Chemistry 125 Lecture 44 January 26 2011 Nucleophilic







![Ethyl [1] n-Propyl 0. 82 iso-Butyl 0. 036 -Methylation Neopentyl 0. 000012 No way Ethyl [1] n-Propyl 0. 82 iso-Butyl 0. 036 -Methylation Neopentyl 0. 000012 No way](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-20.jpg)

![“bridgehead” chloride Cl boat c-hexane with a bridge bicyclo[2. 2. 1]heptane Flattening would generate “bridgehead” chloride Cl boat c-hexane with a bridge bicyclo[2. 2. 1]heptane Flattening would generate](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-23.jpg)
![Cycloalkyl Halides (e. g. J&F Table 7. 2) krelative ~109° 60° [1] <0. 0001 Cycloalkyl Halides (e. g. J&F Table 7. 2) krelative ~109° 60° [1] <0. 0001](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-26.jpg)

![Rate Constant Dependance on Nucleophile Nu R-L Leaving Substrate Group krel p. Ka [1] Rate Constant Dependance on Nucleophile Nu R-L Leaving Substrate Group krel p. Ka [1]](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-28.jpg)

- Slides: 29

Chemistry 125: Lecture 44 January 26, 2011 Nucleophilic Substitution and Mechanistic Tools: Stereochemistry, Rate Law, Substrate, Nucleophile This For copyright notice see final page of this file

SN 2 Nucleophilic Substitution Generality of Nucleophilic Substitution Solvent Nu: R-L Nucleophile Substrate (+) (-) Nu-R L Product Leaving Group But there are different mechanisms! the Pragmatic Logic of Proving a Mechanism with Experiment & Theory (mostly by disproving all alternative mechanisms)

"It is an old maxim of mine that when you have excluded the impossible, whatever remains, however improbable, must be the truth. " The Adventure of the Beryl Coronet

SN 2 Nucleophilic Substitution Nu: R-L (+) Nu-R (-) L Break bond Make bond the Pragmatic Logic (Dissociation) (Association) Simultaneous of Proving a Mechanism D then A A then D “Concerted” with Experiment &(make-as-you-break) Theory (mostly by disproving all alternative mechanisms)

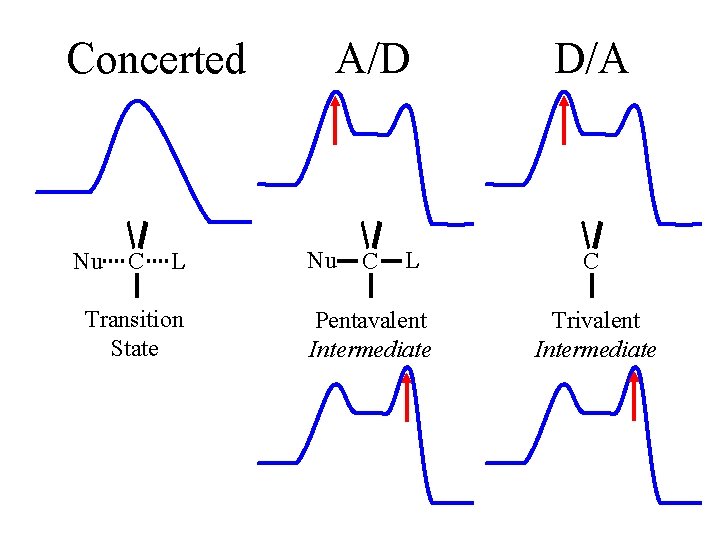
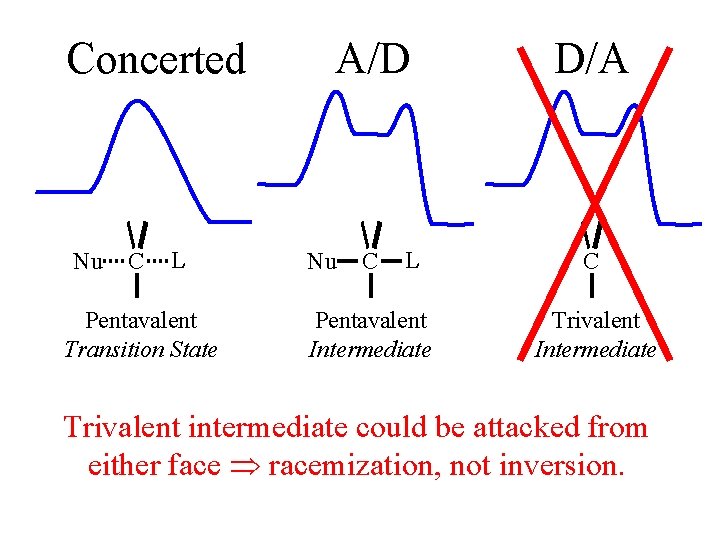
Concerted Nu C L Transition State A/D Nu C L Pentavalent Intermediate D/A C Trivalent Intermediate

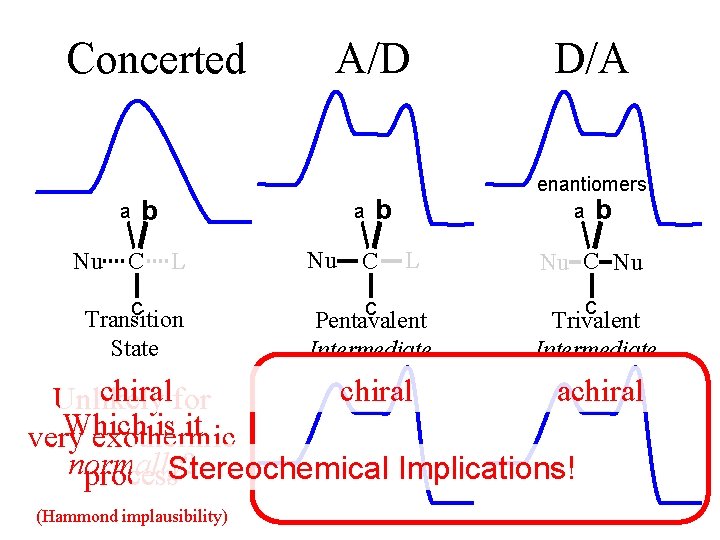
Concerted a Nu A/D b C a L c Transition State Nu enantiomers a b b C D/A L c Pentavalent Intermediate Nu C Nu c Trivalent Intermediate chiralfor chiral achiral Unlikely Which is it very exothermic normally? process. Stereochemical Implications! ( (Hammond implausibility)

Tools for Testing (i. e. Excluding) Mechanisms: Stereochemistry (J&F sec 7. 4 b) Rate Law (J&F sec 7. 4 a) Rate Constant (J&F sec 7. 4 cdefg) Structure X-Ray and Quantum Mechanics

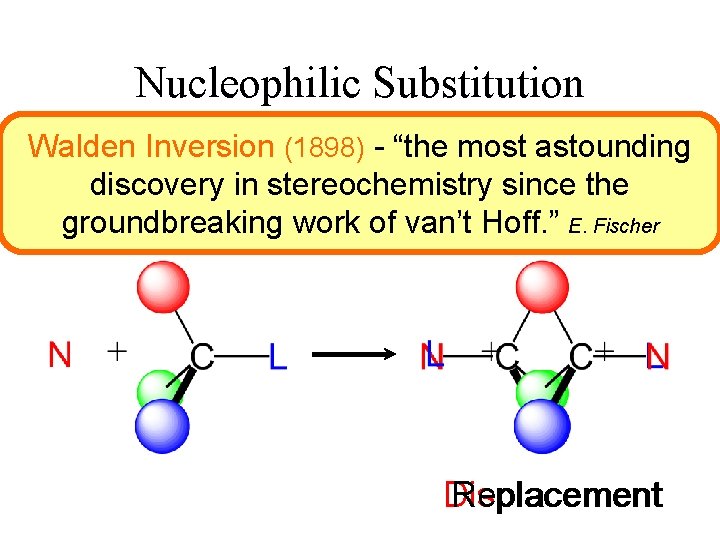
Nucleophilic Substitution Walden N Inversion + RL (1898) - “the most L +astounding RN discovery in stereochemistry since the groundbreaking work of van’t Hoff. ” E. Fischer Displacement Replacement

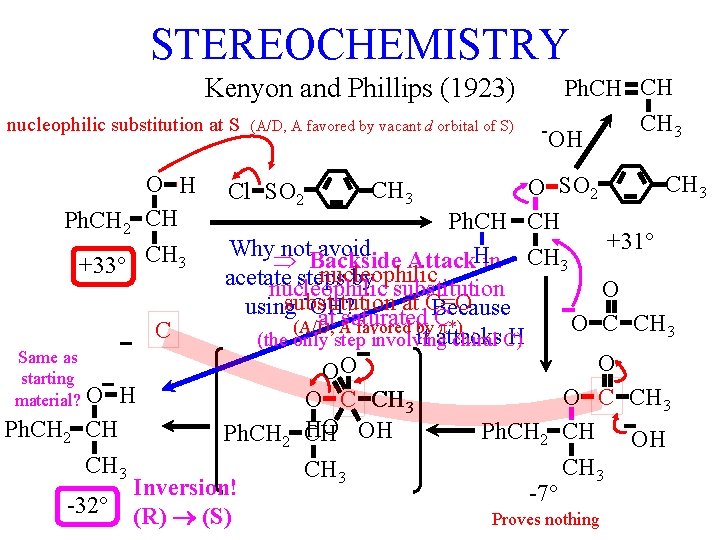
STEREOCHEMISTRY Kenyon and Phillips (1923) nucleophilic substitution at S O H Ph. CH 2 CH +33° CH 3 (A/D, A favored by vacant d orbital of S) Ph. CH CH CH 3 -OH CH 3 O SO 2 Ph. CH 2 CH +31° Why not. Backside avoid H Attack in CH 3 nucleophilic acetate steps by substitution nucleophilic O substitution at C=O using OH? Because at saturated O C CH 3 (A/D, A favored by. C. *) C it attacks H (the only step involving chiral C) Same as O O O starting O C CH 3 material? O H O C CH 3 HO OH Ph. CH 2 CH CH 3 Inversion! -7° -32° (R) (S) Proves nothing Cl SO 2 CH 3

Concerted Nu C L Pentavalent Transition State A/D Nu C L Pentavalent Intermediate D/A C Trivalent Intermediate Trivalent intermediate could be attacked from either face racemization, not inversion.

Tools for Testing (i. e. Excluding) Mechanisms: Stereochemistry Rate Law Rate Constant Structure X-Ray and Quantum Mechanics

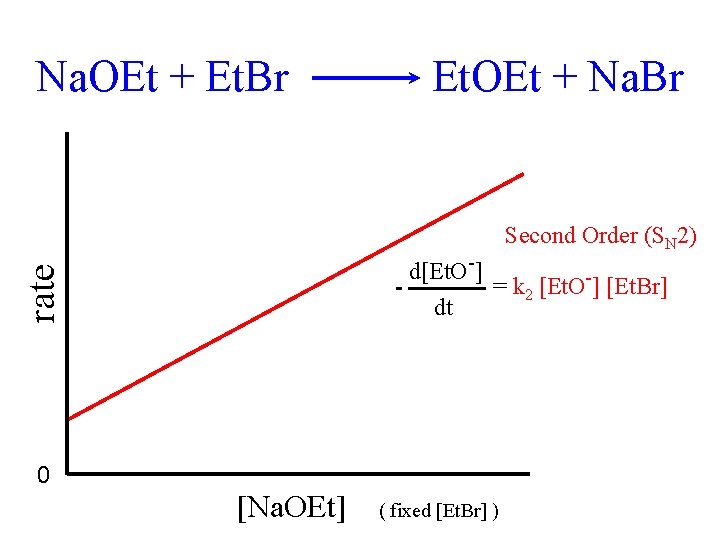
Na. OEt + Et. Br Et. OEt + Na. Br Second Order (SN 2) rate d[Et. O-] = k 2 [Et. O-] [Et. Br] dt 0 [Na. OEt] ( fixed [Et. Br] )

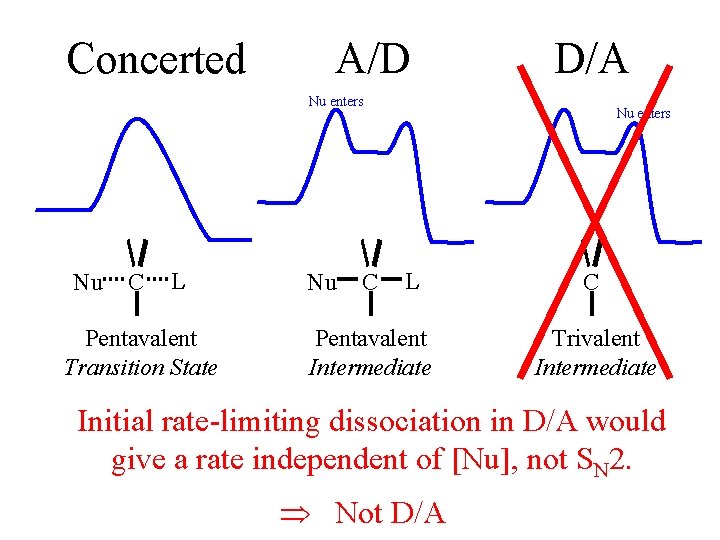
Concerted A/D D/A Nu enters Nu C L Pentavalent Transition State Nu C Nu enters L Pentavalent Intermediate C Trivalent Intermediate Initial rate-limiting dissociation in D/A would give a rate independent of [Nu], not SN 2. Not D/A

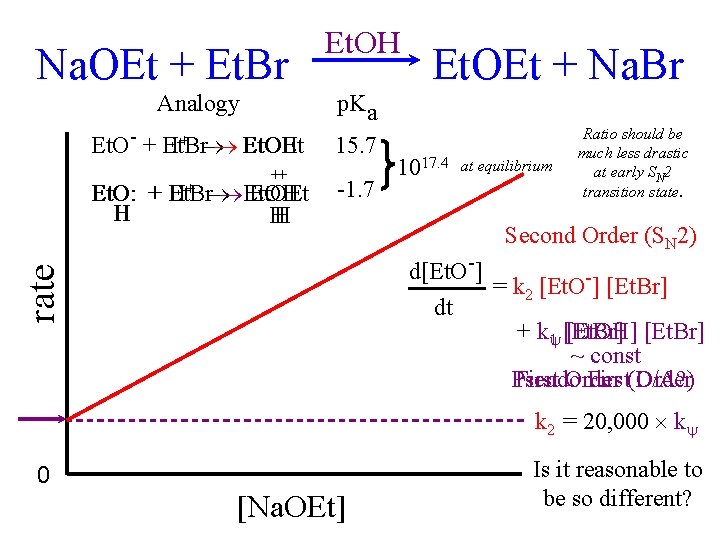
Na. OEt + Et. Br Analogy Et. O- + Et. Br H+ Et. OH Et. OEt ++ Et. O: + Et. Br H+ Et. OH Et. OEt H H H Et. OEt + Na. Br p. Ka 15. 7 -1. 7 1017. 4 at equilibrium Ratio should be much less drastic at early SN 2 transition state. Second Order (SN 2) rate d[Et. O-] = k 2 [Et. O-] [Et. Br] dt + k 1 [Et. Br] [Et. OH] [Et. Br] ~ const Pseudo First Order First(D/A? ) Order k 2 = 20, 000 k 0 [Na. OEt] Is it reasonable to be so different?

Tools for Testing (i. e. Excluding) Mechanisms: Stereochemistry Rate Law Rate Constant Structure X-Ray and Quantum Mechanics

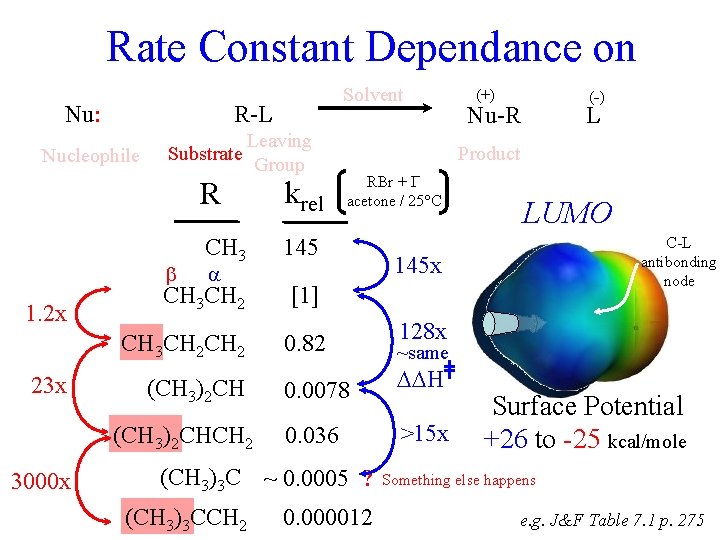
Rate Constant Dependance on Nu: R-L Nucleophile Substrate 23 x Leaving Group R krel CH 3 145 CH 3 CH 2 [1] CH 3 CH 2 0. 82 1. 2 x (CH 3)2 CHCH 2 3000 x Solvent Nu-R L Product RBr + Iacetone / 25°C C-L antibonding node 128 x ~same H 0. 036 >15 x 0. 000012 LUMO 145 x 0. 0078 (CH 3)3 C ~ 0. 0005 ? (CH 3)3 CCH 2 (-) (+) Surface Potential +26 to -25 kcal/mole Something else happens e. g. J&F Table 7. 1 p. 275

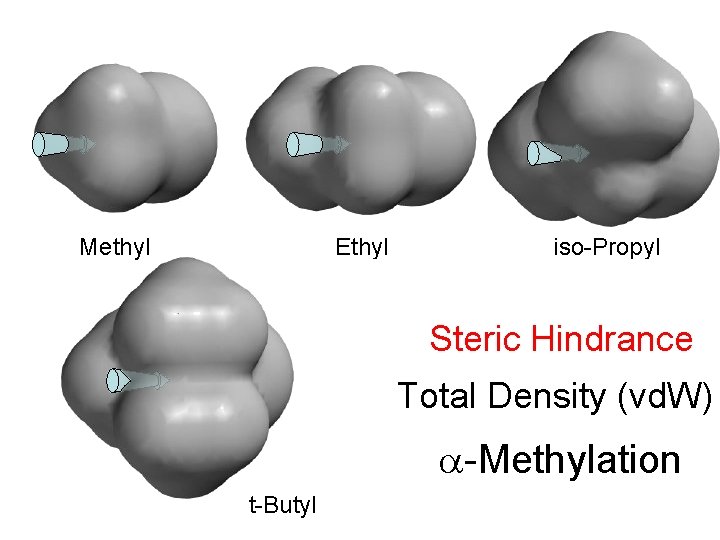
Methyl Ethyl iso-Propyl Steric Hindrance Total Density (vd. W) -Methylation t-Butyl

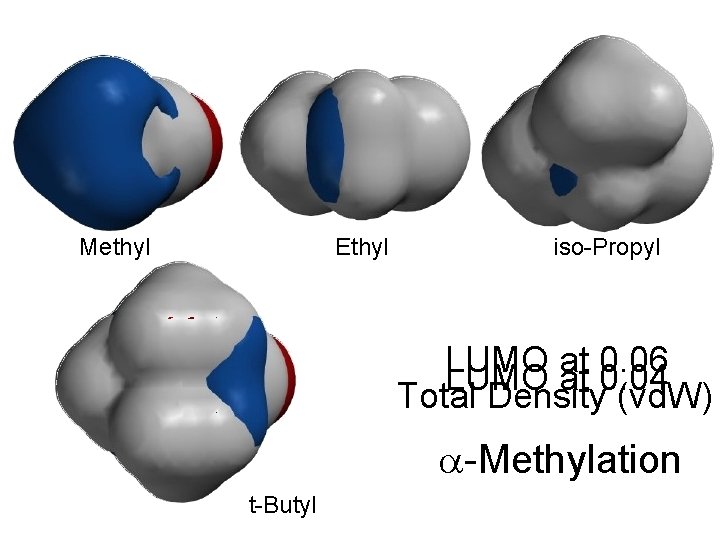
Methyl Ethyl iso-Propyl LUMO at 0. 06 LUMO at 0. 04 Total Density (vd. W) -Methylation t-Butyl

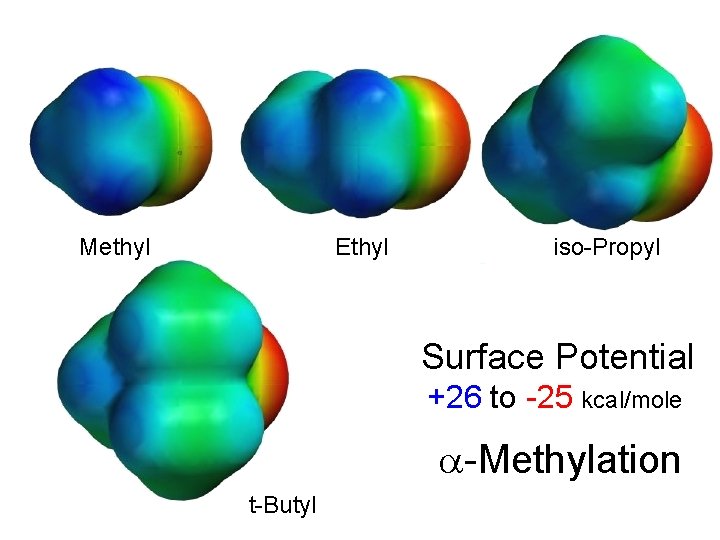
Methyl Ethyl iso-Propyl Surface Potential +26 to -25 kcal/mole -Methylation t-Butyl
![Ethyl 1 nPropyl 0 82 isoButyl 0 036 Methylation Neopentyl 0 000012 No way Ethyl [1] n-Propyl 0. 82 iso-Butyl 0. 036 -Methylation Neopentyl 0. 000012 No way](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-20.jpg)
Ethyl [1] n-Propyl 0. 82 iso-Butyl 0. 036 -Methylation Neopentyl 0. 000012 No way to avoid the third -CH 3

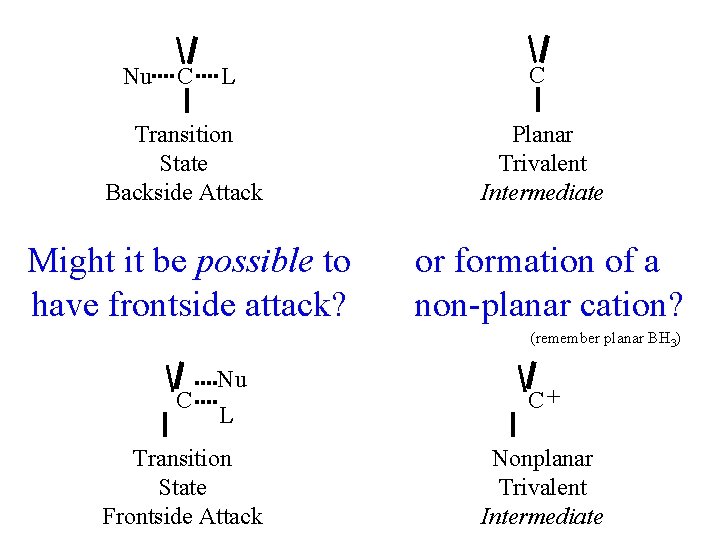
Nu C L C Transition State Backside Attack Planar Trivalent Intermediate Might it be possible to have frontside attack? or formation of a non-planar cation? (remember planar BH 3) C Nu L Transition State Frontside Attack C+ Nonplanar Trivalent Intermediate

“In 1939 Bartlett and Knox published Molecule specifically the account of their work on the bridgedesigned and prepared head chloride, apocamphyl chloride. I to test these believed then, and I believe now, that this mechanistic questions was a fantastically influential paper. For thirty years afterwards, no one really accepted any mechanism unless it had been tested out on a bridgehead case. Indeed, the Bartlett-Knox paper shaped the interests and viewpoint of many chemists about the kind of physical organic they wanted to do. ” John D. Roberts Caltech 1975 Bartlett and Knox * (J. Am. Chem. Soc. - 1939)
![bridgehead chloride Cl boat chexane with a bridge bicyclo2 2 1heptane Flattening would generate “bridgehead” chloride Cl boat c-hexane with a bridge bicyclo[2. 2. 1]heptane Flattening would generate](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-23.jpg)
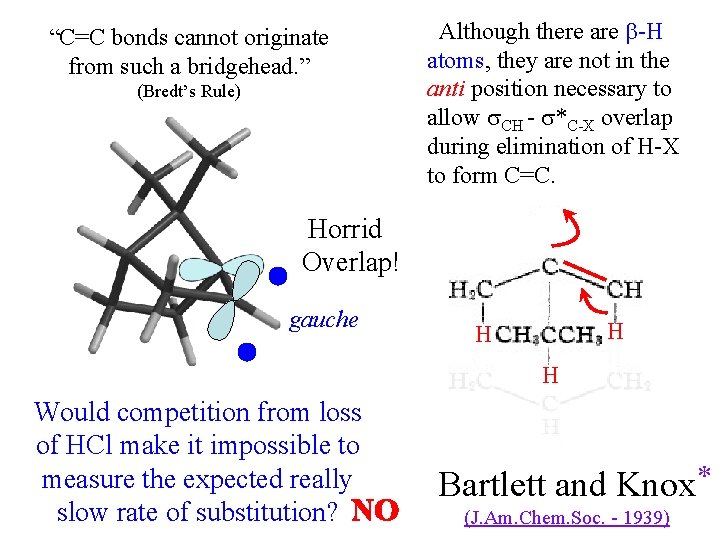
“bridgehead” chloride Cl boat c-hexane with a bridge bicyclo[2. 2. 1]heptane Flattening would generate highly strained angles (estimated >23 kcal/mole). Cation would not be planar. Backside of *C-Cl is inaccessible, and inversion would be impossible. Attack would have to be frontside. Bartlett and Knox * (J. Am. Chem. Soc. - 1939)

“C=C bonds cannot originate from such a bridgehead. ” (Bredt’s Rule) Although there are -H atoms, they are not in the anti position necessary to allow CH - *C-X overlap during elimination of H-X to form C=C. Horrid Overlap! gauche H H H Would competition from loss of HCl make it impossible to measure the expected really slow rate of substitution? NO Bartlett and Knox * (J. Am. Chem. Soc. - 1939)

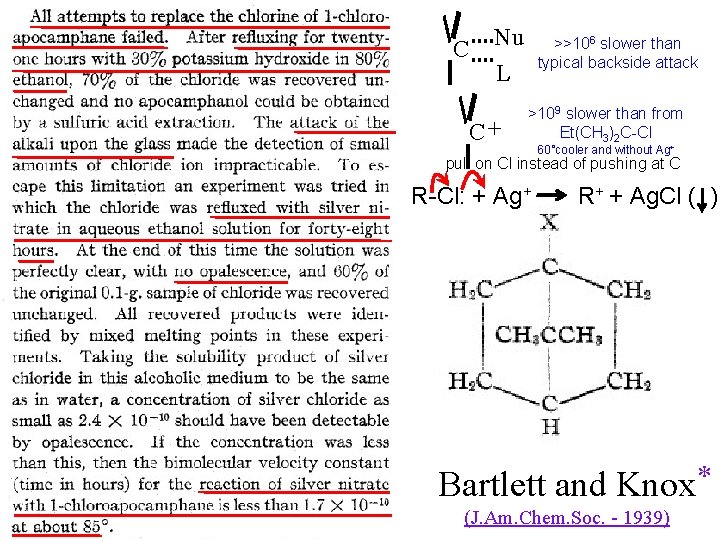
C Nu >>106 slower than typical backside attack L C+ >109 slower than from Et(CH 3)2 C-Cl 60°cooler and without Ag+ pull on Cl instead of pushing at C R-Cl: + Ag+ R+ + Ag. Cl ( ) Bartlett and Knox * (J. Am. Chem. Soc. - 1939)
![Cycloalkyl Halides e g JF Table 7 2 krelative 109 60 1 0 0001 Cycloalkyl Halides (e. g. J&F Table 7. 2) krelative ~109° 60° [1] <0. 0001](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-26.jpg)
Cycloalkyl Halides (e. g. J&F Table 7. 2) krelative ~109° 60° [1] <0. 0001 strain in starting material ? ? ? 90° 109° Br 120° C C C H sp 2 I increased strain in transition state 0. 008 1. 6 0. 01 OK bent

Rate Constant Dependance on Nu: Nucleophile Nu Solvent R-L (+) Nu-R Leaving Substrate Group krel (-) L Product p. Ka (Nu. H+) H 2 O [1] -1. 7 F- 80 3. 2 Cl- 1, 000 -8 Br- 10, 000 -9 HO- 16, 000 15. 7 I- 80, 000 -10 HS- 126, 000 7 For first-row elements nucleophilicity (attack C-L ) parallels basicity (attack H+). Both require high HOMO. But as atoms get bigger, they get better at attacking C-L (compared to attacking H+) e. g. J&F Sec. 7. 4 d, Table 7. 3
![Rate Constant Dependance on Nucleophile Nu RL Leaving Substrate Group krel p Ka 1 Rate Constant Dependance on Nucleophile Nu R-L Leaving Substrate Group krel p. Ka [1]](https://slidetodoc.com/presentation_image_h2/b3e10dce0e364bfbf71da2195579ea08/image-28.jpg)
Rate Constant Dependance on Nucleophile Nu R-L Leaving Substrate Group krel p. Ka [1] -1. 7 F- 80 3. 2 Cl- 1, 000 -8 Br- 10, 000 -9 HO- 16, 000 15. 7 I- 80, 000 -10 HS- 126, 000 7 Nu-R L Polar solvents accelerate reactions that generate (or concentrate) charge, and vice versa. (Nu. H+) krel CH 3 I in H 2 O harder [1] to break 14 H-bonds to smaller ions 160 krel CH 3 Br in Acetone Backwards H 2 O (-) (+) 11 5 Sensible Nu: Solvent [1] e. g. J&F Sec. 7. 4 dg

End of Lecture 44 Jan. 26, 2011 Copyright © J. M. Mc. Bride 2011. Some rights reserved. Except for cited third-party materials, and those used by visiting speakers, all content is licensed under a Creative Commons License (Attribution-Non. Commercial-Share. Alike 3. 0). Use of this content constitutes your acceptance of the noted license and the terms and conditions of use. Materials from Wikimedia Commons are denoted by the symbol . Third party materials may be subject to additional intellectual property notices, information, or restrictions. The following attribution may be used when reusing material that is not identified as third-party content: J. M. Mc. Bride, Chem 125. License: Creative Commons BY-NC-SA 3. 0