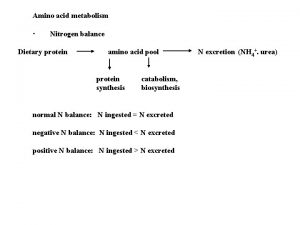
AMINO ACID METABOLISM Jana Novotn Department of the

AMINO ACID METABOLISM Jana Novotná Department of the Medical Chemistry and Biochemistry The 2 nd Faculty of Medicine, Charles Univ.
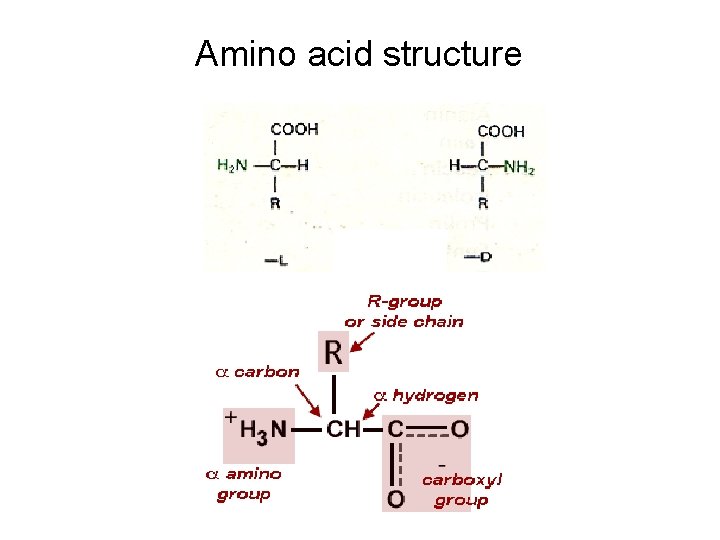
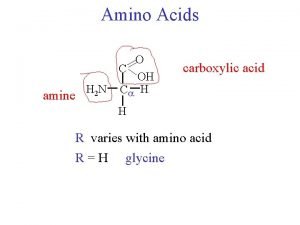
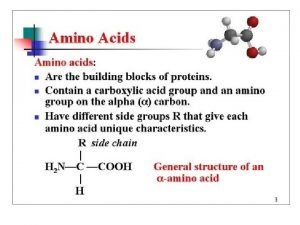
Amino acid structure
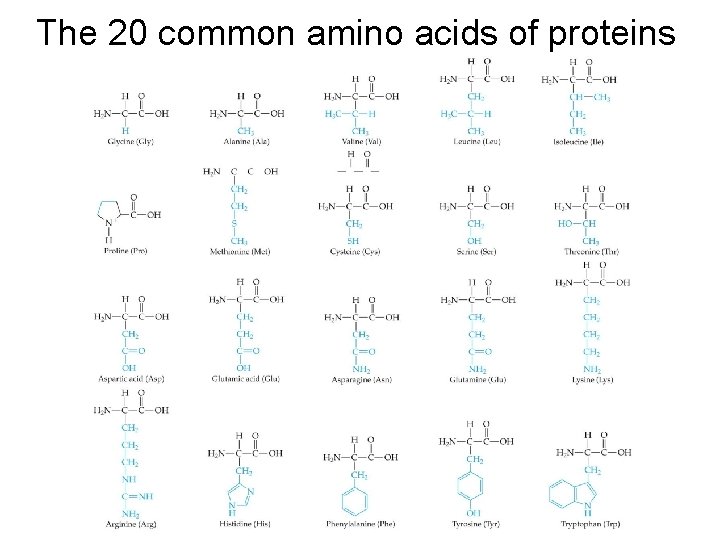
The 20 common amino acids of proteins
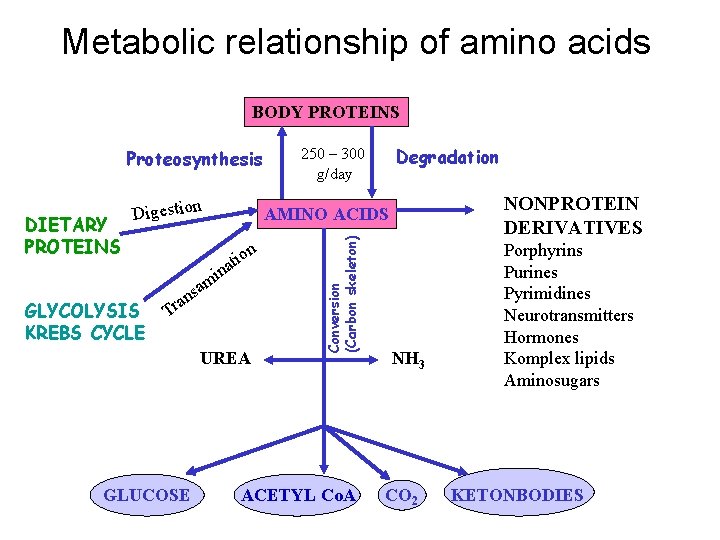
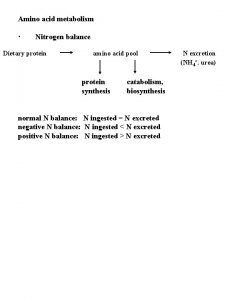
Metabolic relationship of amino acids BODY PROTEINS Proteosynthesis on i t ina am GLYCOLYSIS KREBS CYCLE s ran T UREA GLUCOSE NONPROTEIN DERIVATIVES AMINO ACIDS Conversion (Carbon skeleton) DIETARY PROTEINS Digestion Degradation 250 – 300 g/day ACETYL Co. A NH 3 CO 2 Porphyrins Purines Pyrimidines Neurotransmitters Hormones Komplex lipids Aminosugars KETONBODIES

Enzymes cleaving the peptide bond Endopeptidases – hydrolyse the peptide bond inside a chain: pepsin, trypsin, chymotrypsin Exopeptidases – split the peptide bond at the end of a protein molecule: aminopeptidase, carboxypeptidases Dipeptidases pepsin (p. H 1. 5 – 2. 5) – peptide bond derived from Tyr, Phe, bonds between Leu and Glu trypsin (p. H 7. 5 – 8. 5) – bonds between Lys a Arg chymotrypsin (p. H 7. 5 – 8. 5) – bonds between Phe a Tyr

Essential amino acids in humans Arginine* Histidine* Isoleucine Leucine Valine Lysine Methionine Threonine Phenylalanine Tryptophan *Required to some degree in young growing period and/or sometimes during illness.

Non-essential and nonessential amino acids in humans Can be formed from a-keto acids by transamination and subsequent reactions. Alanine Asparagine Aspartate Glutamine Glycine Proline Serine Cysteine (from Met*) Tyrosine (from Phe*) * Essential amino acids
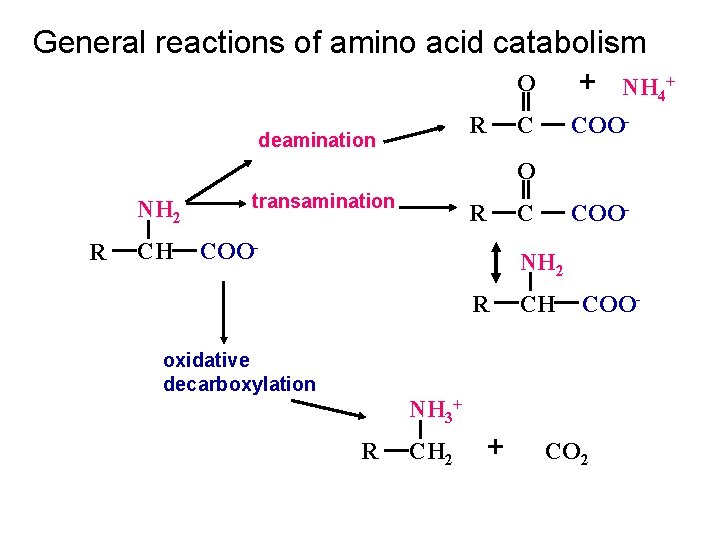
General reactions of amino acid catabolism O + NH 4+ R deamination C COO- O NH 2 R CH transamination R COO- C COO- NH 2 R CH COO- oxidative decarboxylation NH 3+ R CH 2 + CO 2
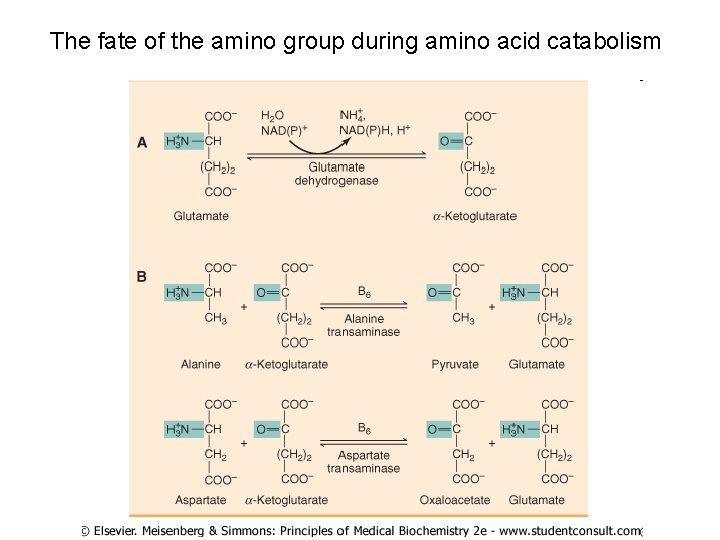
The fate of the amino group during amino acid catabolism

Transamination reaction The first step in the catabolism of most amino acids is removal of a-amino groups by enzymes transaminases or aminotransferases All aminotransferases have the same prostethic group and the same reaction mechanism. The prostethic group is pyridoxal phosphate (PPL), the coenzyme form of pyridoxine (vitamin B 6)
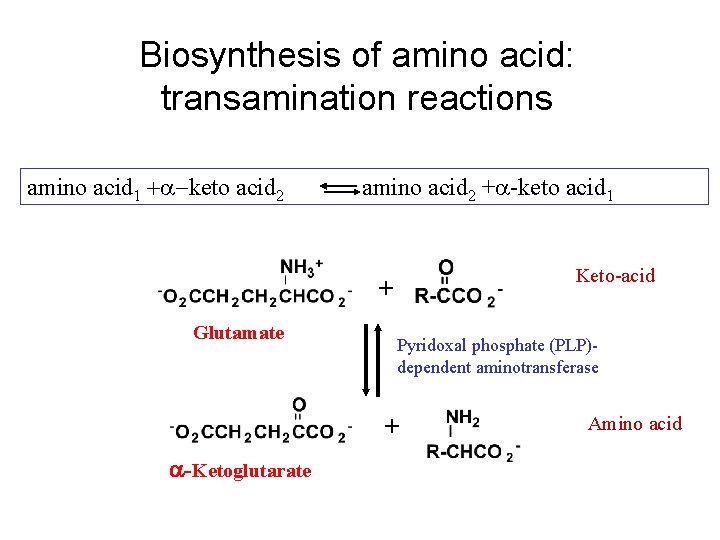
Biosynthesis of amino acid: transamination reactions amino acid 1 +a-keto acid 2 amino acid 2 +a-keto acid 1 Keto-acid + Glutamate Pyridoxal phosphate (PLP)dependent aminotransferase + a-Ketoglutarate Amino acid
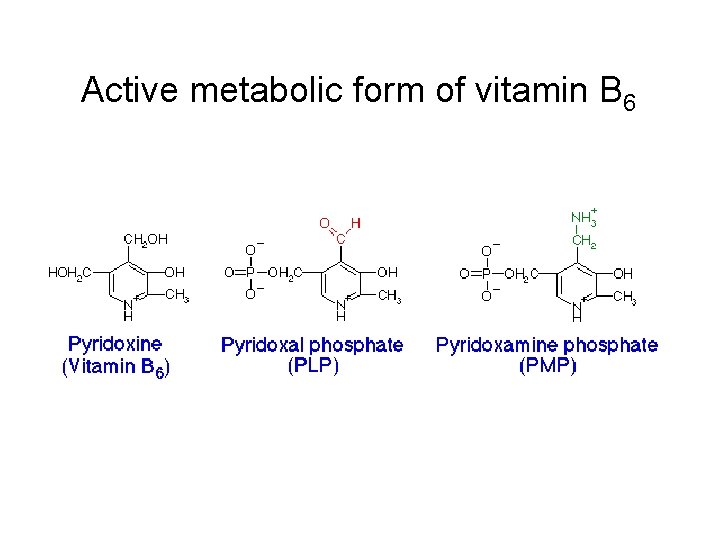
Active metabolic form of vitamin B 6
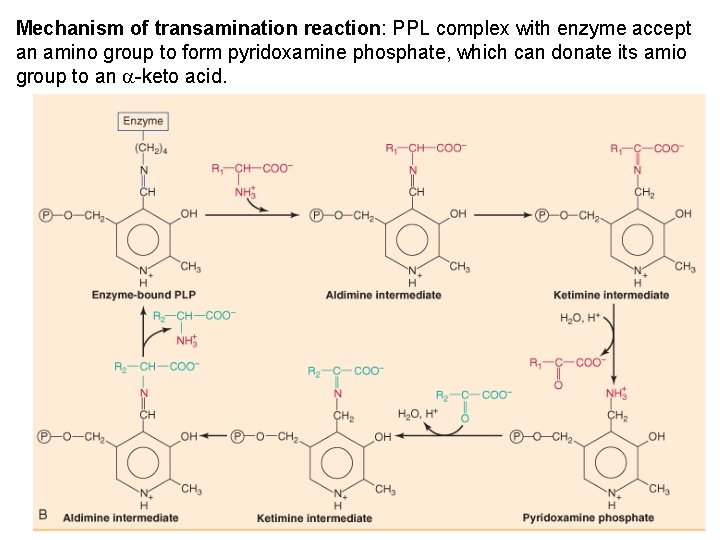
Mechanism of transamination reaction: PPL complex with enzyme accept an amino group to form pyridoxamine phosphate, which can donate its amio group to an a-keto acid.

All amino acids except threonine, lysine, and proline can be transaminated Transaminases are differ in their specificity for L-amino acids. The enzymes are named for the amino group donor.
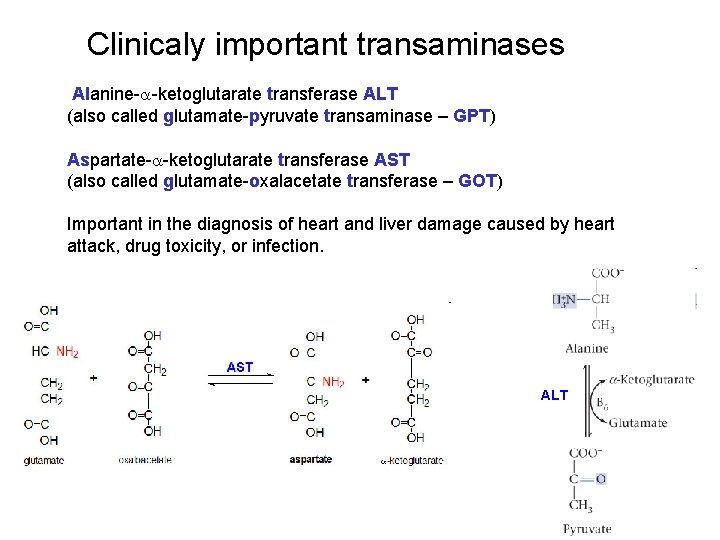
Clinicaly important transaminases Alanine-a-ketoglutarate transferase ALT (also called glutamate-pyruvate transaminase – GPT) Aspartate-a-ketoglutarate transferase AST (also called glutamate-oxalacetate transferase – GOT) Important in the diagnosis of heart and liver damage caused by heart attack, drug toxicity, or infection. ALT
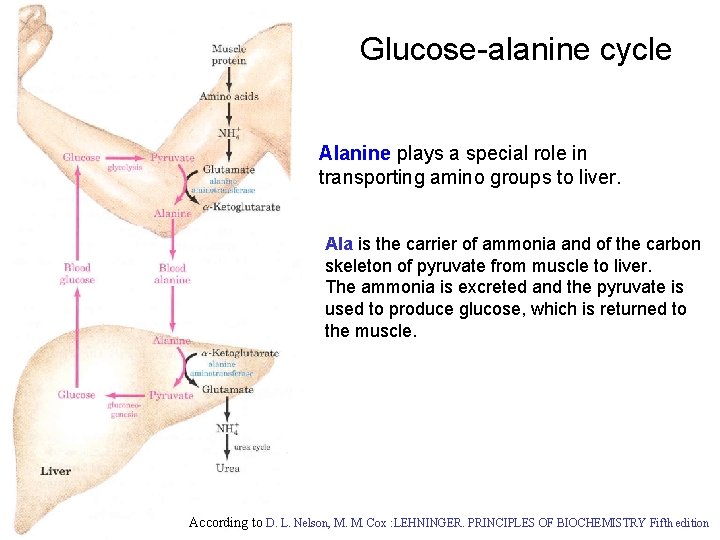
Glucose-alanine cycle Alanine plays a special role in transporting amino groups to liver. Ala is the carrier of ammonia and of the carbon skeleton of pyruvate from muscle to liver. The ammonia is excreted and the pyruvate is used to produce glucose, which is returned to the muscle. According to D. L. Nelson, M. M. Cox : LEHNINGER. PRINCIPLES OF BIOCHEMISTRY Fifth edition

Glutamate releases its amino group as ammonia in the liver The amino groups from many of the a-amino acids are collected in the liver in the form of the amino group of L-glutamate molecules. q Glutamate undergoes oxidative deamination catalyzed by L-glutamate dehydrogenase. q Enzyme is present in mitochondrial matrix. q It is the only enzyme that can use either NAD+ or NADP+ as the acceptor of reducing equivalents. q Combine action of an aminotransferase and glutamate dehydrogenase referred to as transdeamination.
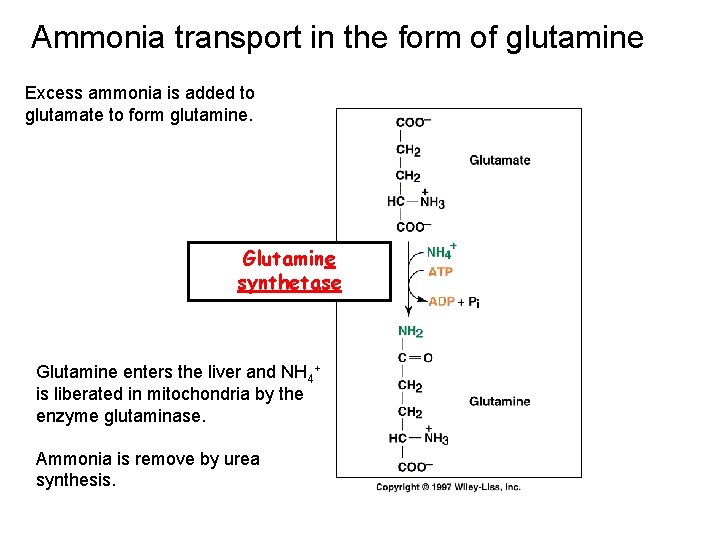
Ammonia transport in the form of glutamine Excess ammonia is added to glutamate to form glutamine. Glutamine synthetase Glutamine enters the liver and NH 4+ is liberated in mitochondria by the enzyme glutaminase. Ammonia is remove by urea synthesis.
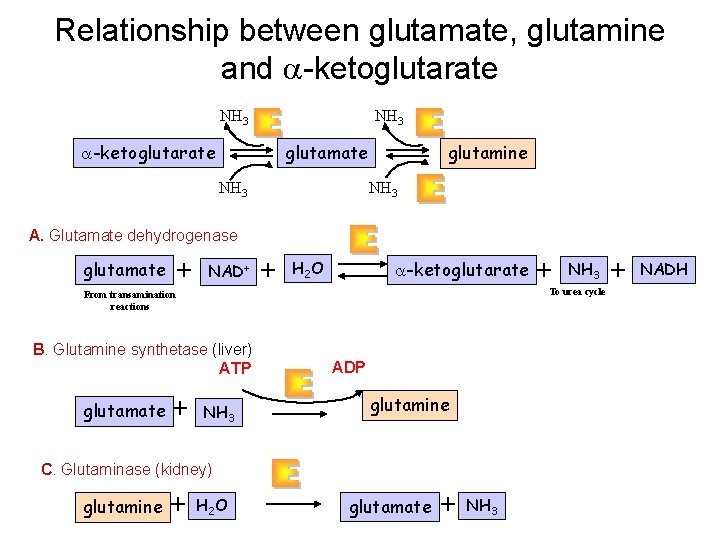
Relationship between glutamate, glutamine and a-ketoglutarate NH 3 glutamate a-ketoglutarate NH 3 glutamine NH 3 A. Glutamate dehydrogenase glutamate + NAD+ + H 2 O a-ketoglutarate B. Glutamine synthetase (liver) ATP + NH 3 ADP glutamine C. Glutaminase (kidney) glutamine + NH 3 To urea cycle From transamination reactions glutamate + H 2 O glutamate + NH 3 + NADH
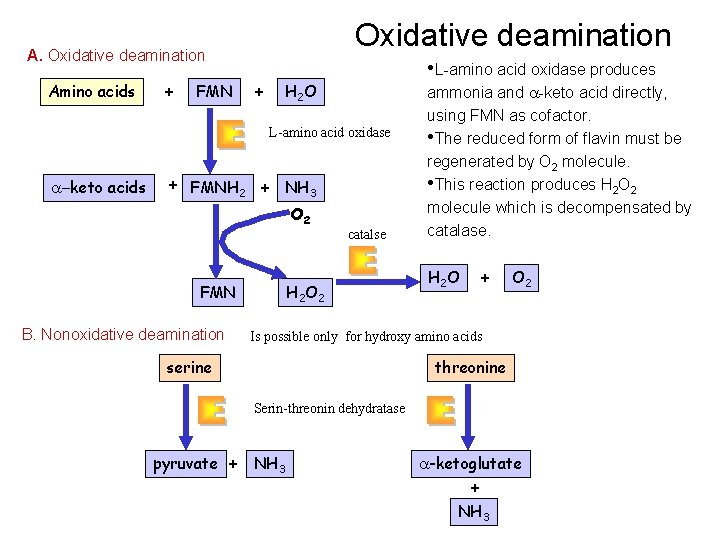
Oxidative deamination Amino acids + FMN + • L-amino acid oxidase produces H 2 O L-amino acid oxidase a-keto acids + FMNH 2 + NH 3 O 2 FMN B. Nonoxidative deamination catalse H 2 O 2 ammonia and a-keto acid directly, using FMN as cofactor. • The reduced form of flavin must be regenerated by O 2 molecule. • This reaction produces H 2 O 2 molecule which is decompensated by catalase. H 2 O + O 2 Is possible only for hydroxy amino acids serine threonine Serin-threonin dehydratase pyruvate + NH 3 a-ketoglutate + NH 3
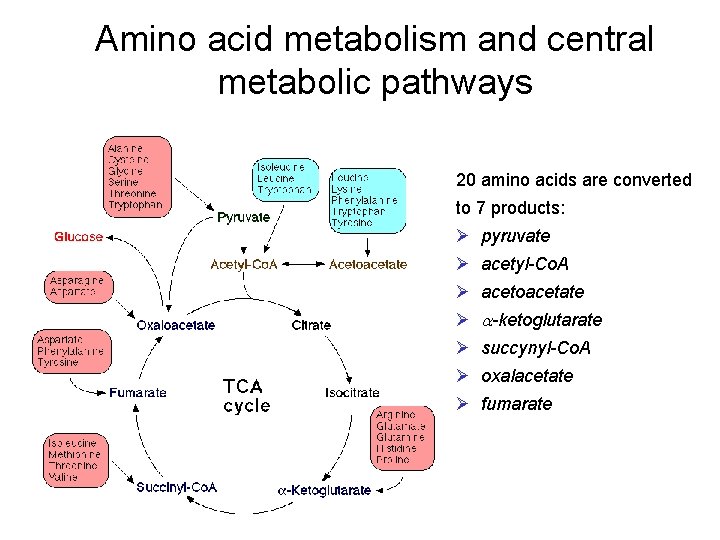
Amino acid metabolism and central metabolic pathways 20 amino acids are converted to 7 products: Ø pyruvate Ø acetyl-Co. A Ø acetoacetate Ø a-ketoglutarate Ø succynyl-Co. A Ø oxalacetate Ø fumarate

Glucogenic Amino Acids formed: a-ketoglutarate, pyruvate, oxaloacetate, fumarate, or succinyl-Co. A Aspartate Asparagine Arginine Phenylalanine Tyrosine Isoleucine Methionine Valine Glutamate Proline Histidine Alanine Serine Cysteine Glycine Threonine Tryptophan

Ketogenic Amino Acids formed acetyl Co. A or acetoacetate Lysine Leucine

Both glucogenic and ketogenic amino acids formed: a-ketoglutarate, pyruvate, oxaloacetate, fumarate, or succinyl-Co. A in addition to acetyl Co. A or acetoacetate Isoleucine Threonine Tryptophan Phenylalanine Tyrosine
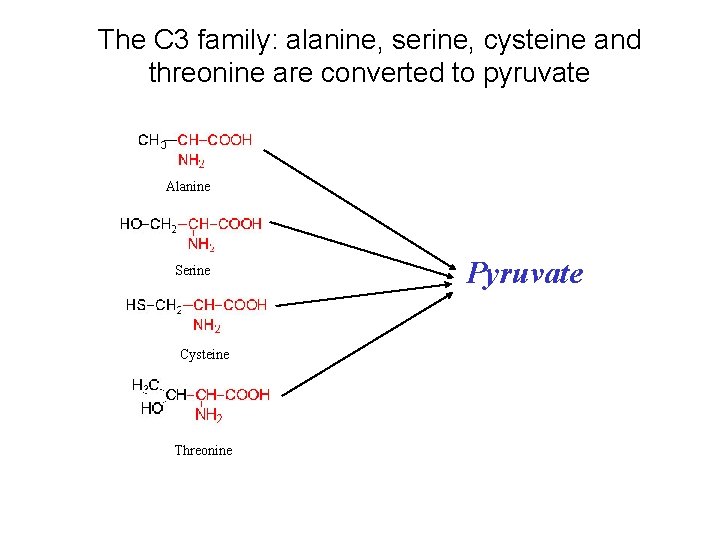
The C 3 family: alanine, serine, cysteine and threonine are converted to pyruvate Alanine Serine Cysteine Threonine Pyruvate
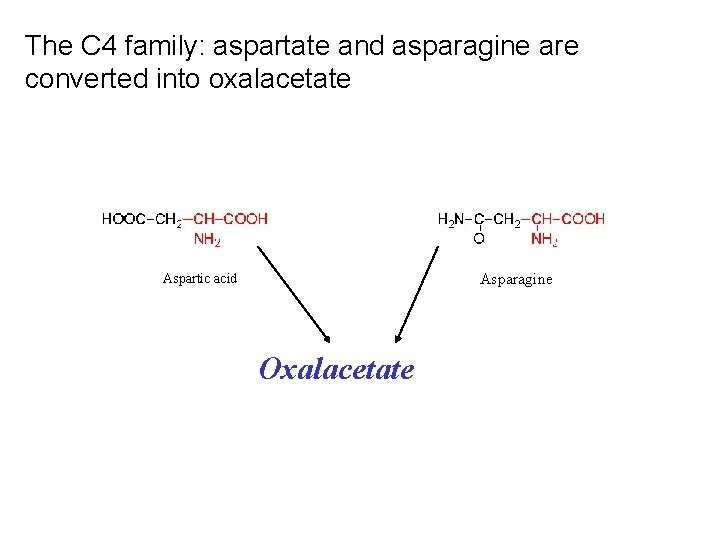
The C 4 family: aspartate and asparagine are converted into oxalacetate Asparagine Aspartic acid Oxalacetate
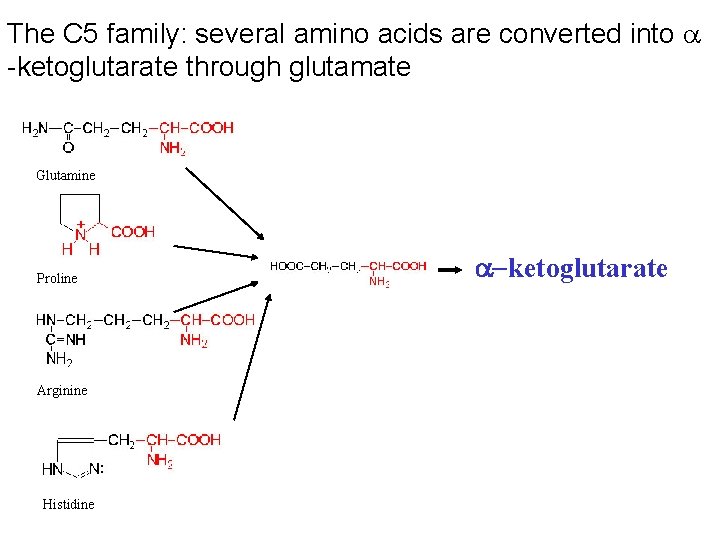
The C 5 family: several amino acids are converted into a -ketoglutarate through glutamate Glutamine Proline Arginine Histidine a-ketoglutarate
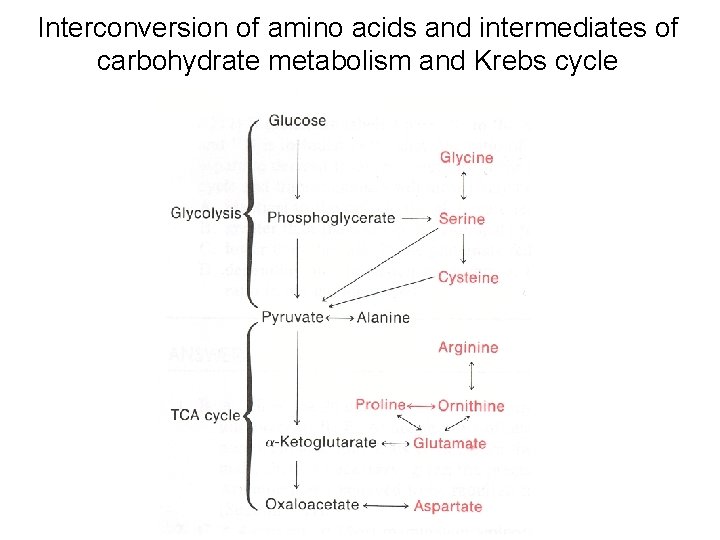
Interconversion of amino acids and intermediates of carbohydrate metabolism and Krebs cycle

Metabolism of some selected amino acids
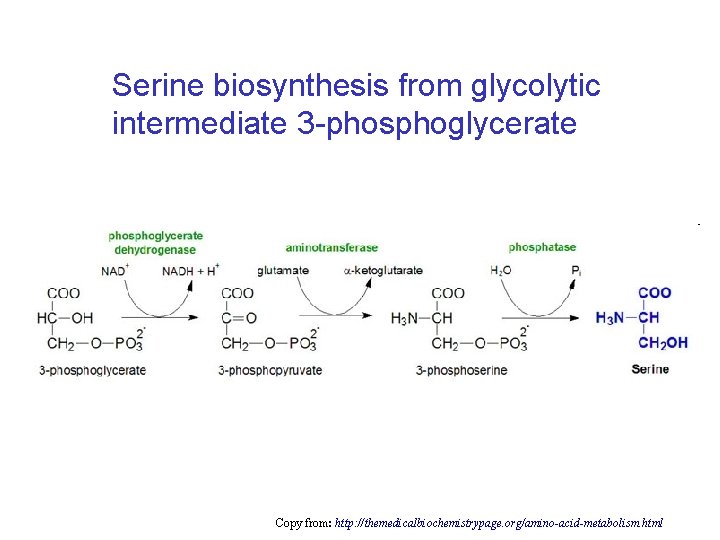
Serine biosynthesis from glycolytic intermediate 3 -phosphoglycerate Copy from: http: //themedicalbiochemistrypage. org/amino-acid-metabolism. html
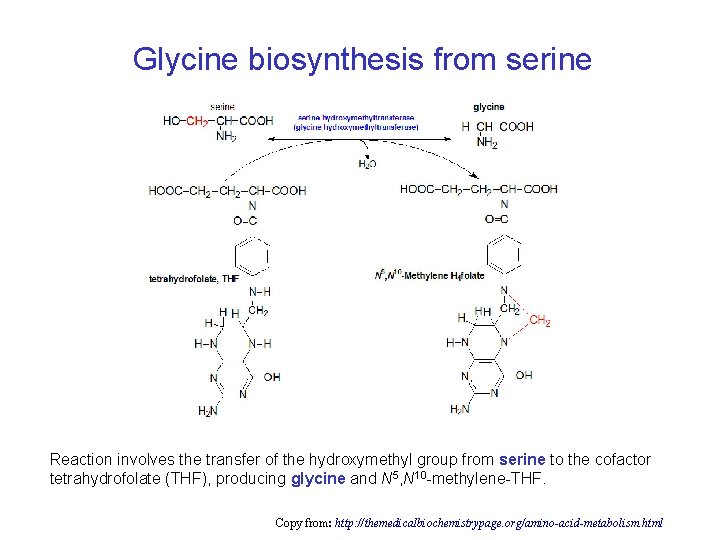
Glycine biosynthesis from serine Reaction involves the transfer of the hydroxymethyl group from serine to the cofactor tetrahydrofolate (THF), producing glycine and N 5, N 10 -methylene-THF. Copy from: http: //themedicalbiochemistrypage. org/amino-acid-metabolism. html
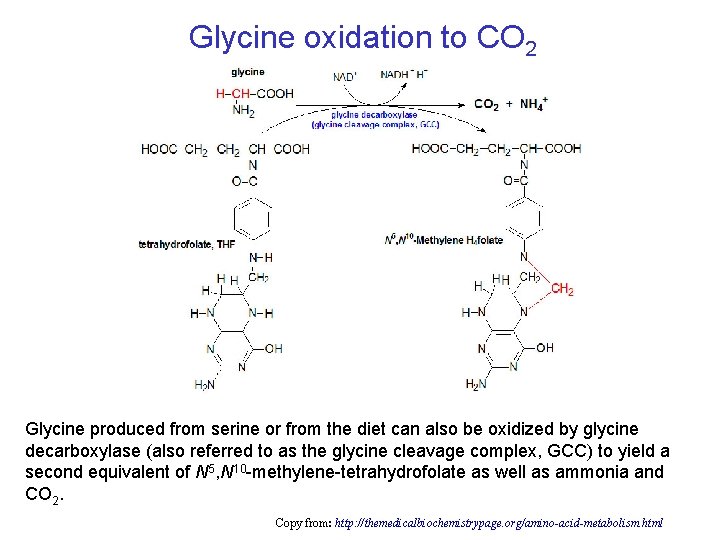
Glycine oxidation to CO 2 Glycine produced from serine or from the diet can also be oxidized by glycine decarboxylase (also referred to as the glycine cleavage complex, GCC) to yield a second equivalent of N 5, N 10 -methylene-tetrahydrofolate as well as ammonia and CO 2. Copy from: http: //themedicalbiochemistrypage. org/amino-acid-metabolism. html
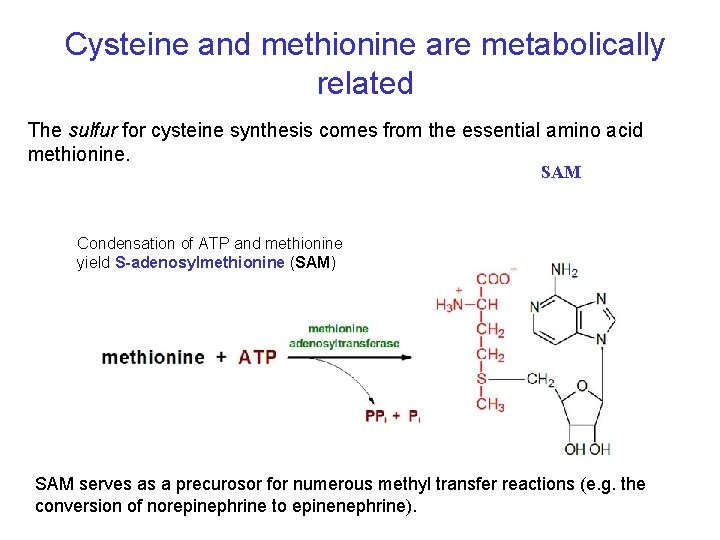
Cysteine and methionine are metabolically related The sulfur for cysteine synthesis comes from the essential amino acid methionine. SAM Condensation of ATP and methionine yield S-adenosylmethionine (SAM) SAM serves as a precurosor for numerous methyl transfer reactions (e. g. the conversion of norepinephrine to epinenephrine).
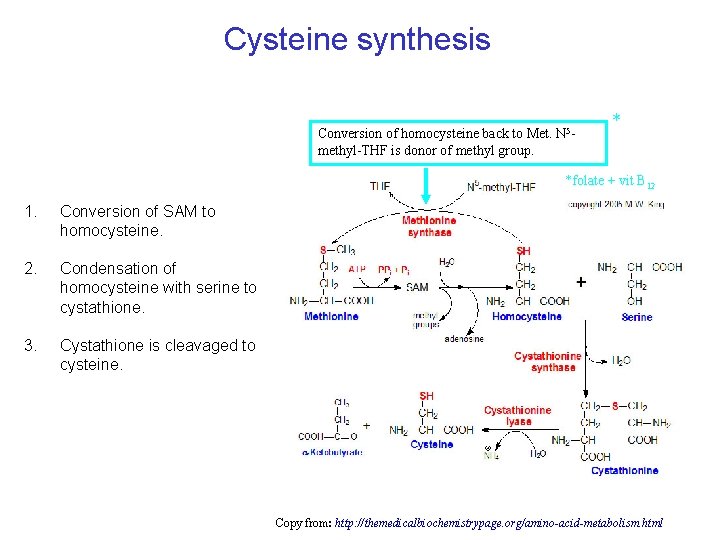
Cysteine synthesis Conversion of homocysteine back to Met. N 5 methyl-THF is donor of methyl group. * *folate + vit B 12 1. Conversion of SAM to homocysteine. 2. Condensation of homocysteine with serine to cystathione. 3. Cystathione is cleavaged to cysteine. Copy from: http: //themedicalbiochemistrypage. org/amino-acid-metabolism. html

Homocystinuria Genetic defects for both the synthase and the lyase. Missing or impaired cystathionine synthase leads to homocystinuria. High concentration of homocysteine and methionine in the urine. Homocysteine is highly reactive molecule. Disease is often associated with mental retardation, multisystemic disorder of connective tissue, muscle, CNS, and cardiovascular system.
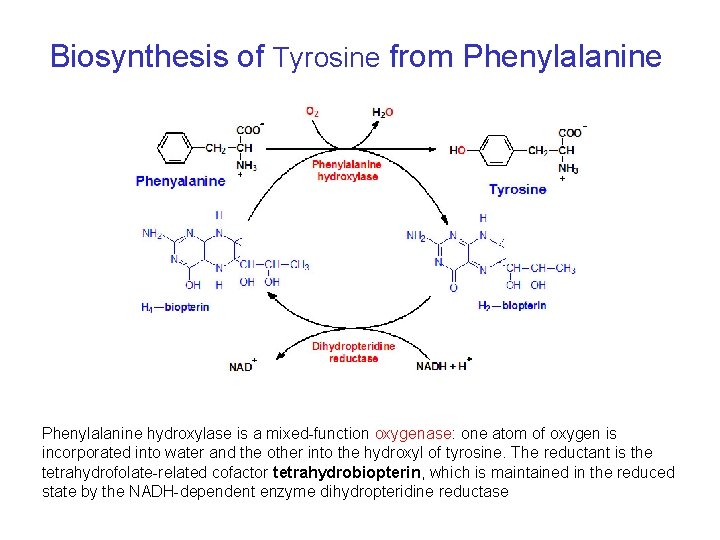
Biosynthesis of Tyrosine from Phenylalanine hydroxylase is a mixed-function oxygenase: one atom of oxygen is incorporated into water and the other into the hydroxyl of tyrosine. The reductant is the tetrahydrofolate-related cofactor tetrahydrobiopterin, which is maintained in the reduced state by the NADH-dependent enzyme dihydropteridine reductase

Phenylketonuria Hyperphenylalaninemia - complete deficiency of phenylalanine hydroxylase (plasma level of Phe raises from normal 0. 5 to 2 mg/d. L to more than 20 mg/d. L). The mental retardation is caused by the accumulation of phenylalanine, which becomes a major donor of amino groups in aminotransferase activity and depletes neural tissue of α-ketoglutarate. Absence of α-ketoglutarate in the brain shuts down the TCA cycle and the associated production of aerobic energy, which is essential to normal brain development. Newborns are routinelly tested for blood concentration of Phe. The diet with low-phenylalanine diet.
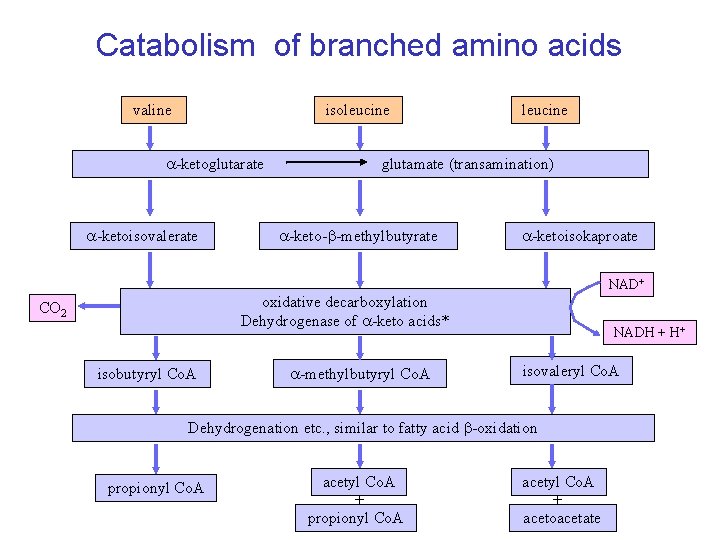
Catabolism of branched amino acids valine isoleucine a-ketoglutarate a-ketoisovalerate leucine glutamate (transamination) a-keto-b-methylbutyrate a-ketoisokaproate NAD+ oxidative decarboxylation Dehydrogenase of a-keto acids* CO 2 isobutyryl Co. A a-methylbutyryl Co. A NADH + H+ isovaleryl Co. A Dehydrogenation etc. , similar to fatty acid b-oxidation propionyl Co. A acetyl Co. A + acetoacetate

Branched-chain aminoaciduria Disease also called Maple Syrup Urine Disease (MSUD) (because of the characteristic odor of the urine in affected individuals). Deficiency in an enzyme, branched-chain α-keto acid dehydrogenase leads to an accumulation of three branchedchain amino acids and their corresponding branched-chain α-keto acids which are excreted in the urine. There is only one dehydrogenase enzyme for all three amino acids. Mental retardation in these cases is extensive.
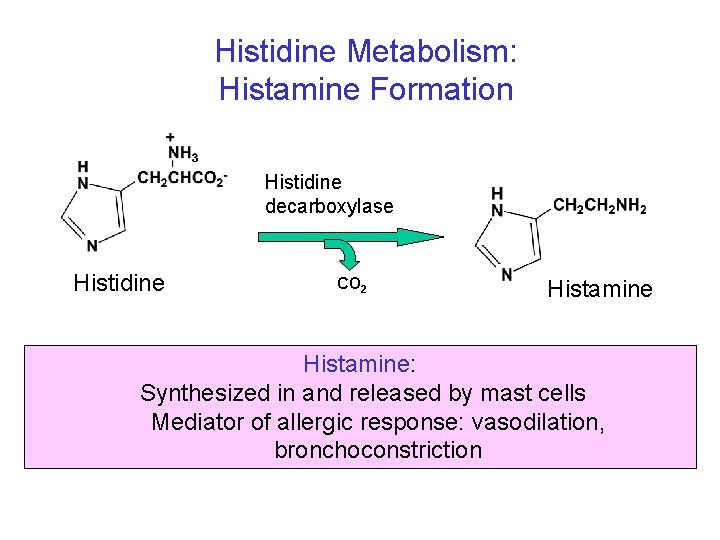
Histidine Metabolism: Histamine Formation Histidine decarboxylase Histidine CO 2 Histamine: Synthesized in and released by mast cells Mediator of allergic response: vasodilation, bronchoconstriction
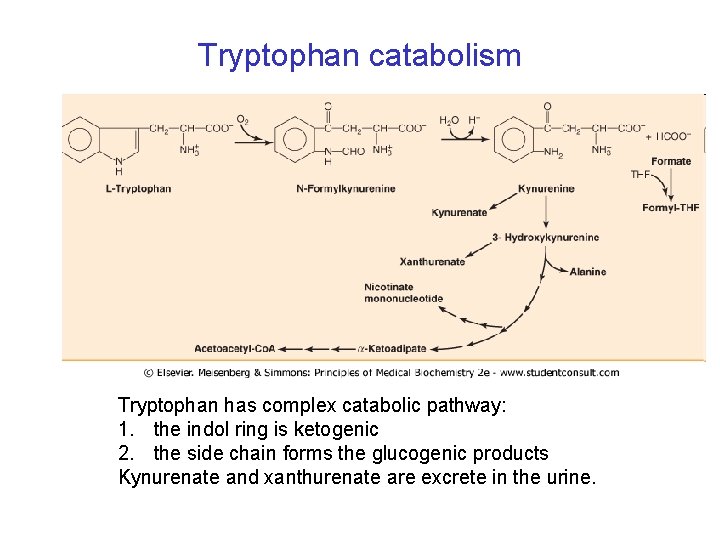
Tryptophan catabolism Tryptophan has complex catabolic pathway: 1. the indol ring is ketogenic 2. the side chain forms the glucogenic products Kynurenate and xanthurenate are excrete in the urine.

Enzymes which metabolised amino acides containe vitamines as cofactors THIAMINE B 1 (thiamine diphosphate) oxidative decarboxylation of a-ketoacids RIBOFLAVIN B 2 (flavin mononucleotide FMN, flavin adenine dinucleotide FAD) oxidses of a-aminoacids NIACIN B 3 – nicotinic acid (nikotinamide adenine dinucleotide NAD+ nikotinamide adenine dinukleotide phosphate NADP+) dehydrogenases, reductase PYRIDOXIN B 6 (pyridoxalphosphate) transamination reaction and decarboxylation FOLIC ACID (tetrahydropholate) Meny enzymes of amino acid metabolism

Helpful website http: //themedicalbiochemistrypage. org/amino-acid-metabolism. html
- Slides: 43