ORGANIC MOLECULES FUNCTIONAL GROUPS VITALISM Anything that was






































- Slides: 82

ORGANIC MOLECULES & FUNCTIONAL GROUPS

VITALISM • Anything that was alive possessed a vital spark, while things that weren't vital spark alive. . . didn’t. • So what is a vital spark? • Well, you can't see it, taste it, feel it, hear it, or smell it. • You can't capture it in a bottle or any other vessel; you can't transfer it from one object to another. You can't measure it or detect it. The only way to determine if an object had one was to determine if it was alive or not. MECHANISM • Biologists don't believe that there any substances or materials which are exclusive to living things. • What makes something alive is not what it's made of; it's how it's put together and what activities (i. e, chemistry) go on within its structures.

adaptation Response to the environment order reproduction Energy processing regulation Growth and development

Miller Urey Experiment Demonstrated that organic compounds can be created by fairly simple physical processes from inorganic substances. The experiment used conditions then thought to provide an approximate representation of those present on the primordial Earth.

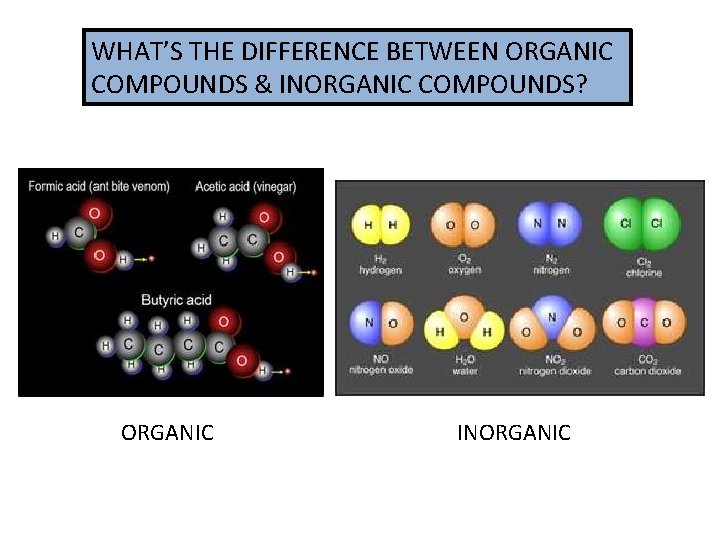
WHAT’S THE DIFFERENCE BETWEEN ORGANIC COMPOUNDS & INORGANIC COMPOUNDS? ORGANIC INORGANIC

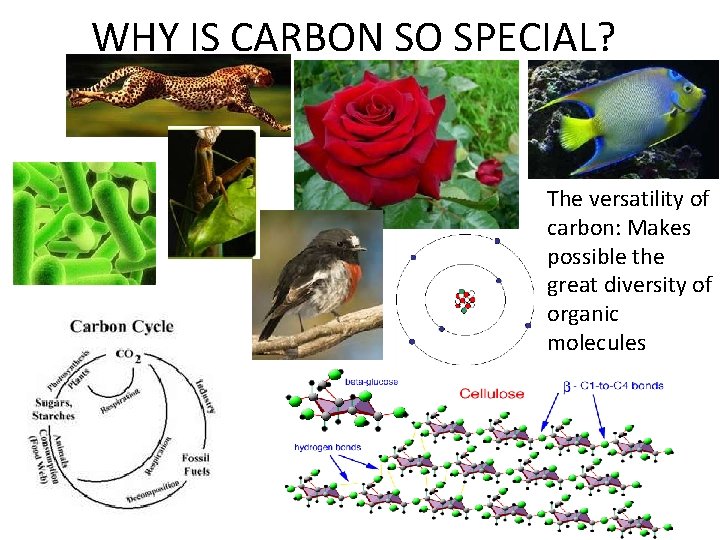
WHY IS CARBON SO SPECIAL? The versatility of carbon: Makes possible the great diversity of organic molecules

Where does the source of carbon for all organic molecules come from?

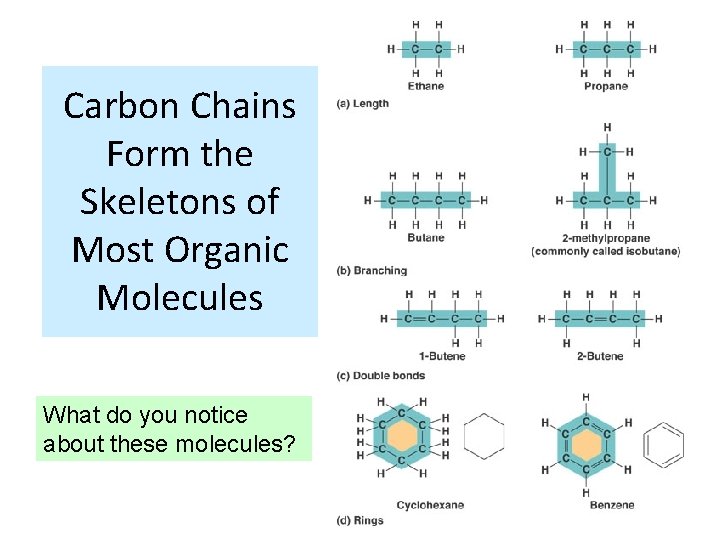
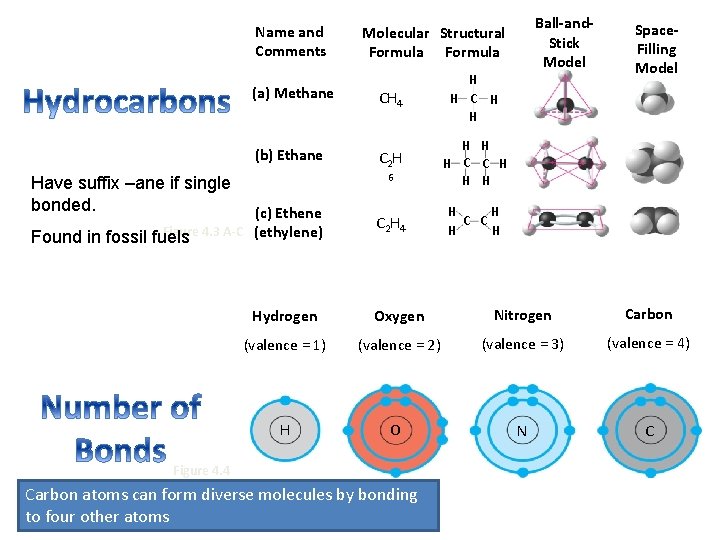
Carbon Chains Form the Skeletons of Most Organic Molecules What do you notice about these molecules?

Name and Comments (a) Methane CH 4 H H C H H (b) Ethane C 2 H 6 H H H C C H H H C 2 H 4 H H C C H H Have suffix –ane if single bonded. (c) Ethene Figure 4. 3 A-C (ethylene) Found in fossil fuels Ball-and. Stick Model Molecular Structural Formula Space. Filling Model Hydrogen Oxygen Nitrogen Carbon (valence = 1) (valence = 2) (valence = 3) (valence = 4) H O N C Figure 4. 4 Carbon atoms can form diverse molecules by bonding to four other atoms

MOLECULAR SHAPE & FUNCTION • Shapes are determined by the positions of the atoms’ orbital. • Molecular shape is very important in living cells. – It determines how the molecules recognize & respond to each other. • If they are complimentary; they will bond


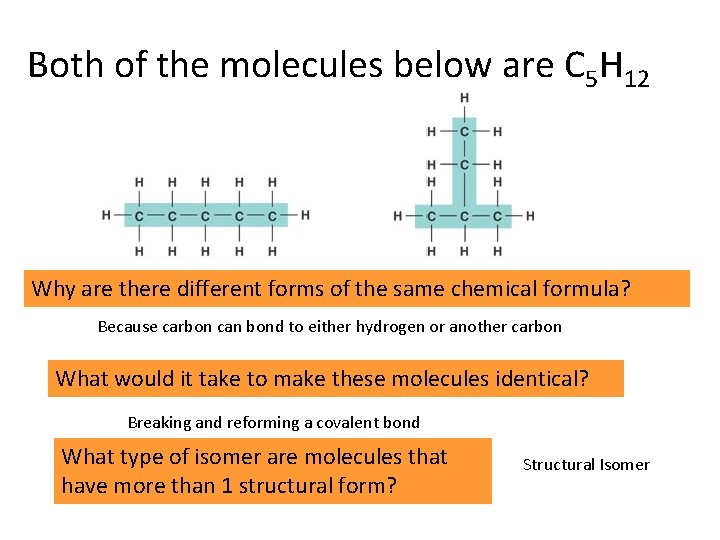
ISOMERS COMPOUNDS THAT HAVE THE SAME NUMBERS OF ATOMS OF THE SAME ELEMENTS BUT DIFFERENT STRUCTURES. (THESE MOLECULES THEREFORE HAVE DIFFERENT PROPERTIES)

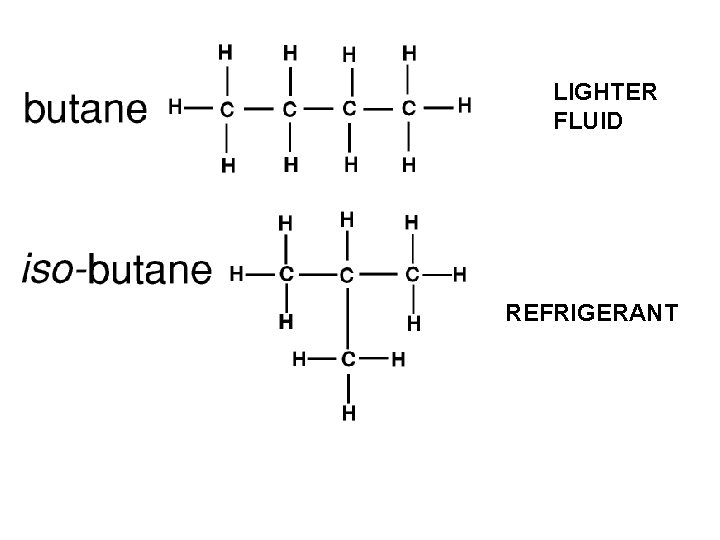
Both of the molecules below are C 5 H 12 Why are there different forms of the same chemical formula? Because carbon can bond to either hydrogen or another carbon What would it take to make these molecules identical? Breaking and reforming a covalent bond What type of isomer are molecules that have more than 1 structural form? Structural Isomer

LIGHTER FLUID REFRIGERANT

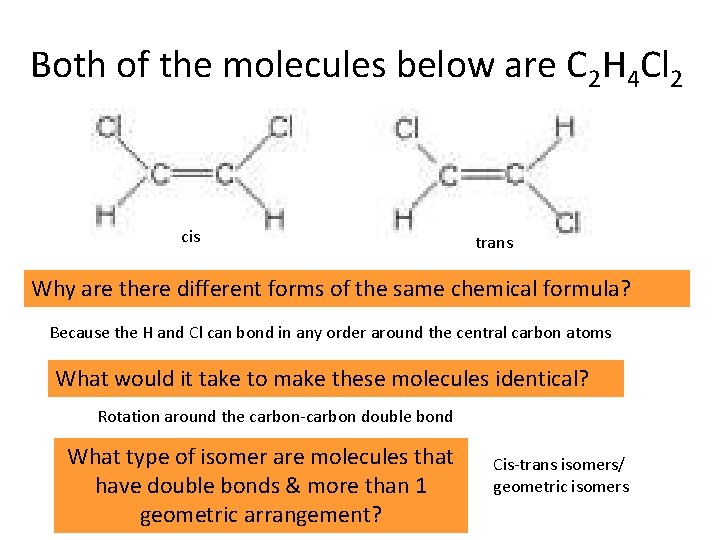
Both of the molecules below are C 2 H 4 Cl 2 cis trans Why are there different forms of the same chemical formula? Because the H and Cl can bond in any order around the central carbon atoms What would it take to make these molecules identical? Rotation around the carbon-carbon double bond What type of isomer are molecules that have double bonds & more than 1 geometric arrangement? Cis-trans isomers/ geometric isomers

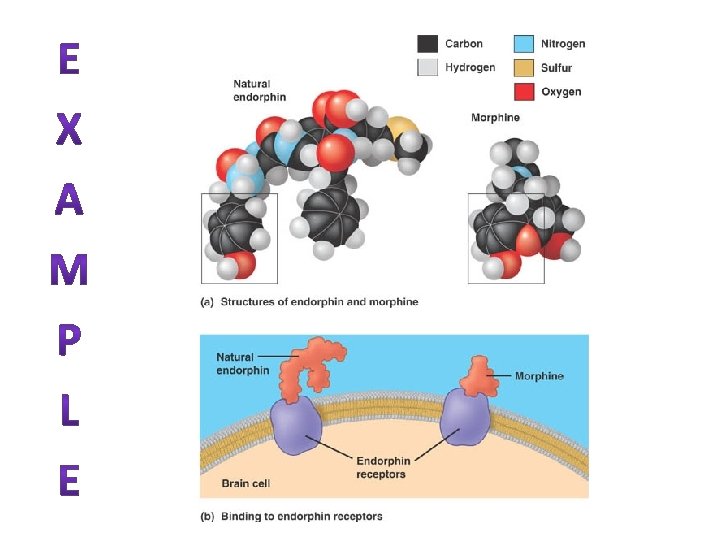
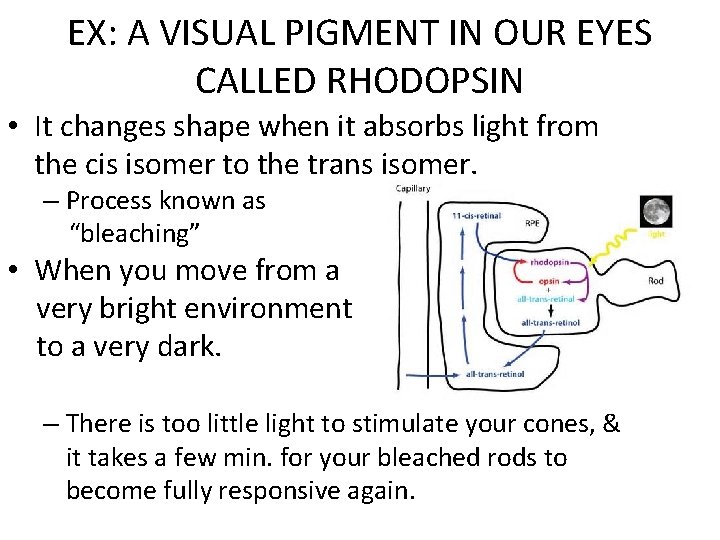
EX: A VISUAL PIGMENT IN OUR EYES CALLED RHODOPSIN • It changes shape when it absorbs light from the cis isomer to the trans isomer. – Process known as “bleaching” • When you move from a very bright environment to a very dark. – There is too little light to stimulate your cones, & it takes a few min. for your bleached rods to become fully responsive again.

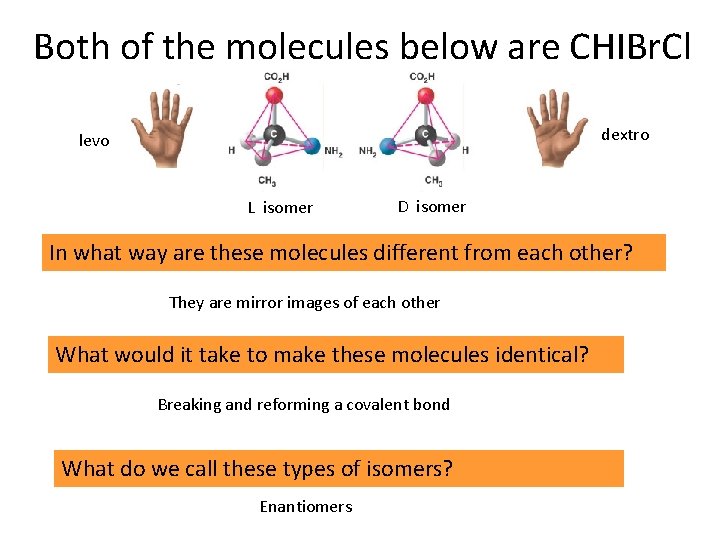
Both of the molecules below are CHIBr. Cl dextro levo L isomer D isomer In what way are these molecules different from each other? They are mirror images of each other What would it take to make these molecules identical? Breaking and reforming a covalent bond What do we call these types of isomers? Enantiomers


Functional groups Are the chemically reactive groups of atoms within an organic molecule THE COMPONENTS OF ORGANIC MOLECULES THAT ARE MOST COMMONLY INVOLVED IN CHEMICAL REACTIONS

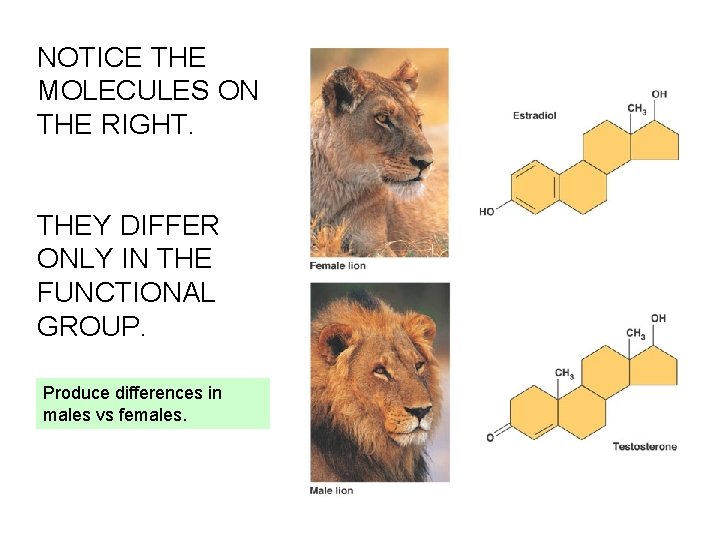
NOTICE THE MOLECULES ON THE RIGHT. THEY DIFFER ONLY IN THE FUNCTIONAL GROUP. Produce differences in males vs females.

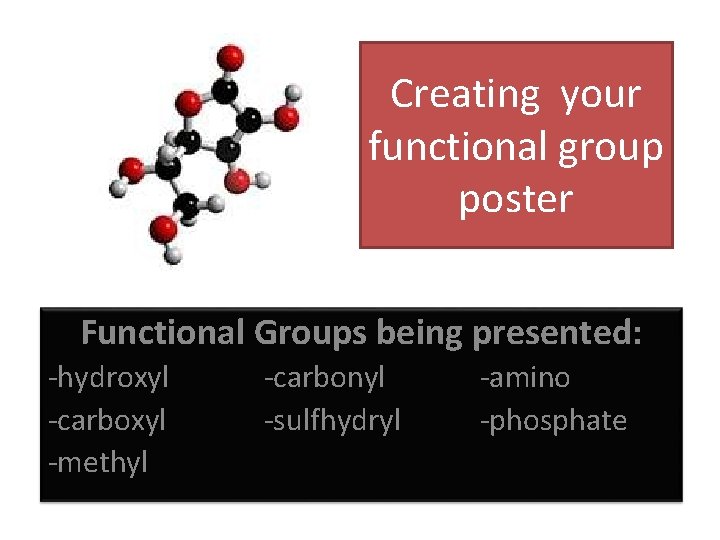
Creating your functional group poster Functional Groups being presented: -hydroxyl -carboxyl -methyl -carbonyl -sulfhydryl -amino -phosphate

Create a large visual aid that conveys the following information: • Draw your functional group • Describe the physical properties your functional group adds to a molecule. • Give the chemical naming suffix • List examples • Record any additional information associated with your functional group. YOU HAVE 15 MINUTES TO COMPLETE YOUR POSTER FOR PRESENTING

A Few Discussion Questions • Can a molecule have more than one functional group? Yes, most do. EX: amino acids have both the amino group and the carboxyl group. • Which group(s) would change the p. H of a solution? In what way? –Carboxylic acids & amines (bases) • Which of the functional groups would hydrogen bond? All of them • Which groups are hydrophillic? All of them…except methyl

Macromolecule Group Project

Carbohydrates (2 groups) • • • Monomer Polymer Chemical structural differences Examples of each & why they are important One group will present simple carbohydrates One group will present complex carbohydrates – Glycosidic linkage

Lipids • • • 3 major types What is different about each of them Chemical structure differences Saturated vs unsaturated Examples of why/how they are important

Nucleic acids • • Monomer Polymer 2 types Chemical structure Differences between them Similarities between them Examples of why/how they are important

Proteins Monomer Polymer Basic chemical structure of a monomer What is different about the different about each type of monomer? • Examples and why/how they are important • •

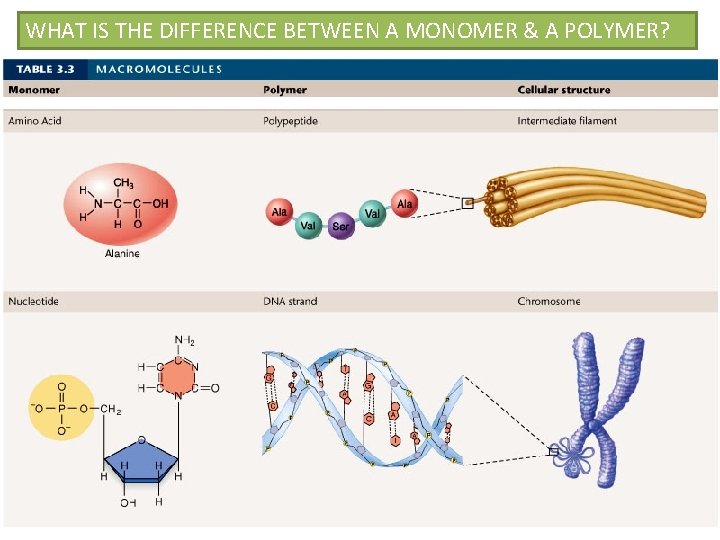
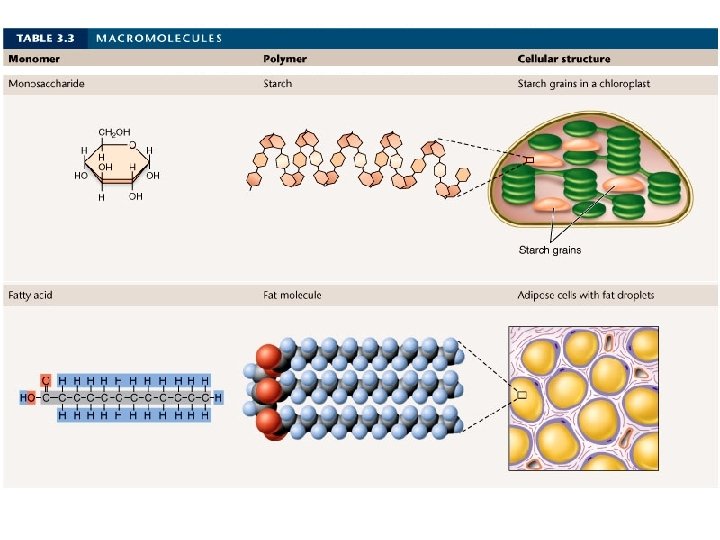
WHAT IS THE DIFFERENCE BETWEEN A MONOMER & A POLYMER?


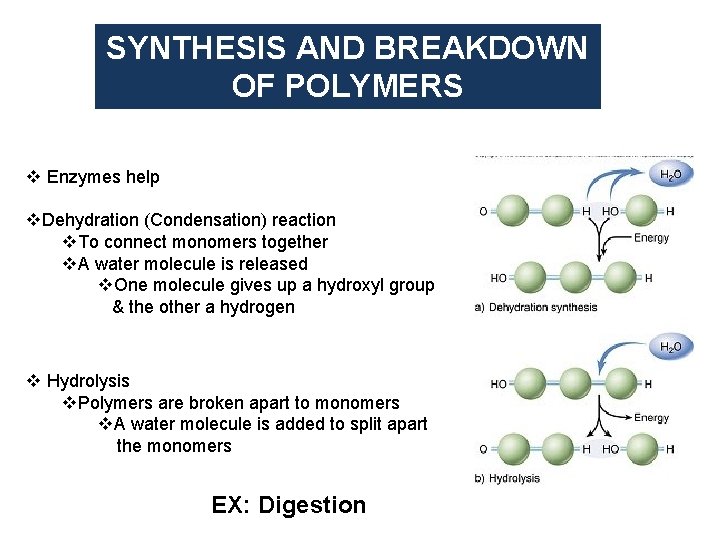
SYNTHESIS AND BREAKDOWN OF POLYMERS v Enzymes help v. Dehydration (Condensation) reaction v. To connect monomers together v. A water molecule is released v. One molecule gives up a hydroxyl group & the other a hydrogen v Hydrolysis v. Polymers are broken apart to monomers v. A water molecule is added to split apart the monomers EX: Digestion

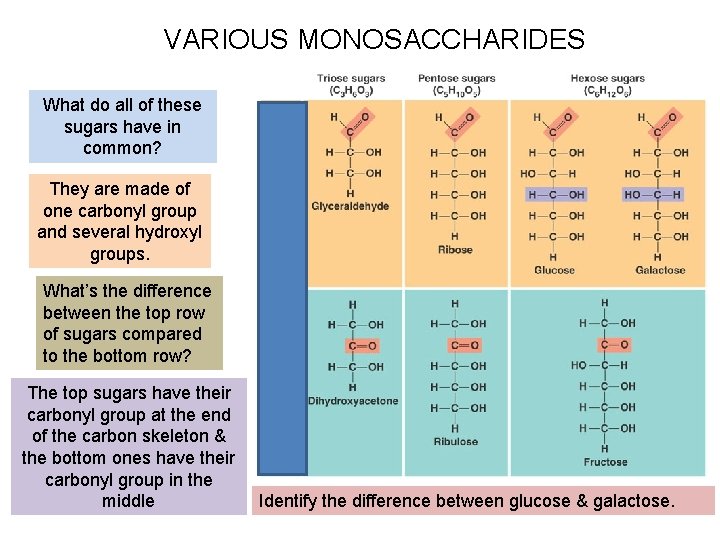
VARIOUS MONOSACCHARIDES What do all of these sugars have in common? They are made of one carbonyl group and several hydroxyl groups. What’s the difference between the top row of sugars compared to the bottom row? The top sugars have their carbonyl group at the end of the carbon skeleton & the bottom ones have their carbonyl group in the middle Identify the difference between glucose & galactose.

What are the monomers used to form? Disaccharides and polysaccharides

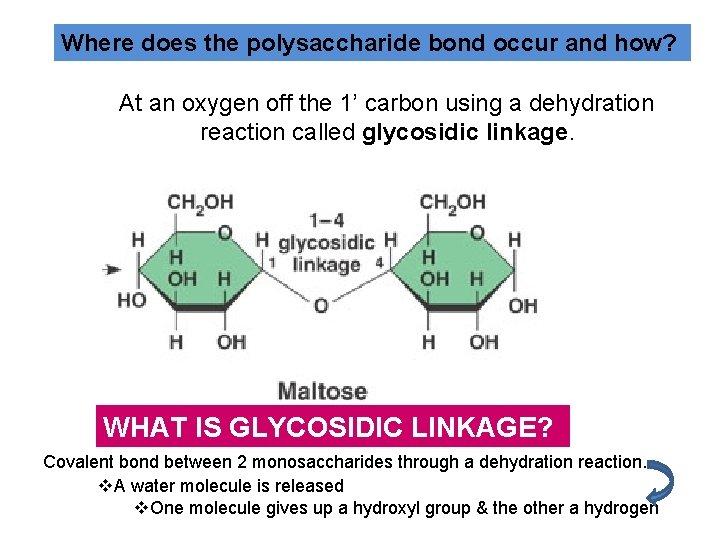
Where does the polysaccharide bond occur and how? At an oxygen off the 1’ carbon using a dehydration reaction called glycosidic linkage. WHAT IS GLYCOSIDIC LINKAGE? Covalent bond between 2 monosaccharides through a dehydration reaction. v. A water molecule is released v. One molecule gives up a hydroxyl group & the other a hydrogen

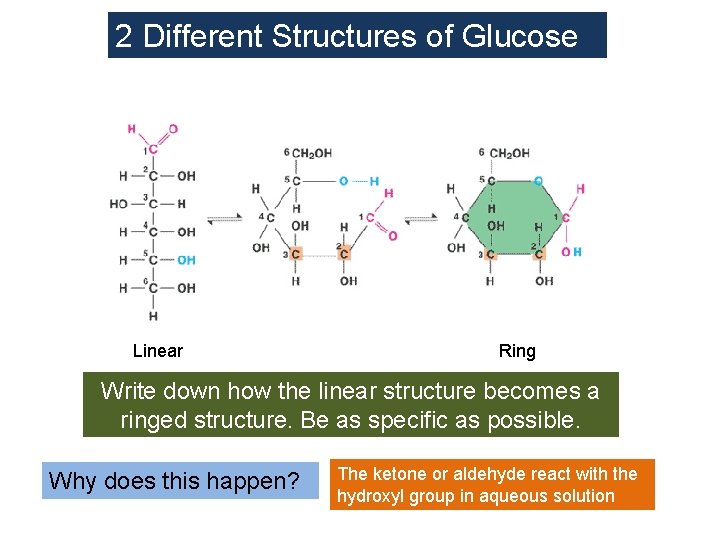
2 Different Structures of Glucose Linear Ring Write down how the linear structure becomes a ringed structure. Be as specific as possible. Why does this happen? The ketone or aldehyde react with the hydroxyl group in aqueous solution

Carbohydrates • Also referred to as sugars • Provide building materials and energy storage • Are molecules that contain carbon, hydrogen and oxygen in a 1: 2: 1 ratio • Are of two main types – Simple carbohydrates – Complex carbohydrates

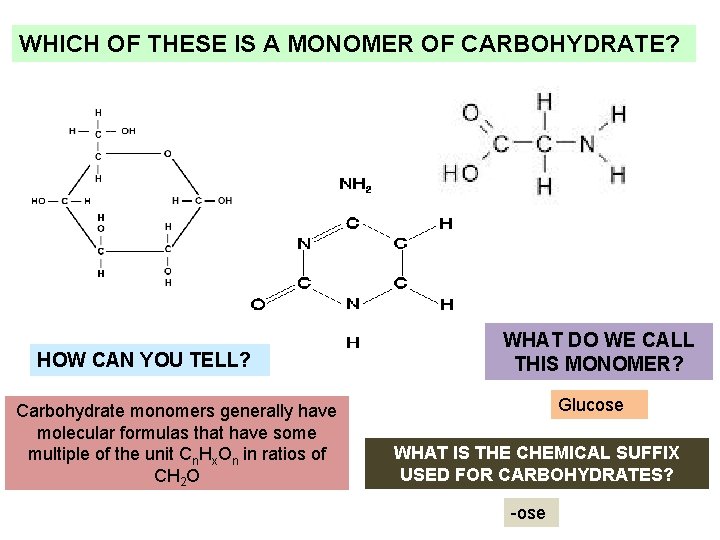
WHICH OF THESE IS A MONOMER OF CARBOHYDRATE? HOW CAN YOU TELL? Carbohydrate monomers generally have molecular formulas that have some multiple of the unit Cn. Hx. On in ratios of CH 2 O WHAT DO WE CALL THIS MONOMER? Glucose WHAT IS THE CHEMICAL SUFFIX USED FOR CARBOHYDRATES? -ose

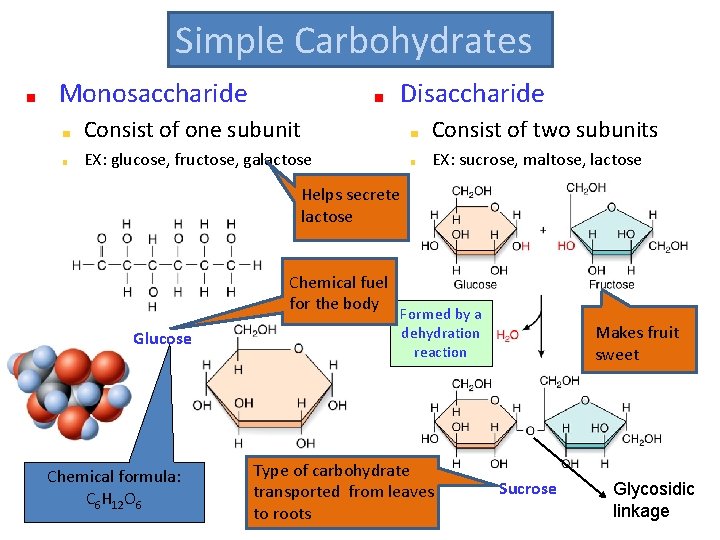
Simple Carbohydrates Monosaccharide Disaccharide Consist of one subunit Consist of two subunits EX: glucose, fructose, galactose EX: sucrose, maltose, lactose Helps secrete lactose Chemical fuel for the body Glucose Chemical formula: C 6 H 12 O 6 Formed by a dehydration reaction Type of carbohydrate transported from leaves to roots Makes fruit sweet Sucrose Glycosidic linkage

Complex Carbohydrates Consist of long polymers of sugar subunits Few hundred to a few thousand Also termed polysaccharides Serve as energy storage and/or building material for structure and/or protection

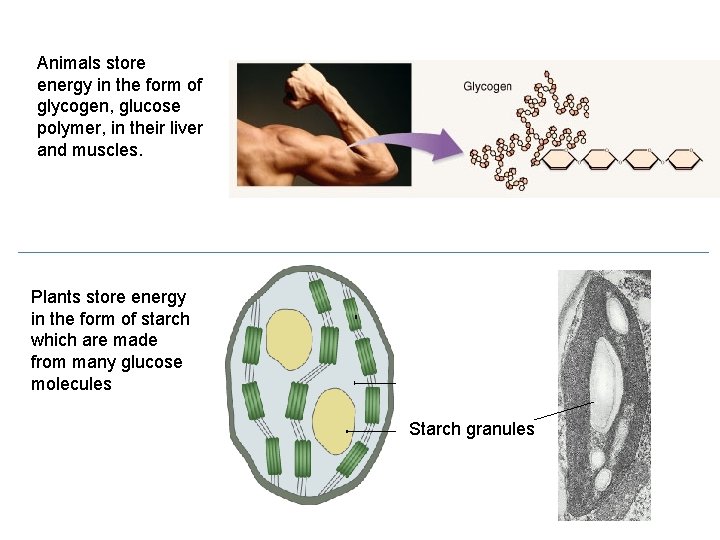
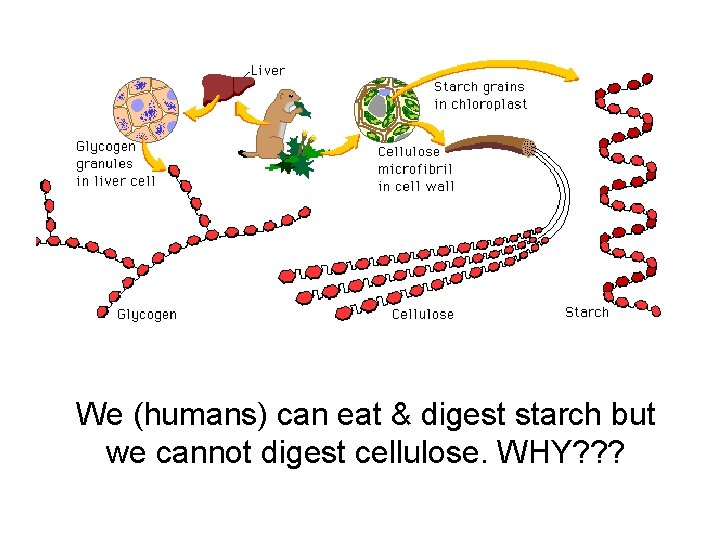
Animals store energy in the form of glycogen, glucose polymer, in their liver and muscles. Plants store energy in the form of starch which are made from many glucose molecules Starch granules

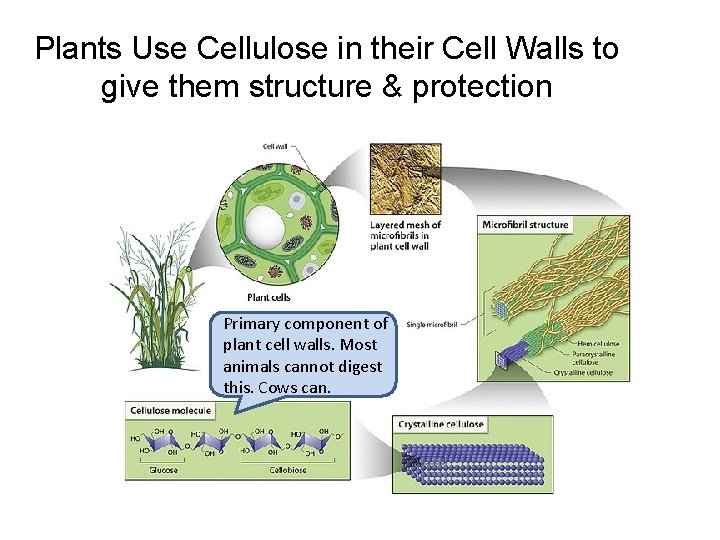
Plants Use Cellulose in their Cell Walls to give them structure & protection Primary component of plant cell walls. Most animals cannot digest this. Cows can.

We (humans) can eat & digest starch but we cannot digest cellulose. WHY? ? ?

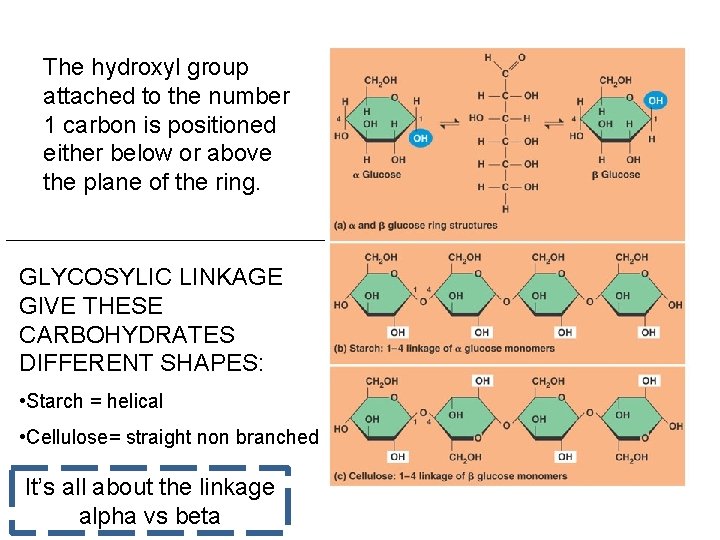
The hydroxyl group attached to the number 1 carbon is positioned either below or above the plane of the ring. GLYCOSYLIC LINKAGE GIVE THESE CARBOHYDRATES DIFFERENT SHAPES: • Starch = helical • Cellulose= straight non branched It’s all about the linkage alpha vs beta

• Your body contains enzymes (amylase) that break starch down into glucose to fuel your body. But we humans don't have enzymes that can break down cellulose. • Some animals do:

• Cellulose is a lot stronger than starch. cellulose is strong enough to make fibers from, and make rope, clothing, etc. • Cellulose doesn't dissolve in water the way starch will, and doesn't break down as easily. • This is a good thing… since our clothes are made of cellulose, wooden park benches and wooden houses would all dissolve after one good rain.

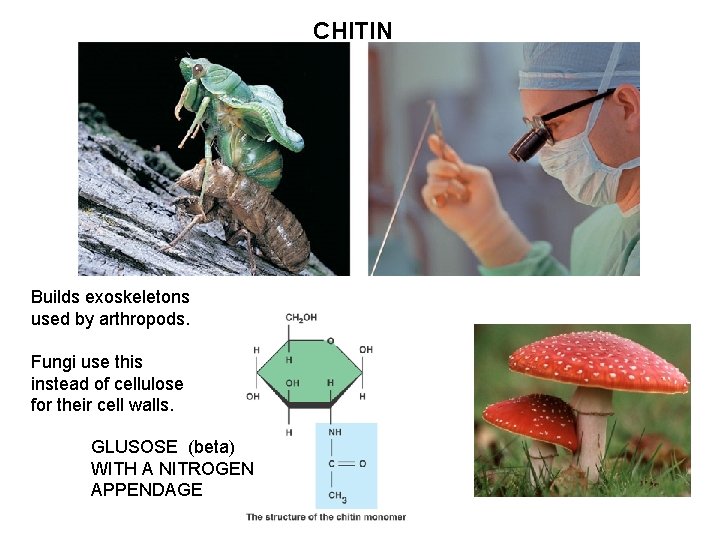
CHITIN Builds exoskeletons used by arthropods. Fungi use this instead of cellulose for their cell walls. GLUSOSE (beta) WITH A NITROGEN APPENDAGE

Lipids • Large nonpolar molecules that are insoluble in water • They are NOT polymers but they are large molecules assembled from smaller molecules. • Three major types – Triglycerides – Phospholipids – Steroids

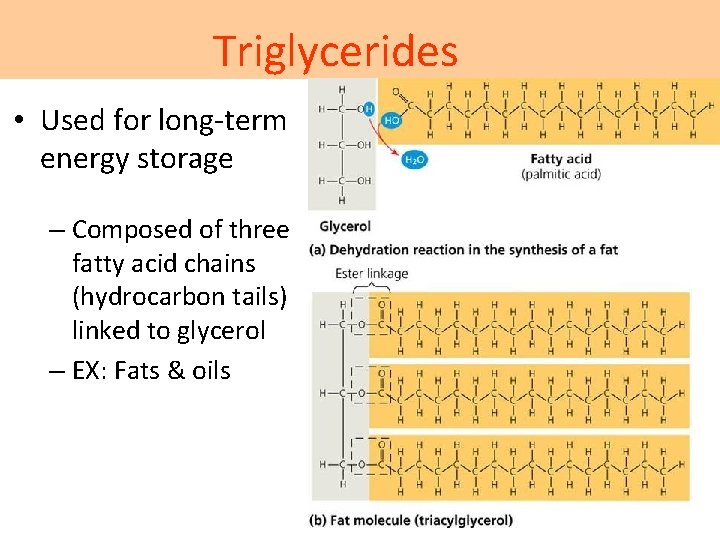
Triglycerides • Used for long-term energy storage – Composed of three fatty acid chains (hydrocarbon tails) linked to glycerol – EX: Fats & oils

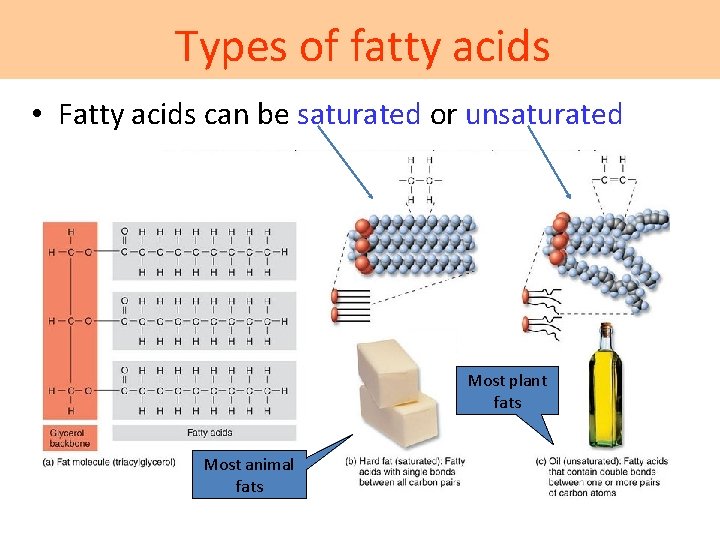
Types of fatty acids • Fatty acids can be saturated or unsaturated Most plant fats Most animal fats

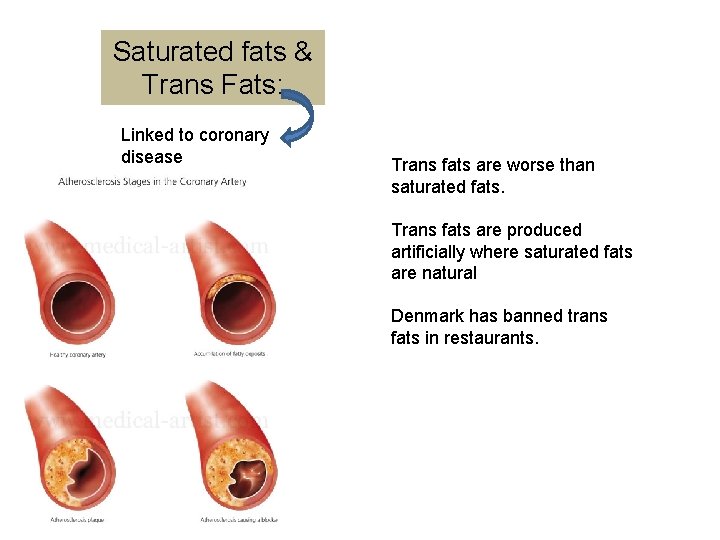
Saturated fats & Trans Fats: Linked to coronary disease Trans fats are worse than saturated fats. Trans fats are produced artificially where saturated fats are natural Denmark has banned trans fats in restaurants.

We need fat in our diet!! Why? • Essential fatty acids – Dietary fats that are essential for growth development and cell functions, but cannot be made by our body’s processes • Proper functioning of nerves and brain- fats are part of myelin- a fatty material which wraps around our nerve cells so that they can send electrical messages. Our brains contain large amounts of essential fats • Maintaining healthy skin and other tissues. All our body cells need to contain some fats as essential parts of cell membranes, controlling what goes in and out of our cells • Transporting fat-soluble vitamins A, D, E and K through the bloodstream to where they are needed • Forming steroid hormones needed to regulate many bodily processes

A GREAT SOURCE OF HEALTHY FAT TO EAT ARE THE OMEGAS • • Prevent coronary heart disease Prevent stroke Prevent diabetes Promote healthy nerve activity Improve vitamin absorption Maintain a healthy immune system Promote cell development MAIN FUNCTION OF FATS IS ENERGY STORAGE -lipids store twice as much chemical energy as the carbohydrate.

Lipids are needed for thermal insulation

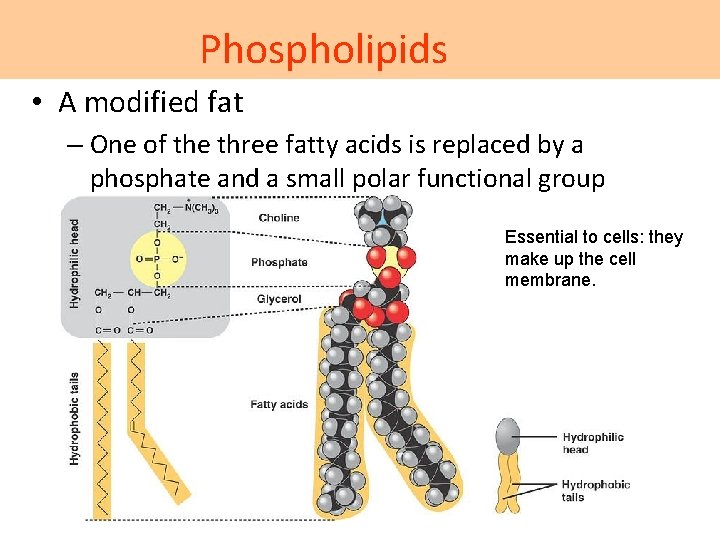
Phospholipids • A modified fat – One of the three fatty acids is replaced by a phosphate and a small polar functional group Essential to cells: they make up the cell membrane.

Steroids • Composed of four carbon rings Examples: Cholesterol Found in most animal cell membranes & in vertebrates it is synthesized in the liver Male and female sex hormones Progesterone, estrogen, & testosterone The attaching chemical groups to the 4 carbon rings is what gives variety.

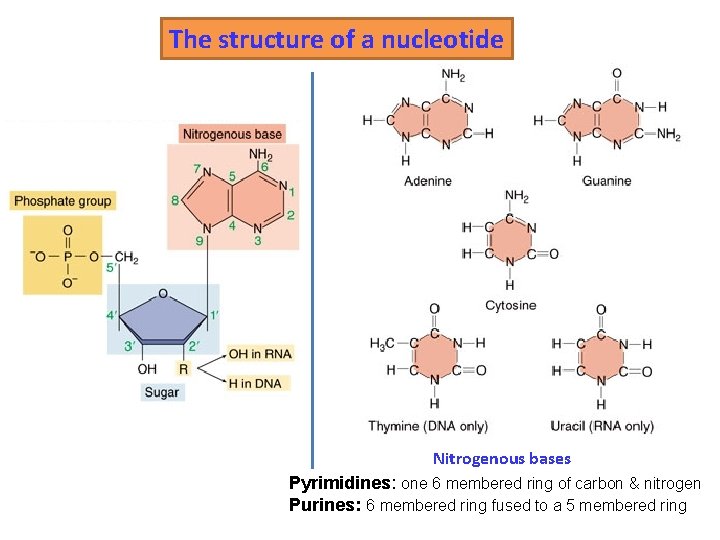
Nucleic Acids • Serve as information storage molecules • Store, transmit and help express hereditary information • Long polymers of repeating subunits termed nucleotides • A nucleotide is composed of three parts – Five-carbon sugar – Nitrogen-containing base – Phosphate

The structure of a nucleotide Nitrogenous bases Pyrimidines: one 6 membered ring of carbon & nitrogen Purines: 6 membered ring fused to a 5 membered ring

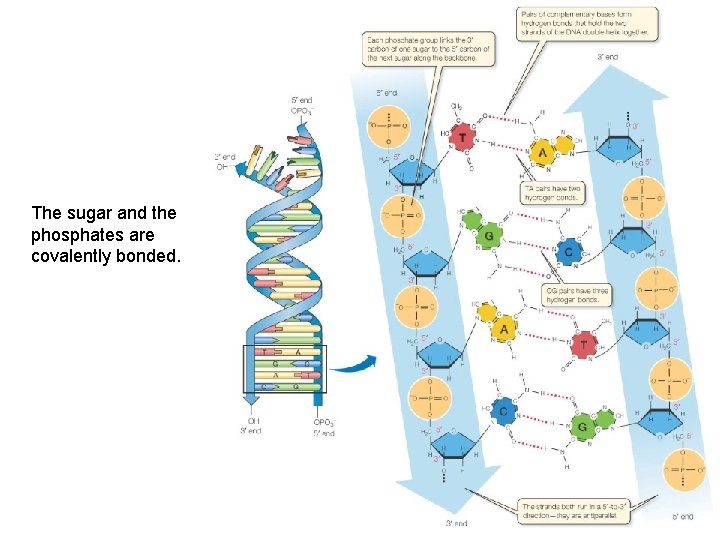
The sugar and the phosphates are covalently bonded.

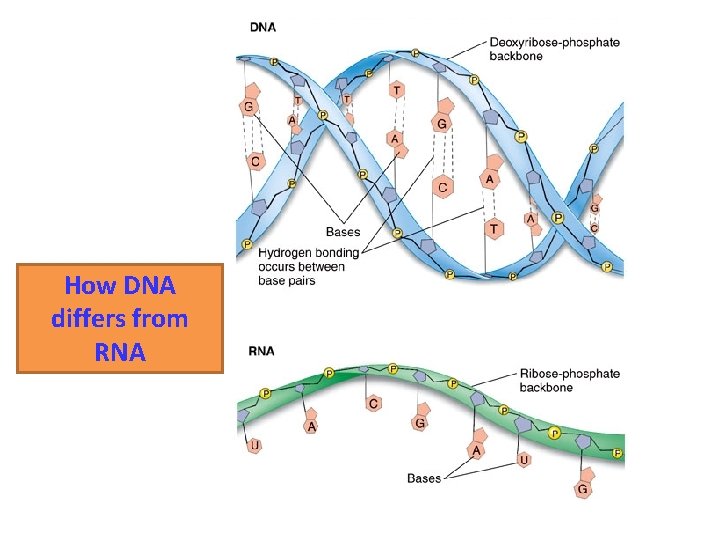
How DNA differs from RNA

Nucleic Acids • Two varieties – Deoxyribonucleic acid (DNA) – Ribonucleic acid (RNA) DNA Sugar = Deoxyribose Bases = A, G, C, T Double-stranded Can’t leave the nucleus RNA Sugar = Ribose Bases = A, G, C, U Single-stranded Can travel outside the nucleus

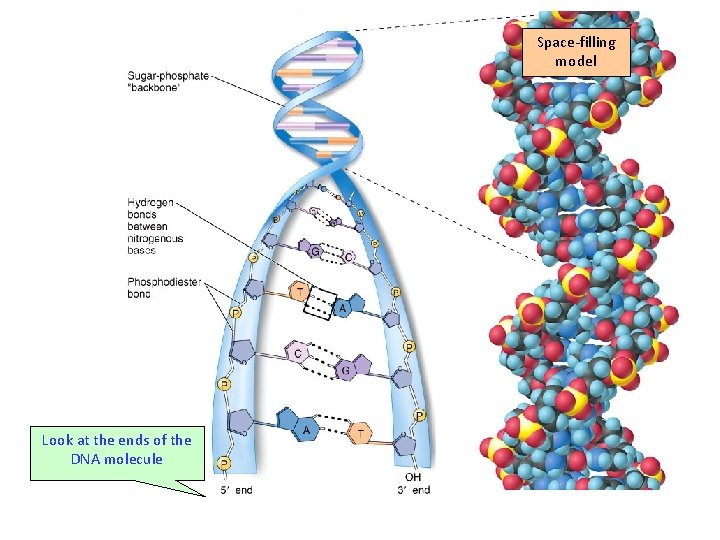
Space-filling model Look at the ends of the DNA molecule

AMONG THE DNA SEQUENCE ARE GENES WHICH ARE CODES FOR SPECIFIC PROTEINS. 5’ –ATTGCAATGGCTAGGGCCAATGC- 3’ 3’ -TAACGTTACCGATCCCGGTTACG- 5’ In the above DNA sequence the code is different • DNA is read in one direction (5’ to 3’)

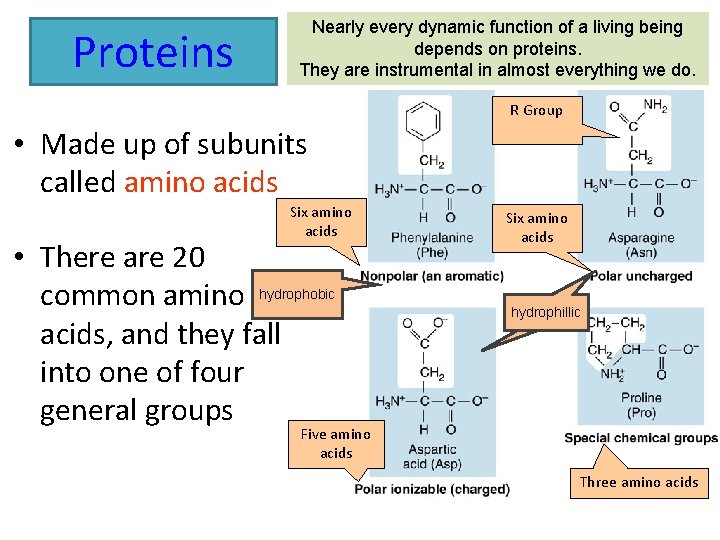
Proteins Nearly every dynamic function of a living being depends on proteins. They are instrumental in almost everything we do. R Group • Made up of subunits called amino acids Six amino acids • There are 20 common amino hydrophobic acids, and they fall into one of four general groups Six amino acids hydrophillic Five amino acids Three amino acids

Polar and non-polar amino acids • Amino acids with non-polar chains are found in regions of proteins that are linked to the hydrophobic area of the cell membrane. • Amino acids with polar side chains are found in regions of proteins that are exposed to water. • Membrane proteins: create a hydrophillic channel in proteins through which polar substances can move.

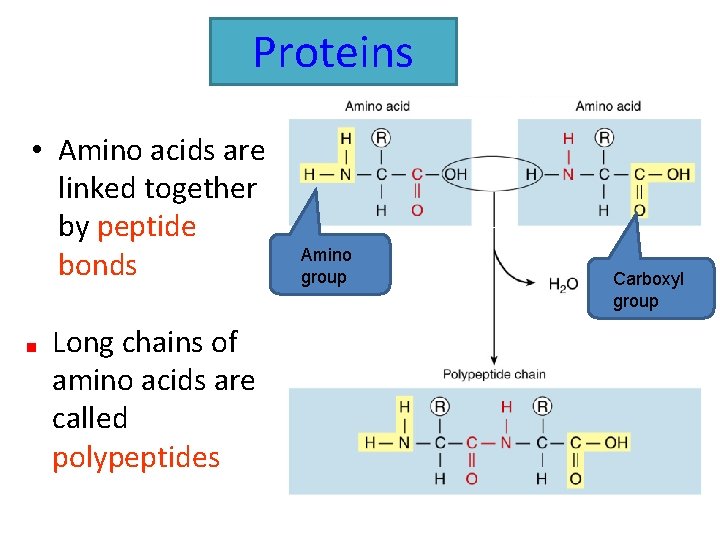
Proteins • Amino acids are linked together by peptide bonds Long chains of amino acids are called polypeptides Amino group Carboxyl group

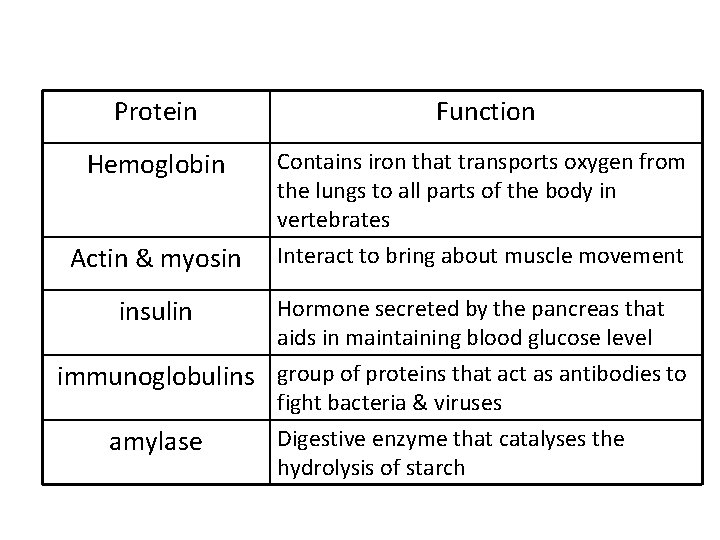
Protein Function Hemoglobin Contains iron that transports oxygen from the lungs to all parts of the body in vertebrates Actin & myosin Interact to bring about muscle movement insulin Hormone secreted by the pancreas that aids in maintaining blood glucose level immunoglobulins group of proteins that act as antibodies to fight bacteria & viruses amylase Digestive enzyme that catalyses the hydrolysis of starch

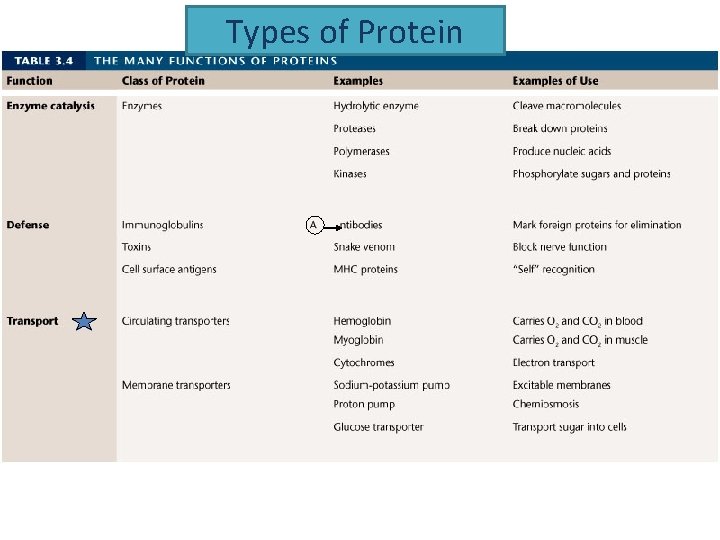
Types of Protein

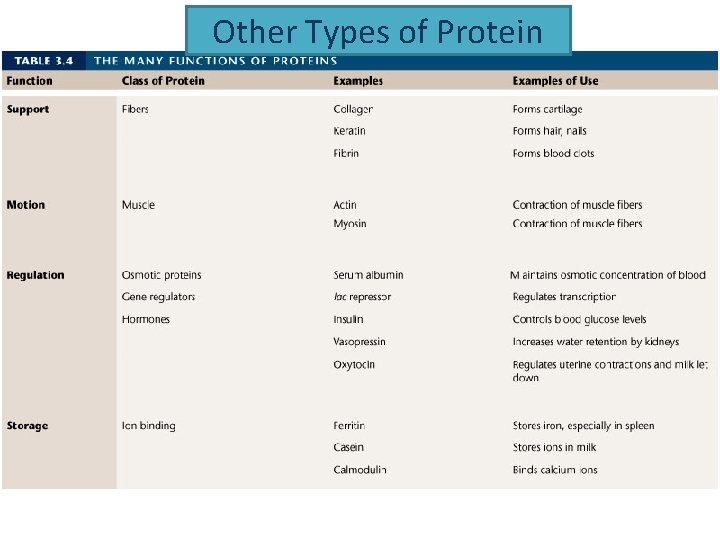
Other Types of Protein

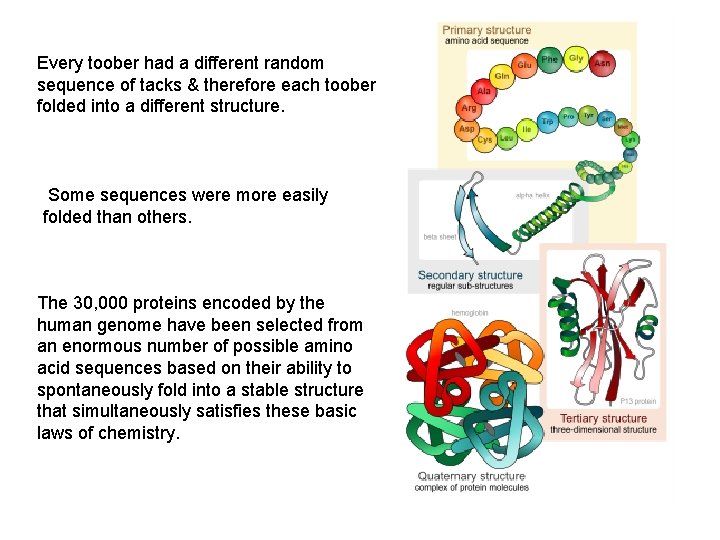
Protein Structure • Determined by the sequence of its amino acids • There are four general levels – Primary – Secondary – Tertiary – Quaternary

Building a Protein • Go to the back and grab a blue toober and a bag of thumb tacks • Make sure your bag of thumb tacks have the following: – 2 blue – 2 red – 2 green – 3 white – 6 yellow

Building a Protein • Take your 15 tacks and place them in any order along the toober. Just make sure they are equal distance from each other and they are on the same side. Fold your protein so that all of the hydrophobic sidechains (yellow tacks) are buried on the inside of your protein, where they will be hidden from polar water molecules.

Fold your protein so the acidic and basic (charged) sidechains are on the outside surface of the protein and pair one negative sidechain (red tack) with one positive sidechain (blue tack) so that they come within one inch of each other and neutralize each other. This positive-negative pairing helps stabilize your protein.

Continue to fold your protein making sure that your polar side-chains (white tacks) are also on the outside surface of your protein where they can hydrogen bond with water. Last, fold your protein so that the two cysteine side-chains (green tacks) are positioned opposite each other on the inside of the protein where they can form a covalent disulfide bond that helps stabilize your protein.

Every toober had a different random sequence of tacks & therefore each toober folded into a different structure. Some sequences were more easily folded than others. The 30, 000 proteins encoded by the human genome have been selected from an enormous number of possible amino acid sequences based on their ability to spontaneously fold into a stable structure that simultaneously satisfies these basic laws of chemistry.

Protein Structure • Primary structure – The specific amino acid sequence of a protein • Secondary structure – The initial folding of the amino acid chain by hydrogen bonding • Tertiary structure – The final three-dimensional shape of the protein • Quaternary structure – The spatial arrangement of polypeptides in a multicomponent protein

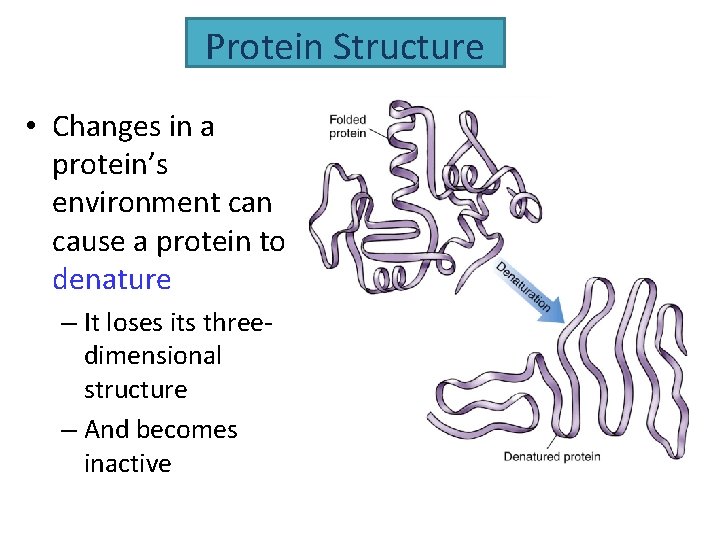
Protein Structure • Changes in a protein’s environment can cause a protein to denature – It loses its threedimensional structure – And becomes inactive

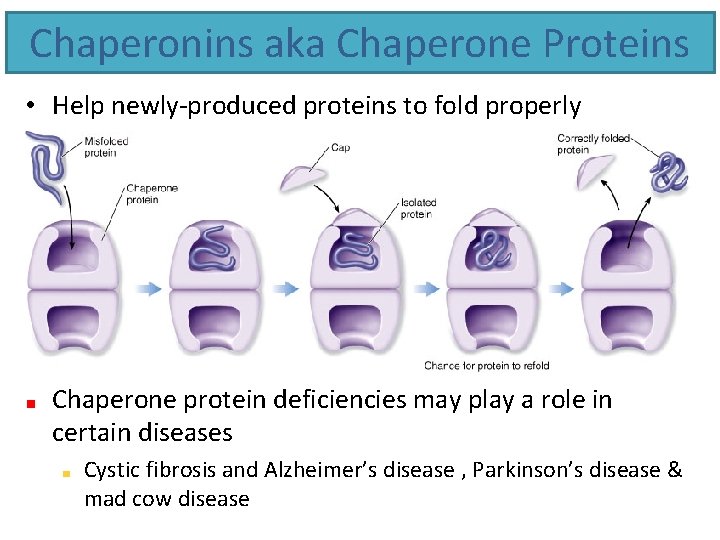
Chaperonins aka Chaperone Proteins • Help newly-produced proteins to fold properly Chaperone protein deficiencies may play a role in certain diseases Cystic fibrosis and Alzheimer’s disease , Parkinson’s disease & mad cow disease

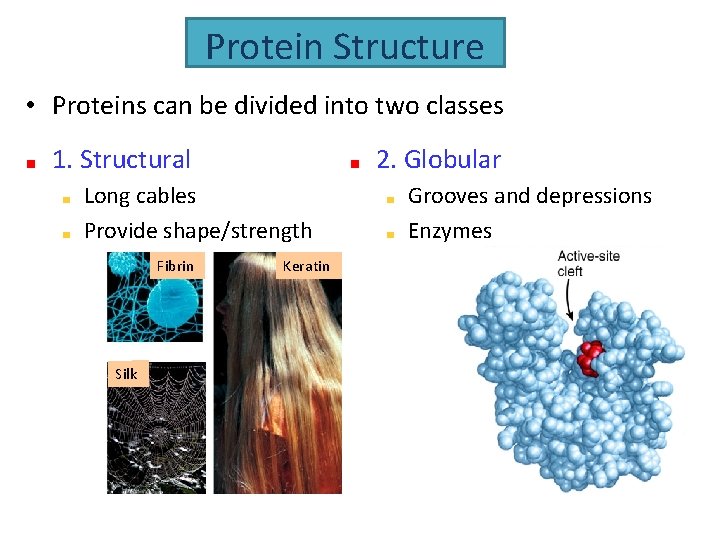
Protein Structure • Proteins can be divided into two classes 1. Structural 2. Globular Long cables Provide shape/strength Fibrin Silk Keratin Grooves and depressions Enzymes

ENZYMES ORGANIC MOLECULES WHICH ACT AS CATALYSTS

Enzymes • Are proteins – So they are made up of ? • Somewhere in this protein is an area that’s designed to match a specific molecule = active site. • The molecule that matches the active site is called the enzyme’s substrate.

Enzymes • Influence the rate of reaction • A set of reactants present with enzymes will form products at a faster rate than without enzymes. • Enzymes cannot force reactions to occur that would not normally occur • The enzymes role is to lower the energy level needed to start the reaction. – Enzymes lower the activation energy of reactions • Enzymes are not used up during the reaction

WHAT FACTORS MIGHT AFFECT ENZYME CATALYST REACTIONS? ENZYME LAB TIME!!!