Nucleophilic Substitution Nucleophilic Substitution Y R X Y

Nucleophilic Substitution

Nucleophilic Substitution – Y: + R X Y R + – : X Nucleophile is a Lewis base (electron-pair donor), often negatively charged and used as Na+ or K+ salt. Substrate is usually an alkyl halide.
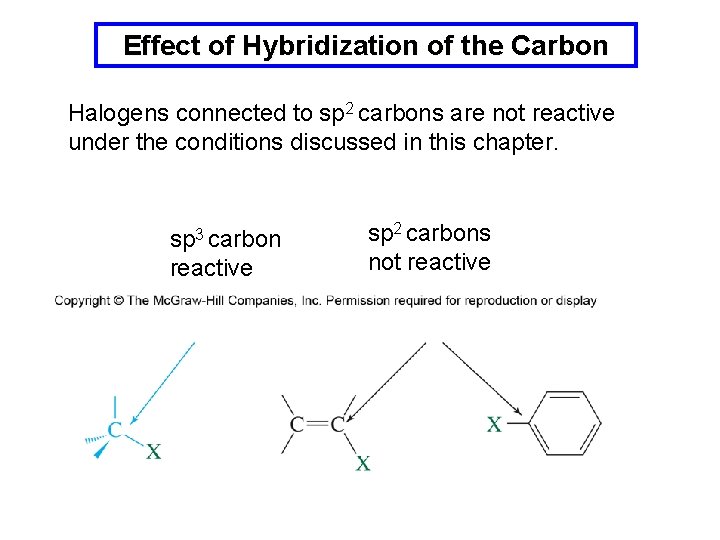
Effect of Hybridization of the Carbon Halogens connected to sp 2 carbons are not reactive under the conditions discussed in this chapter. sp 3 carbon reactive sp 2 carbons not reactive

Nucleophilic Substitution Therefore, substrate cannot be an a vinylic halide or an aryl halide, except under certain conditions to be discussed in Chapter 12.
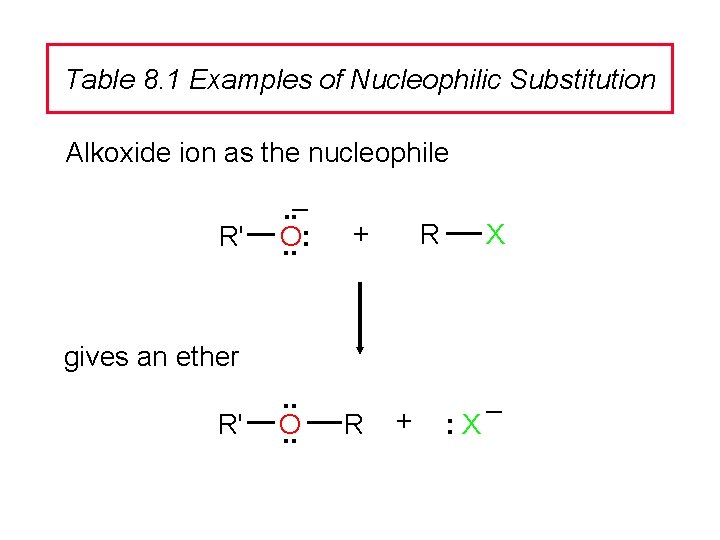
Table 8. 1 Examples of Nucleophilic Substitution Alkoxide ion as the nucleophile R' . . – O: . . + R X gives an ether R' . . O. . R + : X –
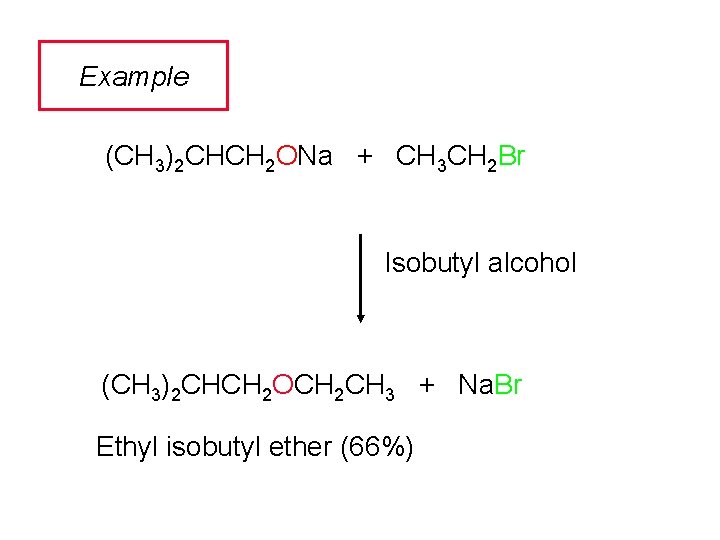
Example (CH 3)2 CHCH 2 ONa + CH 3 CH 2 Br Isobutyl alcohol (CH 3)2 CHCH 2 OCH 2 CH 3 + Na. Br Ethyl isobutyl ether (66%)
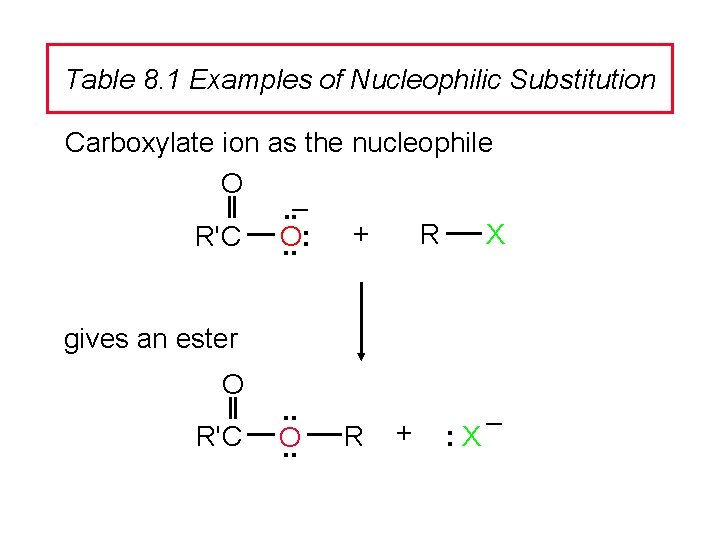
Table 8. 1 Examples of Nucleophilic Substitution Carboxylate ion as the nucleophile O. . – + R X R'C O: . . gives an ester O R'C . . O. . R + : X –
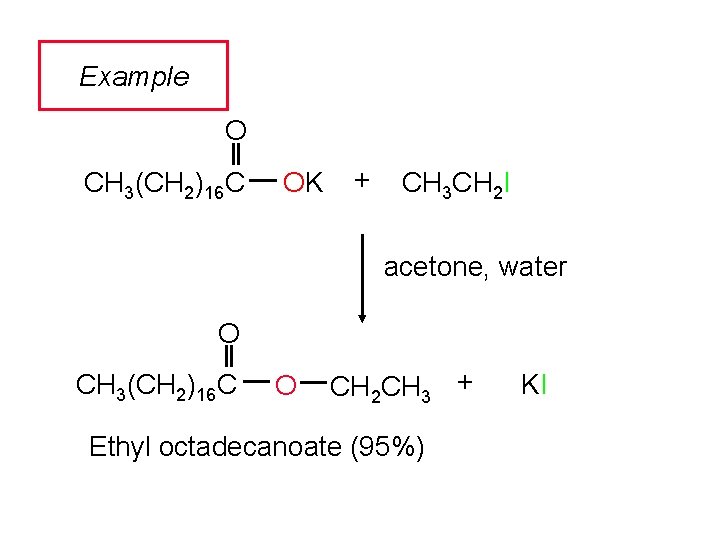
Example O CH 3(CH 2)16 C OK + CH 3 CH 2 I acetone, water O CH 3(CH 2)16 C O CH 2 CH 3 + Ethyl octadecanoate (95%) KI
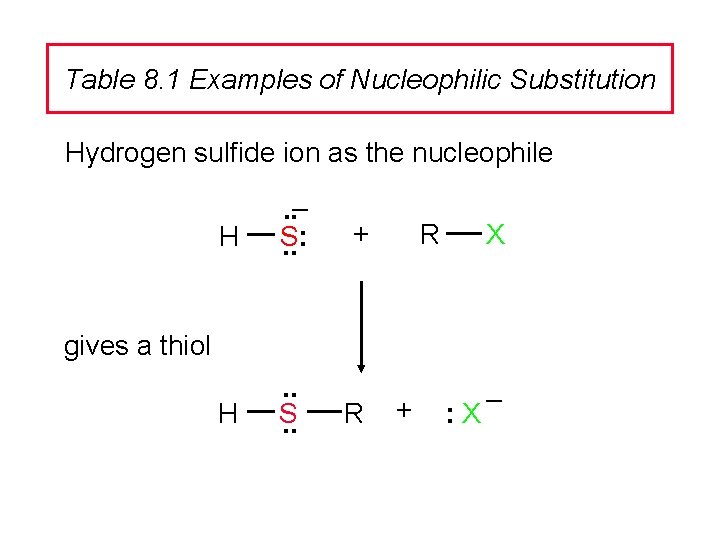
Table 8. 1 Examples of Nucleophilic Substitution Hydrogen sulfide ion as the nucleophile H . . – S: . . H . . S. . + R X gives a thiol R + : X –
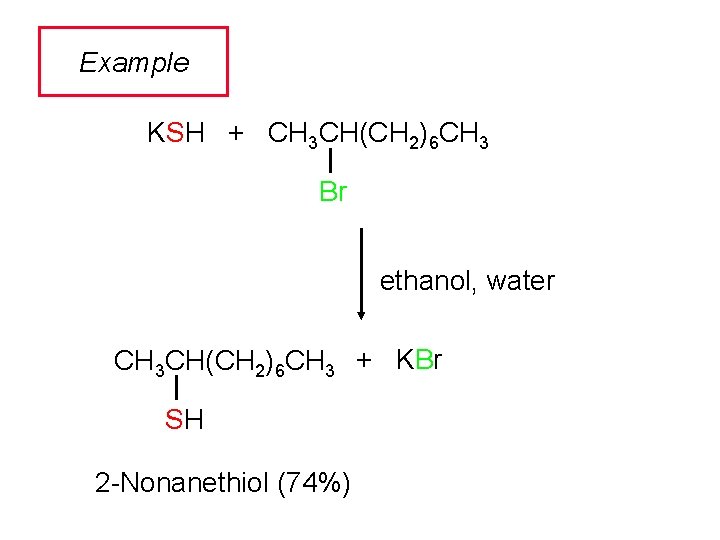
Example KSH + CH 3 CH(CH 2)6 CH 3 Br ethanol, water CH 3 CH(CH 2)6 CH 3 + KBr SH 2 -Nonanethiol (74%)

Table 8. 1 Examples of Nucleophilic Substitution Cyanide ion as the nucleophile : N – C: + R X gives a nitrile : N C R + : X –
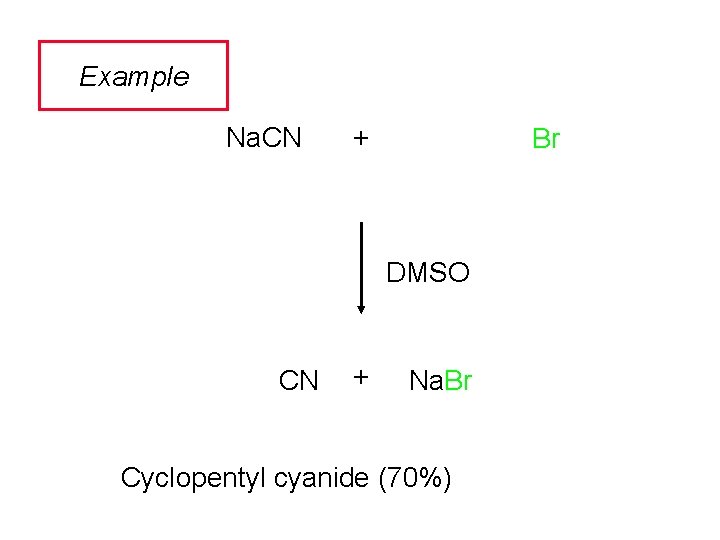
Example Na. CN + Br DMSO CN + Na. Br Cyclopentyl cyanide (70%)

Table 8. 1 Examples of Nucleophilic Substitution Azide ion as the nucleophile – : N. . + N – N. . : gives an alkyl azide + – : N N N. . + R R + X : X –
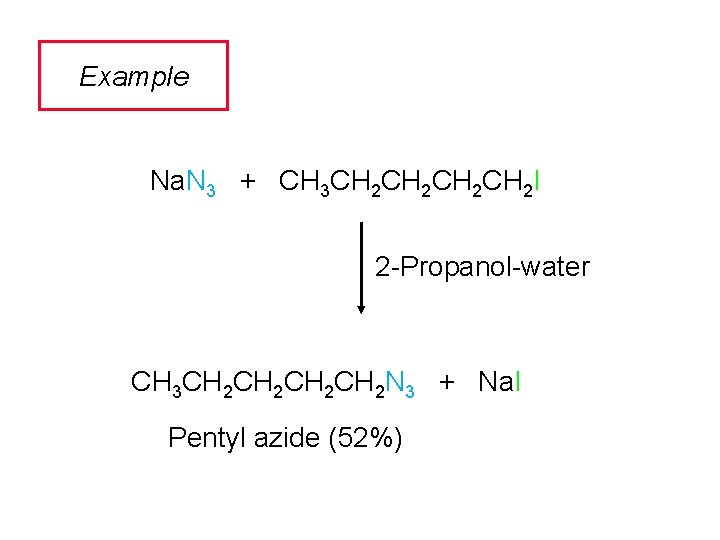
Example Na. N 3 + CH 3 CH 2 CH 2 I 2 -Propanol-water CH 3 CH 2 CH 2 N 3 + Na. I Pentyl azide (52%)

Table 8. 1 Examples of Nucleophilic Substitution Iodide ion as the nucleophile. . – : . . I: + R X gives an alkyl iodide. . : . . I R + : X –
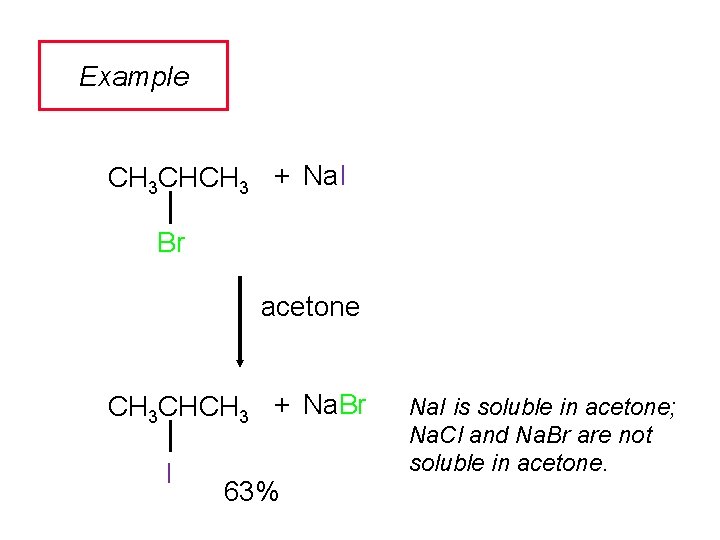
Example CH 3 CHCH 3 + Na. I Br acetone CH 3 CHCH 3 + Na. Br I 63% Na. I is soluble in acetone; Na. Cl and Na. Br are not soluble in acetone.

Relative Reactivity of Halide Leaving Group Alkyl iodides are most reactive since: (a) the carbon-iodine bond is weakest for the halogens; (b) iodide is the weakest base of the halides (this implies that iodide is most stable) since HI is the strongest acid.

SN 2 Mechanism of Nucleophilic Substitution Since the rate of reaction depends on the halide the rate determining step must involve breaking of the carbonhalogen bond. Kinetic studies of the above reaction showed that the rate is also dependent on the hydroxide concentration: The rate determining step therefore involves both the nucleophile and the alkyl halide.
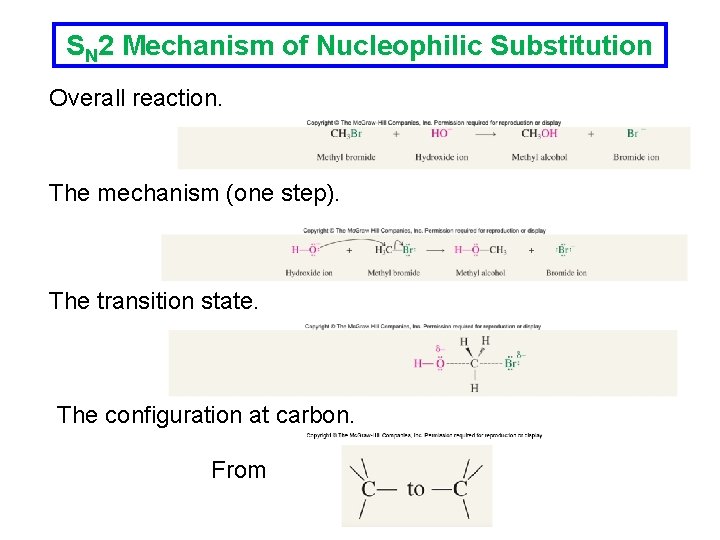
SN 2 Mechanism of Nucleophilic Substitution Overall reaction. The mechanism (one step). The transition state. The configuration at carbon. From
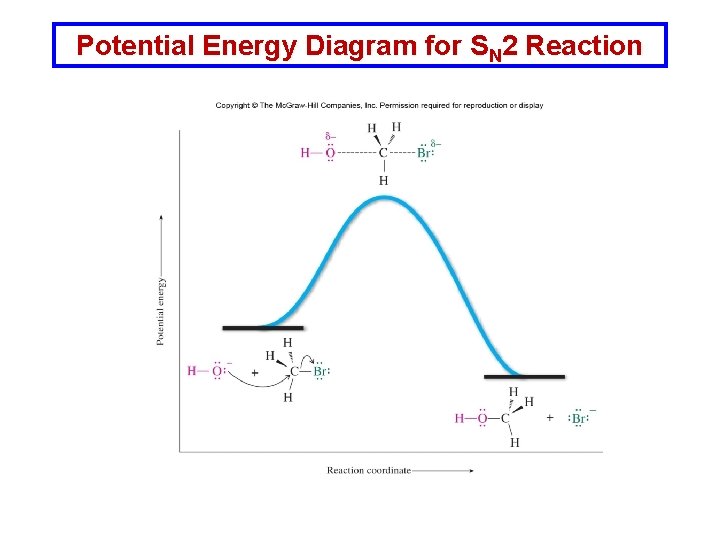
Potential Energy Diagram for SN 2 Reaction
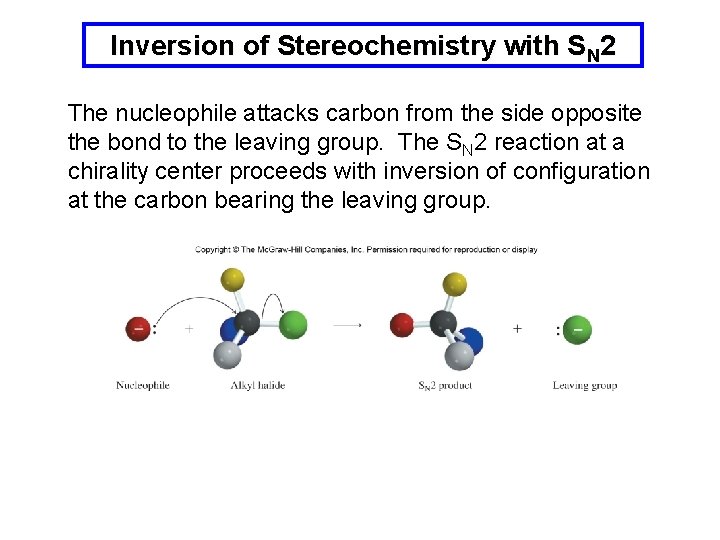
Inversion of Stereochemistry with SN 2 The nucleophile attacks carbon from the side opposite the bond to the leaving group. The SN 2 reaction at a chirality center proceeds with inversion of configuration at the carbon bearing the leaving group.
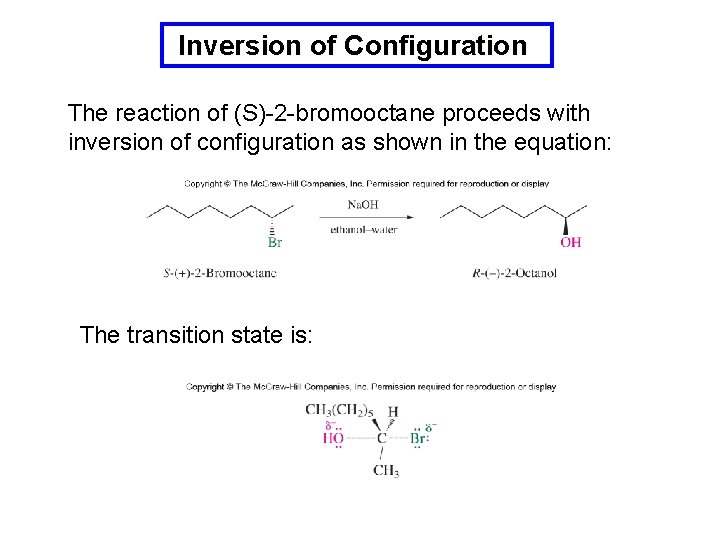
Inversion of Configuration The reaction of (S)-2 -bromooctane proceeds with inversion of configuration as shown in the equation: The transition state is:

Steric Effects and SN 2 Reaction Rates

Effect of the Alkyl Group on the Rate The rate of the reaction: RBr + Li. I RI + Li. Br decreases with increasing steric hindrance. The reaction is fastest with the least hindered methylbromide. The rate of SN 2 reactions normally decreases in the order: CH 3 X > primary > secondary > tertiary
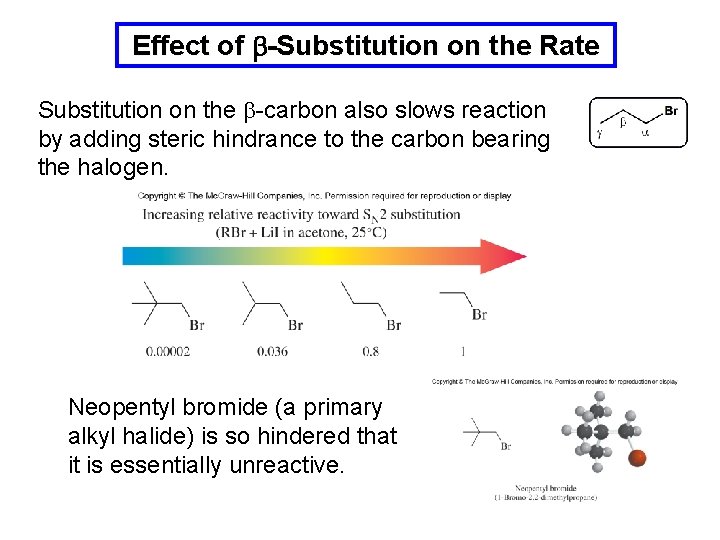
Effect of b-Substitution on the Rate Substitution on the b-carbon also slows reaction by adding steric hindrance to the carbon bearing the halogen. Neopentyl bromide (a primary alkyl halide) is so hindered that it is essentially unreactive.

Nucleophiles and Nucleophilicity
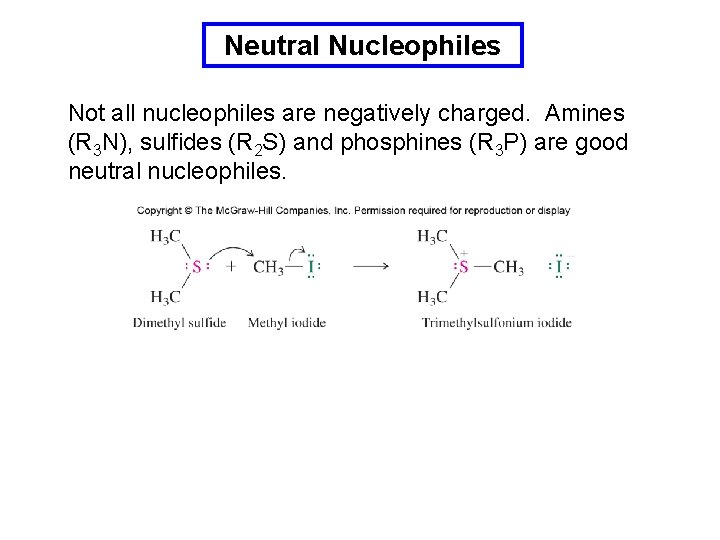
Neutral Nucleophiles Not all nucleophiles are negatively charged. Amines (R 3 N), sulfides (R 2 S) and phosphines (R 3 P) are good neutral nucleophiles.

Neutral Nucleophiles Solvolysis reactions with water (hydrolysis) and alcohols also involve neutral nucleophiles SN 2 hydrolysis reaction mechanism:
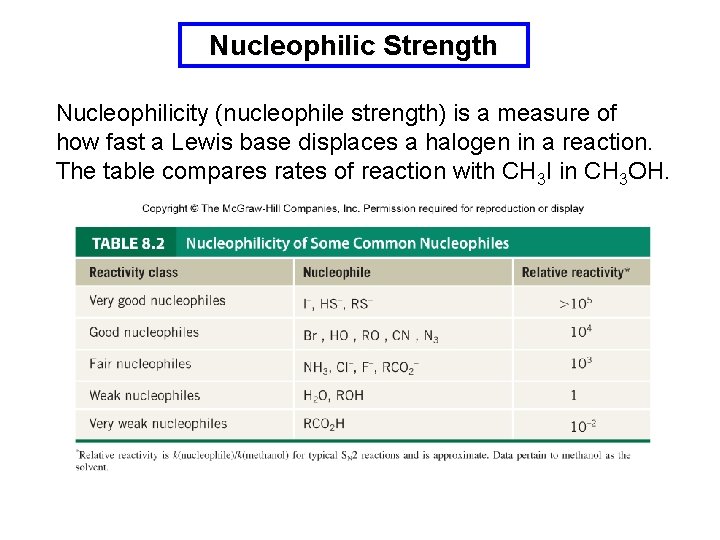
Nucleophilic Strength Nucleophilicity (nucleophile strength) is a measure of how fast a Lewis base displaces a halogen in a reaction. The table compares rates of reaction with CH 3 I in CH 3 OH.

Nucleophilicity When comparing nucleophiles with the same nucleophilic atom then the stronger base is the stronger nucleophile. The stronger base is the conjugate base of the weaker acid. This generalization holds when comparing nucleophiles in the same row of the Periodic table

Nucleophilic Strength When comparing nucleophiles that have the same nucleophilic atom the charged nucleophile is stronger.

Nucleophilic Strength When comparing nucleophiles in the same group of the Periodic Table the most important factor is solvation of the nucleophile. Iodide is the weakest base of the halogens but the best nucleophile. The smaller chloride is a stronger base but is more solvated because it has higher charge density.

The SN 1 Mechanism of Nucleophilic Substitution

A question. . . Tertiary alkyl halides are very unreactive in substitutions that proceed by the SN 2 mechanism. Do they undergo nucleophilic substitution at all? Yes. But by a mechanism different from SN 2. The most common examples are seen in solvolysis reactions.

Nucleophilic Substitution of Tertiary Alkyl halides The reaction (CH 3)3 CBr + 2 H 2 O (CH 3)3 COH + H 3 O+ + Br- follows a first order rate law: Rate = k[(CH 3)3 CBr] And the reaction is termed SN 1.
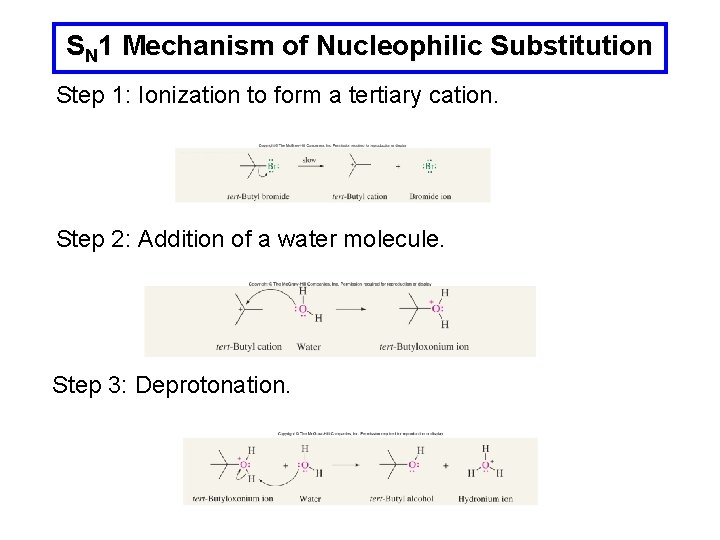
SN 1 Mechanism of Nucleophilic Substitution Step 1: Ionization to form a tertiary cation. Step 2: Addition of a water molecule. Step 3: Deprotonation.
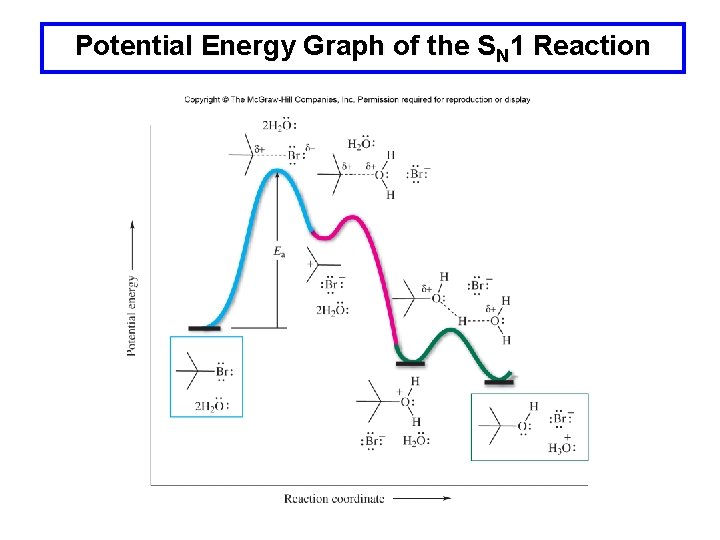
Potential Energy Graph of the SN 1 Reaction
![Characteristics of the SN 1 mechanism first order kinetics: rate = k[RX] unimolecular rate-determining Characteristics of the SN 1 mechanism first order kinetics: rate = k[RX] unimolecular rate-determining](http://slidetodoc.com/presentation_image_h2/532725c01a1389a7f2d5f16d24c99bde/image-38.jpg)
Characteristics of the SN 1 mechanism first order kinetics: rate = k[RX] unimolecular rate-determining step carbocation intermediate rate follows carbocation stability rearrangements sometimes observed reaction is not stereospecific much racemization in reactions of optically active alkyl halides

The Alkyl Halide and the Rate of SN 1 Reaction Comparison of a series of alkyl bromides under SN 1 reaction conditions (solvolysis) reveals that tertiary alkyl halides react fastest. In general, methyl and primary alkyl halides never react by the SN 1 mechanism and tertiary alkyl halides never react by SN 2.
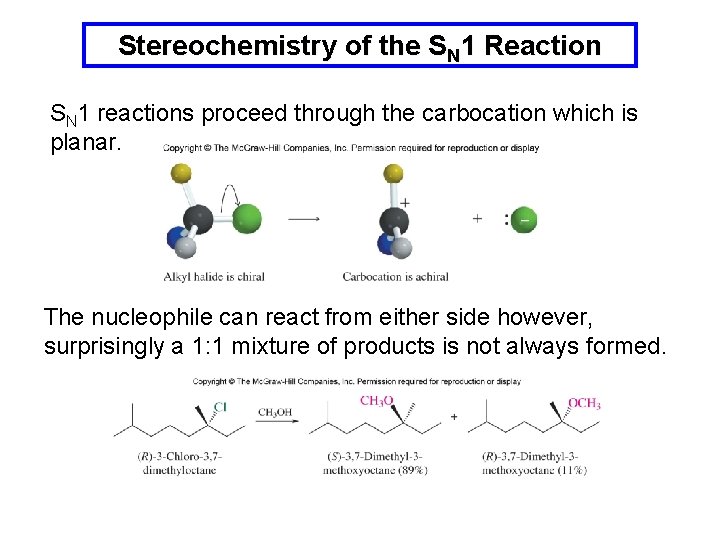
Stereochemistry of the SN 1 Reaction SN 1 reactions proceed through the carbocation which is planar. The nucleophile can react from either side however, surprisingly a 1: 1 mixture of products is not always formed.
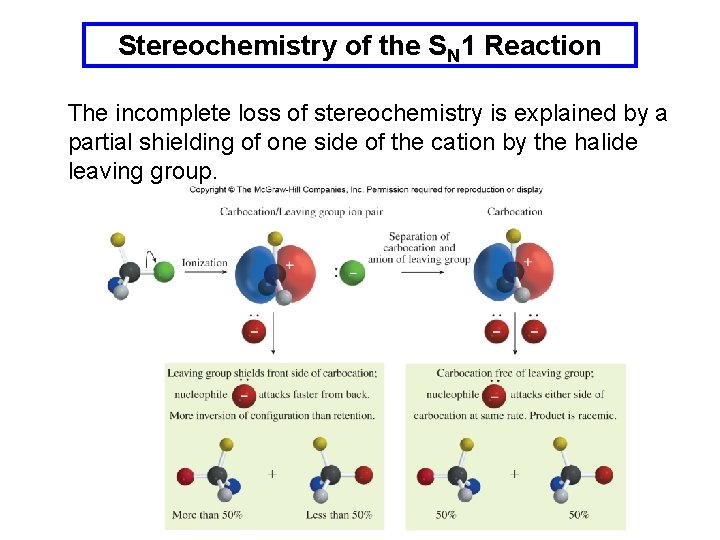
Stereochemistry of the SN 1 Reaction The incomplete loss of stereochemistry is explained by a partial shielding of one side of the cation by the halide leaving group.

Carbocation Rearrangements in SN 1 Reactions Rearrangements are evidence for carbocation intermediates and serve to confirm the SN 1 reaction mechanism.
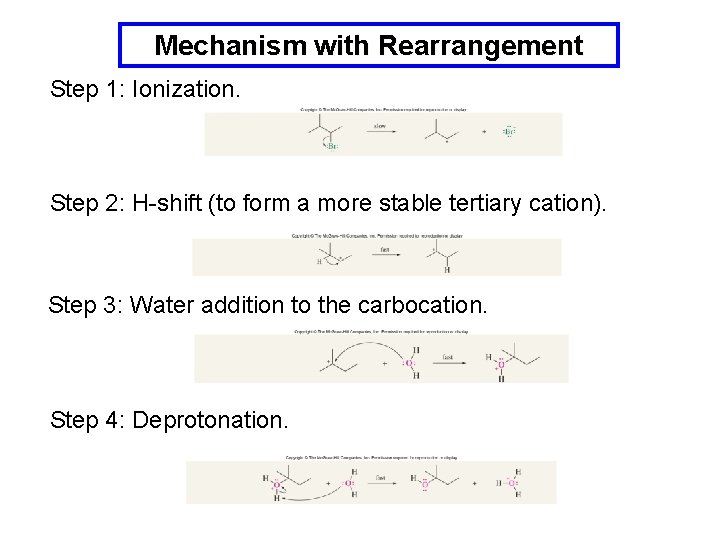
Mechanism with Rearrangement Step 1: Ionization. Step 2: H-shift (to form a more stable tertiary cation). Step 3: Water addition to the carbocation. Step 4: Deprotonation.

Effect of Solvent on Substitution Solvent affects the rate of a reaction not the products formed and the questions are: 1. What properties of the solvent influence the rate most? 2. How does the rate-determining step of the mechanism respond to the properties of the solvent?

Classification of Solvents Protic solvents are those that are capable of hydrogen bonding. Normally they have an –OH group or an N-H group. Aprotic solvents are not hydrogen bond donors. Polarity of a solvent is related to its dielectric constant (e). Solvents with high dielectric constants are considered polar and those with low dielectric constants are non polar.
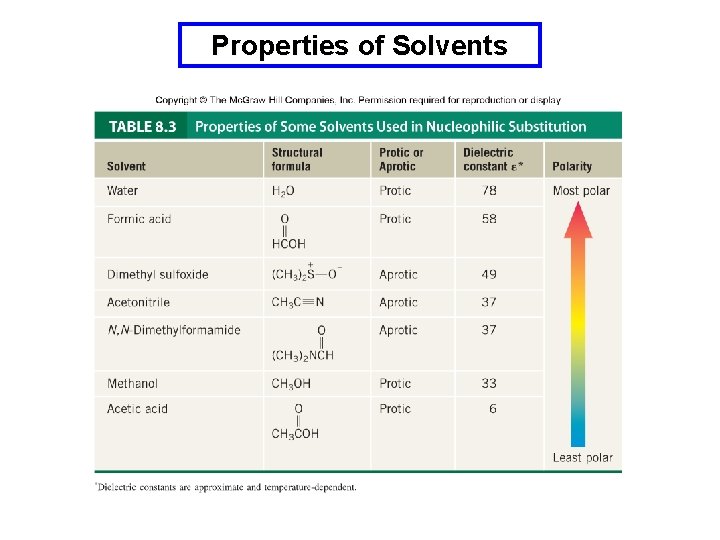
Properties of Solvents

Solvents and SN 2 Reactions Polar protic solvents hydrogen bond to, and solvate, the nucleophile and suppress its nucleophilicity and reduce the rate of reaction. Polar aprotic solvents cannot solvate the nucleophile and the nucleophile is free to react.
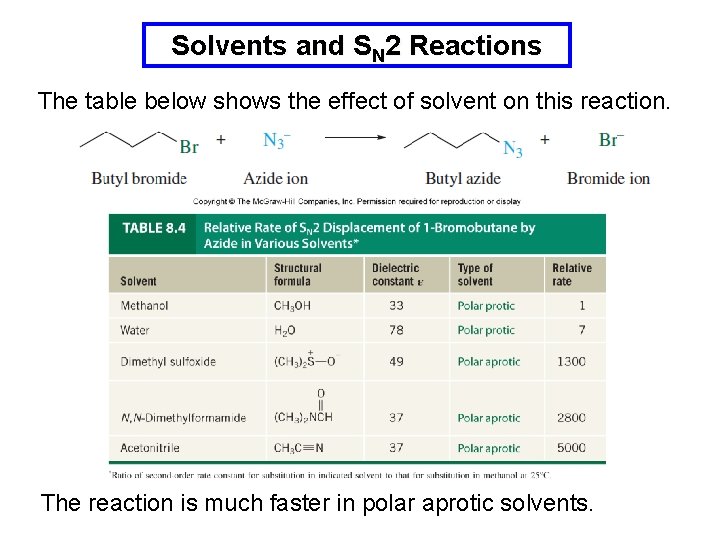
Solvents and SN 2 Reactions The table below shows the effect of solvent on this reaction. The reaction is much faster in polar aprotic solvents.

Solvents and SN 2 Reactions This reaction was studied under two different conditions. Reaction of hexyl bromide in a polar protic solvent required heating for 24 hours to form 76% of hexyl cyanide. With a polar aprotic solvent dimethyl sulfoxide and the less reactive hexyl chloride at room temperature for 20 minutes yielded 91 % of hexyl cyanide.
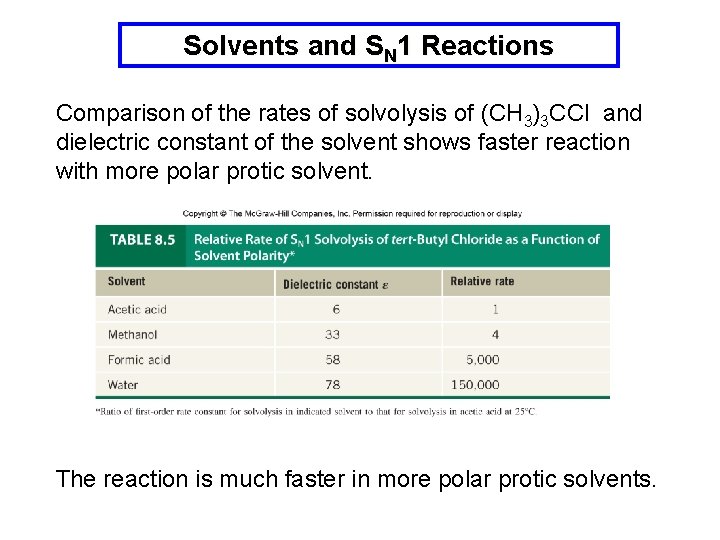
Solvents and SN 1 Reactions Comparison of the rates of solvolysis of (CH 3)3 CCl and dielectric constant of the solvent shows faster reaction with more polar protic solvent. The reaction is much faster in more polar protic solvents.
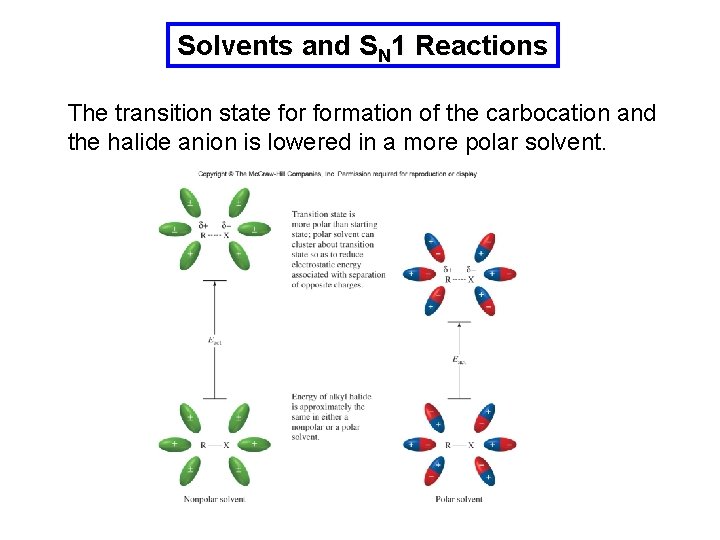
Solvents and SN 1 Reactions The transition state formation of the carbocation and the halide anion is lowered in a more polar solvent.

When. . . Primary alkyl halides undergo nucleophilic substitution: they always react by the SN 2 mechanism. Tertiary alkyl halides undergo nucleophilic substitution: they always react by the SN 1 mechanism. Secondary alkyl halides undergo nucleophilic substitution: they react by the SN 1 mechanism in the presence of a weak nucleophile (solvolysis). SN 2 mechanism in the presence of a good nucleophile.

Substitution and Elimination as Competing Reactions
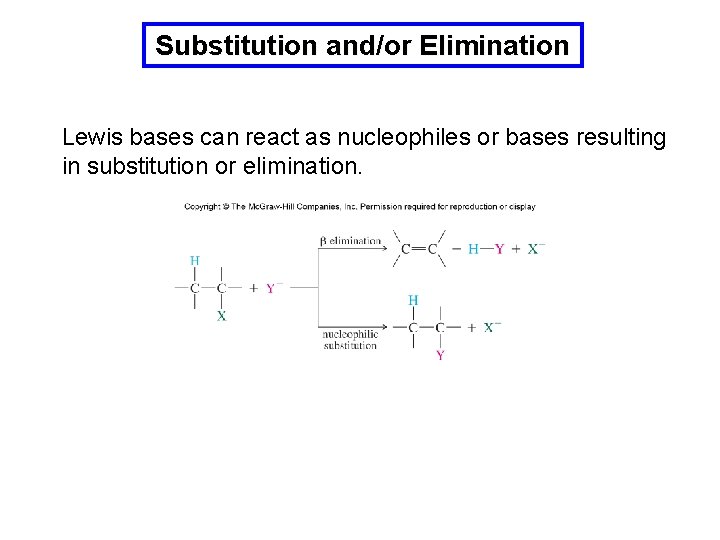
Substitution and/or Elimination Lewis bases can react as nucleophiles or bases resulting in substitution or elimination.
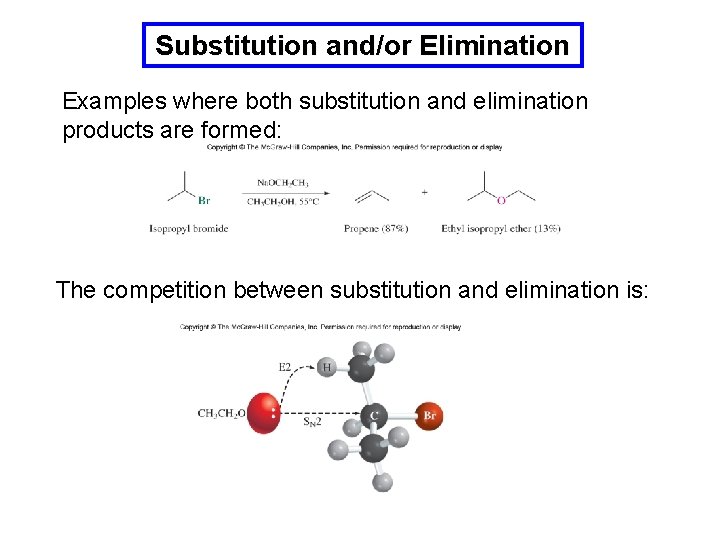
Substitution and/or Elimination Examples where both substitution and elimination products are formed: The competition between substitution and elimination is:

Steric Effect on the SN 2/E 2 Reactions The balance between substitution and elimination can be affected by changing from an unhindered base to a more hindered base. Unhindered base (CH 3 CH 2 ONa): predominantly substitution. Hindered base (CH 3)3 COK: predominantly elimination
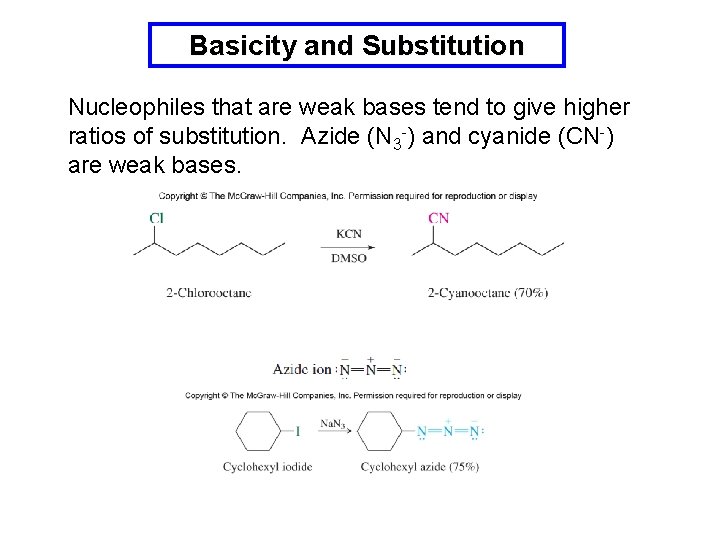
Basicity and Substitution Nucleophiles that are weak bases tend to give higher ratios of substitution. Azide (N 3 -) and cyanide (CN-) are weak bases.
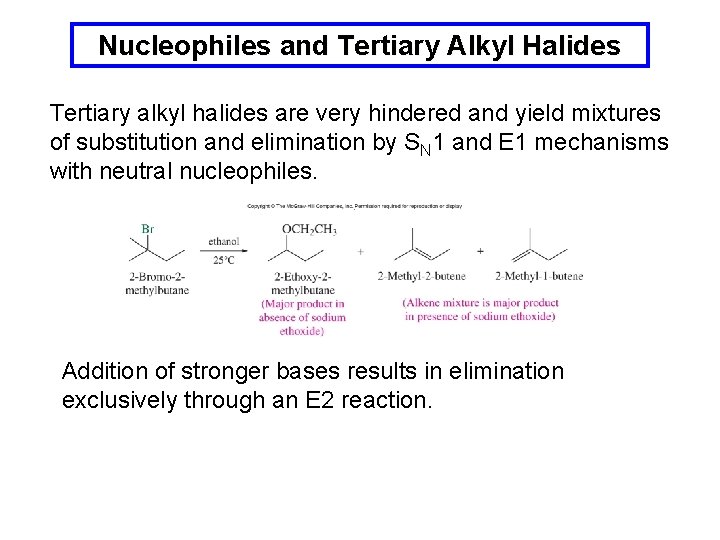
Nucleophiles and Tertiary Alkyl Halides Tertiary alkyl halides are very hindered and yield mixtures of substitution and elimination by SN 1 and E 1 mechanisms with neutral nucleophiles. Addition of stronger bases results in elimination exclusively through an E 2 reaction.

Nucleophilic Substitution of Alkyl Sulfonates
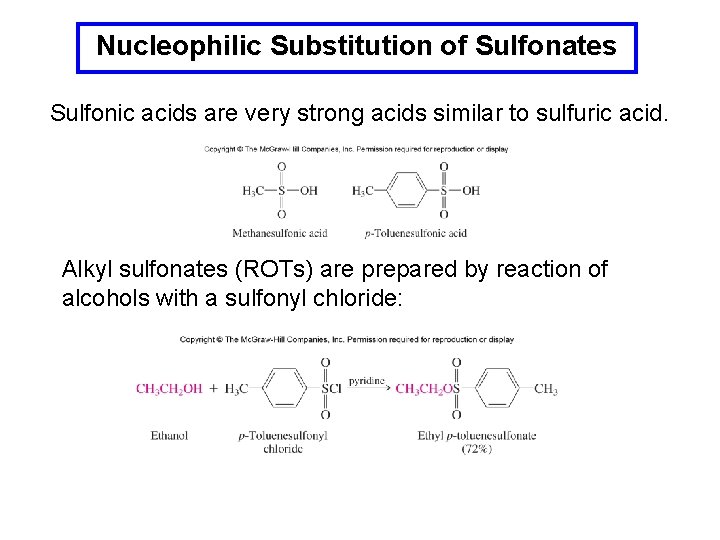
Nucleophilic Substitution of Sulfonates Sulfonic acids are very strong acids similar to sulfuric acid. Alkyl sulfonates (ROTs) are prepared by reaction of alcohols with a sulfonyl chloride:
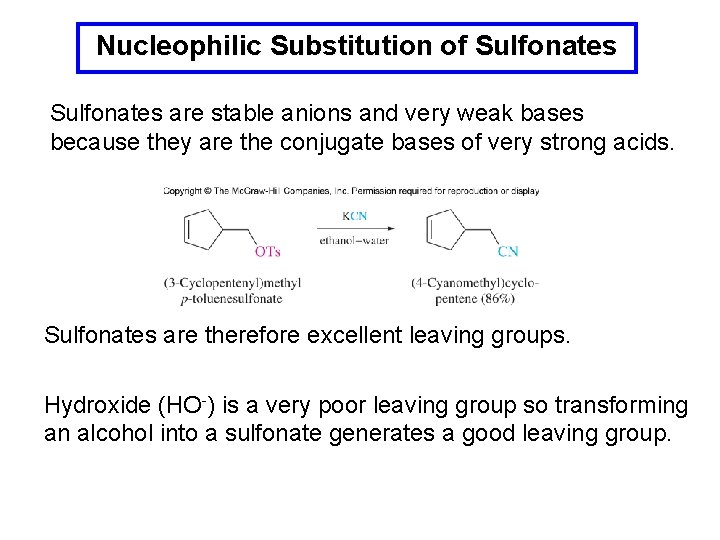
Nucleophilic Substitution of Sulfonates are stable anions and very weak bases because they are the conjugate bases of very strong acids. Sulfonates are therefore excellent leaving groups. Hydroxide (HO-) is a very poor leaving group so transforming an alcohol into a sulfonate generates a good leaving group.
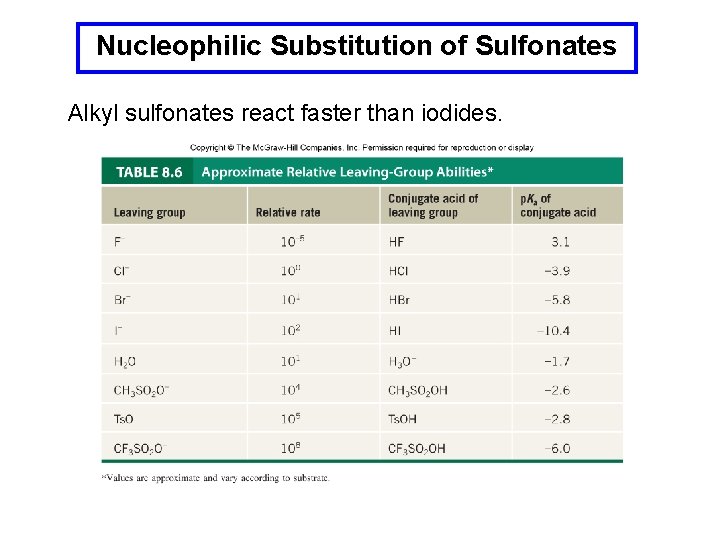
Nucleophilic Substitution of Sulfonates Alkyl sulfonates react faster than iodides.

Nucleophilic Substitution of Sulfonates are better leaving groups than halides so they can be used to form alkyl halides by substitution.
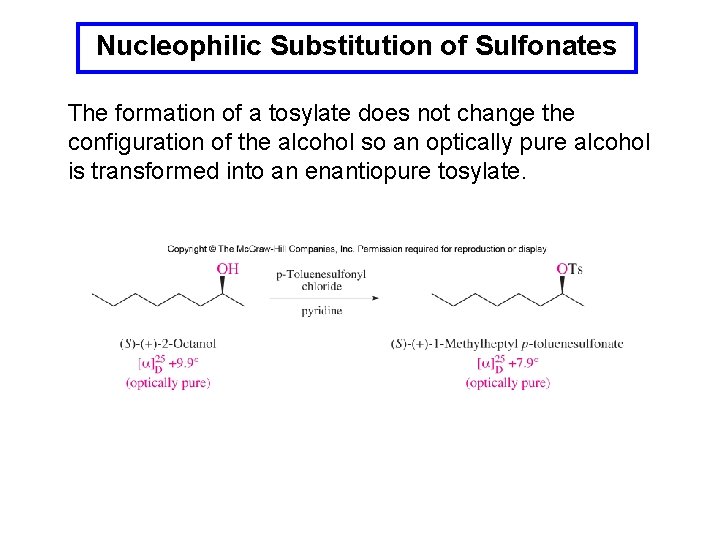
Nucleophilic Substitution of Sulfonates The formation of a tosylate does not change the configuration of the alcohol so an optically pure alcohol is transformed into an enantiopure tosylate.
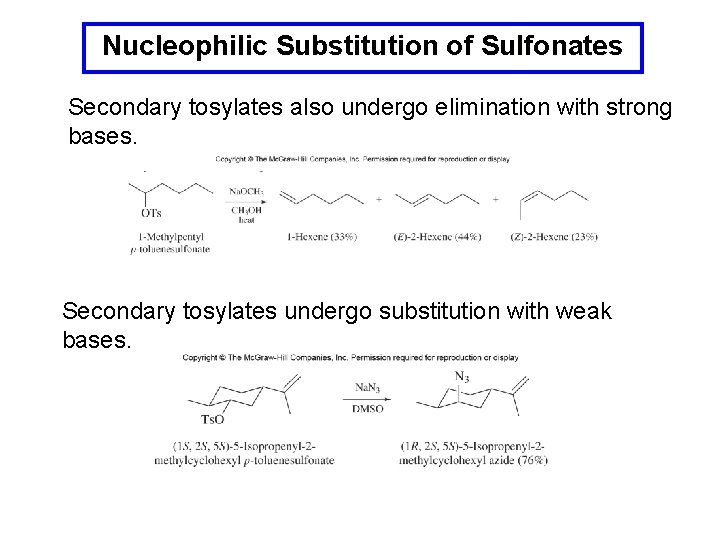
Nucleophilic Substitution of Sulfonates Secondary tosylates also undergo elimination with strong bases. Secondary tosylates undergo substitution with weak bases.

Nucleophilic Substitution and Retrosynthetic Analysis
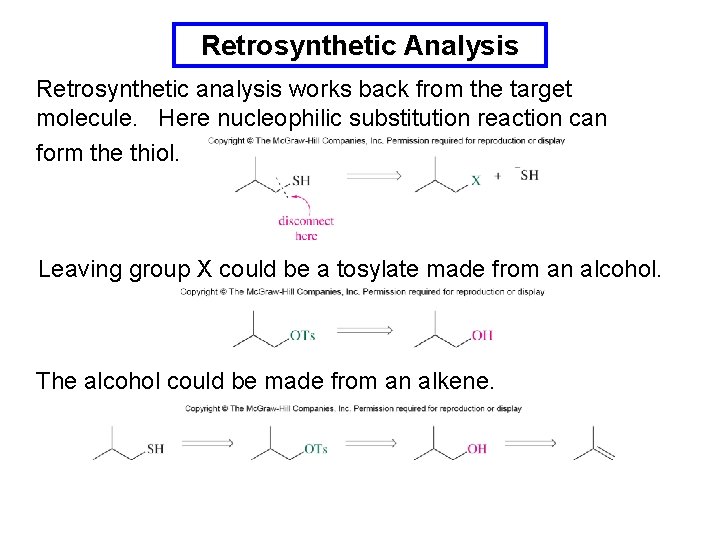
Retrosynthetic Analysis Retrosynthetic analysis works back from the target molecule. Here nucleophilic substitution reaction can form the thiol. Leaving group X could be a tosylate made from an alcohol. The alcohol could be made from an alkene.

Retrosynthetic Analysis to Synthesis The full retrosynthetic analysis is: Once the retrosynthetic analysis has been developed simply write the synthesis starting from the alkene. Anti-Markovnikov addition of H-OH
- Slides: 68