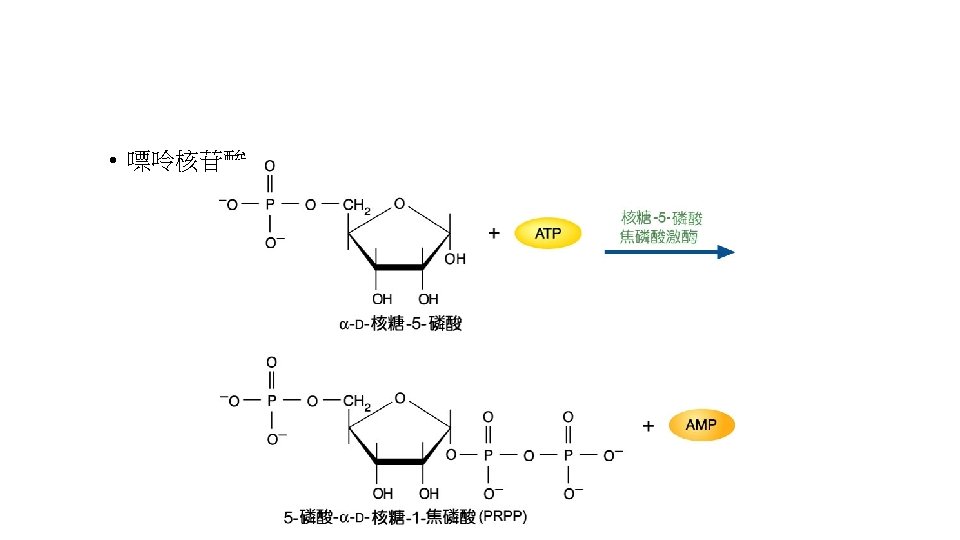
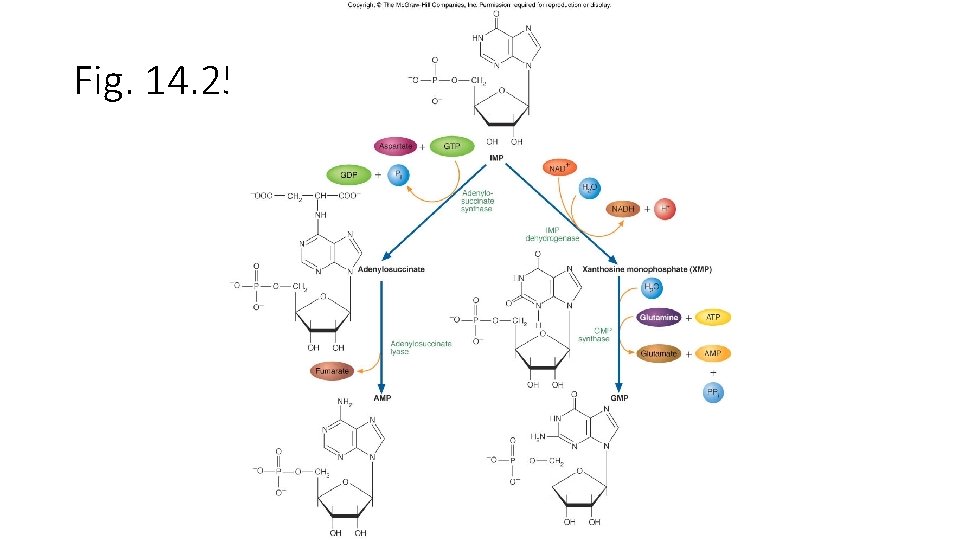
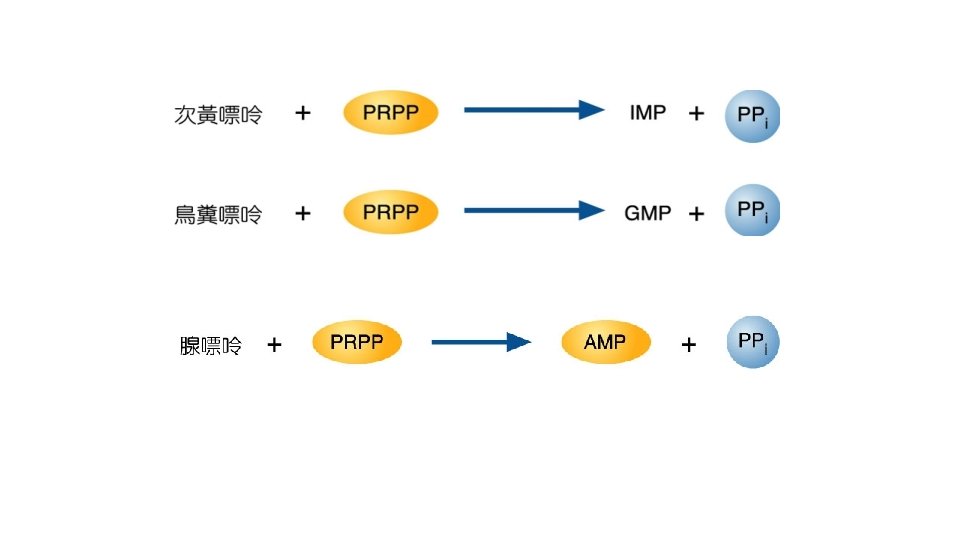
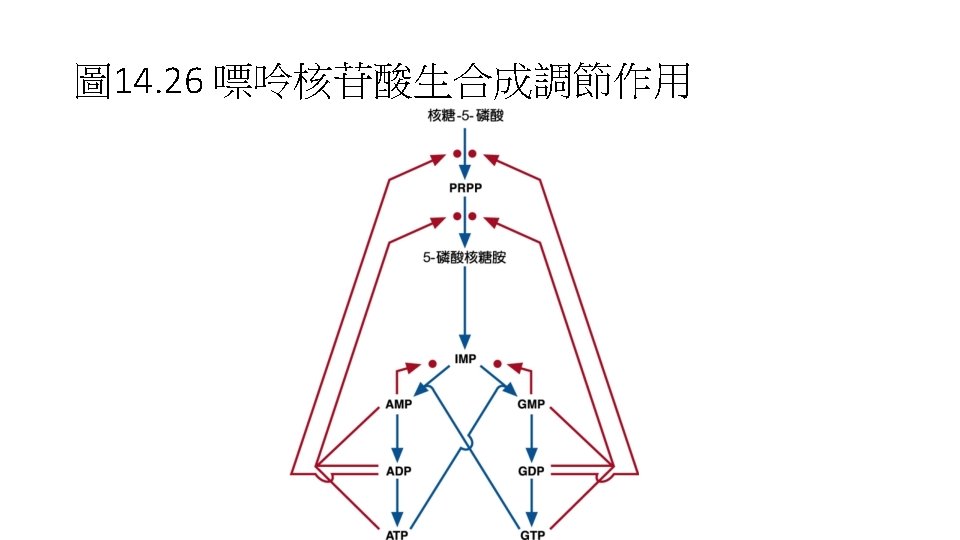
• Purine nucleotide合成 • -D-ribose-5 -phosphate + ATP PRPP + AMP • -D-ribose-5 -phosphate是來自Pentose phosphate pathway。反應是由PRPP synthetase,再經過9個步驟後 形成IMP,IMP再合成AMP或GMP。 • AMP + ATP 2 ADP • NMP + ATP NDP + ADP • 再由Nucleotide diphosphate kinase合成NTP • N 1 DP + N 2 TP N 1 TP + N 2 DP


Fig. 14. 24

Fig. 14. 25




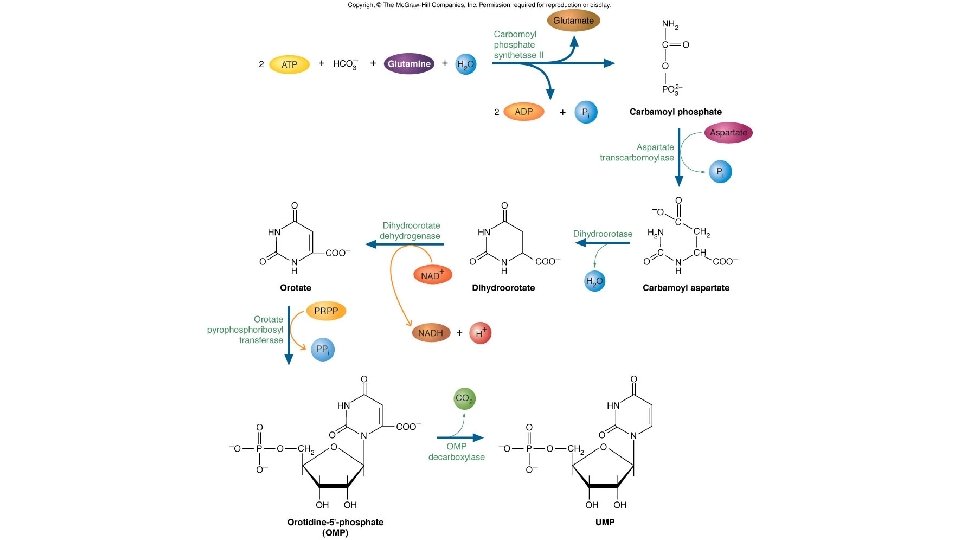
Pyrimidine nucleotides的合成 • Carbamoyl phosphate synthetase II位於細胞質中催化 生成Carbamoyl phosphate反應如下: • 是合成pyrimidine途徑的第一反應,也是關鍵酵素。 • Carbamoyl phosphate synthetase I在粒線體中,是合 成Urea第一個反應。

Fig. 14. 27

• pyrimidine的合成與purine不一樣,先合成出乳清酸 (orotate) • orotate + PRPP OMP UMP • • UMP是合成CTP的先驅物 • UMP UTP CTP •


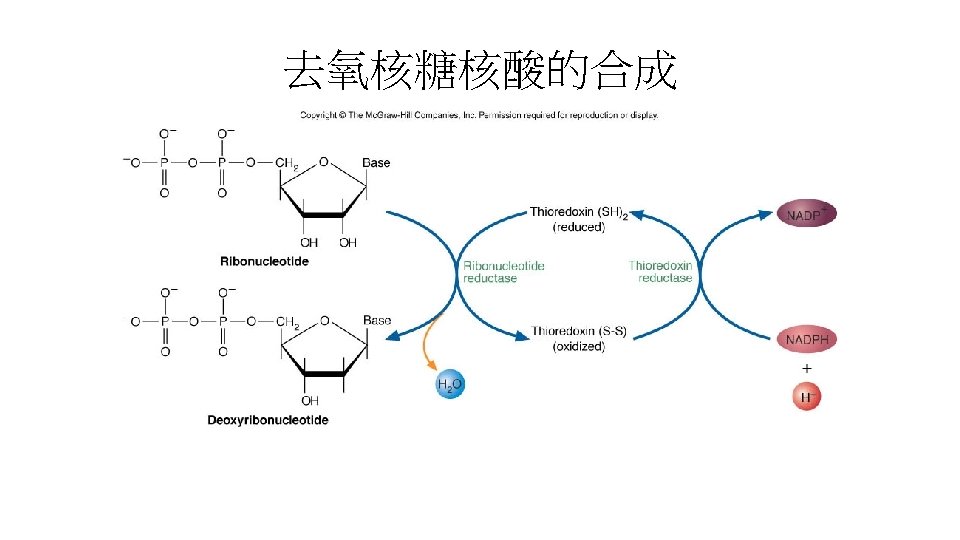
• Deoxyribonucleotides的合成: • 以上所提及都是ribonucleotide(A、G、U與C)是組成RNA的 建材分子(building block)。 • 而DNA的合成是由 2’-deoxyribonucleotide(A、T、C、G) 組成。 • 2’-deoxyribonucleotide是由NDP(nucleotide diphosphate)還原合成而來。 反應是由還原態的Ribonucleotide reductase催化, Thioredoxin reductase(13 k. Da NADPH是其輔因子)再催化氧 化態的Ribonucleotide reductase成還原態。

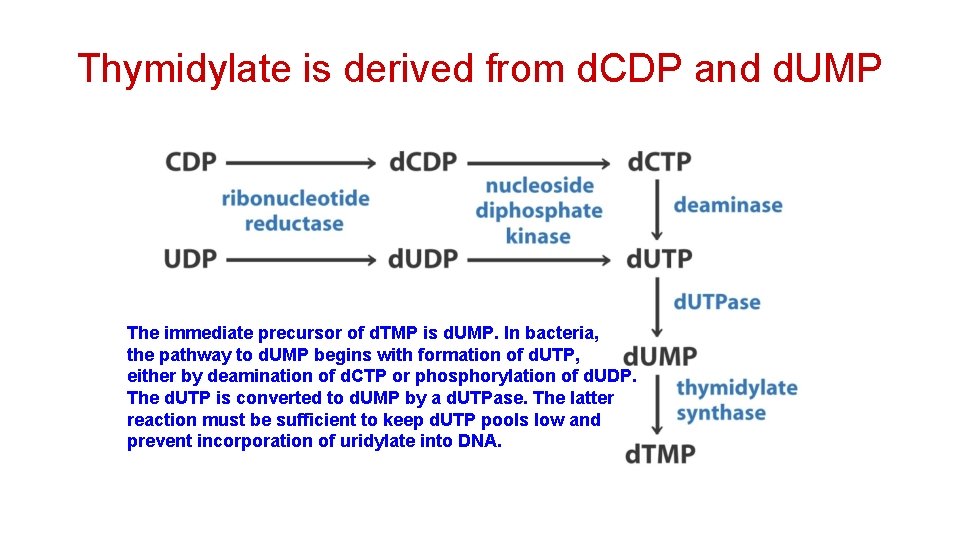
Thymidylate is derived from d. CDP and d. UMP The immediate precursor of d. TMP is d. UMP. In bacteria, the pathway to d. UMP begins with formation of d. UTP, either by deamination of d. CTP or phosphorylation of d. UDP. The d. UTP is converted to d. UMP by a d. UTPase. The latter reaction must be sufficient to keep d. UTP pools low and prevent incorporation of uridylate into DNA.

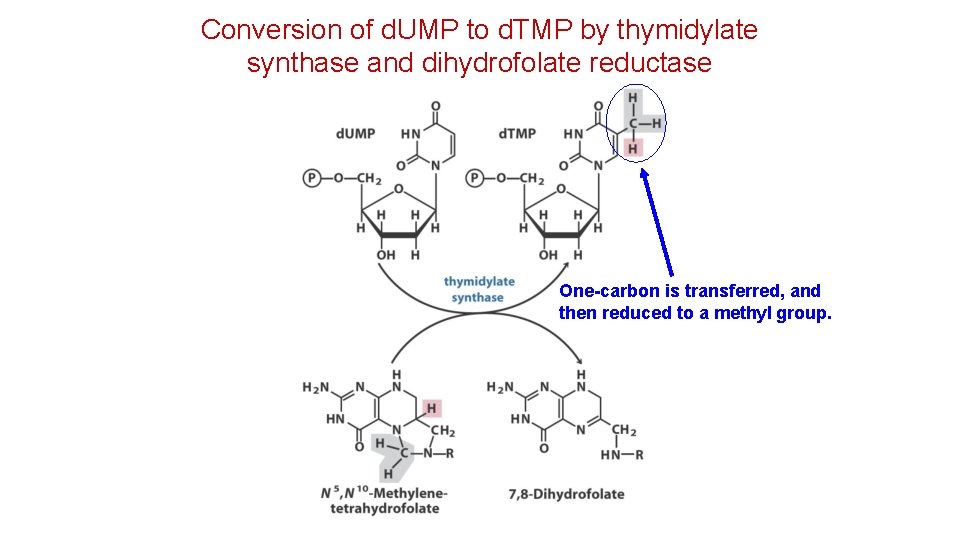
Conversion of d. UMP to d. TMP by thymidylate synthase and dihydrofolate reductase One-carbon is transferred, and then reduced to a methyl group.


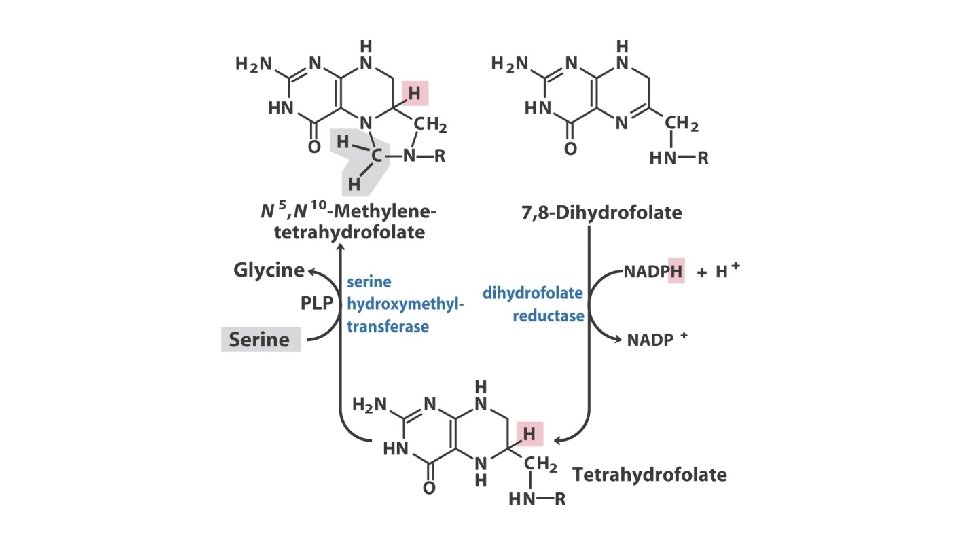
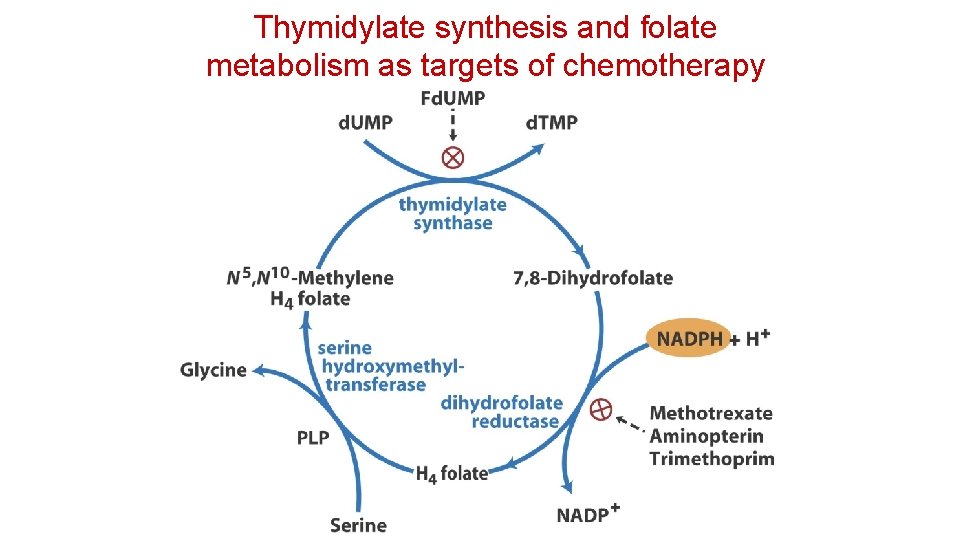
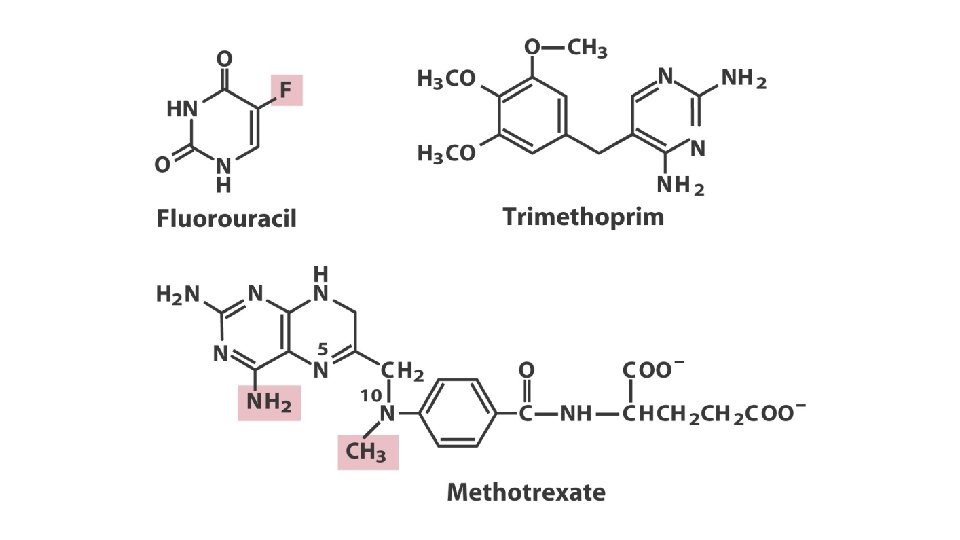
Thymidylate synthesis and folate metabolism as targets of chemotherapy • Another prominent chemotherapeutic agent, methotrexate, is an inhibitor of dihydrofolate reductase. • This folate analog acts as a competitive inhibitor; the enzyme binds methotrexate with about 100 times higher affinity than dihydrofolate. • Aminopterin is a related compound that acts similarity.

Thymidylate synthesis and folate metabolism as targets of chemotherapy
