DIETARY MINERALS Microminerals An Overview n Inorganic atoms





































- Slides: 44

DIETARY MINERALS

Microminerals : An Overview n �� Inorganic atoms or molecules n �� Microminerals or trace elements n �� < 100 mg/day needed

Bioavailability & Regulation of Trace Minerals n Bioavailability influenced by: ¨ Genetics ¨ Nutritional status ¨ Nutrient interactions ¨ Aging Absorbed in small intestine n Circulated in blood n Deficiencies & toxicities rare n ¨ Except genetic disorders & environmental exposure

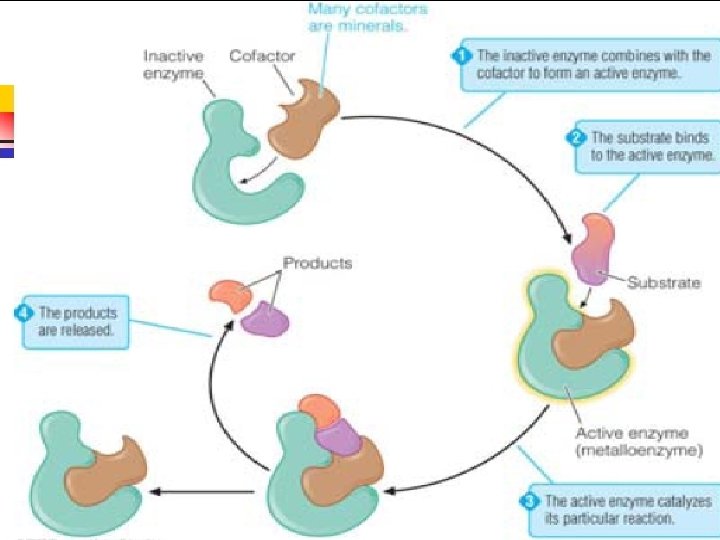
Functions of Trace Minerals in the Body Cofactors ¨Metalloenzyme n Components of nonenzymatic molecules n Provide structure to mineralized tissues n


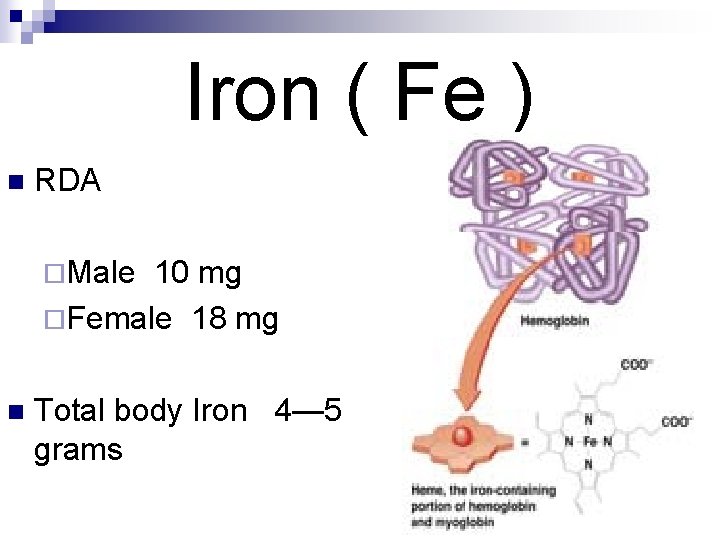
Iron ( Fe ) n RDA ¨Male 10 mg ¨Female 18 mg n Total body Iron 4— 5 grams

Dietary Sources n Animal Sources ¨ Meat, liver, kidney, egg yolk, fish n Plant Sources ¨ Green leafy vegetables, spinach, nuts, dates, beans

Iron in the Body 70% of iron in body is functional; found in enzymes and other molecules n ¨ �� >80% of this found in red blood cells 30% of iron is in storage depots or transport proteins n Iron absorption, transport, storage and loss is highly regulated n

Distribution of Iron in Body 1. n n n 2. n n Functional Iron Hb 900 grams contains 3 grams of iron Mb 40 grams contains 0. 13 grams of iron Enzymes 10— 20 grams contains 0. 04– 0. 08 grams of iron Storage iron Transferrin contains 0. 04 grams of iron Ferritin contains 0. 4– 0. 8 grams of iron

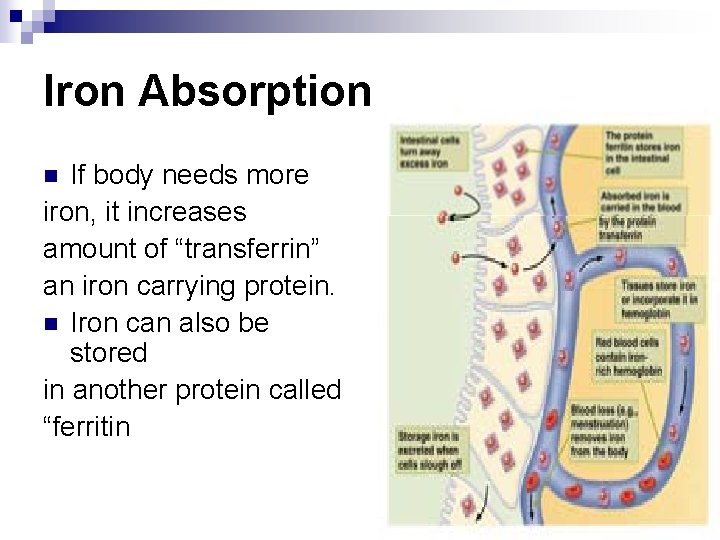
Iron Absorption If body needs more iron, it increases amount of “transferrin” an iron carrying protein. n Iron can also be stored in another protein called “ferritin n

Iron Absorption n Transport across ¨ �� Brush border ¨ �� Basolateral membrane n Heme iron ¨ �� Chemical modification not needed n Nonheme iron ¨ �� Reduced n Ferritin to ferrous form

Absorption, cont. Iron from animal sources much better absorbed than that from plant sources n Absorption of iron from plant sources increased by ¨ �� Vitamin C ¨ �� Meat in diet n Absorption is decreased by ¨ �� Phytates (grain products) ¨ �� Polyphenols (tea, coffee) ¨ �� Other minerals (calcium, zinc) n

Effect of Iron Status on Iron Absorption n Iron deficiency ¨�� Increases production of transport proteins ¨�� Decreases ferritin production n Adequate or excess iron ¨�� Decreases production of transport proteins

Iron Circulation, Uptake Into Cells, & Storage n Iron storage compounds in liver, bone marrow, and spleen ¨Ferritin n �� Main storage form ¨ Hemosiderin n �� Long-term storage

Bioavailability of Iron Influenced by: n Form ¨ �� Heme > Ferrous > Ferric n Iron status n Presence/absence of other dietary components n Only 10 % of dietary iron is absorbed

Excretion of iron Mostly in feces n Small amount in sweat n Very small amount in urine n Average excretion n ¨< I mg/ day in males ¨ 1. 5 – 2 mg /day in females n In nephrotic syndrome excretion increases

Functions of Iron n �� Oxygen transport: hemoglobin n �� Iron reservoir: myoglobin n �� Cellular energy metabolism

Oxygen Transport: Hemoglobin �� Most abundant protein in red blood cells n �� 4 protein subunits + 4 iron-containing heme groups n �� Delivers oxygen to cells n �� Picks up carbon dioxide n

Iron Reservoir: Myoglobin �� Found in muscle cells n �� Heme group + protein subunit n �� Releases oxygen to cells when needed for: n ¨ �� ATP production ¨ �� Muscle contraction

Cellular Energy Metabolism n �� Cytochromes ¨ �� Heme-containing complexes ¨ �� Function in electron transport chain ¨ �� Allow conversion of ADP to ATP n �� Iron as cofactor ¨ �� Electron transport chain ¨ �� Citric acid cycle ¨ �� Gluconeogensis

Other Roles of Iron Cytochrome P 450 enzymes n Cofactor for antioxidant enzymes ¨�� Protects DNA, cell membranes, proteins n Cofactor for enzyme to make DNA n

Iron Deficiency Most common nutritional deficiency n At-risk groups ¨�� Infants, growing children, pregnant women n

Mild Iron Deficiency n �� Signs ¨ �� Fatigue ¨ �� Impaired physical work performance ¨ �� Behavioral abnormalities ¨ �� Impaired intellectual abilities in children ¨ �� Body temperature regulation ¨ �� Influences immune system

Severe Iron Deficiency: Iron. Deficiency Anemia �� Microcytic hypochromic anemia ¨ �� Small, pale red blood cells ¨�� Inability to produce enough heme ¨�� Decreased ability to carry oxygen ¨�� Decreased ATP synthesis n �� Infants, children, pregnant and lactating women most at risk. n

Iron Toxicity Excess deposited in liver, heart, muscles n Most common cause of childhood poisoning n Symptoms n ¨ Vomiting, diarrhea, constipation, black stools ¨ �� Death

Iron Toxicity n Hemosiderosis ¨ Microscopically visible form is hemosiderosis ¨ Iron overload without tissue injury n Hemochromatosis ¨ Macroscopically visible form ¨ Iron overload with tissue injury ¨ Bronze diabetes n Bronze pigmentation of skin and tissues

Copper ( Cu ) n Dietary sources ¨ Oyster ¨ Nuts ¨ Legumes ¨ Mushrooms ¨ Meat ¨ Dry fruit ¨ Potatoes

Absorption, Metabolism, & Regulation of Copper Absorbed in small intestine & stomach as free and bound Cu n Amino acids bound Cu is also absorbable n Influenced by Cu status n ¨ Body stores and requirement ¨ Form : Cupric or Cuprous Excess incorporated into bile & eliminated in feces n

Absorption, Metabolism, & Regulation of Copper n Bioavailability decreases with ¨ Antacids ¨ Iron ¨ Ca ¨ Zn

Transport of Cu Ceruloplasmin 96 % n Remaining with albumin and globulin n n Functions of ceruloplasmin ¨ Cu transport ¨ Conversion of iron from ferrous to ferric form

Functions of Copper Needed to absorb and utilize iron n Part of anti oxidant enzyme superoxidase dimutase n ATP synthesis n ¨ Cytochrome n C oxidase Connective tissue synthesis

Excretion Unabsorbed in feces n Bile n Sweat n Urine n Menstrual blood n

Copper Deficiency & Toxicity n Deficiency ¨ �� Hospitalized patients & preterm infants ¨ �� Antacids n Signs & Symptoms ¨ �� Defective problems n �� Toxicity ¨ �� Rare connective tissue, anemia, neural

Copper Deficiency n Menke’s Disease ¨ X-linked recessive genetic disorder ¨ Poor copper absorption ¨ Steely hair syndrome ¨ Growth retardation

Copper Deficiency n Wilson’s disease ¨ Autosomal recessive disorder ¨ Increased Cu absorption ¨ Liver Cu level increases ¨ Excessive urinary excretion of Cu ¨ Serum Cu level decreases ¨ Cu accumulates in liver , brain and kidney ¨ CNS lesions with muscular incoordination

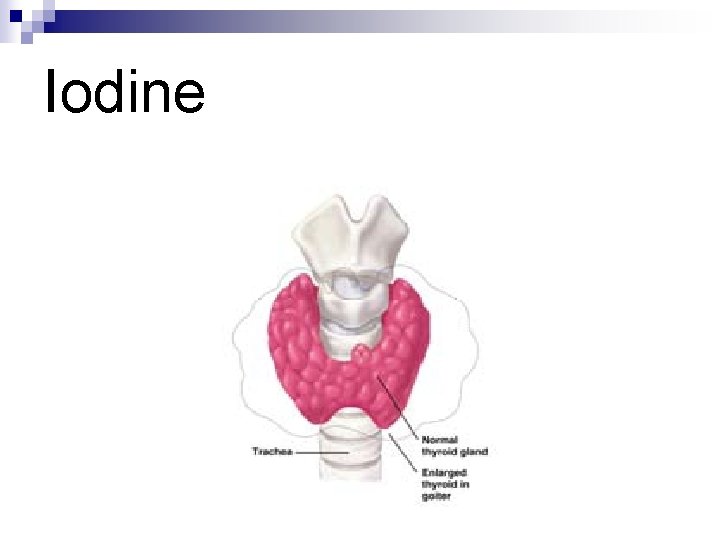
Iodine

Iodine Essential for the formation of thyroid hormones n Total iodine content 20— 40 mg ( 20 % in thyroid gland) n Plasma iodide level 0. 3— 0. 5 micro gm/ dl n Daily intake 500 micro gm n Almost all absorbed n Remaining excreted in urine n

Dietary Sources Seafood n Milk/dairy products n Iodized salt n Cod liver oil n

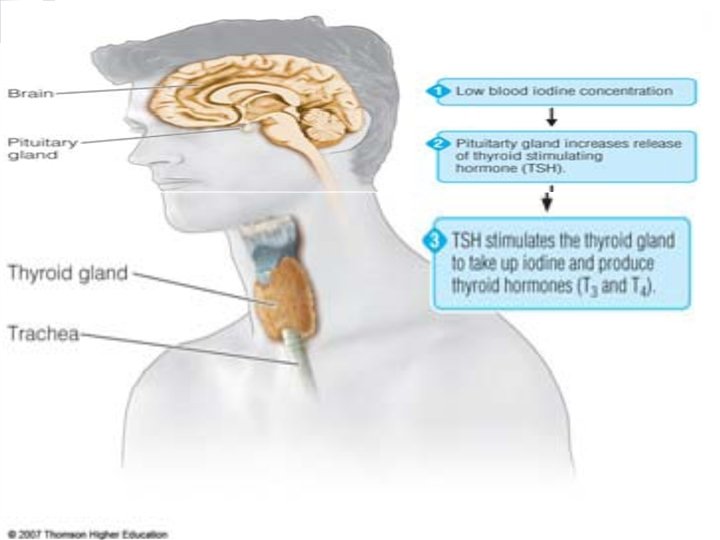
Absorption, Metabolism, &Regulation of Iodine Absorbed in small intestine & stomach n Also absorbed in lungs, mucous membrane and skin n Taken up by thyroid gland n Thyroid-stimulating hormone regulates uptake n


Functions of Iodine n Component of: ¨ �� Thyroxine (T 4) ¨ �� Triiodothyronine (T 3) Regulates energy metabolism, growth, development n Signs of deficiency n ¨ �� Severe fatigue ¨ �� Lethargy

Iodine Deficiency n Goiter (less severe) ¨ �� Enlarged thyroid gland due to body’s attempt to increase thyroid hormone production

Iodine Deficiency n Cretinism (more severe) ¨ �� Severe iodine deficiency during pregnancy, serious problems in baby ¨ �� Stunted growth, deaf, mute, mentally retarded

Iodine Toxicity n �� Hypothyroidism �� Hyperthyroidism ( Thyrotoxicosis ) n �� Formation of goiters n