Crystalline state Symmetry in nature Symmetry in arts



- Slides: 33

Crystalline state • Symmetry in nature • Symmetry in arts and industry • Description of symmetry – basic concepts • Crystallography of two dimensions • Crystallography of three dimensions

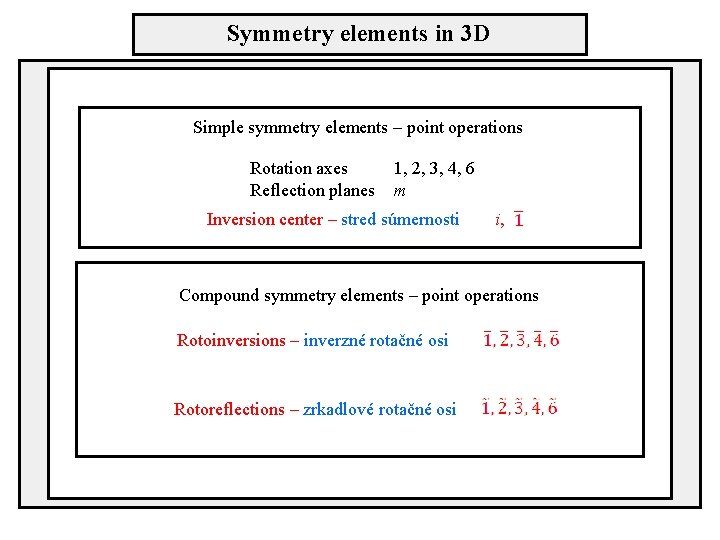
Symmetry elements in 3 D Simple symmetry elements – point operations Rotation axes Reflection planes 1, 2, 3, 4, 6 m Inversion center – stred súmernosti i, Compound symmetry elements – point operations Rotoinversions – inverzné rotačné osi Rotoreflections – zrkadlové rotačné osi

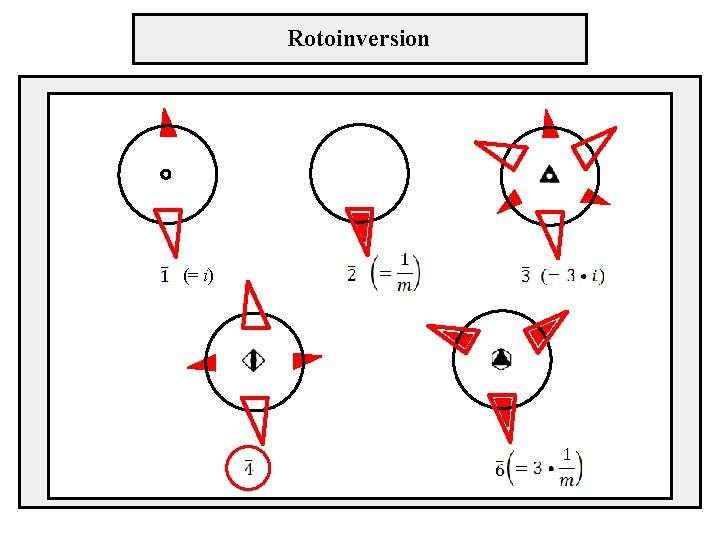
Rotoinversion (= i)

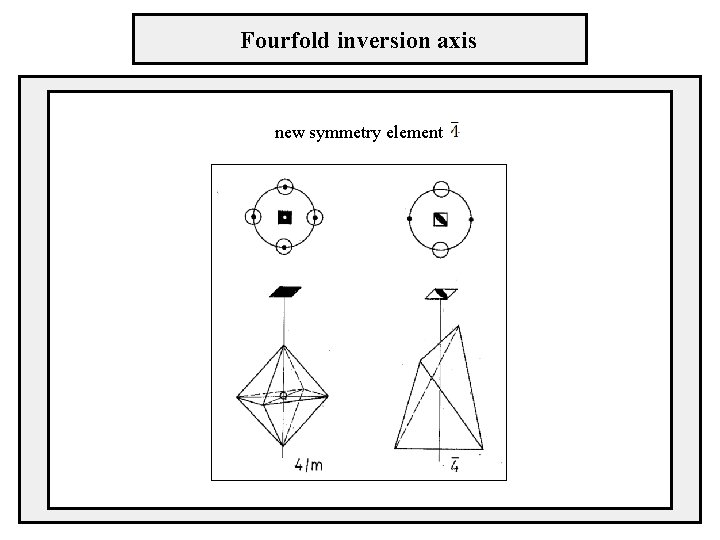
Fourfold inversion axis new symmetry element

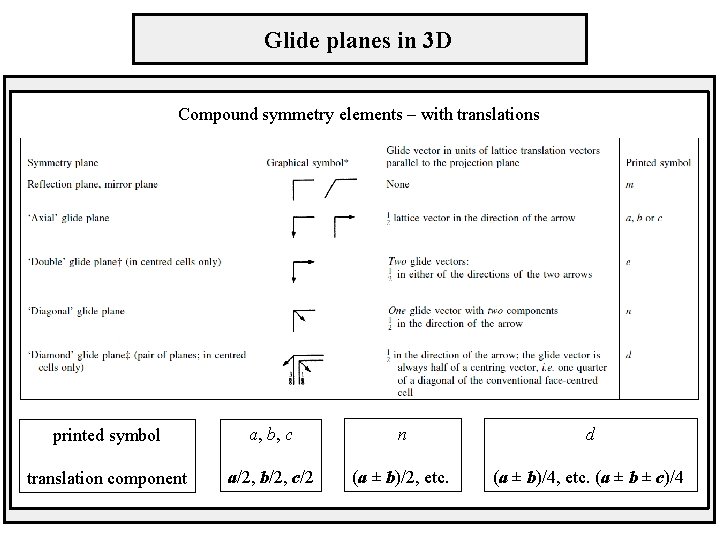
Glide planes in 3 D Compound symmetry elements – with translations printed symbol a, b, c n d translation component a/2, b/2, c/2 (a ± b)/2, etc. (a ± b)/4, etc. (a ± b ± c)/4

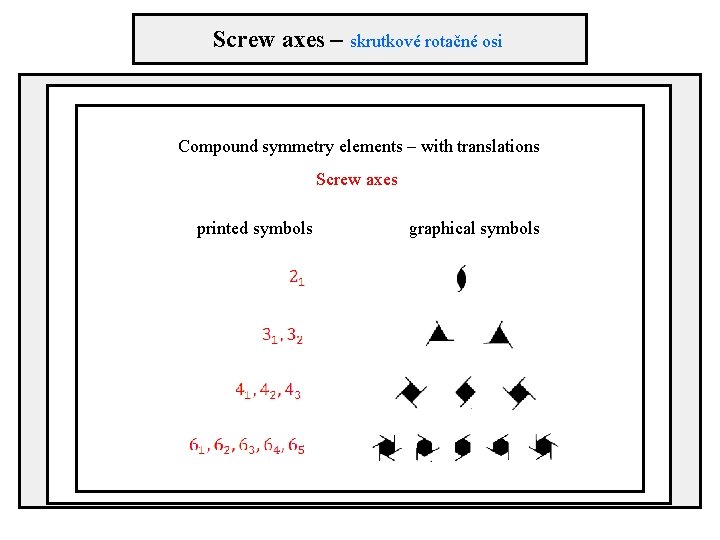
Screw axes – skrutkové rotačné osi Compound symmetry elements – with translations Screw axes printed symbols graphical symbols

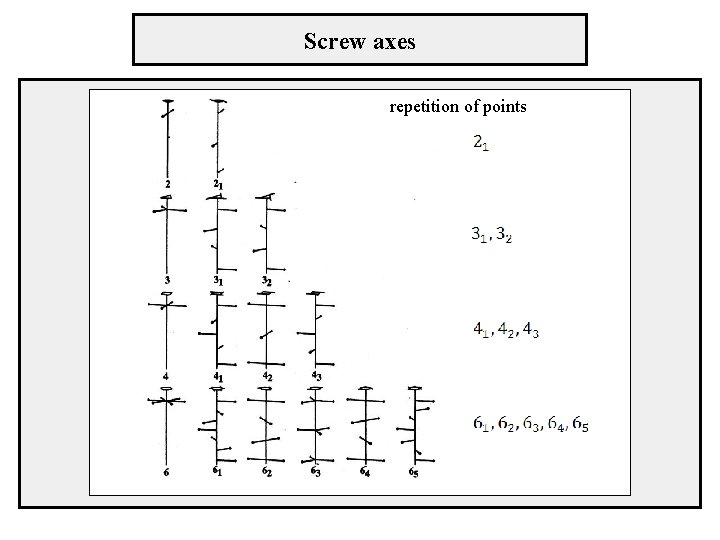
Screw axes repetition of points

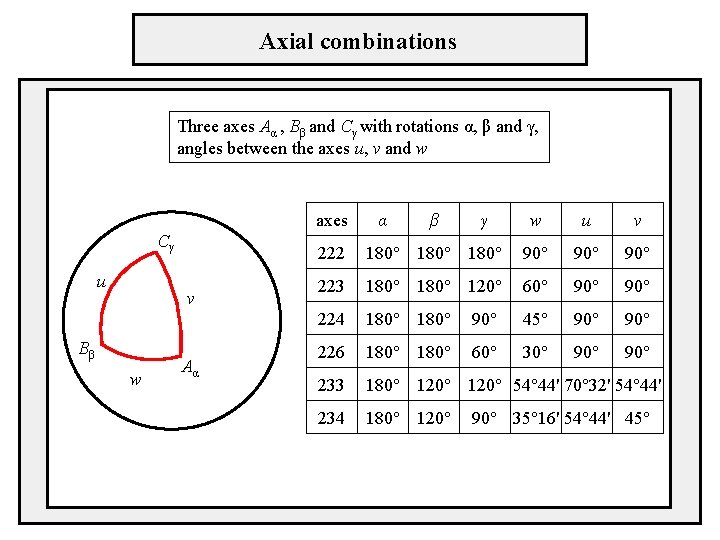
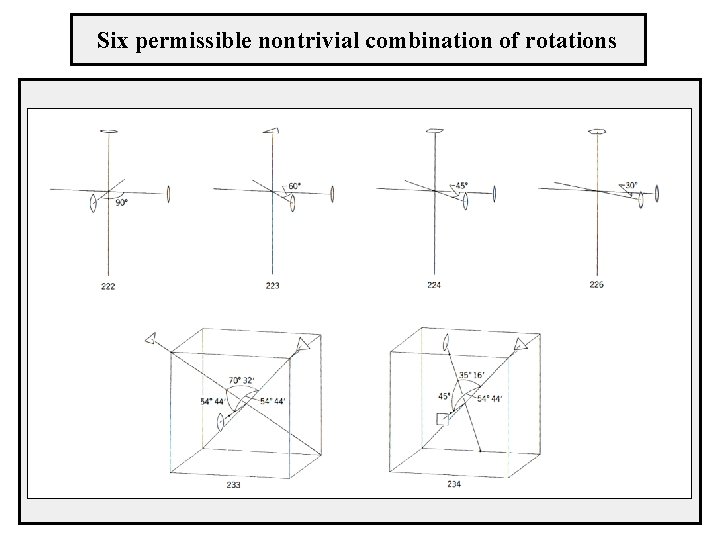
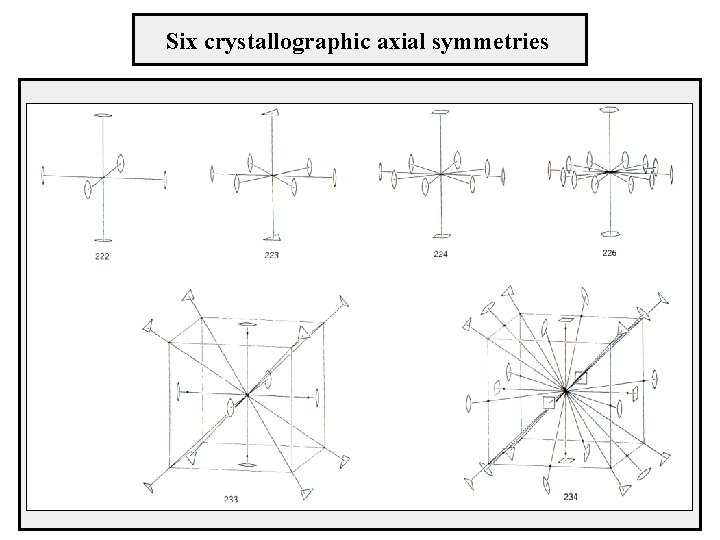
Combination of intersecting axes - every two rotations around intersecting axes can be replaced by one appropriate rotation - the angles and the orientation of the axes are arbitrary - strong limitation for crystal structures - the rotation angles can acquire only the values 0°, 60°, 90°, 120°, 180° - the same holds for the resulting rotation which axis combinations are allowed? there are 35 triplets of axes but only the following combinations are allowed 222, 223, 224, 226, 233, 234

Axial combinations Three axes Aα , Bβ and Cγ with rotations α, β and γ, angles between the axes u, v and w axes Cγ u v Bβ w Aα α β γ w u v 222 180° 90° 90° 223 180° 120° 60° 90° 224 180° 90° 45° 90° 226 180° 60° 30° 90° 233 180° 120° 54° 44' 70° 32' 54° 44' 234 180° 120° 90° 35° 16' 54° 44' 45°

Six permissible nontrivial combination of rotations

Six crystallographic axial symmetries

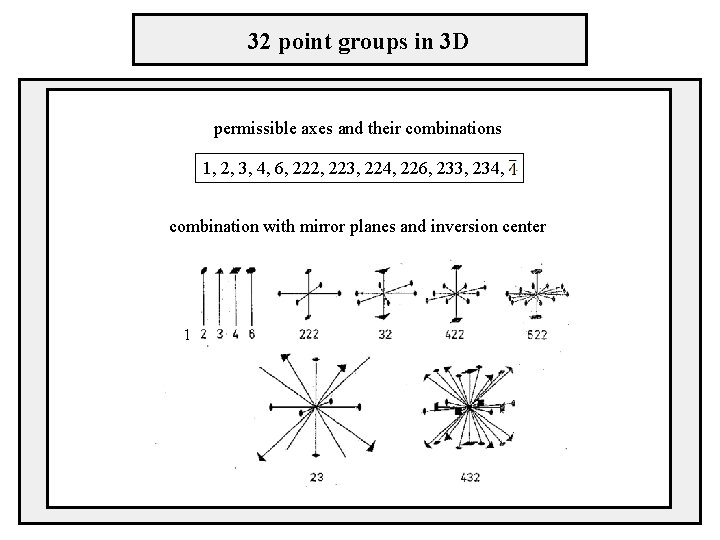
32 point groups in 3 D permissible axes and their combinations 1, 2, 3, 4, 6, 222, 223, 224, 226, 233, 234, combination with mirror planes and inversion center 1

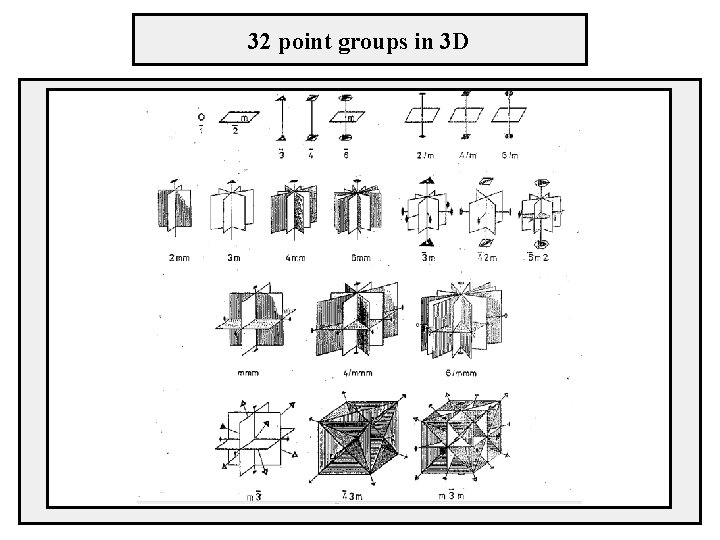
32 point groups in 3 D

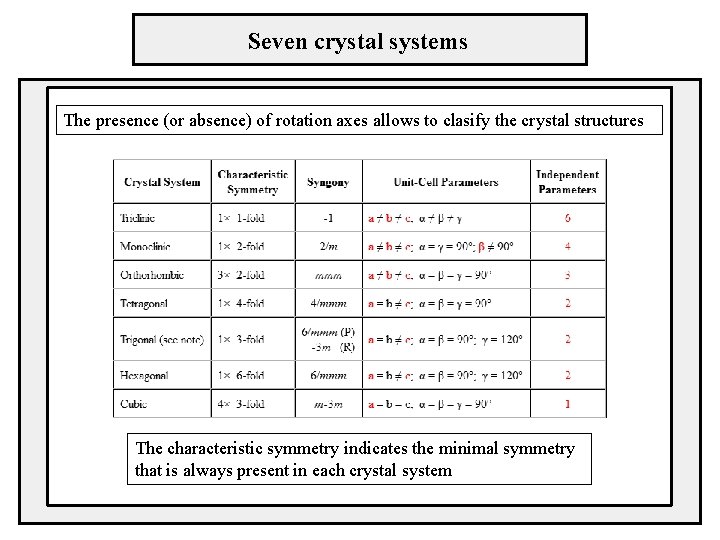
Seven crystal systems The presence (or absence) of rotation axes allows to clasify the crystal structures The characteristic symmetry indicates the minimal symmetry that is always present in each crystal system

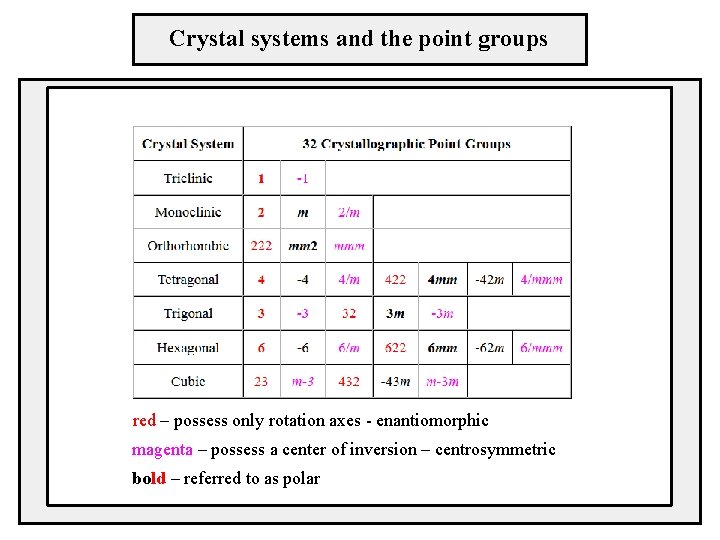
Crystal systems and the point groups red – possess only rotation axes - enantiomorphic magenta – possess a center of inversion – centrosymmetric bold – referred to as polar

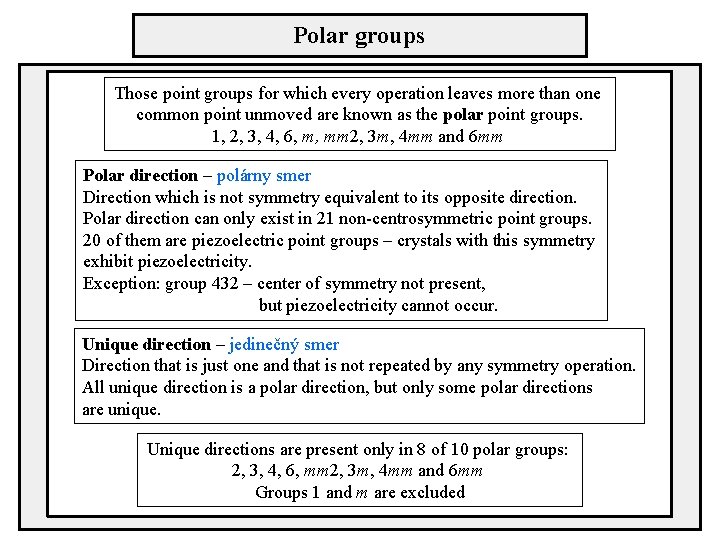
Polar groups Those point groups for which every operation leaves more than one common point unmoved are known as the polar point groups. 1, 2, 3, 4, 6, m, mm 2, 3 m, 4 mm and 6 mm Polar direction – polárny smer Direction which is not symmetry equivalent to its opposite direction. Polar direction can only exist in 21 non-centrosymmetric point groups. 20 of them are piezoelectric point groups – crystals with this symmetry exhibit piezoelectricity. Exception: group 432 – center of symmetry not present, but piezoelectricity cannot occur. Unique direction – jedinečný smer Direction that is just one and that is not repeated by any symmetry operation. All unique direction is a polar direction, but only some polar directions are unique. Unique directions are present only in 8 of 10 polar groups: 2, 3, 4, 6, mm 2, 3 m, 4 mm and 6 mm Groups 1 and m are excluded

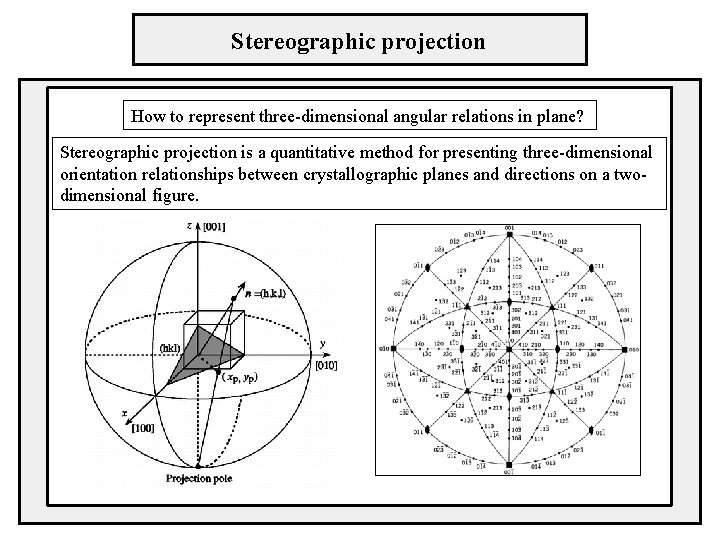
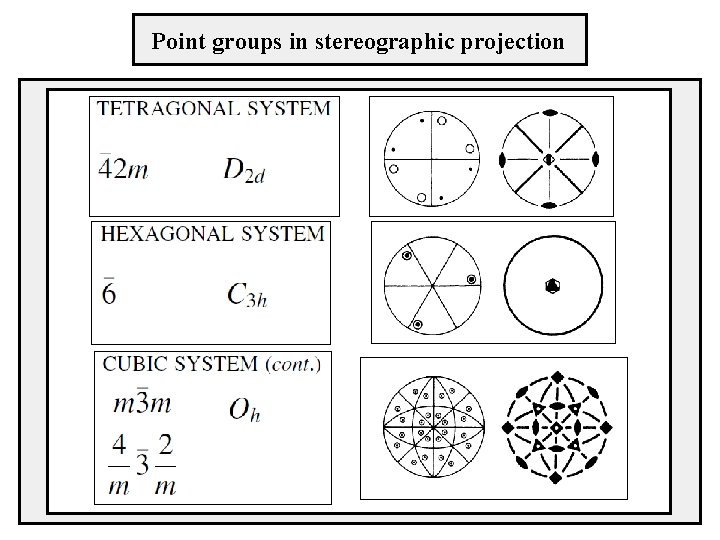
Stereographic projection How to represent three-dimensional angular relations in plane? Stereographic projection is a quantitative method for presenting three-dimensional orientation relationships between crystallographic planes and directions on a twodimensional figure.

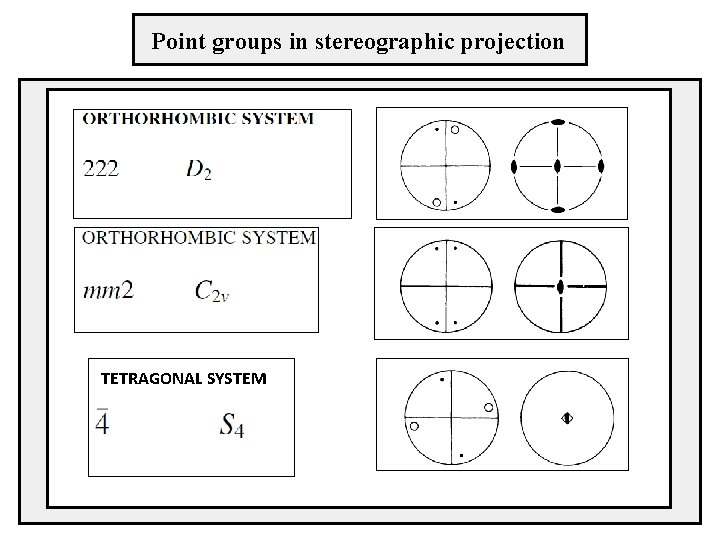
Point groups in stereographic projection TETRAGONAL SYSTEM

Point groups in stereographic projection

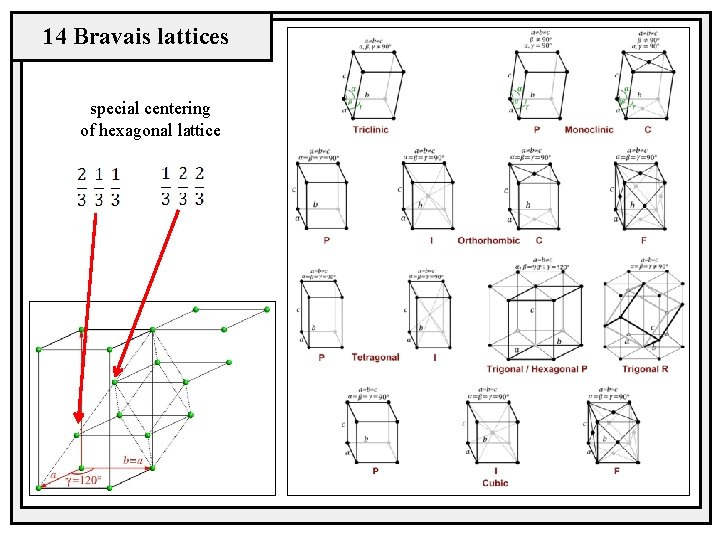
14 Bravais lattices special centering of hexagonal lattice

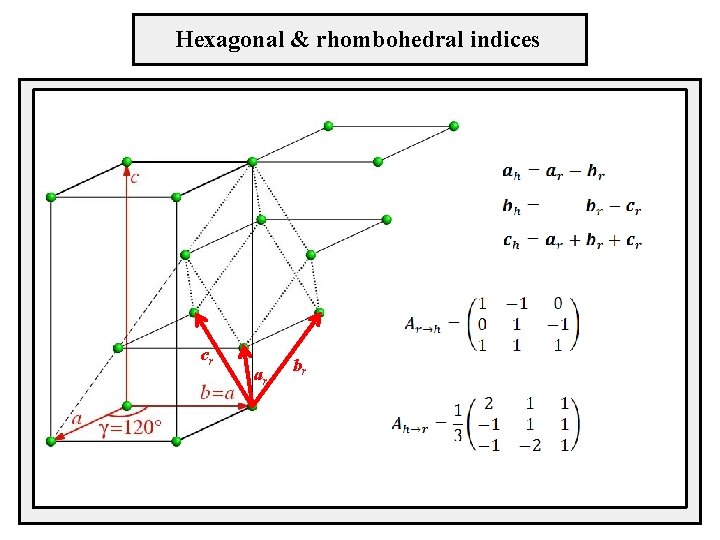
Hexagonal & rhombohedral indices cr ar br

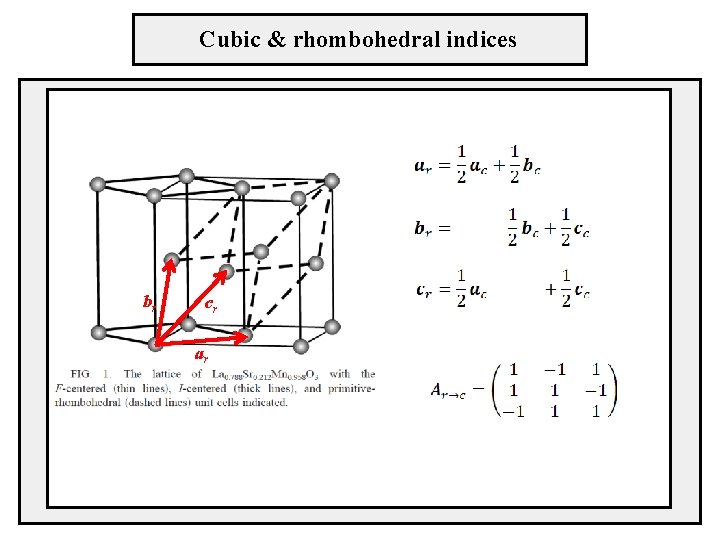
Cubic & rhombohedral indices br cr ar

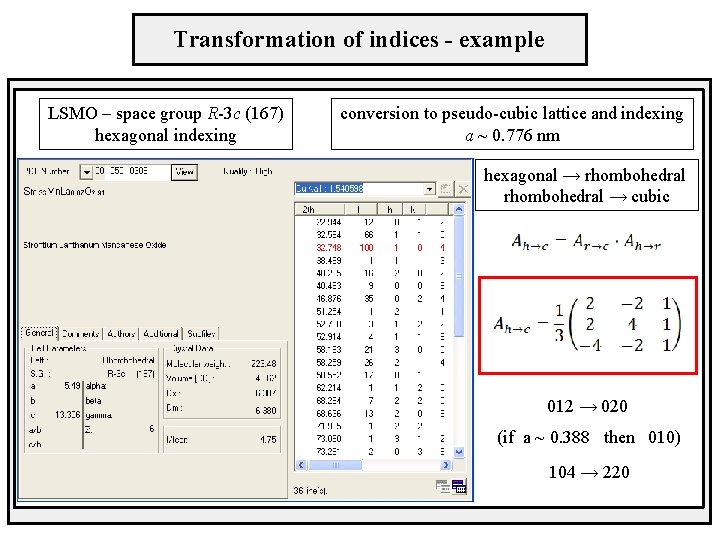
Transformation of indices - example LSMO – space group R-3 c (167) hexagonal indexing conversion to pseudo-cubic lattice and indexing a ~ 0. 776 nm hexagonal → rhombohedral → cubic 012 → 020 (if a ~ 0. 388 then 010) 104 → 220

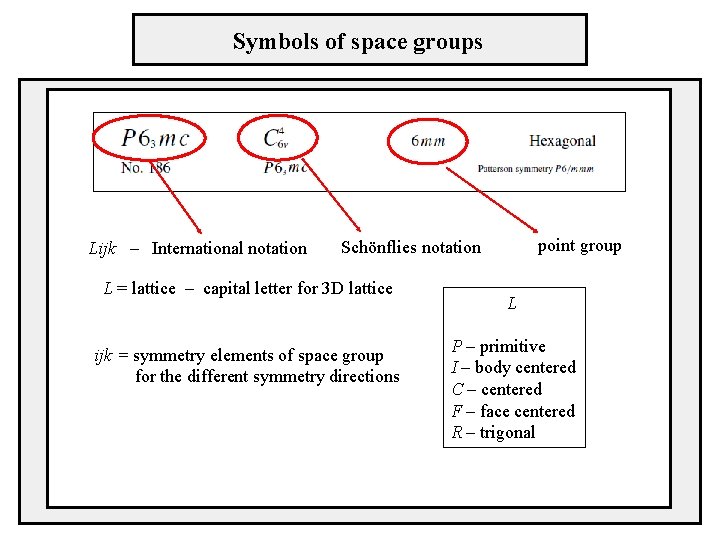
Symbols of space groups Lijk – International notation point group Schönflies notation L = lattice – capital letter for 3 D lattice ijk = symmetry elements of space group for the different symmetry directions L P – primitive I – body centered C – centered F – face centered R – trigonal

International Tables for Crystallography

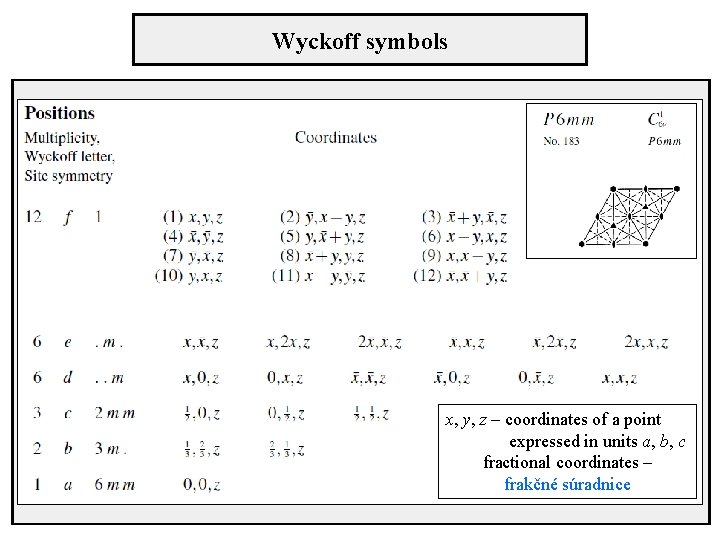
Wyckoff symbols x, y, z – coordinates of a point expressed in units a, b, c fractional coordinates – frakčné súradnice

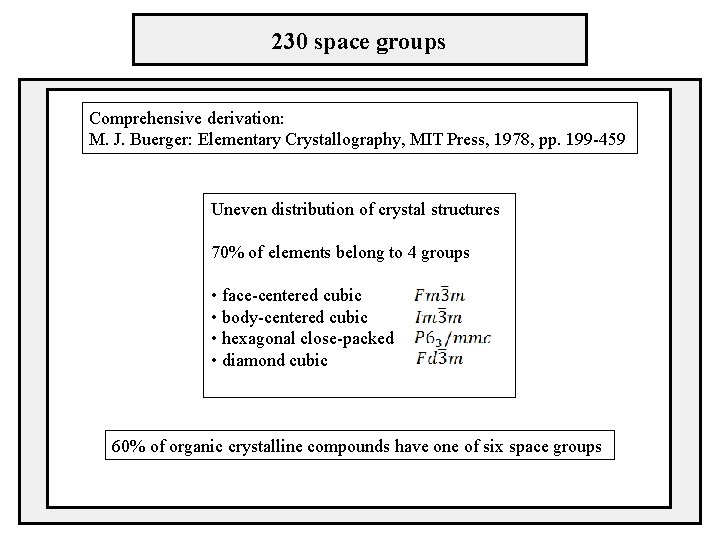
230 space groups Comprehensive derivation: M. J. Buerger: Elementary Crystallography, MIT Press, 1978, pp. 199 -459 Uneven distribution of crystal structures 70% of elements belong to 4 groups • face-centered cubic • body-centered cubic • hexagonal close-packed • diamond cubic 60% of organic crystalline compounds have one of six space groups

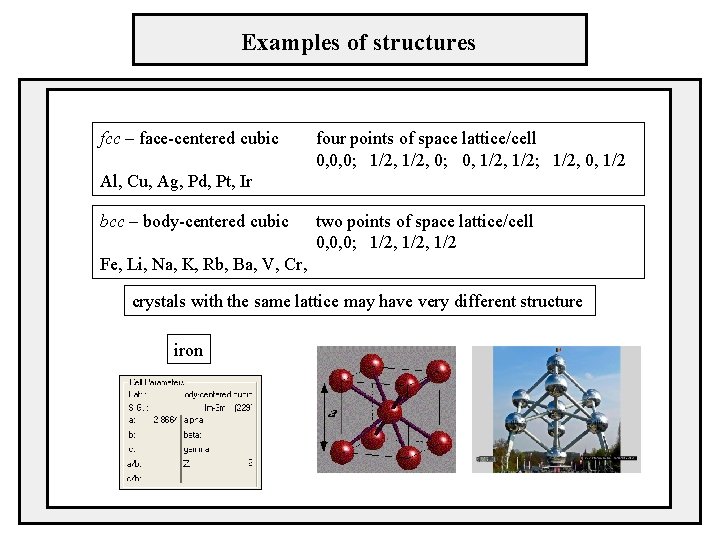
Examples of structures fcc – face-centered cubic four points of space lattice/cell 0, 0, 0; 1/2, 0; 0, 1/2; 1/2, 0, 1/2 Al, Cu, Ag, Pd, Pt, Ir bcc – body-centered cubic two points of space lattice/cell 0, 0, 0; 1/2, 1/2 Fe, Li, Na, K, Rb, Ba, V, Cr, crystals with the same lattice may have very different structure iron

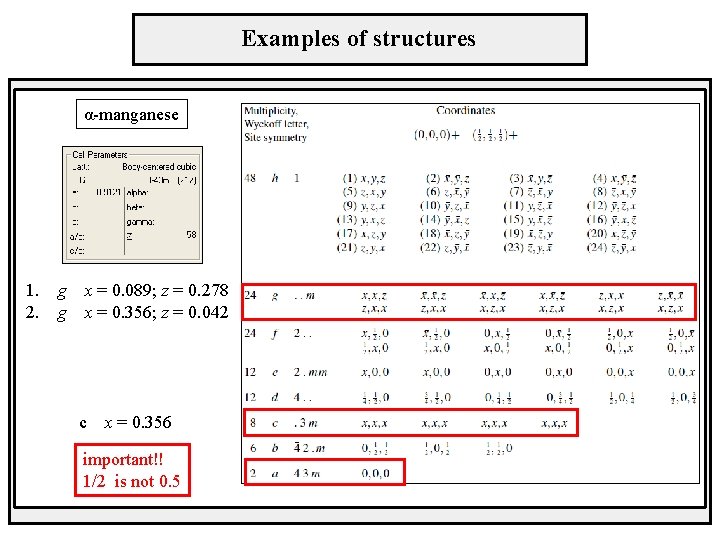
Examples of structures α-manganese 1. g x = 0. 089; z = 0. 278 2. g x = 0. 356; z = 0. 042 c x = 0. 356 important!! 1/2 is not 0. 5

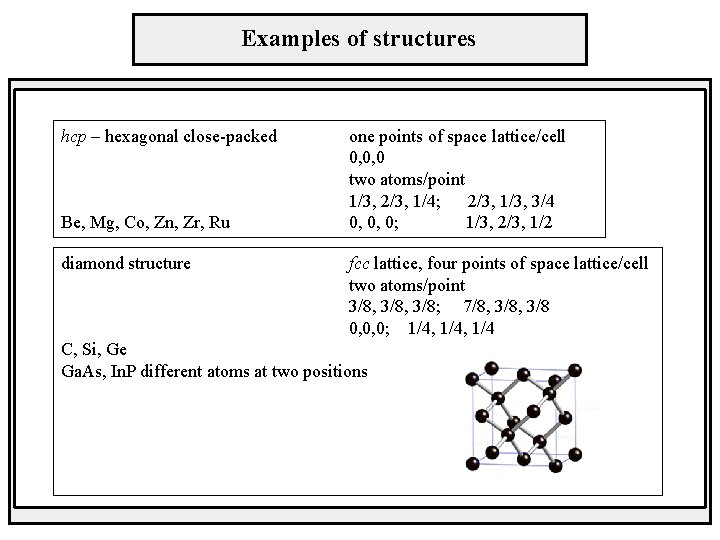
Examples of structures hcp – hexagonal close-packed Be, Mg, Co, Zn, Zr, Ru diamond structure one points of space lattice/cell 0, 0, 0 two atoms/point 1/3, 2/3, 1/4; 2/3, 1/3, 3/4 0, 0, 0; 1/3, 2/3, 1/2 fcc lattice, four points of space lattice/cell two atoms/point 3/8, 3/8; 7/8, 3/8 0, 0, 0; 1/4, 1/4 C, Si, Ge Ga. As, In. P different atoms at two positions

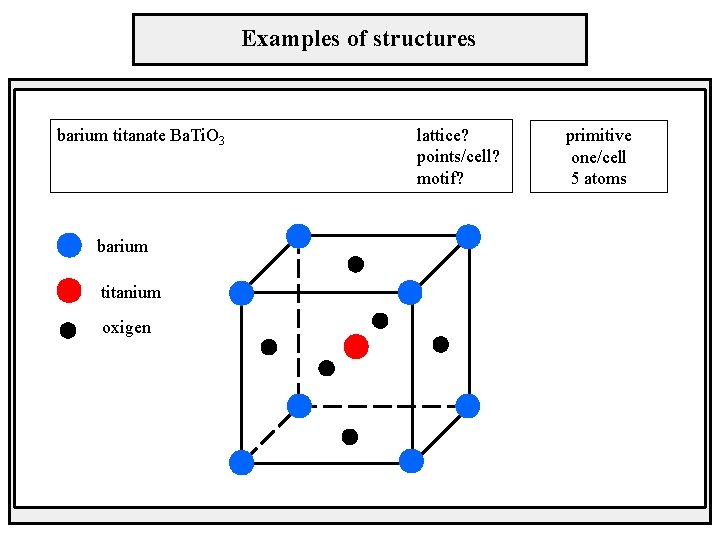
Examples of structures barium titanate Ba. Ti. O 3 barium titanium oxigen lattice? points/cell? motif? primitive one/cell 5 atoms

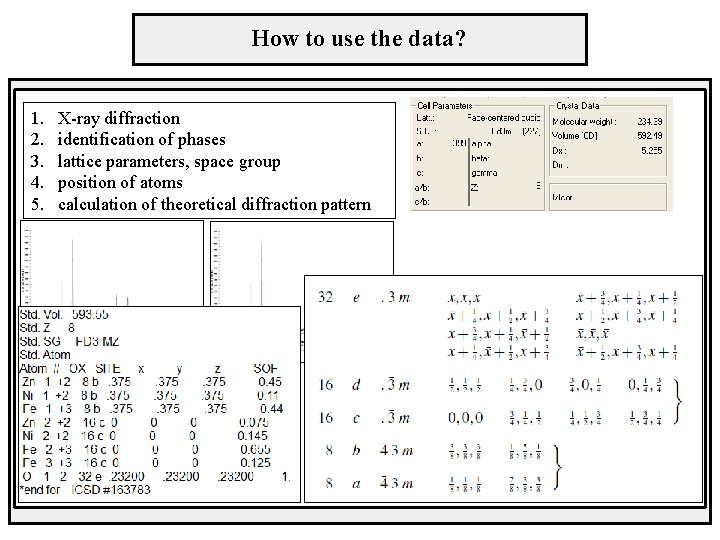
How to use the data? 1. 2. 3. 4. 5. X-ray diffraction identification of phases lattice parameters, space group position of atoms calculation of theoretical diffraction pattern
