Organic Chemistry 4 th Edition Paula Yurkanis Bruice
Organic Chemistry 4 th Edition Paula Yurkanis Bruice Chapter 10 Substitution Reactions of Alkyl Halides Irene Lee Case Western Reserve University Cleveland, OH © 2004, Prentice Hall

What is a substitution reaction? The atom or group that is substituted or eliminated in these reactions is called a leaving group

Alkyl halides have relatively good leaving groups How do alkyl halides react? Alternatively …

Because a nucleophile substitutes for the halogen, these reactions are known as nucleophilic substitution reactions The reaction mechanism which predominates depends on the following factors: • the structure of the alkyl halide • the reactivity of the nucleophile • the concentration of the nucleophile • the solvent of the reaction
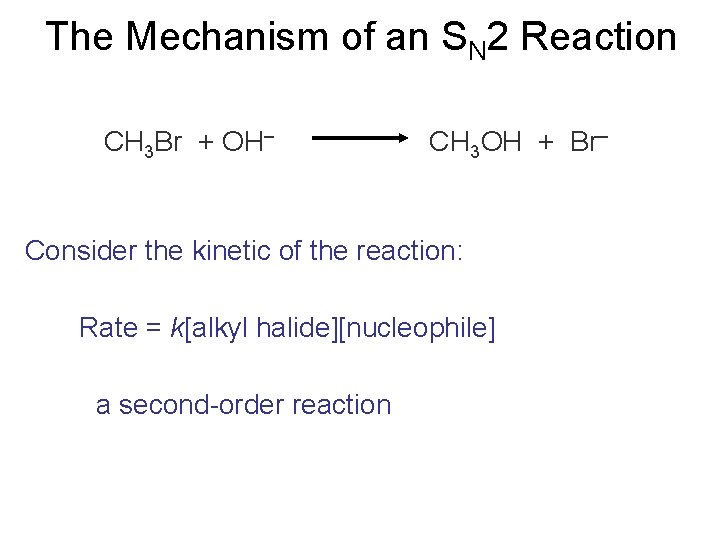
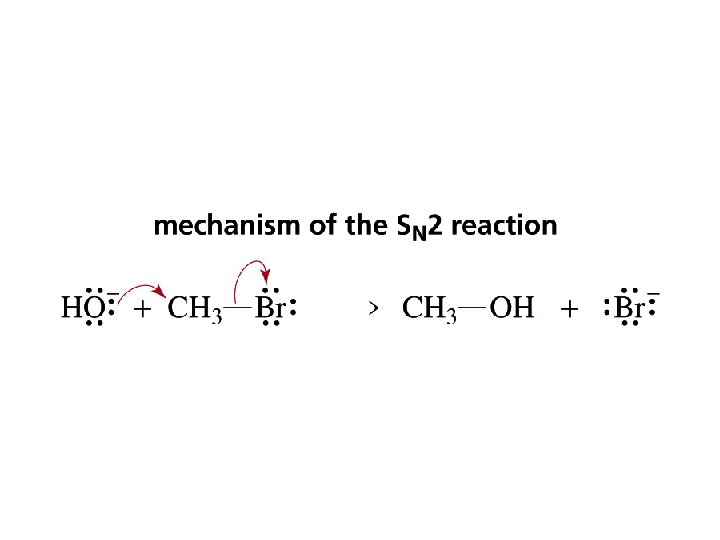
The Mechanism of an SN 2 Reaction CH 3 Br + OH– CH 3 OH + Br– Consider the kinetic of the reaction: Rate = k[alkyl halide][nucleophile] a second-order reaction

Three Experimental Evidences Support an SN 2 Reaction Mechanism 1. The rate of the reaction is dependent on the concentration of the alkyl halides and the nucleophile 2. The rate of the reaction with a given nucleophile decreases with increasing size of the alkyl halides 3. The configuration of the substituted product is inverted compared to the configuration of the reacting chiral alkyl halide

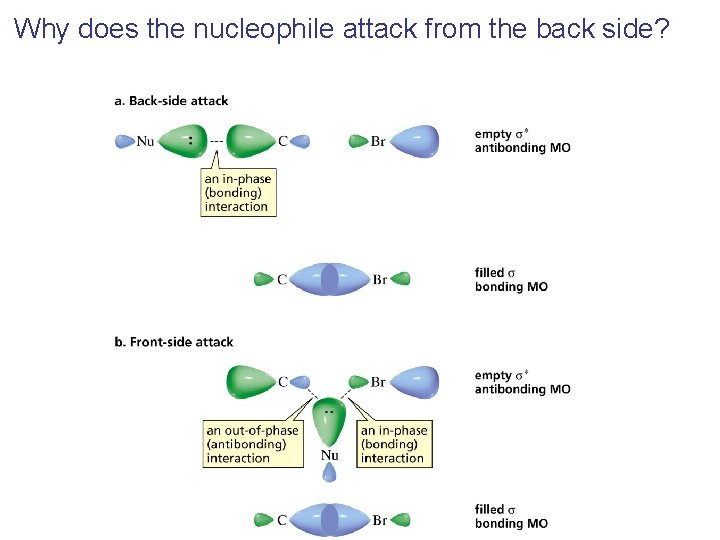
Why does the nucleophile attack from the back side?
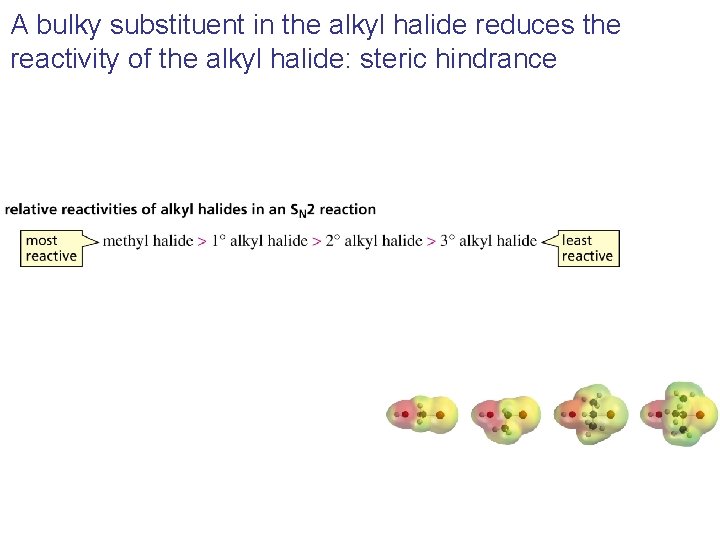
A bulky substituent in the alkyl halide reduces the reactivity of the alkyl halide: steric hindrance
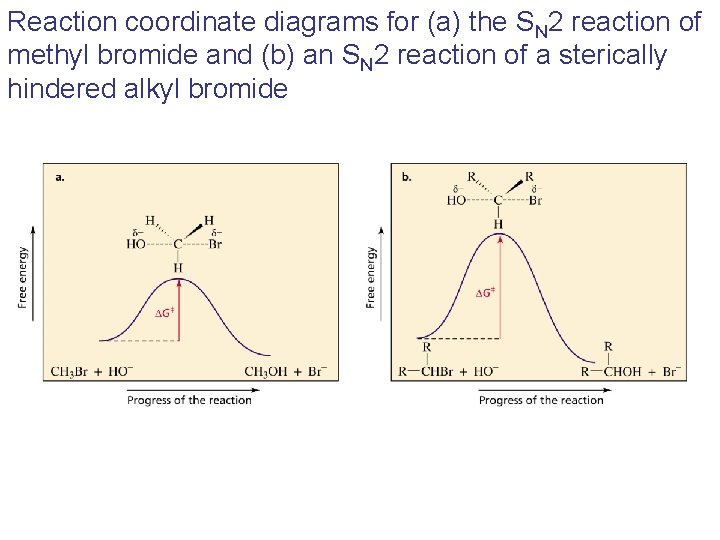
Reaction coordinate diagrams for (a) the SN 2 reaction of methyl bromide and (b) an SN 2 reaction of a sterically hindered alkyl bromide
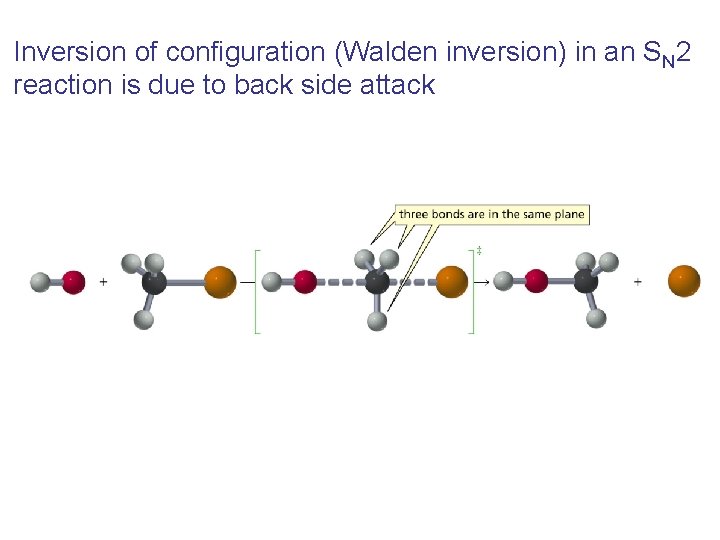
Inversion of configuration (Walden inversion) in an SN 2 reaction is due to back side attack

SN 2 Reactions Are Affected by the Leaving Group

The weaker the base, the better it is as a leaving group

Carbon and iodide have the same electronegativity Why is RI the most reactive? Large atoms are more polarizable than small atoms The high polarizability of a large iodide atom causes it to react as if it were polar

The Nucleophile Affects an SN 2 Reaction Nucleophilicity is a measure of how readily a compound (a nucleophile) is able to attack an electron -deficient atom Nucleophilicity is measured by a rate constant (k) UNLIKE Basicity is a measure of how well a compound (a base) shares its lone pair with a proton Basicity is measured by the acid dissociation constant (Ka)

When comparing molecules with the same attacking atom stronger base, better nucleophile OH– CH 3 O– –NH 2 CH 3 CH 2 NH– weaker base, poorer nucleophile > > H 2 O CH 3 OH NH 3 CH 3 CH 2 NH 2
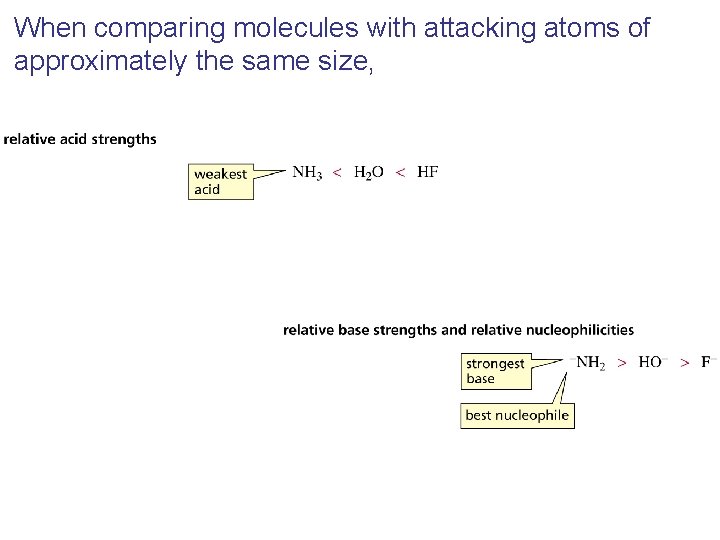
When comparing molecules with attacking atoms of approximately the same size,
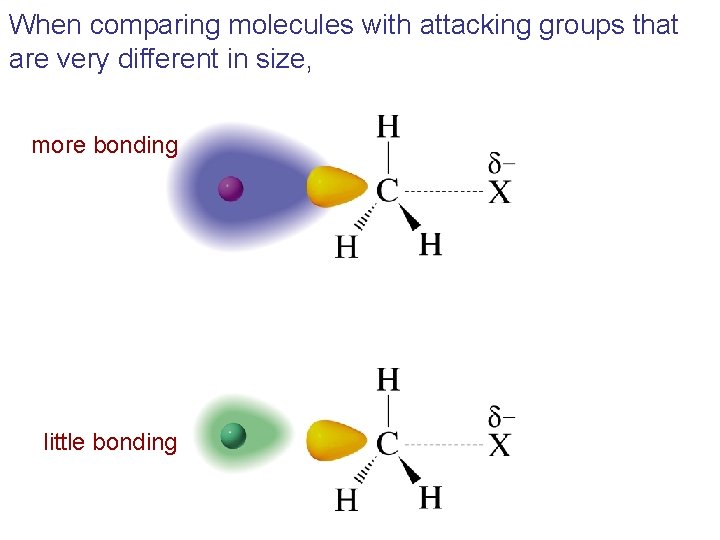
When comparing molecules with attacking groups that are very different in size, more bonding little bonding
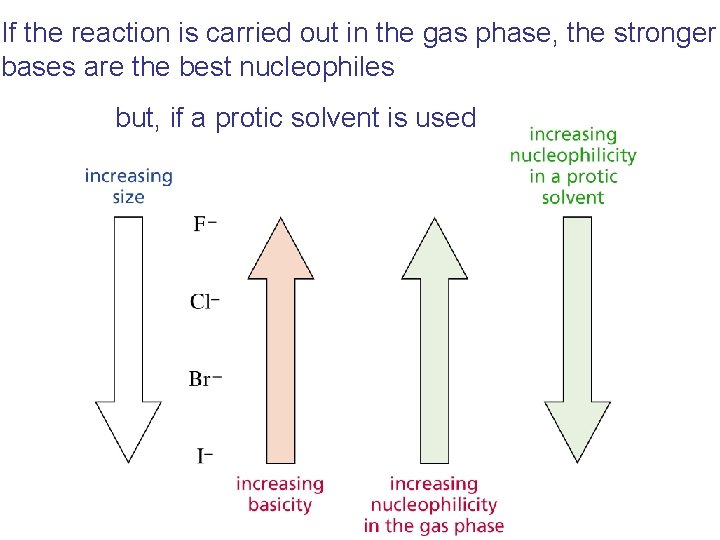
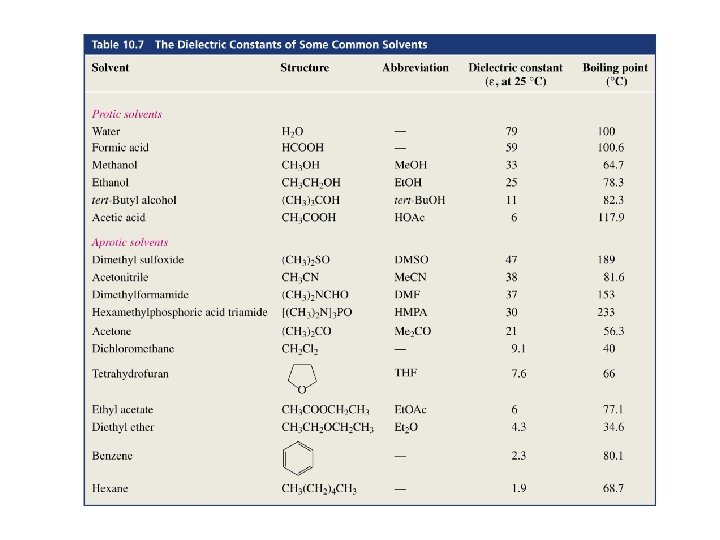
If the reaction is carried out in the gas phase, the stronger bases are the best nucleophiles but, if a protic solvent is used

The Effect of Solvent on Nucleophilicity How does a protic solvent make strong bases less nucleophilic?
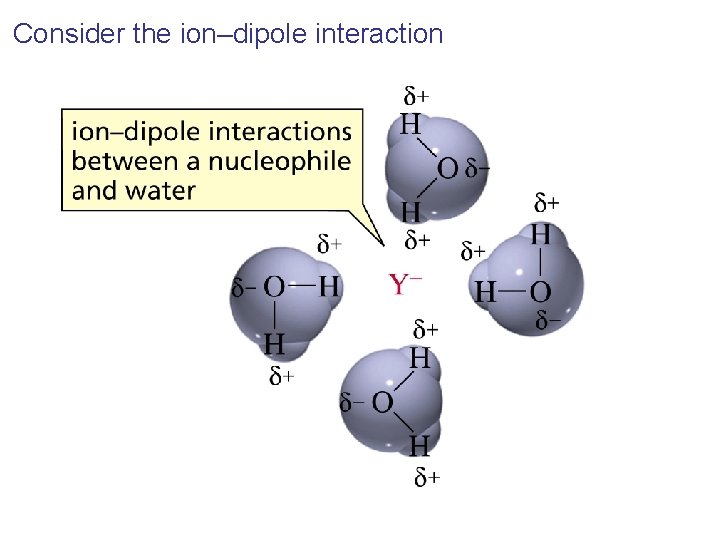
Consider the ion–dipole interaction

It is easier to break the ion-dipole interactions between a weak base and the solvent than between a strong base and the solvent Therefore, fluoride is a better nucleophile in nonpolar solvent Also, aprotic polar solvents such as DMSO and DMF facilitate the reaction of ionic compounds because they solvate cations

Nucleophilicity Is Affected by Steric Effects Steric effects affect nucleophilicity, but not basicity

An SN 2 reaction proceeds in the direction that allows the strongest base to displace the weaker base

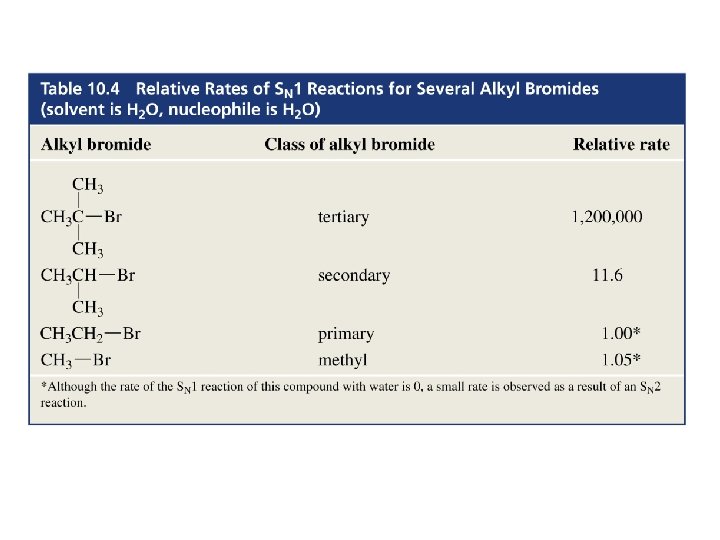
Experimental Evidence for an SN 1 Reaction 1. The rate of the reaction depends only on the concentration of the alkyl halide 2. The rate of the reaction is favored by the bulkiness of the alkyl substituent 3. In the substitution of a chiral alkyl halide, a racemic mixture of product is obtained
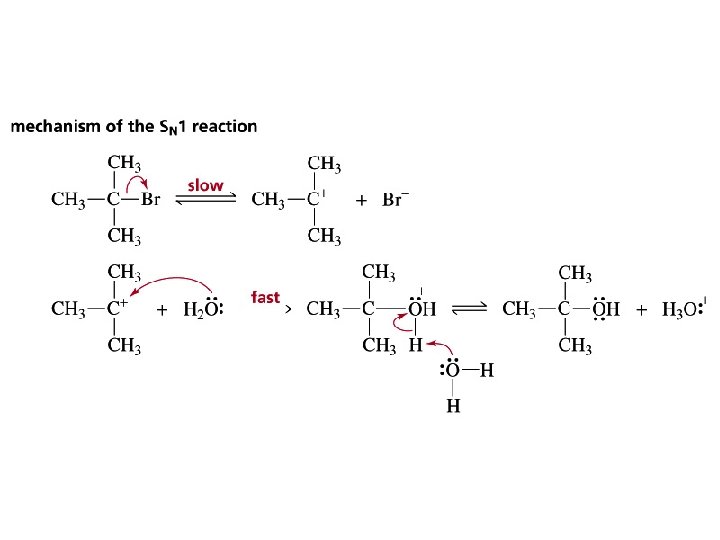
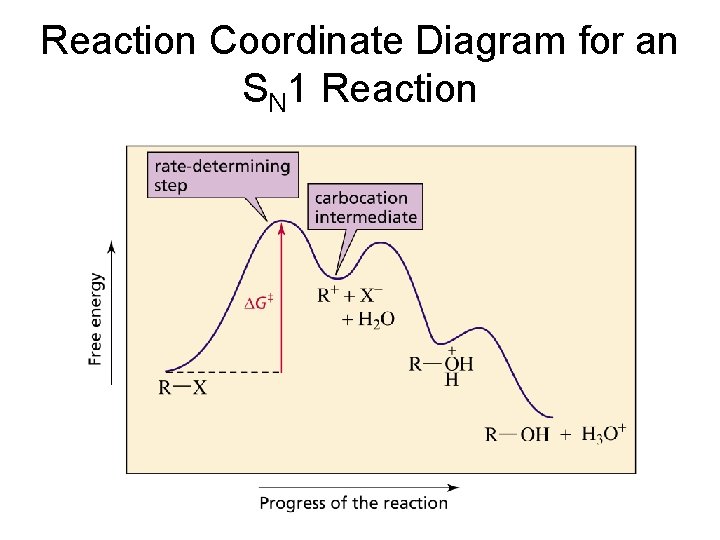
Reaction Coordinate Diagram for an SN 1 Reaction

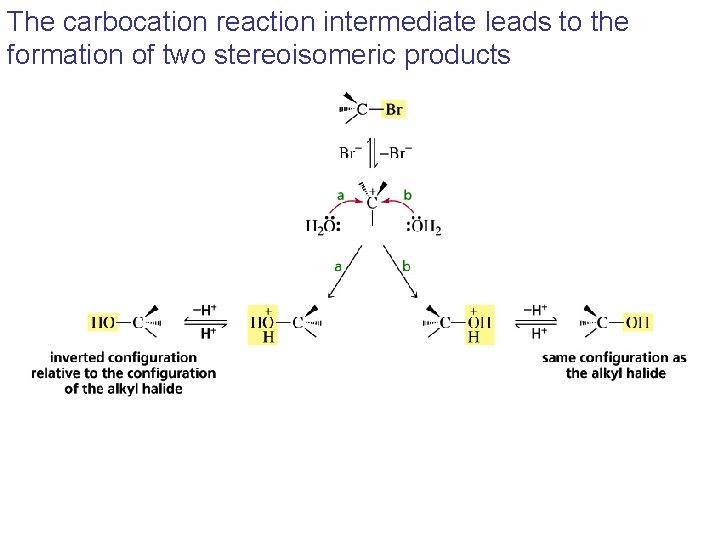
The carbocation reaction intermediate leads to the formation of two stereoisomeric products

The Effect of the Leaving Group on an SN 1 Reaction The nucleophile has no effect on the rate of an SN 1 reaction
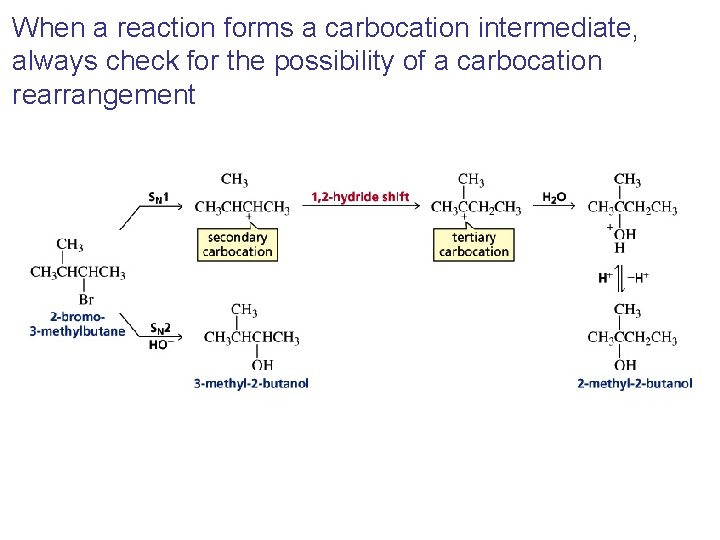
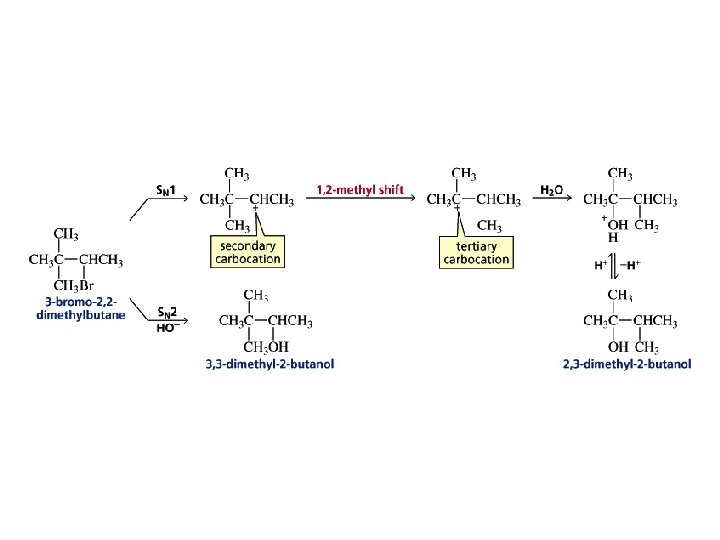
When a reaction forms a carbocation intermediate, always check for the possibility of a carbocation rearrangement
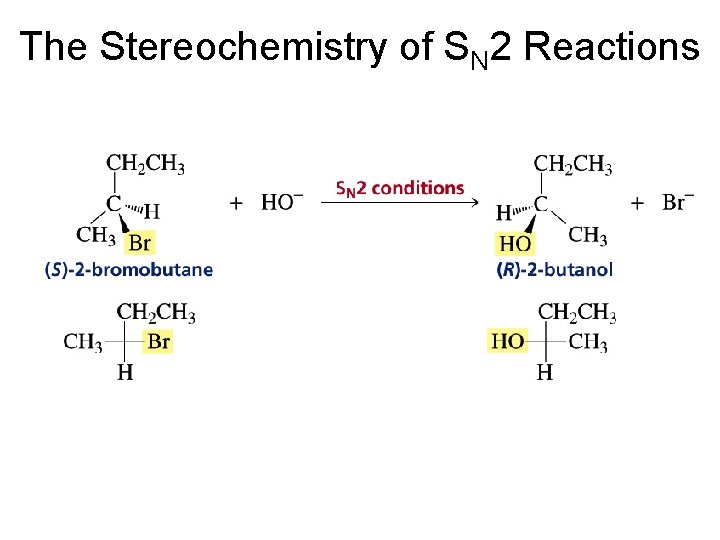
The Stereochemistry of SN 2 Reactions
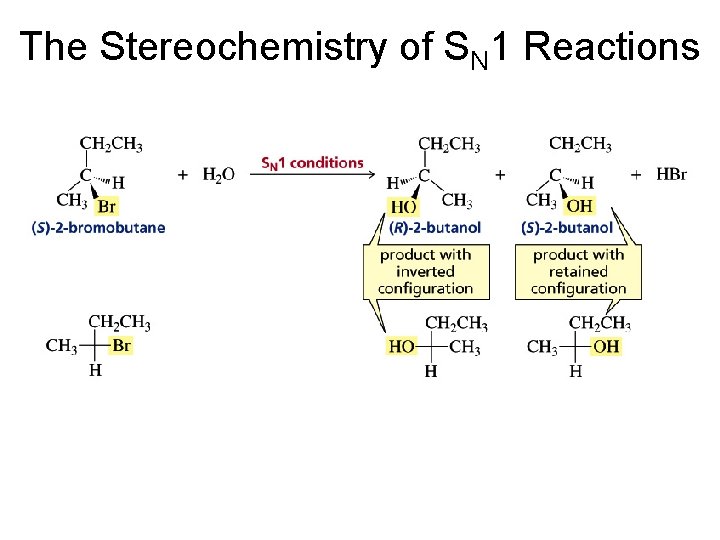
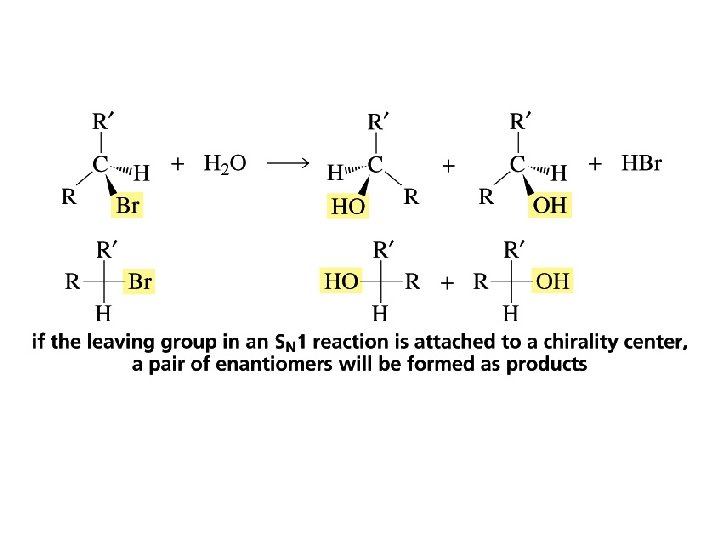
The Stereochemistry of SN 1 Reactions
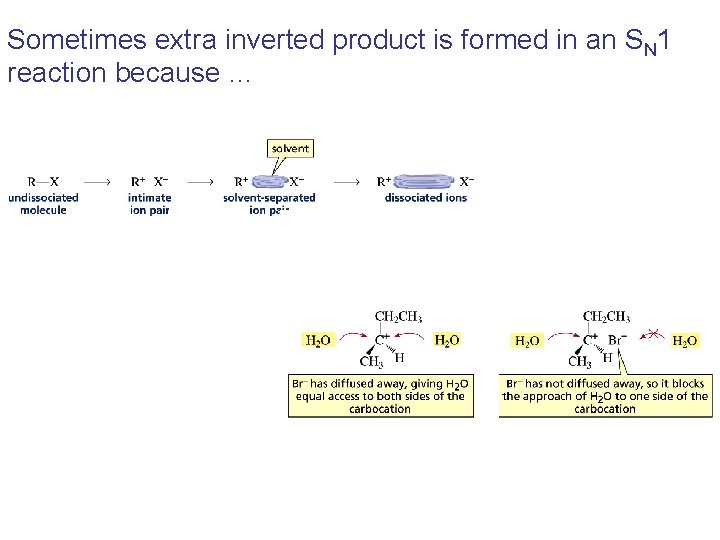
Sometimes extra inverted product is formed in an SN 1 reaction because …
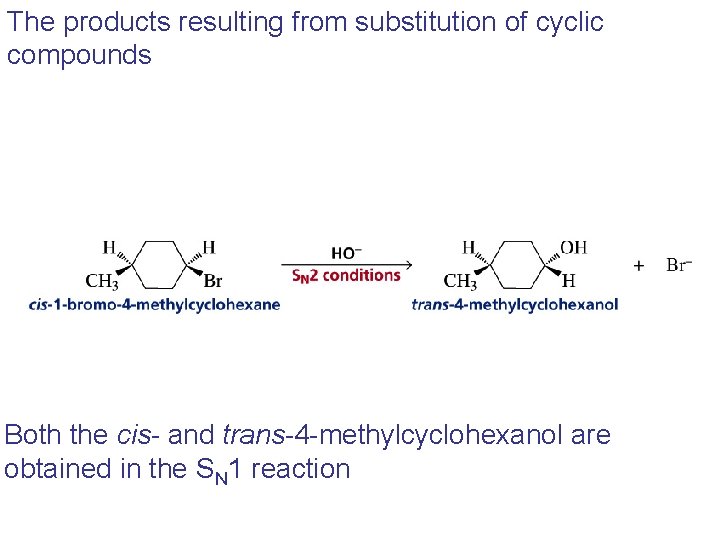
The products resulting from substitution of cyclic compounds Both the cis- and trans-4 -methylcyclohexanol are obtained in the SN 1 reaction
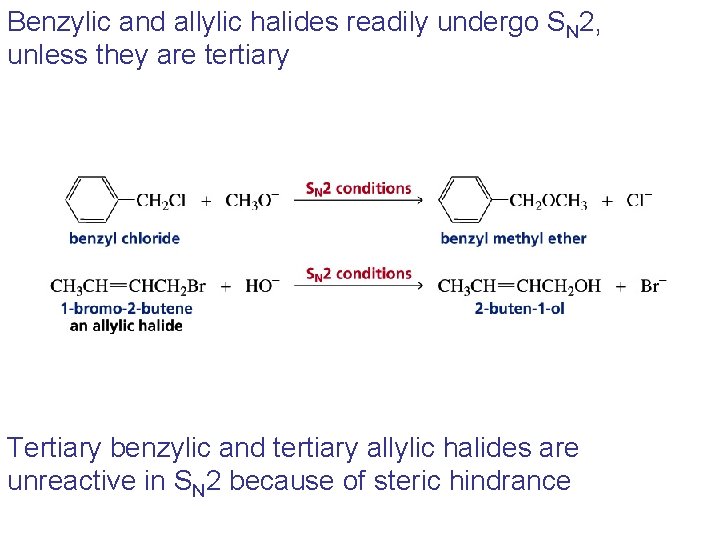
Benzylic and allylic halides readily undergo SN 2, unless they are tertiary Tertiary benzylic and tertiary allylic halides are unreactive in SN 2 because of steric hindrance
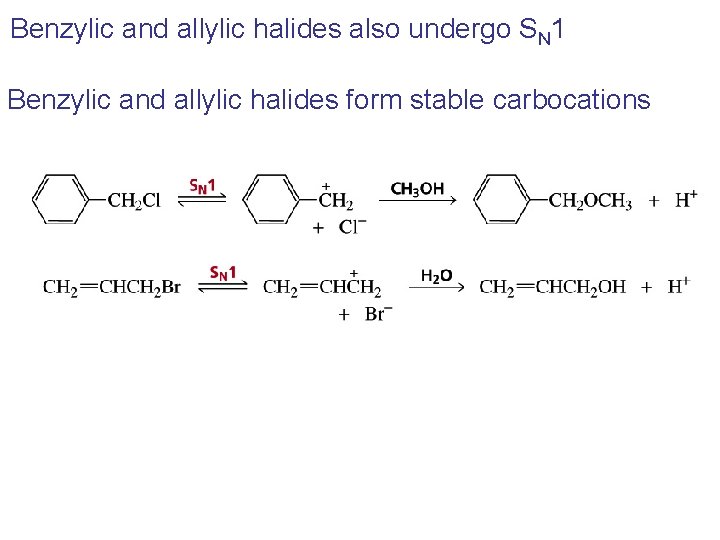
Benzylic and allylic halides also undergo SN 1 Benzylic and allylic halides form stable carbocations
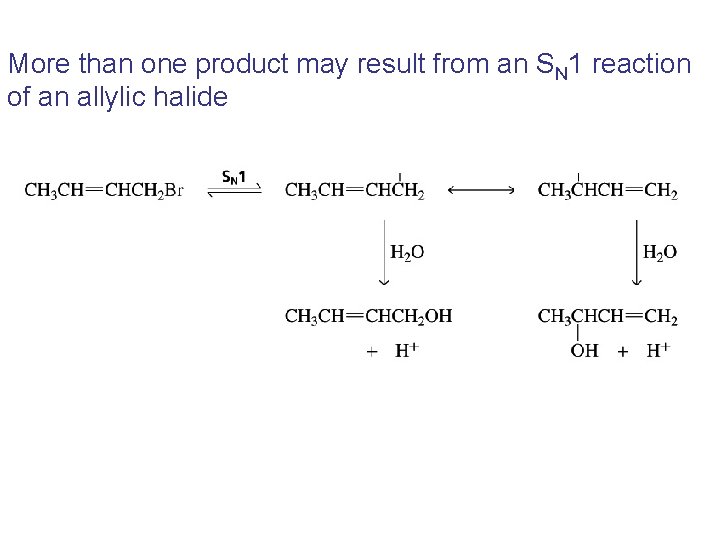
More than one product may result from an SN 1 reaction of an allylic halide
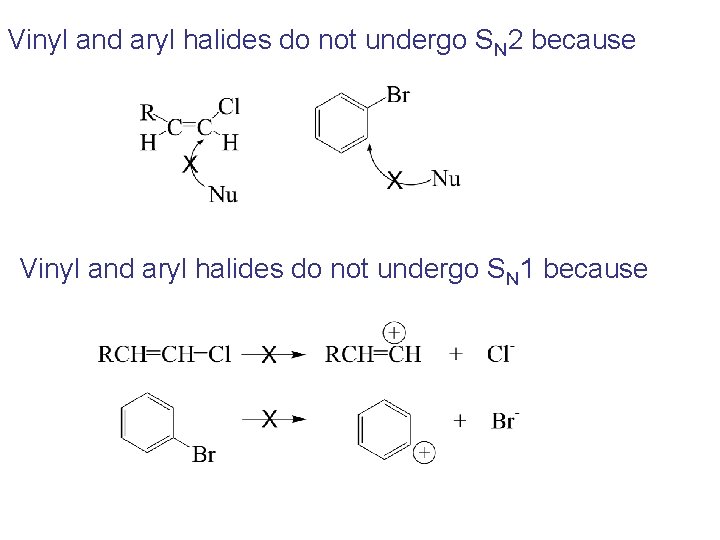
Vinyl and aryl halides do not undergo SN 2 because Vinyl and aryl halides do not undergo SN 1 because



When an alkyl halide can undergo either SN 1 or SN 2, the concentration of the nucleophile, the reactivity of the nucleophile, and the solvent of the reaction will determine which reaction will predominate An SN 2 reaction is favored by a high concentration of a good nucleophile An SN 1 reaction is favored by a low concentration of a nucleophile or by a poor nucleophile

The Role of Solvent in SN 2 and in SN 1 Reactions one or more reactants charged in the rate-limiting step none of the reactants is charged in the rate-limiting step increase the polarity of the solvent decrease the rate of the reaction increase the rate of the reaction
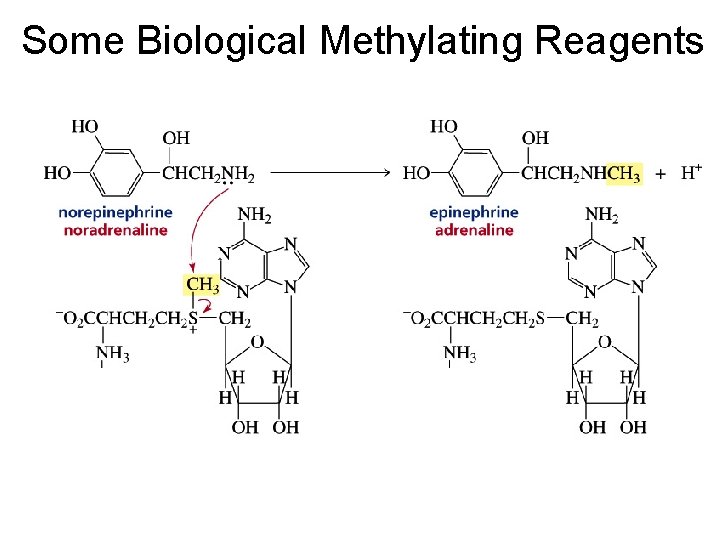
Some Biological Methylating Reagents
- Slides: 49