Determination of the Empirical Formula of Magnesium Oxide















- Slides: 20

Determination of the Empirical Formula of Magnesium Oxide Experiment 2

Experiment 2 Goal: n To study stoichiometric relationships governing mass and amount in chemical formulas. Method: n n n Measure masses before and after the oxidation reaction of Mg metal and O 2 gas Use masses to calculate the experimental empirical formula of magnesium oxide Compare the experimental empirical formula to theoretical empirical formula

Practical Chemistry Atoms / molecules: Formulas / equations: hard to count written based on numerical relationships (not masses) By using mass relationship to moles: n Make something “unmeasurable” measurable. n Relate numbers of atoms and molecules to “real world” values (masses).

Overview of Concepts Law of conservation of mass n Total mass of products must equal total mass of reactants Law of constant composition n Any portion of compound has same ratio of masses as elements in compound (constant mass ratio) Molecular composition expressed 3 ways n n n Mass of each element per mole of compound Mass of each element present relative to total mass (mass %) # each type of atom of per molecule/formula unit (formula)

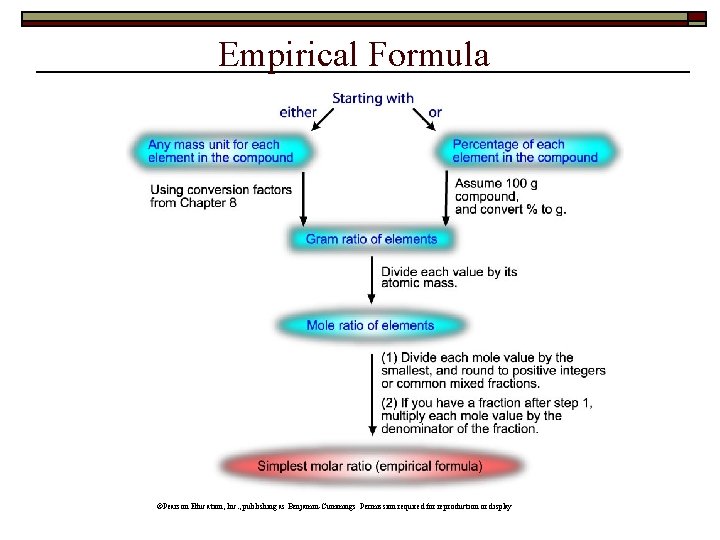
Empirical and Molecular Formulas Empirical Formula n n n “Simplest” formula Agrees with elemental analysis Smallest set of whole # ratio of atoms Molecular Formula n n May be same as empirical May be multiple of empirical “Na 2 Cl 2” = Na. Cl H 2 O 2 ≠ HO

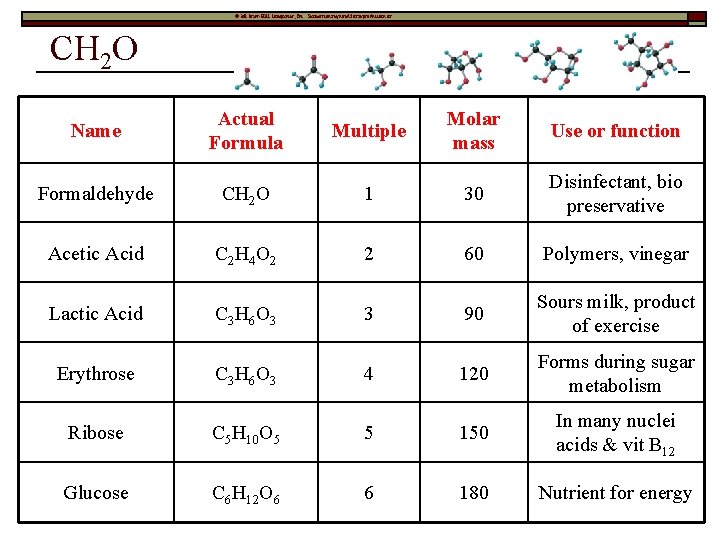
© Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display CH 2 O Name Actual Formula Multiple Molar mass Use or function Formaldehyde CH 2 O 1 30 Disinfectant, bio preservative Acetic Acid C 2 H 4 O 2 2 60 Polymers, vinegar Lactic Acid C 3 H 6 O 3 3 90 Sours milk, product of exercise Erythrose C 3 H 6 O 3 4 120 Forms during sugar metabolism Ribose C 5 H 10 O 5 5 150 In many nuclei acids & vit B 12 Glucose C 6 H 12 O 6 6 180 Nutrient for energy

Reaction Summary Heating Mg metal in air: 1) Mg(s) + N 2(g) + O 2(g) Mg. O(s) + Mg 3 N 2(s) Converting Mg 3 N 2 (add H 2 O): 2) Mg. O(s)+Mg 3 N 2(s)+H 2 O(l) Mg. O(s)+Mg(OH)2(s)+ NH 3(g) Heating (conversion to magnesium oxide): 3) Mg. O(s) + Mg(OH)2(s) Mgx. Oy(s) + H 2 O(g)

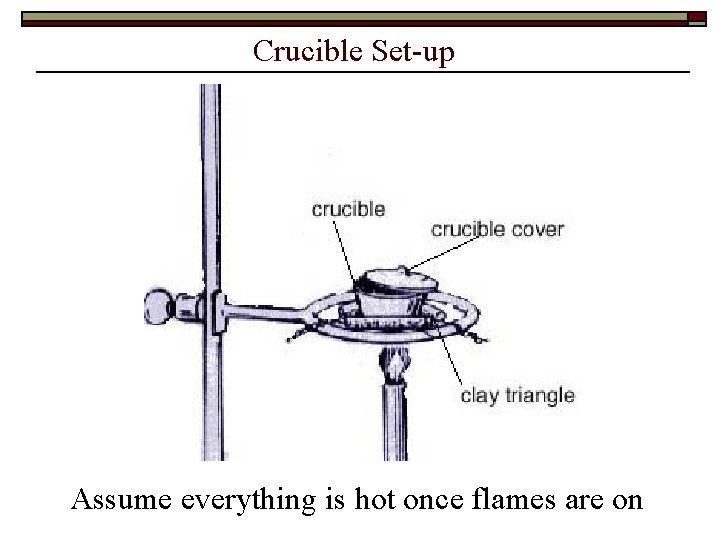
Crucible Set-up Assume everything is hot once flames are on

Procedure Heat empty crucible + lid n Mass cool crucible + lid Obtain ~0. 3 g Mg ribbon n Mass crucible + lid + Mg Place lid with ~0. 5 cm gap mcr+lid+Mg Heat gently ~1 min; strongly ~10 min Cool; add ~ 1 m. L distilled H 2 O (gray powder) Heat gently ~2 -3 min; strongly ~5 min n Mass cool crucible + lid (gray powder) mcr+lid+product

Mass balance and moles Final product: Mgx. Oy n Mass Mg: n Mass O: Empirical formula: n lowest whole-# ratio Mg: O Mgx. Oy

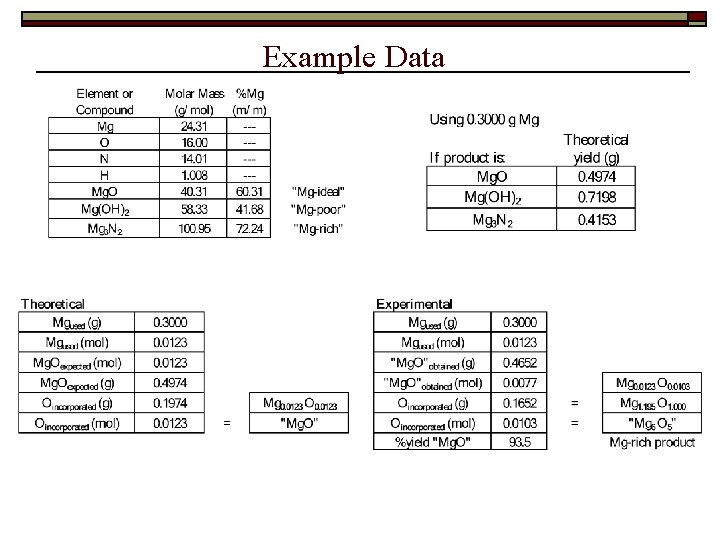
Example Data

Share your results Report your empirical formula on the board Record class formulas Discuss sources of error (include in discussion) n n n Does your formula match expected one? Mg. O Does class data generally show expected results? Effects of: Incomplete conversion to Mg. O Mg, Mg 3 N 2, Mg(OH)2, … Product losses – likely source of error? Residual water – likely source of error? n Brainstorm: Keep logical; discard unlikely

Data/Results Result summary / sample calculations: 1. 2. 3. 4. 5. 6. 7. 8. mass of Mg metal used theoretical yield of Mg. O: Mg(s) + ½O 2(g) → Mg. O(s) mass of Mgx. Oy produced mass of O incorporated into product moles of Mg and O in Mgx. Oy empirical formula of the oxide (individual; class) percent by mass of Mg and O in Mgx. Oy percent yield of the reaction: Mg + ½ O 2 Mg. O

Discussion Compare experimental empirical formula to theoretical empirical formula Primary sources of error/deviation Effects of factors such as: n n incomplete conversion of Mg 3 N 2 to Mg. O, or residual Mg(OH)2 in the product? Does this method a valid way to determine empirical formula of metal oxides?

Report Abstract Results Sample calculations Discussion Review questions

Example #1 2. 448 g of an iron oxide was analyzed and found to contain 1. 771 g Fe and 0. 6766 g O. Calculate the empirical formula. 1. Calculate no. moles each 2. Insert moles as subscripts 3. Divide by smallest no. moles 4. Multiply by 2, 3…until whole #s Formula: Fe 3 O 4

Example #2 Elemental analysis of a sample of an ionic compound gave the following results: 2. 82 g of Na, 4. 35 g of Cl, and 7. 83 g of O. What is the empirical formula? 1. Calculate no. moles each 2. Insert moles as subscripts 3. Divide by smallest no. moles 4. Multiply by until whole #s

Example #3 Lactic acid (90. 08 g/mol) forms in muscle tissue and causes muscle soreness. It is 40. 0 mass% C, 6. 71 mass% H, and 53. 3 mass% O. Find the empirical formula. 1. Calculate no. moles each 2. Insert moles as subscripts 3. Divide by smallest no. moles 4. Multiply by until whole #s

Empirical Formula ©Pearson Education, Inc. , publishing as Benjamin-Cummings Permission required for reproduction or display

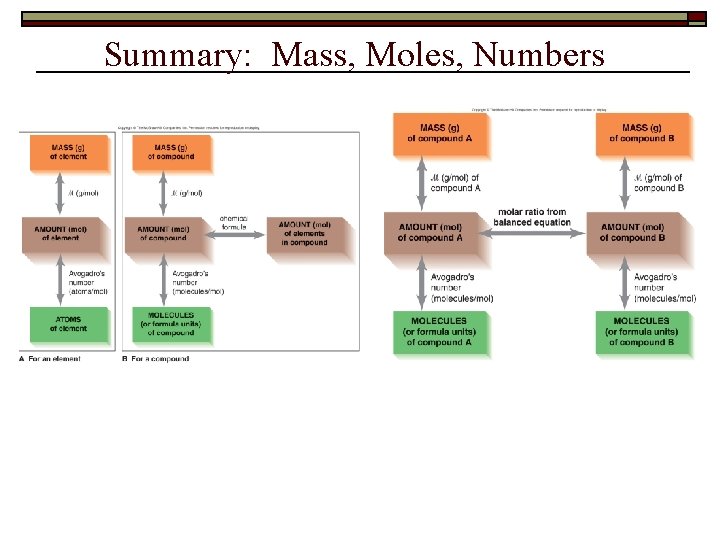
Summary: Mass, Moles, Numbers