Biochemistry of Atherosclerosis Jana Novotn Atherosclerosis Atherosclerosis is

Biochemistry of Atherosclerosis Jana Novotná

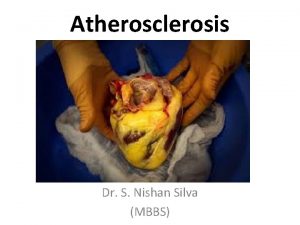
Atherosclerosis • Atherosclerosis is a disease of large and medium-sized muscular arteries and is characterized by: – endothelial dysfunction – vascular inflammation – the buildup of lipids, cholesterol, calcium, and cellular debris within the intima of the vessel wall • It leads to the narrowing of arteries or complete blockage. • Its main components are endothelial disfunction, lipid deposition, inflammatory reaction in the vascular wall. • Remodeling of vessel wall.

Atherosclerosis • Intense cross-talk between EC, VSMC, plasma-derived inflammatory cells, lymphocytes (involves array of chemokines, cytokines, growth factors). • Attraction of cells to the sites of atherosclerotic lesion. • Migration, proliferation, apoptosis, excess production of extracellular matrix.
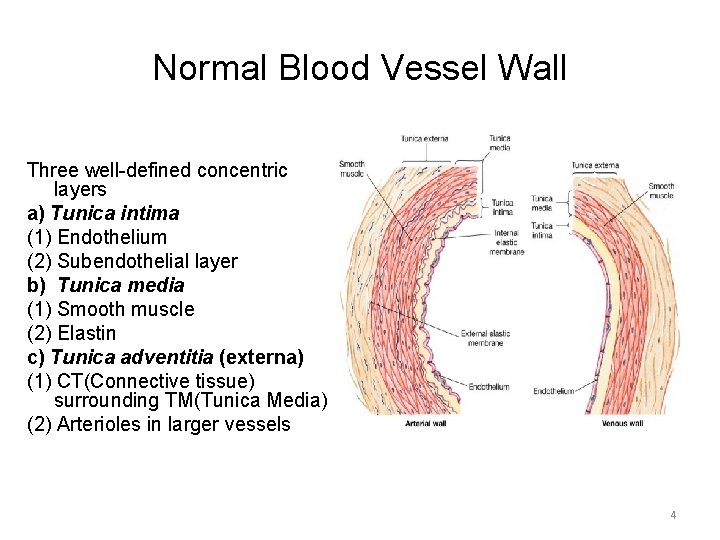
Normal Blood Vessel Wall Three well-defined concentric layers a) Tunica intima (1) Endothelium (2) Subendothelial layer b) Tunica media (1) Smooth muscle (2) Elastin c) Tunica adventitia (externa) (1) CT(Connective tissue) surrounding TM(Tunica Media) (2) Arterioles in larger vessels 4

Normal Blood Vessel Wall • The lumen of healthy arterial wall is lined by confluent layer of endothelial cells. • Arterial endothelium repels cells and inhibits blood clotting. • Endothelium controls important function: 1. the ability of blood vessels to dilatate (vasodilatation) 2. the ability of blood vessels to constrict (vasoconstriction) • Endothelium regulates tissue and organ blood flow, releases variety substances to control vasomotor tone: – – prostacyclines hyperpolarizing factor endothelin NO
Normal Blood Vessel Wall • In the case of intact endothelium, the stimulus for vasodilatation are: – mechanical stimulation by blood flow – catecholamines, bradykinin, platelets-released serotonin stimulate specific receptors • In the case of endothelium disfunction: – direct vasoconstrictor action of the stimuli on the VSMC outweighs the endothelium-dependent vasodilatator effect – this action leads to paradoxial vasoconstriction (Hypercholesterolemia and other cardiovascular risk factors are associated with endothelial disfunction).
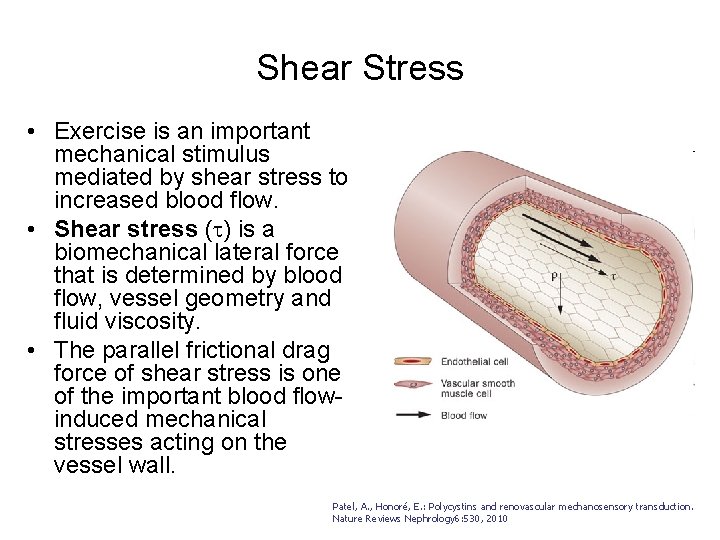
Shear Stress • Exercise is an important mechanical stimulus mediated by shear stress to increased blood flow. • Shear stress (t) is a biomechanical lateral force that is determined by blood flow, vessel geometry and fluid viscosity. • The parallel frictional drag force of shear stress is one of the important blood flowinduced mechanical stresses acting on the vessel wall. Patel, A. , Honoré, E. : Polycystins and renovascular mechanosensory transduction. Nature Reviews Nephrology 6: 530, 2010

Shear Stress • Shear stress activates the endothelium and induces nitric oxide release, which causes vasorelaxation. • Maintenance of a physiologic, laminar shear stress is crucial for normal vascular functioning, • Shear stress regulates vascular caliber, inhibits proliferation, thrombosis and inflammation of the vessel wall. • Thus, shear stress is atheroprotective.

Nitric Oxide (NO) • Factor which controls vasodilatation: endothelium-derived relaxing factor (EDRF) • The activity of e. NOS is controlled by intracellular Ca 2+. • e. NOS – endothelial constitutive enzyme (continually expressed) and i. NOS, inducible, from VSMC. • NO cause vasodilatation via stimulation guanylate cyclase and c. GMP production (second messenger for many important cellular functions, particularly for signalling smooth muscle relaxation).

The Development of Atherosclerosis • The key event – damage to the endothelium caused by excess of lipoproteins, hypertension, diabetes, components of cigarette smoke. • Endothelium becomes more permeable to lipoproteins. • Lipoproteins move below the endothelial layer (to intima). • Endothelium loses its cell-repelent quality. • Inflammatory cells move itno the vascular wall.
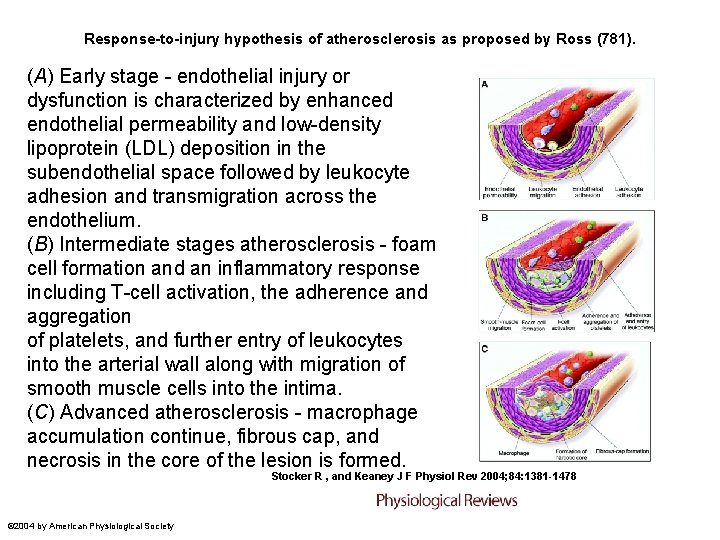
Response-to-injury hypothesis of atherosclerosis as proposed by Ross (781). (A) Early stage - endothelial injury or dysfunction is characterized by enhanced endothelial permeability and low-density lipoprotein (LDL) deposition in the subendothelial space followed by leukocyte adhesion and transmigration across the endothelium. (B) Intermediate stages atherosclerosis - foam cell formation and an inflammatory response including T-cell activation, the adherence and aggregation of platelets, and further entry of leukocytes into the arterial wall along with migration of smooth muscle cells into the intima. (C) Advanced atherosclerosis - macrophage accumulation continue, fibrous cap, and necrosis in the core of the lesion is formed. Stocker R , and Keaney J F Physiol Rev 2004; 84: 1381 -1478 © 2004 by American Physiological Society

The Role of Oxidative Stress in Atherosclerosis • The oxidative modification of low density lipoproteins (LDL) in the arterial wall by reactive oxygen species (ROS) (O 2. -, OH-, ONOO-) • Increased production of free ROS in hypercholesterolemia, diabetes mellitus (DM), arterial hypertension, smoking, age • LDL retained in the intima (in part by binding to proteoglycan) undergoes oxidative modification. • Lipid hydroperoxides, lysophospholipides, carbonyl compounds localize in the lipid fraction. • Oxygen free-radicals inactivate NO rapidly. • NO + superoxide (O 2. -) peroxynitrite (ONOO-). • NO has no longer vasoprotective function.
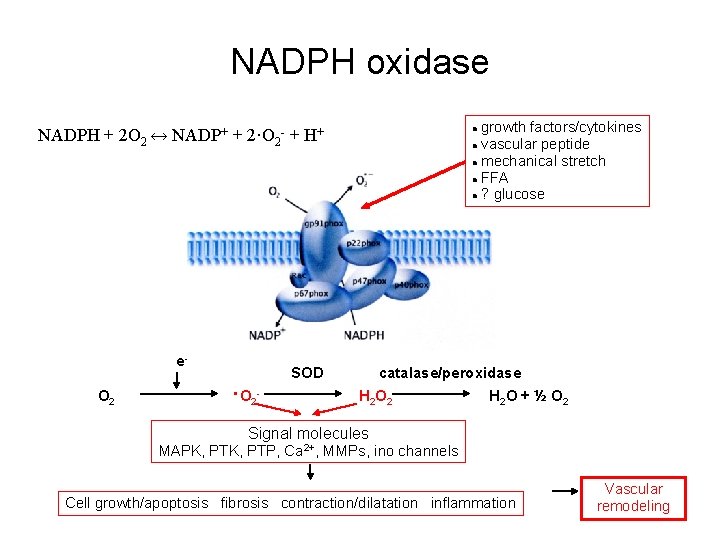
NADPH oxidase growth factors/cytokines vascular peptide mechanical stretch FFA ? glucose NADPH + 2 O 2 ↔ NADP+ + 2·O 2 - + H+ e. O 2 SOD ·O 2 - catalase/peroxidase H 2 O 2 H 2 O + ½ O 2 Signal molecules MAPK, PTP, Ca 2+, MMPs, ino channels Cell growth/apoptosis fibrosis contraction/dilatation inflammation Vascular remodeling
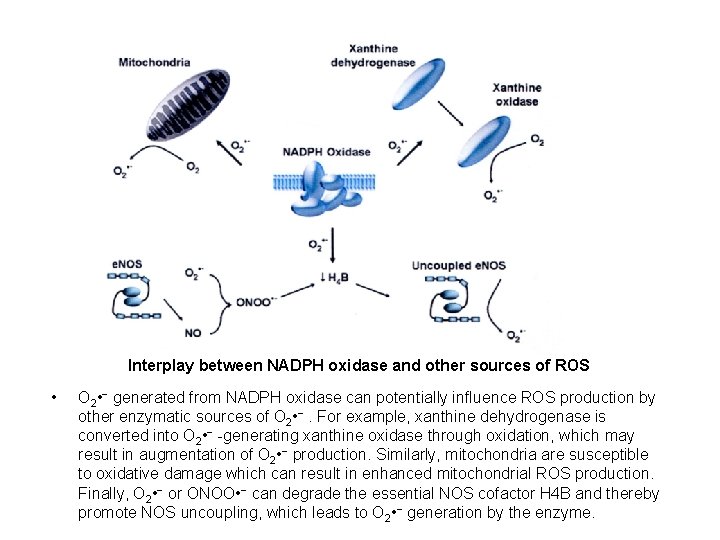
Interplay between NADPH oxidase and other sources of ROS • O 2 • − generated from NADPH oxidase can potentially influence ROS production by other enzymatic sources of O 2 • −. For example, xanthine dehydrogenase is converted into O 2 • − -generating xanthine oxidase through oxidation, which may result in augmentation of O 2 • − production. Similarly, mitochondria are susceptible to oxidative damage which can result in enhanced mitochondrial ROS production. Finally, O 2 • − or ONOO • − can degrade the essential NOS cofactor H 4 B and thereby promote NOS uncoupling, which leads to O 2 • − generation by the enzyme.
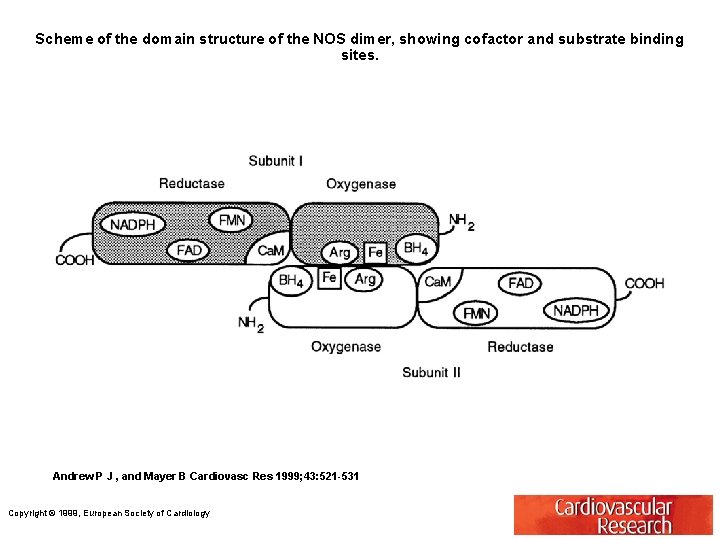
Scheme of the domain structure of the NOS dimer, showing cofactor and substrate binding sites. Andrew P J , and Mayer B Cardiovasc Res 1999; 43: 521 -531 Copyright © 1999, European Society of Cardiology
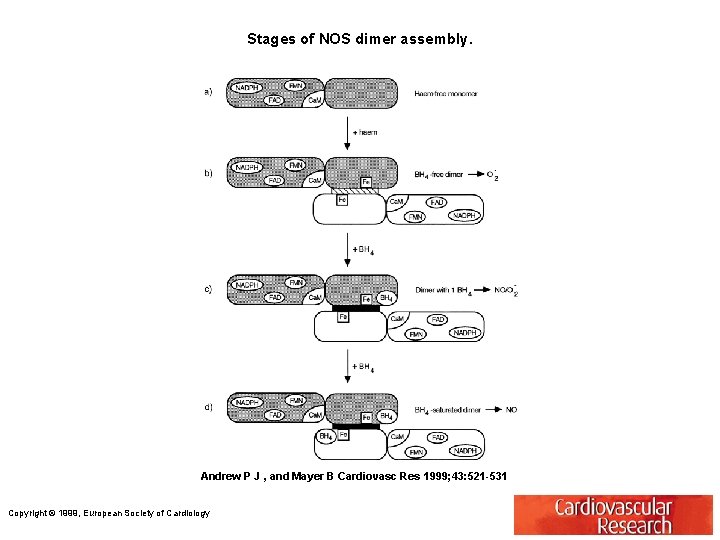
Stages of NOS dimer assembly. Andrew P J , and Mayer B Cardiovasc Res 1999; 43: 521 -531 Copyright © 1999, European Society of Cardiology
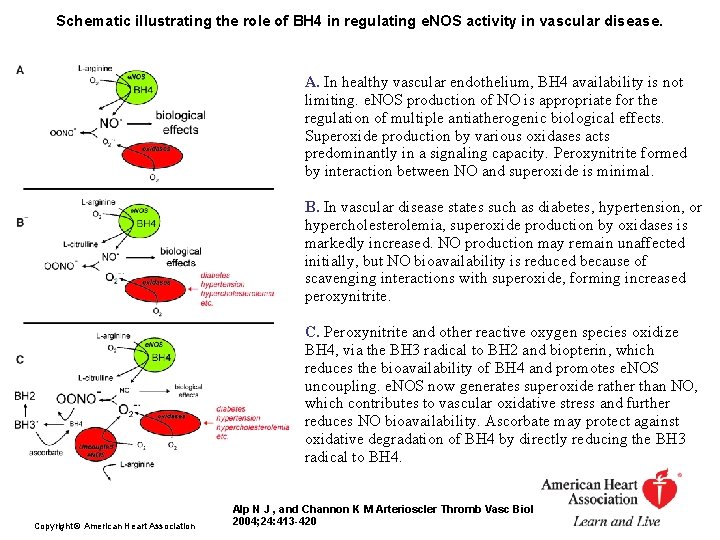
Schematic illustrating the role of BH 4 in regulating e. NOS activity in vascular disease. A. In healthy vascular endothelium, BH 4 availability is not limiting. e. NOS production of NO is appropriate for the regulation of multiple antiatherogenic biological effects. Superoxide production by various oxidases acts predominantly in a signaling capacity. Peroxynitrite formed by interaction between NO and superoxide is minimal. B. In vascular disease states such as diabetes, hypertension, or hypercholesterolemia, superoxide production by oxidases is markedly increased. NO production may remain unaffected initially, but NO bioavailability is reduced because of scavenging interactions with superoxide, forming increased peroxynitrite. C. Peroxynitrite and other reactive oxygen species oxidize BH 4, via the BH 3 radical to BH 2 and biopterin, which reduces the bioavailability of BH 4 and promotes e. NOS uncoupling. e. NOS now generates superoxide rather than NO, which contributes to vascular oxidative stress and further reduces NO bioavailability. Ascorbate may protect against oxidative degradation of BH 4 by directly reducing the BH 3 radical to BH 4. Copyright © American Heart Association Alp N J , and Channon K M Arterioscler Thromb Vasc Biol 2004; 24: 413 -420
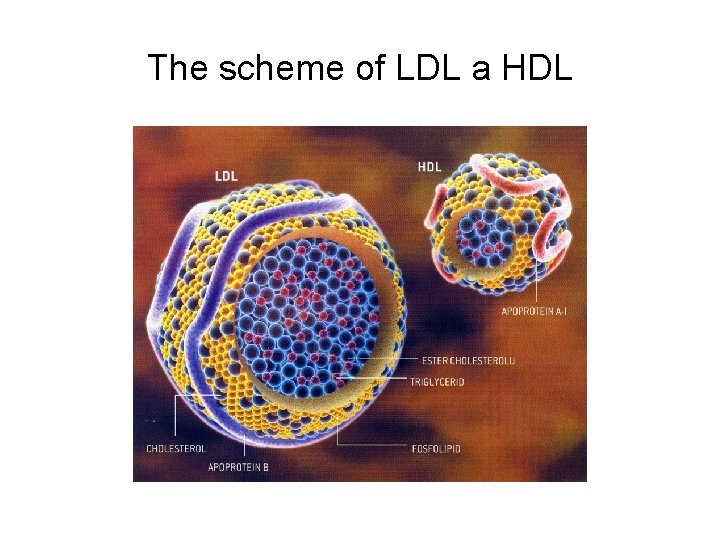
The scheme of LDL a HDL
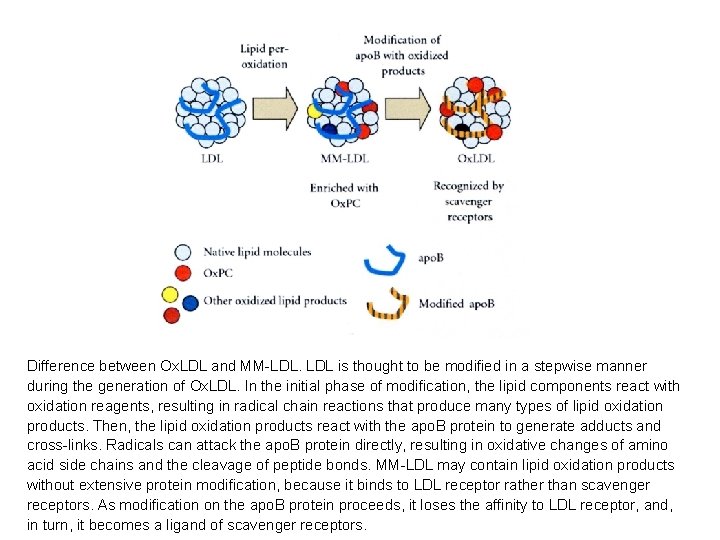
Difference between Ox. LDL and MM-LDL. LDL is thought to be modified in a stepwise manner during the generation of Ox. LDL. In the initial phase of modification, the lipid components react with oxidation reagents, resulting in radical chain reactions that produce many types of lipid oxidation products. Then, the lipid oxidation products react with the apo. B protein to generate adducts and cross-links. Radicals can attack the apo. B protein directly, resulting in oxidative changes of amino acid side chains and the cleavage of peptide bonds. MM-LDL may contain lipid oxidation products without extensive protein modification, because it binds to LDL receptor rather than scavenger receptors. As modification on the apo. B protein proceeds, it loses the affinity to LDL receptor, and, in turn, it becomes a ligand of scavenger receptors.

Lipoprotein(a) or Lp(a) • Lp(a) - LDL like particle with a molecule of Apolipoprotein B-100 linked by a disulphide bridge to Apolipoprotein (a). • The apo(a) component is similar to plasminogen in terms of structure and competes with plasminogen for binding sites resulting in reduced fibrinolysis and the formation of blood clots. • Lp(a) is also thought to speed up the process of atherosclerosis by binding LDL, calcium and other components into an atherosclerotic plaque on the blood vessel wall. • This dual action of Lp(a) explains its role in the promotion of cardiovascular disease.
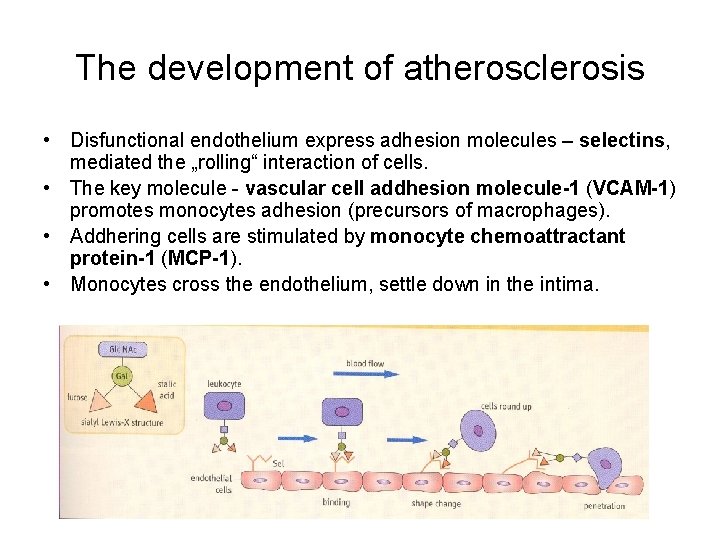
The development of atherosclerosis • Disfunctional endothelium express adhesion molecules – selectins, mediated the „rolling“ interaction of cells. • The key molecule - vascular cell addhesion molecule-1 (VCAM-1) promotes monocytes adhesion (precursors of macrophages). • Addhering cells are stimulated by monocyte chemoattractant protein-1 (MCP-1). • Monocytes cross the endothelium, settle down in the intima.
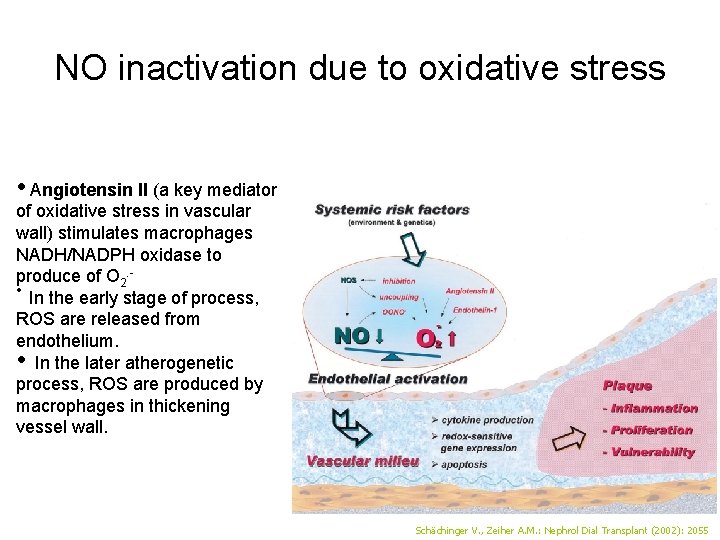
NO inactivation due to oxidative stress • Angiotensin II (a key mediator of oxidative stress in vascular wall) stimulates macrophages NADH/NADPH oxidase to produce of O 2. • In the early stage of process, ROS are released from endothelium. • In the later atherogenetic process, ROS are produced by macrophages in thickening vessel wall. Schächinger V. , Zeiher A. M. : Nephrol Dial Transplant (2002): 2055

Triggers for inflammation in atherosclerosis • Reduced NO bioactivity, increased oxidative stress leads to tyrosine nitration of protein in vessel wall. • Nitrotyrosine and oxidized LDL activate transcriptional factor NF-k. B (nuclear factor kappa-light-chain-enhancer of activated B cells). • NF-k. B increases proliferation of VSMC • Reduced NO bioactivity and enhanced oxidative stress stimulate cytokine production (interleukins, TNFa, MCP 1, interferons) and monocytes attraction.
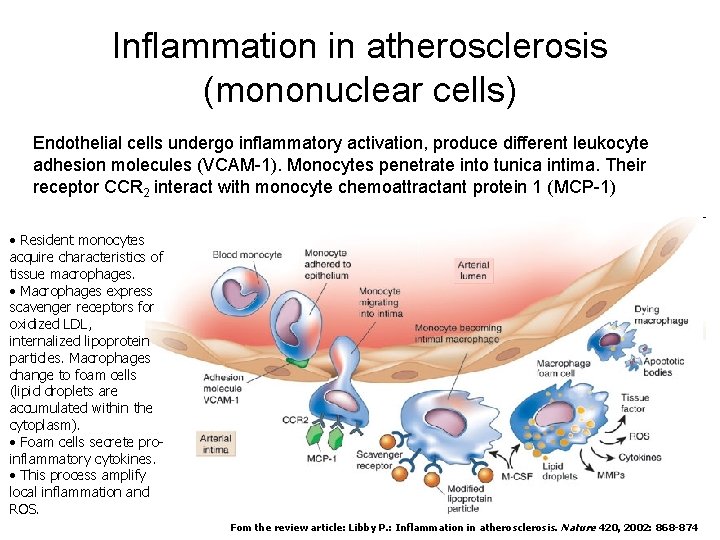
Inflammation in atherosclerosis (mononuclear cells) Endothelial cells undergo inflammatory activation, produce different leukocyte adhesion molecules (VCAM-1). Monocytes penetrate into tunica intima. Their receptor CCR 2 interact with monocyte chemoattractant protein 1 (MCP-1) • Resident monocytes acquire characteristics of tissue macrophages. • Macrophages express scavenger receptors for oxidized LDL, internalized lipoprotein particles. Macrophages change to foam cells (lipid droplets are accumulated within the cytoplasm). • Foam cells secrete proinflammatory cytokines. • This process amplify local inflammation and ROS. Fom the review article: Libby P. : Inflammation in atherosclerosis. Nature 420, 2002: 868 -874
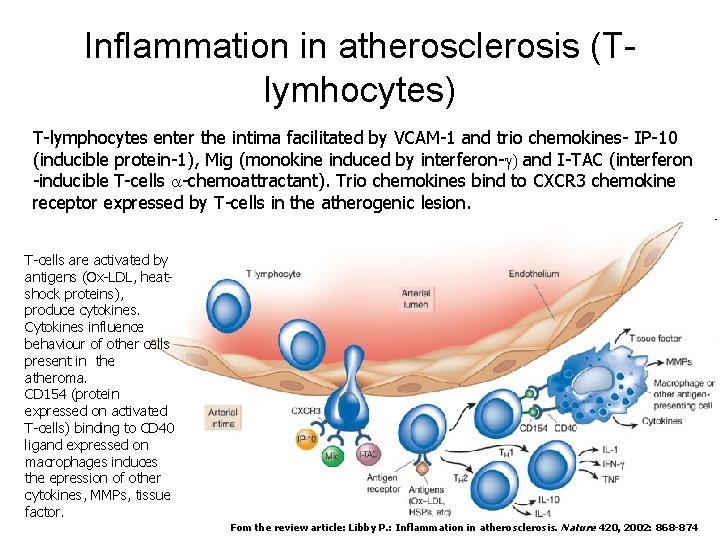
Inflammation in atherosclerosis (Tlymhocytes) T-lymphocytes enter the intima facilitated by VCAM-1 and trio chemokines- IP-10 (inducible protein-1), Mig (monokine induced by interferon-g) and I-TAC (interferon -inducible T-cells a-chemoattractant). Trio chemokines bind to CXCR 3 chemokine receptor expressed by T-cells in the atherogenic lesion. T-cells are activated by antigens (Ox-LDL, heatshock proteins), produce cytokines. Cytokines influence behaviour of other cells present in the atheroma. CD 154 (protein expressed on activated T-cells) binding to CD 40 ligand expressed on macrophages induces the epression of other cytokines, MMPs, tissue factor. Fom the review article: Libby P. : Inflammation in atherosclerosis. Nature 420, 2002: 868 -874
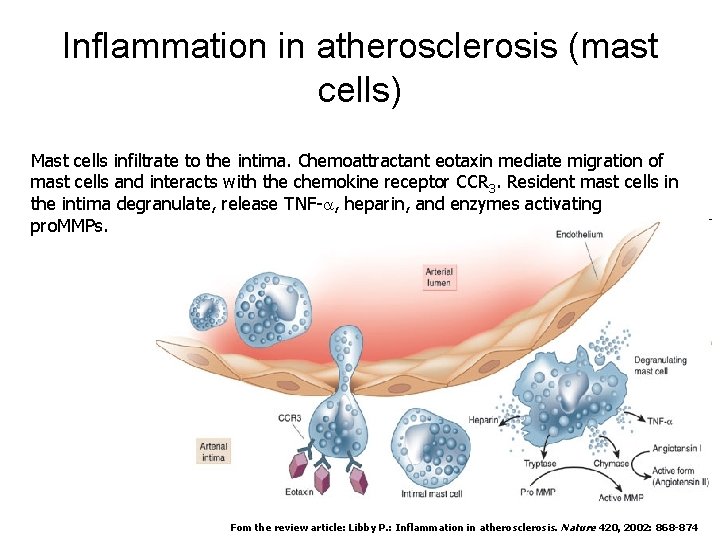
Inflammation in atherosclerosis (mast cells) Mast cells infiltrate to the intima. Chemoattractant eotaxin mediate migration of mast cells and interacts with the chemokine receptor CCR 3. Resident mast cells in the intima degranulate, release TNF-a, heparin, and enzymes activating pro. MMPs. Fom the review article: Libby P. : Inflammation in atherosclerosis. Nature 420, 2002: 868 -874
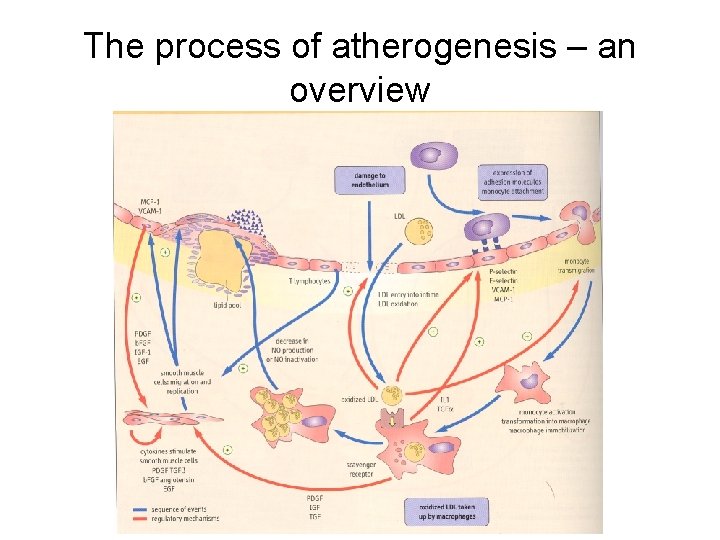
The process of atherogenesis – an overview

The process of atherogenesis • Lipid entry into the arterial wall is a key process in atherogenesis. • Hypercholesterolemia – factor for VCAM-1 and MCP-1 induction. • LDL and VLDL are most atherogenic, enter vascular wall more easily. • LDL – in plasma are protected against oxidation by vit. E, ubiquinon, plasma antioxidants (b-carotene, vit. C). • Out of plasma, LDL phospholipides and fatty acids oxidize.

The process of atherogenesis • Activated macrophages produce enzymes – lipoxygenases, myeloperoxidase, NADPH oxidase. • Oxidized LDL are cytotoxic to endothelial cells, mitogenic for macrophages. • Oxidized LDL apolipoprotein apo. B 100 bind to the scavenger receptor. • Scavenger receptors are not subjected to regulation by intracellular cholesterol level. • Macrophages take up oxidized LDL, overload with lipids.
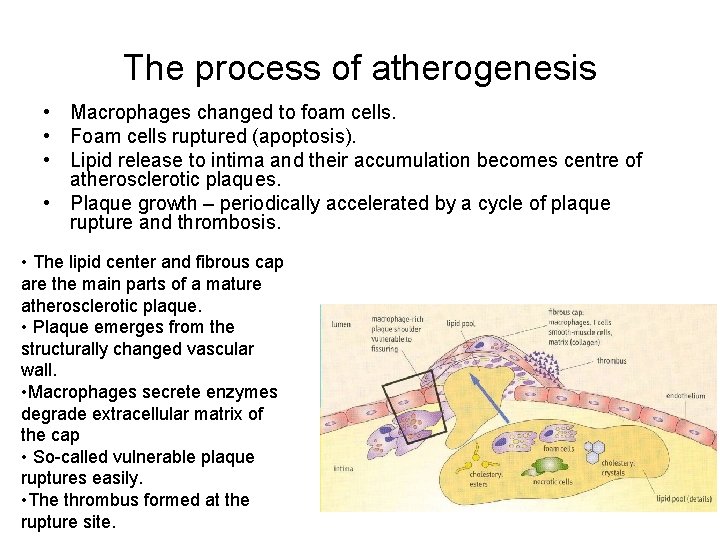
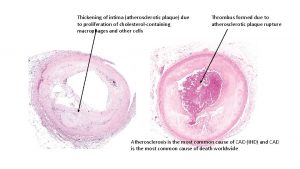
The process of atherogenesis • Macrophages changed to foam cells. • Foam cells ruptured (apoptosis). • Lipid release to intima and their accumulation becomes centre of atherosclerotic plaques. • Plaque growth – periodically accelerated by a cycle of plaque rupture and thrombosis. • The lipid center and fibrous cap are the main parts of a mature atherosclerotic plaque. • Plaque emerges from the structurally changed vascular wall. • Macrophages secrete enzymes degrade extracellular matrix of the cap • So-called vulnerable plaque ruptures easily. • The thrombus formed at the rupture site.

The process of atherogenesis

The process of atherogenesis • IFN-g induces macrophage MMP expression. • IFN-g inhibits VSMC proliferation and collagen synthesis which further weakening the cap. • VSMC undergo apoptosis. • After plaque rupture the area is exposed its interior to the blood. • The interior of the plaque is highly thrombogenic – the smallmolecular-weight glycoprotein (tissue factor) initiates the extrinsic clotting cascade. • Tissue factor complexes with factor VII/VIIa, factor IX and X are activated. • Platelets are activated and thrombus forms quickly on the surface of a ruptured plaque. • Thrombus completely occludes the arterial lumen. It cause tissue necrosis (myocardial infarction or brain stroke).

Cardiovascular risk and its assesment • Plasma concentration of lipoproteins (LDL-cholesterol, HDL-cholesterol, and triacylglycerols) – 5. 2 mmol/L (200 mg/d. L) increases the risk. • Optimal level of LDL-cholesterol - 2. 6 mmol/L (100 mg/d. L)

Cardiovascular risk and its assesment The risk of atherosclerotic event can be decreased by: – – – cholesterol low diet omega-3 fatty acids (abundant in fish oils) exercise smoking cessation control of high pressure drugs statins, fibrates (fibric acid), ezetimibe, antioxidants Statins – inhibit intracellular cholesterol synthase (HMG-Co. A reductase). Fibrates - lower plasma cholesterol by stimulating LPL, decreasing TAG concentration, and increasing HDL. Ezetimibe – inhibitor of intestinal cholesterol transporter. b-carotene, a-tocoferol, vitamin C (such as these contained in fruits or their combinations) have preventive benefit, protect LDL against oxidation.

Summary • Atherosclerosis is an intima-based lesion organized into a fibrous cap and an atheromatous (gruel-like) core and composed of SMCs, ECM, inflammatory cells, lipids, and necrotic debris. • Atherogenesis is driven by an interplay of inflammation and injury to vessel wall cells. • Atherosclerotic plaques accrue slowly over decades but may acutely cause symptoms due to rupture, thrombosis, hemorrhage, or embolization. • Risk factor recognition and reduction can reduce the incidence and severity of atherosclerosis-related disease. 35
- Slides: 35