Inorganic gaseous pollutants IAQ Introduction Inorganic gaseous pollutants



























- Slides: 31

Inorganic gaseous pollutants (IAQ)

Introduction Ø Inorganic gaseous pollutants are the major contributors to Indoor quality problems Ø Combustion appliances and tobacco smoke are the major sources of gases along with other types of pollutants Ø These Inorganic gaseous pollutants include Carbon monoxide. Sulfur dioxide. Nitrous oxide. Nitrogen dioxide.

Factors effecting concentration. Ø The emission patterns and concentrations of the pollutants depend on Following factors: v Types of Fuels used v Combustion efficiency v Appliance design v Ventilation system v Operating conditions v Maintenance v Frequency of use

Appliances based on Fuel type and their application Ø Un-vented space heaters Ø Wood burning stoves v Furnaces v Fire places

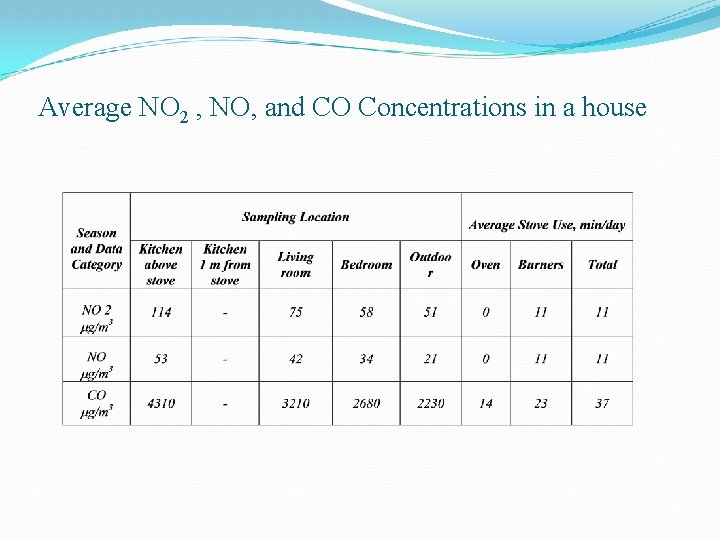
Behavior of combustion appliances Ø Un-vented space heaters v In this type of device the pollutant concentration is maximum within the first hour of furnace operation v The concentrations come to a standard steady state as the device is shut down Ø Wood burning appliances v These appliances emit pollutants intermittently and depend completely on the type of wood used v The other pollutants released due to the devices of this type include organic compounds (like HCHO) v The indoor to outdoor ratio of these Inorganic gaseous pollutants can be well over unity

Average NO 2 , NO, and CO Concentrations in a house

Health Effects and standards

Health Effects of CO 2 Ø The ratio of indoor to outdoor concentration of CO 2 is generally greater than ‘ 1’ Ø The concentration level of CO 2 to cause any serious health problems in the range of 30, 000 ppm Ø Respiration is effected at concentration levels from and above 15, 000 ppm Ø Above 30, 000 ppm it can cause headaches, dizziness and nausea Ø Prolonged exposure to concentration above 5000 ppm can increase the incidence of illness Ø The concentration of 5000 ppm is adopted as TLV for the submarines

Health Effects of CO Ø Health effects due to CO include loss of alertness, impaired perception, learning disorders, sleep deprivation, drowsiness and confusion Ø Health effects due to prolonged exposure to low concentration have been controversial, but acute illness and deaths have been reported Ø CO combines with hemoglobin and myoglobin to stop the supply of oxygen to tissues affecting brain, myocardium and muscle tissues Ø Carboxyhemoglobin cause severe health effects at various percentage levels in blood v Loss of vigilance ability @ 3% - 5% v Loss of hand to eye co-ordination @6% - 10%

Health Effects of NO and NO 2 Ø At high level there can be very serious health effects like coma and eventual death Ø NO and NO 2 are the most reactive species of the nitrogen oxides Ø They combine with other indoor pollutants to form complex toxic substances Ø React with amines like benzo(a)pyrene and pyrelene to form carcinogenic nitrosoamine and mutagens

Health Effects of SO 2 and Cl 2 Ø SO 2, another pollutant found indoor at 0. 25 – 0. 5 ppm can cause bronchioconstriction in exercising asthmatics Ø Short-term exposure to the low level of SO 2 can cause irritation Ø Long term exposure can cause functional changes in lung making the body susceptible to damage by other pollutants Ø Cl 2 gas another gas found when two cleaning agents are mixed together one containing acidic compound another sodium hypochlorite Ø Cl 2 gas can cause coughing, breathlessness, irritation of upper airways, bronco spasms, nauseas and vomiting

Sources and Indoor Concentrations

Sources Unvented space heaters Ø The unvented gas heater is one of the major sources of pollutants like Inorganic gaseous pollutants Ø This device can be classified into two categories v Gas space heaters – heaters of this type use gas as fuel v Kerosene space heaters - heaters that use kerosene as fuel

Classification of Kerosene space heaters Ø Convective Ø Radiant Ø Convective / Radiant Ø Two stage Ø Wickless

Factors affecting Emission rates Ø Emission rates of pollutants from kerosene space heaters depends on the following factors v Usage pattern v Type of heater v Type of fuel v Age of heater

Significant emissions from Kerosene space heaters Ø All heaters emit NO, NO 2, CO 2, respirable particles and formaldehyde Ø Emission rate of CO 2 depends on type of fuel used and duration of burning Ø The average CO 2 emissions rates are: v 49, 8000 +/- 2400 mg/Kg from natural gas heaters v 60, 500 +/- 1200 mg/Kg from a propane heater v 70, 000 mg/Kg from a kerosene heater Ø CO, NO 2 and formaldehyde were emitted in various conditions in majority of the experiments

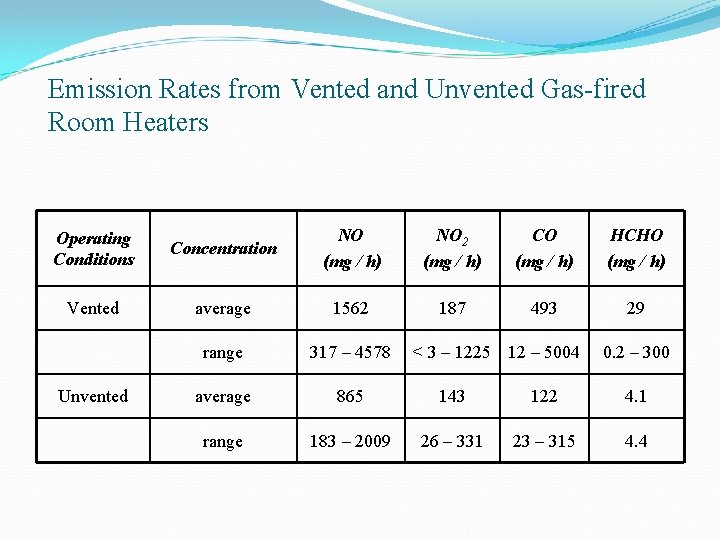
Emission Rates from Vented and Unvented Gas-fired Room Heaters Operating Conditions Concentration NO (mg / h) NO 2 (mg / h) CO (mg / h) HCHO (mg / h) Vented average 1562 187 493 29 range 317 – 4578 average 865 143 122 4. 1 range 183 – 2009 26 – 331 23 – 315 4. 4 Unvented < 3 – 1225 12 – 5004 0. 2 – 300

Wood burning stove’s fireplaces and Furnaces as sources Ø These are potential sources for both indoor and outdoor air Ø They emit NO, NO 2, CO 2, SO 2, respirable suspended particles, benzo(a)pyrene and formaldehyde (which are vented outside) Ø Cracks and leaks in stovepipes, downdrafts, log roll over in fireplaces and negative air pressure cause the indoor pollution Ø The emissions of pollutants per cord of wood are generally: v 0. 5 – 1. 5 lb of sulphur v 0. 7 – 2. 6 lb of NOx v 300 – 1200 lb of CO

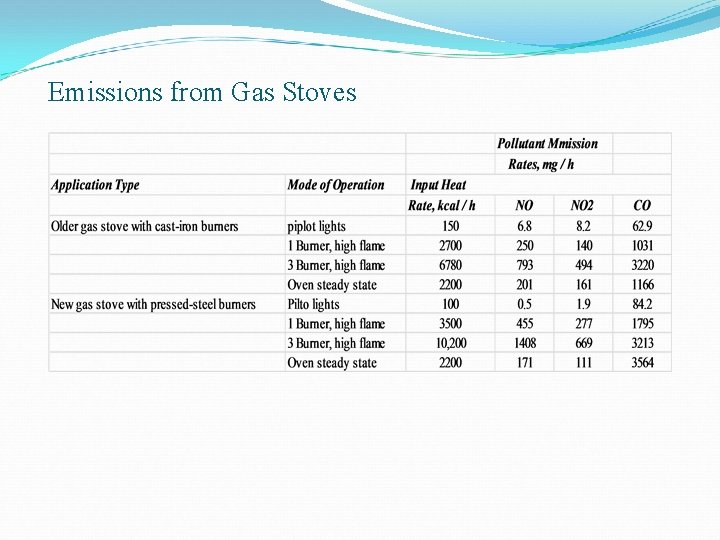
Gas stoves and Ovens Ø Gas stoves produce pollutants like CO, CO 2, NO 2 and aldehyde indoors Ø CO level is increased by 20 – 25 ppm within the first 30 mins of operation, 1. 2 ppm of NO in first 45 mins and 25 ppm of NO 2 Ø CO 2 emissions for ovens in steady state were higher for a new stove compared to an old stove Ø The concentration of indoor pollutant is decided by v Air changes per hour v Number of heaters v Frequency of use v Airspace volume

Emissions from Gas Stoves

Other Combustion sources Ø Other sources of pollutants are water heaters, washers, dryers and attached garages Ø Several hobbies like welding and soldering etc are also sources of indoor air pollution Ø Tobacco smoke is the major contributor of respirable particles Ø Gas water heaters lead to high indoor concentration of Nitrogen oxides

Sampling and Measurement

Measuring Devices Ø Personal monitors are available only for CO, NO 2 and SO 2 and can work in all models Ø The most commonly used monitors are: v CODE – II Ø Portable / Stationary instruments are used to measure inorganic compounds concentration Ø Most common method used to measure CO and CO 2 is nondispersive infrared (NDIR) photometry

Other Measuring Devices Ø Individual concentration of gas in a gaseous mixture is measured by Thermal conductivity Detector (TCD) Ø Thermal conductivity Detector is used for quantifying CO, CO 2 and H 2 O Ø Flame photometric method is used for SO 2 and H 2 S

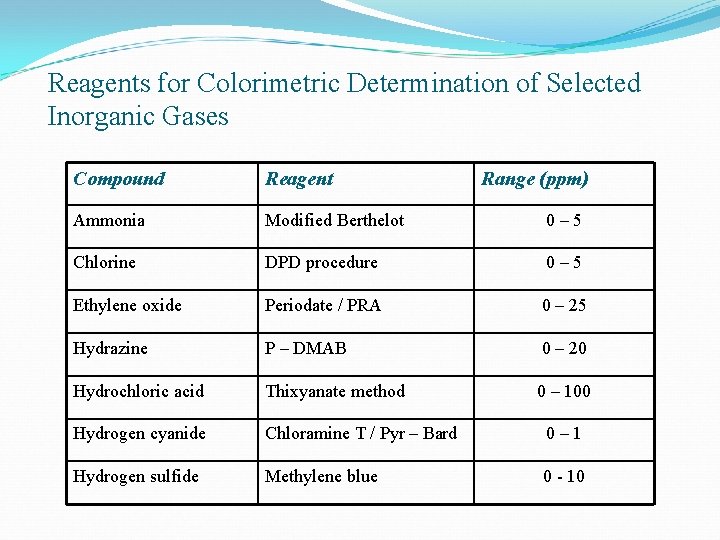
Effective Measuring Methods Ø The Gas filter Correlation method (GFC) is used to measure CO concentration (certified by EPA) Ø Ozone is measured by chemiluminescent method Ø SO 2 concentration is measured by: v Colorimetric method v Flame photometric detection (FPD)

Reagents for Colorimetric Determination of Selected Inorganic Gases Compound Reagent Range (ppm) Ammonia Modified Berthelot 0– 5 Chlorine DPD procedure 0– 5 Ethylene oxide Periodate / PRA 0 – 25 Hydrazine P – DMAB 0 – 20 Hydrochloric acid Thixyanate method 0 – 100 Hydrogen cyanide Chloramine T / Pyr – Bard 0– 1 Hydrogen sulfide Methylene blue 0 - 10

Control Strategies

Source control Ø The major contributors are the wood burning stoves, so removal of them indoors substantially reduces the pollution Ø Replacement of wood burning stoves by oil or gas fired furnaces Ø Modification in the design of the gas and kerosene stoves is another way to subside the indoor pollution Ø Other preventive measures like properation and regular maintenance of the burner could be a possible alternative

Increased ventilation Ø Local and Mechanical ventilation are most effective in removing the pollutants Ø Hood installed over the cooking place is one of the most common local ventilation methods Ø Ductless Cooking ranges with Carbon filters are being used Ø Carbon filters have poor adsorption capacity (which can be improved by careful design) Ø Mechanical ventilation increases the air exchange rate hence decreasing the concentration of the pollutants

Air Cleaning Ø Absorption methods v This method can effectively remove the pollutants indoors v Liquid desiccant based air conditioning systems are being used indoors v Absorber using monoethanolamine is being used in submarines to reduce the concentration of CO 2 v The systems failed when the outdoor concentration was much higher

Air Cleaning Adsorption methods Ø Commonly used adsorbents are: v Silica gel v Activated alumina v Activated carbon v Manganese oxides Ø These adsorbents should be able to remove the moisture from indoor air using solid adsorbents (desiccants)