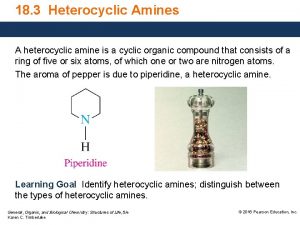
Heterocyclic Chemistry Benzopyridines Quinoline and Isoquinoline Heterocyclic Chemistry
Heterocyclic Chemistry Benzopyridines Quinoline and Isoquinoline: Heterocyclic Chemistry 1
![Introduction : Quinoline Benzo[b]pyridine Isoquinoline Benzo[c]pyridine Ø Quninoline and isoquinoline are two fused heterocycles Introduction : Quinoline Benzo[b]pyridine Isoquinoline Benzo[c]pyridine Ø Quninoline and isoquinoline are two fused heterocycles](http://slidetodoc.com/presentation_image_h/081dca34a06fb40882cc9fe05abc059f/image-2.jpg)
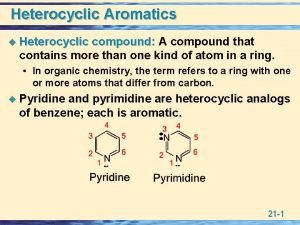
Introduction : Quinoline Benzo[b]pyridine Isoquinoline Benzo[c]pyridine Ø Quninoline and isoquinoline are two fused heterocycles derived by fusion of pyridine ring with a benzene ring. Ø Quinoline is high boiling liquid (b. p. 237°C) and smells like pyridine while isoquinoline is a low melting solid (m. p. 26. 5°C, b. p. 243°C). Heterocyclic Chemistry 2
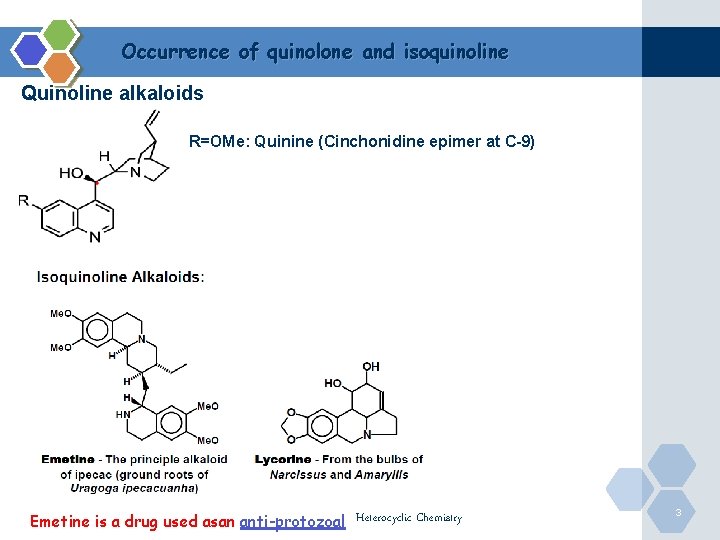
Occurrence of quinolone and isoquinoline Quinoline alkaloids R=OMe: Quinine (Cinchonidine epimer at C-9) Emetine is a drug used asan anti-protozoal Heterocyclic Chemistry 3

Basicity Ø Quinoline and isoquinoline are weak bases but slightly more basic than pyridine but less basic than anilines since the nitrogen in quinoline and isoquinoline is more electronegative being sp 2 hybridized compared to sp 3 hybridized nitrogen of anilines. Heterocyclic Chemistry 4
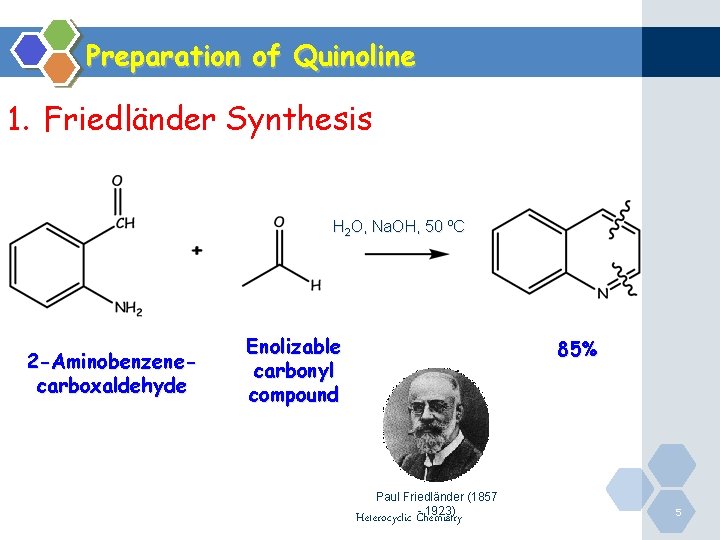
Preparation of Quinoline 1. Friedländer Synthesis H 2 O, Na. OH, 50 ºC 2 -Aminobenzenecarboxaldehyde Enolizable carbonyl compound 85% Paul Friedländer (1857 - 1923) Heterocyclic Chemistry 5
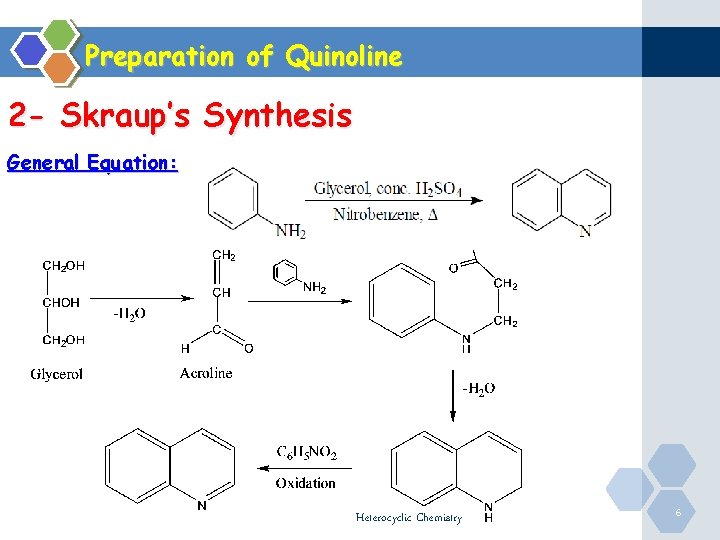
Preparation of Quinoline 2 - Skraup’s Synthesis General Equation: Heterocyclic Chemistry 6
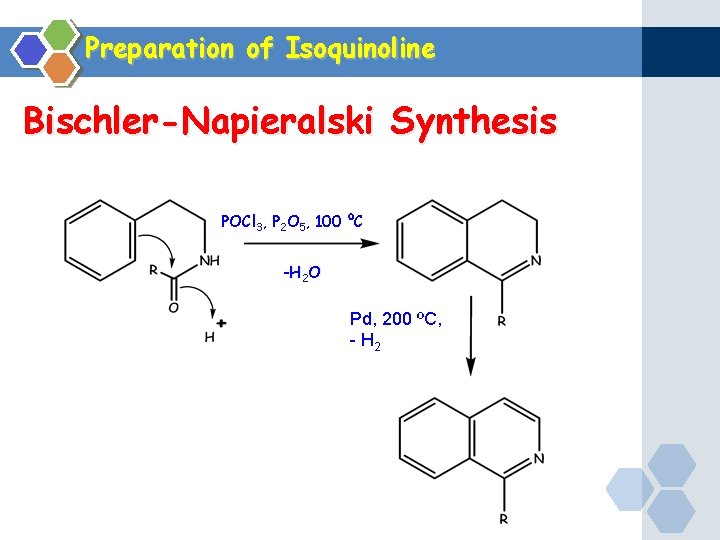
Preparation of Isoquinoline Bischler-Napieralski Synthesis POCl 3, P 2 O 5, 100 ºC -H 2 O Pd, 200 ºC, - H 2
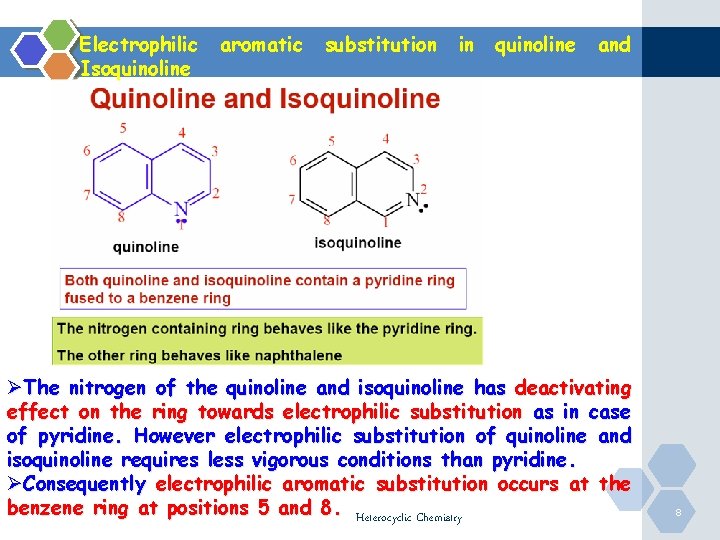
Electrophilic Isoquinoline aromatic substitution in quinoline and ØThe nitrogen of the quinoline and isoquinoline has deactivating effect on the ring towards electrophilic substitution as in case of pyridine. However electrophilic substitution of quinoline and isoquinoline requires less vigorous conditions than pyridine. ØConsequently electrophilic aromatic substitution occurs at the benzene ring at positions 5 and 8. Heterocyclic Chemistry 8
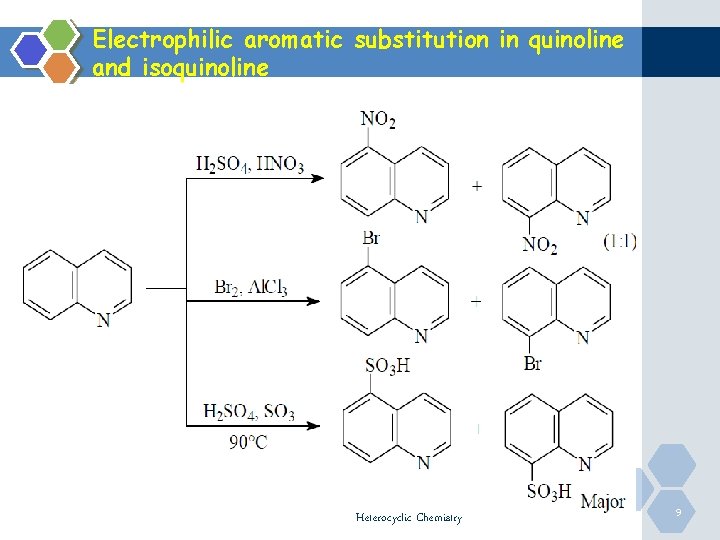
Electrophilic aromatic substitution in quinoline and isoquinoline Heterocyclic Chemistry 9
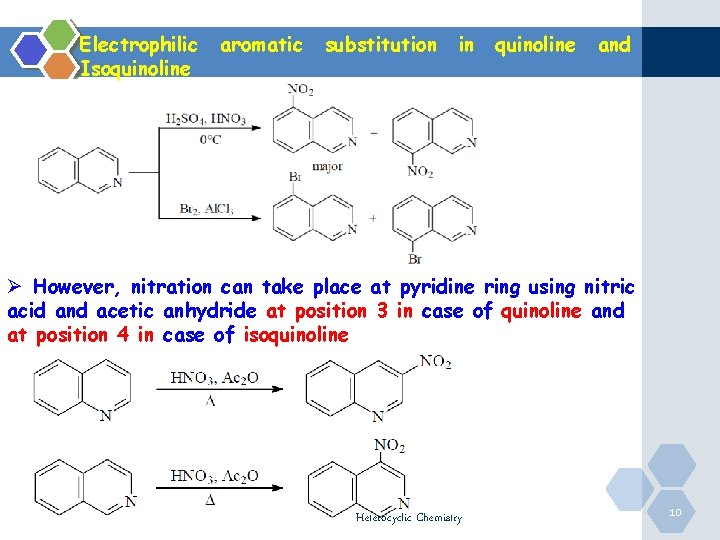
Electrophilic Isoquinoline aromatic substitution in quinoline and Ø However, nitration can take place at pyridine ring using nitric acid and acetic anhydride at position 3 in case of quinoline and at position 4 in case of isoquinoline Heterocyclic Chemistry 10
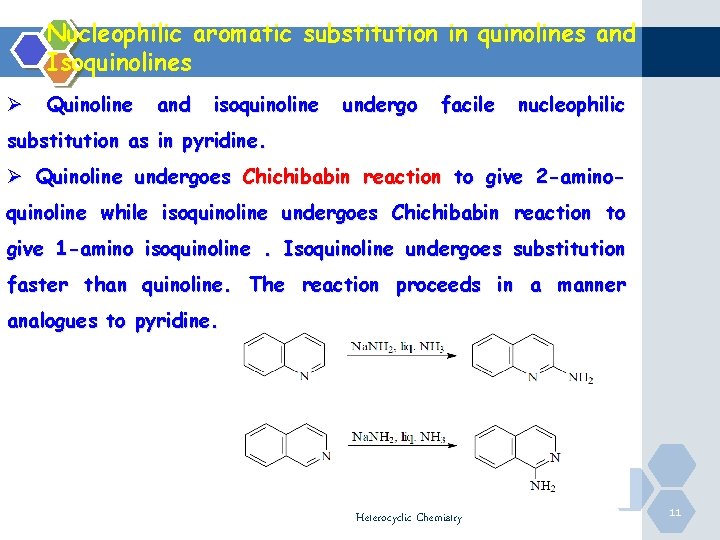
Nucleophilic aromatic substitution in quinolines and Isoquinolines Ø Quinoline and isoquinoline undergo facile nucleophilic substitution as in pyridine. Ø Quinoline undergoes Chichibabin reaction to give 2 -aminoquinoline while isoquinoline undergoes Chichibabin reaction to give 1 -amino isoquinoline. Isoquinoline undergoes substitution faster than quinoline. The reaction proceeds in a manner analogues to pyridine. Heterocyclic Chemistry 11
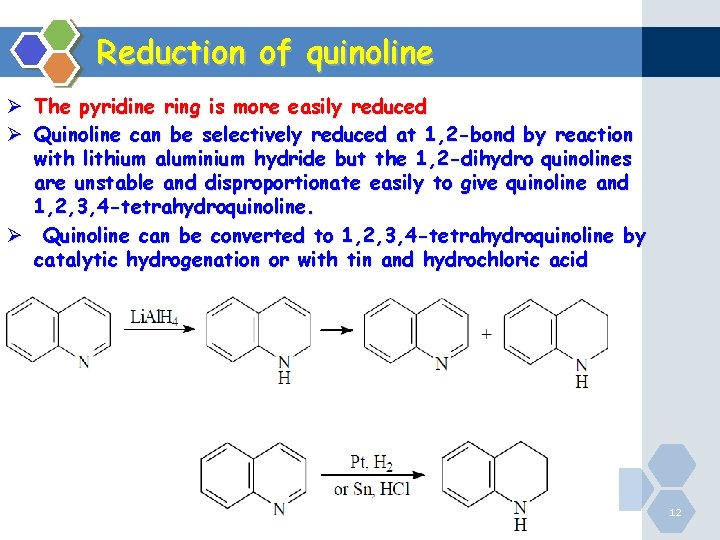
Reduction of quinoline Ø The pyridine ring is more easily reduced Ø Quinoline can be selectively reduced at 1, 2 -bond by reaction with lithium aluminium hydride but the 1, 2 -dihydro quinolines are unstable and disproportionate easily to give quinoline and 1, 2, 3, 4 -tetrahydroquinoline. Ø Quinoline can be converted to 1, 2, 3, 4 -tetrahydroquinoline by catalytic hydrogenation or with tin and hydrochloric acid Heterocyclic Chemistry 12
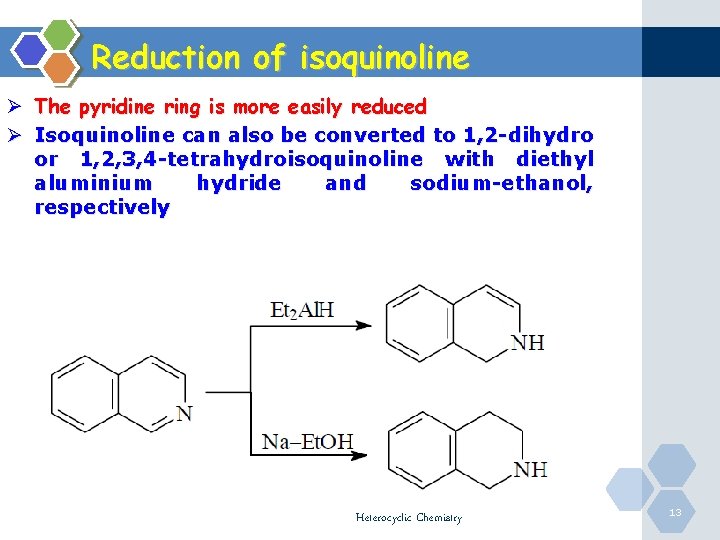
Reduction of isoquinoline Ø The pyridine ring is more easily reduced Ø Isoquinoline can also be converted to 1, 2 -dihydro or 1, 2, 3, 4 -tetrahydroisoquinoline with diethyl aluminium hydride and sodium-ethanol, respectively Heterocyclic Chemistry 13
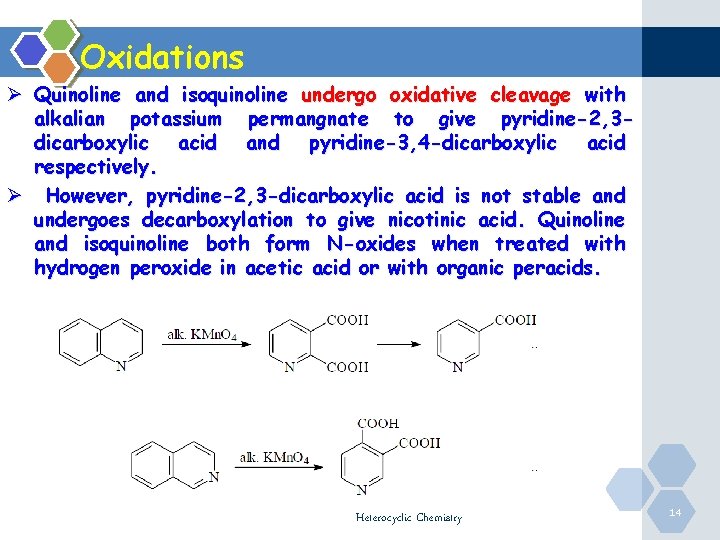
Oxidations Ø Quinoline and isoquinoline undergo oxidative cleavage with alkalian potassium permangnate to give pyridine-2, 3 dicarboxylic acid and pyridine-3, 4 -dicarboxylic acid respectively. Ø However, pyridine-2, 3 -dicarboxylic acid is not stable and undergoes decarboxylation to give nicotinic acid. Quinoline and isoquinoline both form N-oxides when treated with hydrogen peroxide in acetic acid or with organic peracids. Heterocyclic Chemistry 14
- Slides: 14