Chapter 3 Notes Biochemistry the chemistry of life

Chapter 3 Notes Biochemistry: the chemistry of life
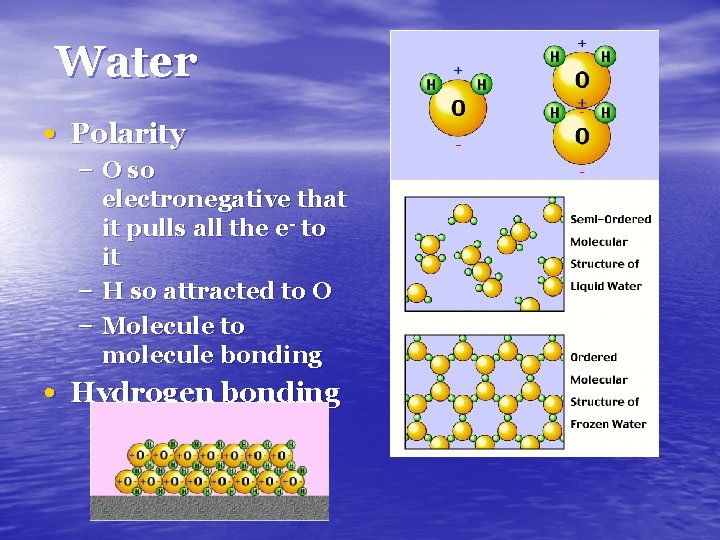
Water • Polarity – O so electronegative that it pulls all the e- to it – H so attracted to O – Molecule to molecule bonding • Hydrogen bonding

Polar Molecules • Solvent • Dissolves other polar molecules • Sugars, proteins, and ionic compounds

Cohesion and Adhesion • Attraction to each other • Attraction to other molecules • Capillarity – Adhesive and cohesive forces – Movement through small tubes – Against gravity – Grass giant redwoods

Properties of Water—so cool! • Cohesive/adhesive behavior • Versatile solvent • Resists changes in temperature • High heat of vaporization and cools • surfaces as it evaporates Expands when it freezes & less dense (floats)

Water and Temperature • Specific heat: amount of energy required • • • to change the temperature Water has high specific heat Allows for moderation of Earth’s climate Helps organisms regulate their body temperature more effectively

SOLUTIONS • Mixture in which One or More Substances are • • • UNIFORMLY Distributed in Another Substance. Solutions can be mixtures of liquids, solids, or gases. Think of Sugar Water (The Solution) - the Sugar is dissolved in the Water and is Uniformly Distributed. SOLUTE (Sugar) is the Substance Dissolved in the Solution (Sugar Water). The particles that compose the Solute may be Ions, Atoms, or Molecules. SOLVENT (Water) is the Substance in which the Solute is dissolved.

Acids and bases • The p. H is a measure of how acidic or basic a solution is. • A scale with values ranging from below 0 to above 14 is used to measure p. H. • Substances with a p. H below 7 are acidic. An acid is any substance that forms ions (H+) in water. • A solution is neutral if its p. H equals seven.

Bases • Substances with p. H above 7 are basic. A base is any substance that forms hydroxide ions (OH-) in water. • Bases tend to FEEL Slippery (soap) and have a BITTER Taste. • Soap, bleach, lye, oven cleaner, baking soda… p. H 11

Acids • Substances with p. H • below 7 are acidic. An acid is any substance that forms hydronium ions (H+ or H 3 O+) in water Acids tend to have a SOUR Taste and in concentrated forms are Highly Corrosive.

Carbon: The Element of Life • • In all organic compounds Four bonding sites Covalent Bonds with other elements and itself Chains, branches, rings Single, double, triple bonds Functional groups—help determine properties of the molecules

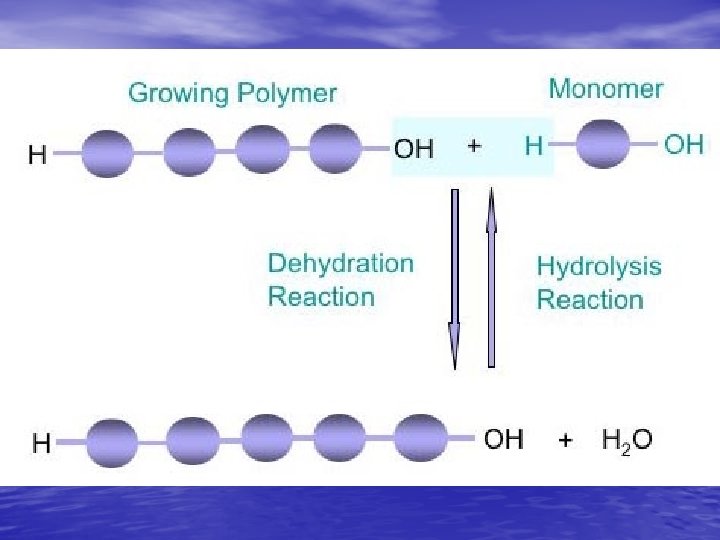
Building Molecules • Monomers – One subunit – 1 brick on the wall • Polymers – Many subunits – A row of bricks to the whole wall… • Condensation reactions—build it • Hydrolysis—break it down
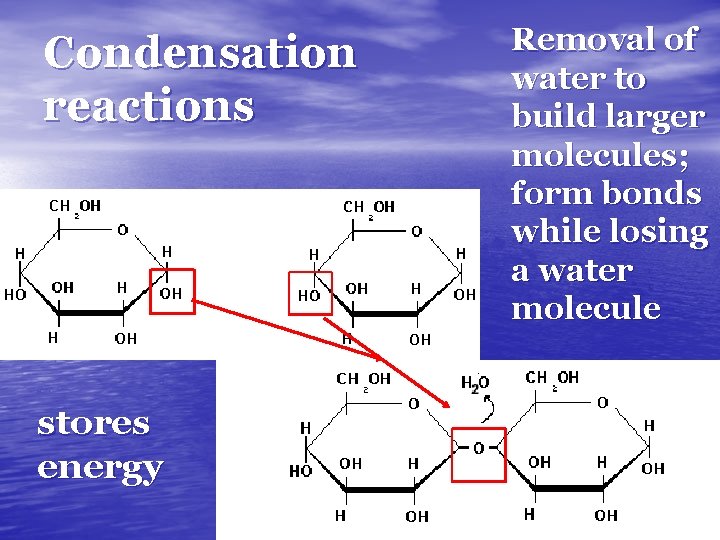
Condensation reactions stores energy Removal of water to build larger molecules; form bonds while losing a water molecule
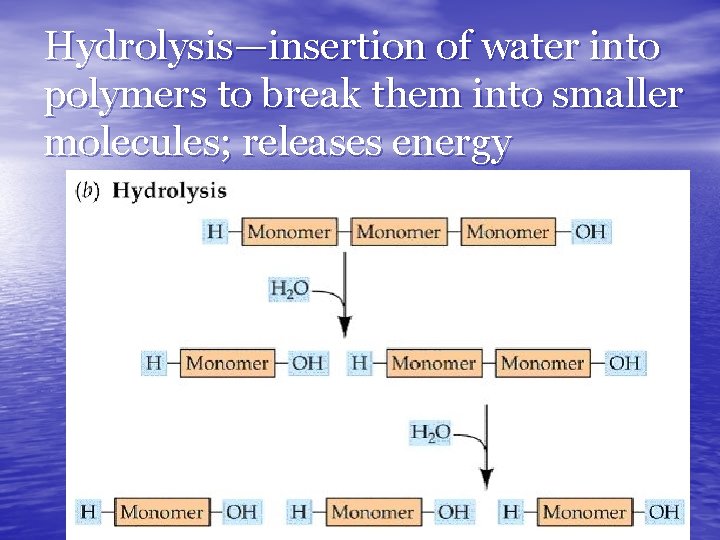
Hydrolysis—insertion of water into polymers to break them into smaller molecules; releases energy

ATP • Adenosine • triphosphate the energy molecule • The secrete is in the • final phosphate The energy molecule because bonds are easy to make and break

Organic Compounds • CARBOHYDRATES, LIPIDS (FAT), PROTEINS, • • • and NUCLEIC ACIDS (DNA, RNA) CHONPS— 6 essential elements of life built from Carbon, Hydrogen, and Oxygen Atoms occur in different Ratios in each class of Compound Despite their similarities, the different classes of compounds have different properties

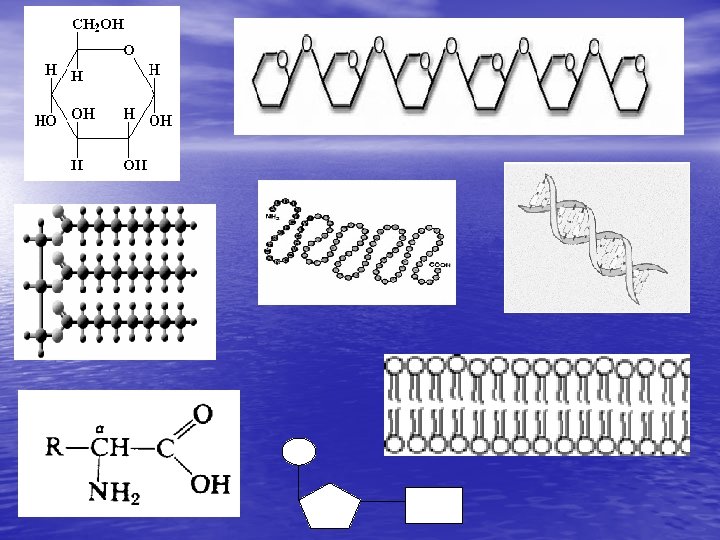
Carbohydrates • human body obtain • • • MOST of their ENERGY from CARBOHYDRATES CHO Sugars, Starches and Cellulose are Carbohydrates Monosaccharides, Disaccharides, & Polysaccharides

MONOSACCHARIDES • Monomer of Carbohydraytes is… • SINGLE SUGARS (Simple Sugar) – – – Glucose—made by plants (photosynthesis) Galactose—found in milk Fructose—fruit sugar • C 6 H 12 O 6 • Differing Structures determine the different • Properties ISOMERS: Compounds with a Single Chemical Formula but Different Forms
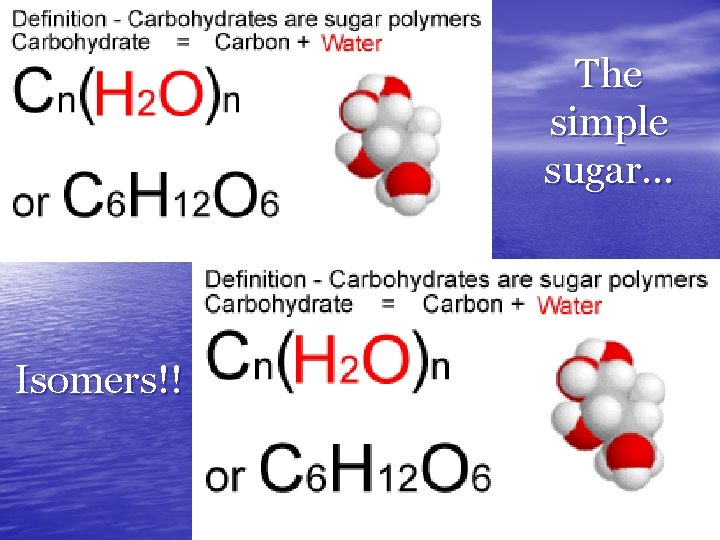
The simple sugar… Isomers!!
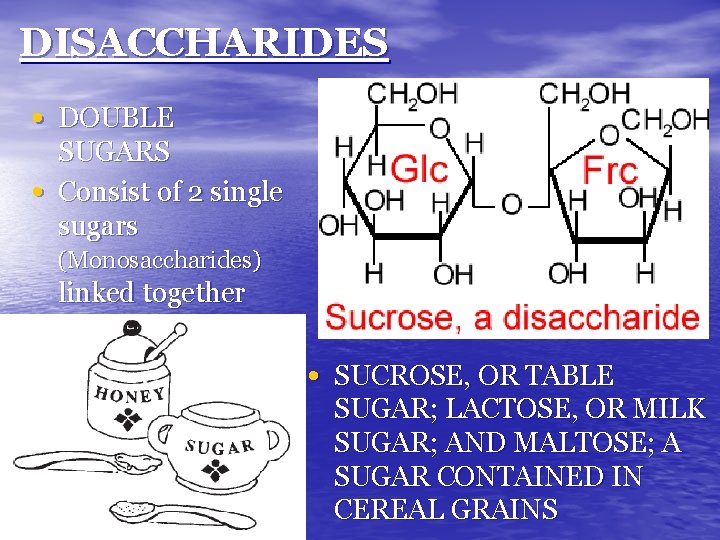
DISACCHARIDES • DOUBLE • SUGARS Consist of 2 single sugars (Monosaccharides) linked together • SUCROSE, OR TABLE SUGAR; LACTOSE, OR MILK SUGAR; AND MALTOSE; A SUGAR CONTAINED IN CEREAL GRAINS

POLYSACCHARIDES • MADE OF LONG • • CHAINS OF SUGARS ("Many Sugars", Three or More Monosaccharides) POLY means "Many“ Starches: BREAD, PASTA, AND POTATOES, ARE POLYSACCHARIDES
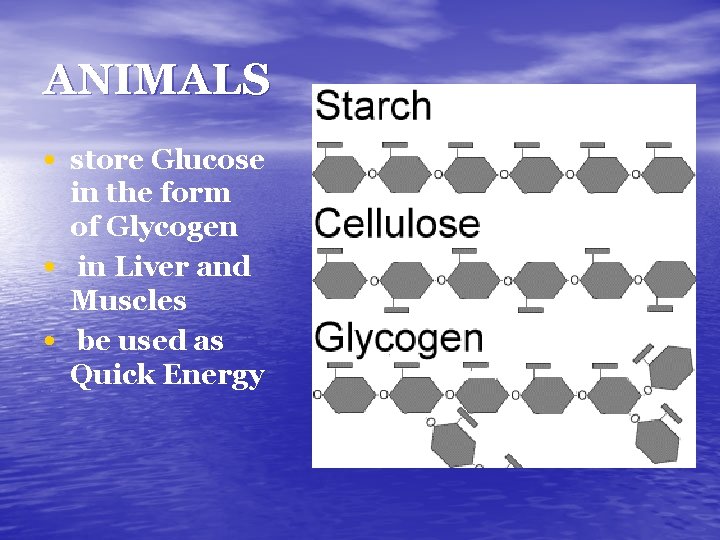
ANIMALS • store Glucose • • in the form of Glycogen in Liver and Muscles be used as Quick Energy
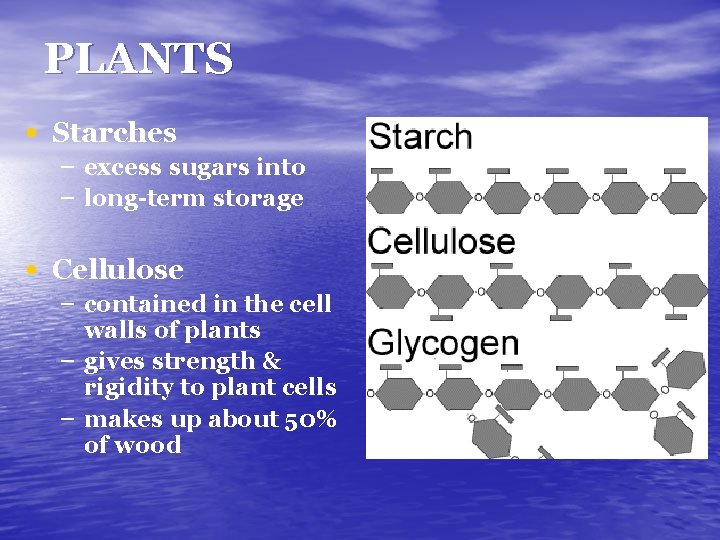
PLANTS • Starches – excess sugars into – long-term storage • Cellulose – contained in the cell walls of plants – gives strength & rigidity to plant cells – makes up about 50% of wood

PROTEINS • composed mainly • of Carbon, Hydrogen, and Nitrogen Construction materials for the body parts such as muscles, skin and blood • Our cells need proteins to make other proteins, such as enzymes

AMINO ACIDS • Proteins are made up of smaller units called amino acids: • monomer/building blocks of protein Our bodies contain thousands of different proteins 20 Different • All these proteins are made from about amino acids • Amino Acids Differ ONLY in the type of R Group they Carry. The difference among the Amino Acid R Groups gives different Proteins Very Different Shapes
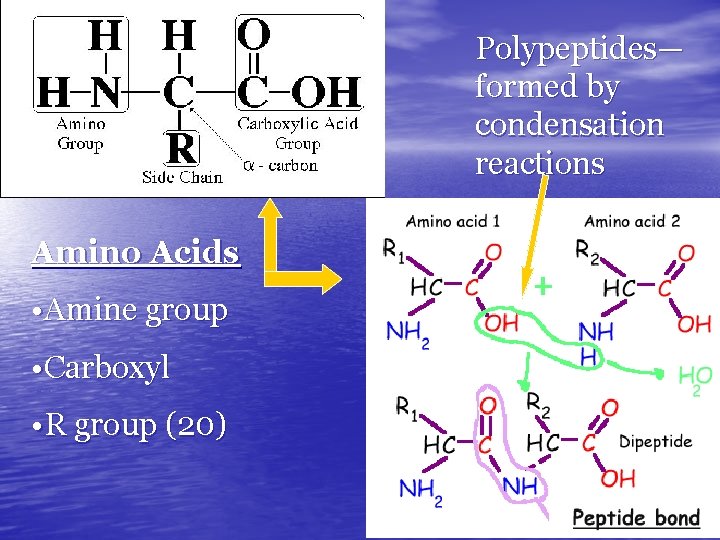
Polypeptides— formed by condensation reactions Amino Acids • Amine group • Carboxyl • R group (20)
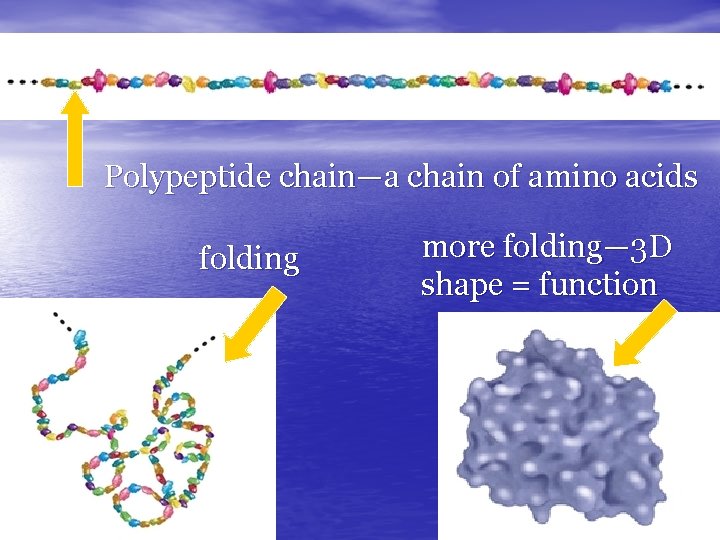
Polypeptide chain—a chain of amino acids folding more folding— 3 D shape = function
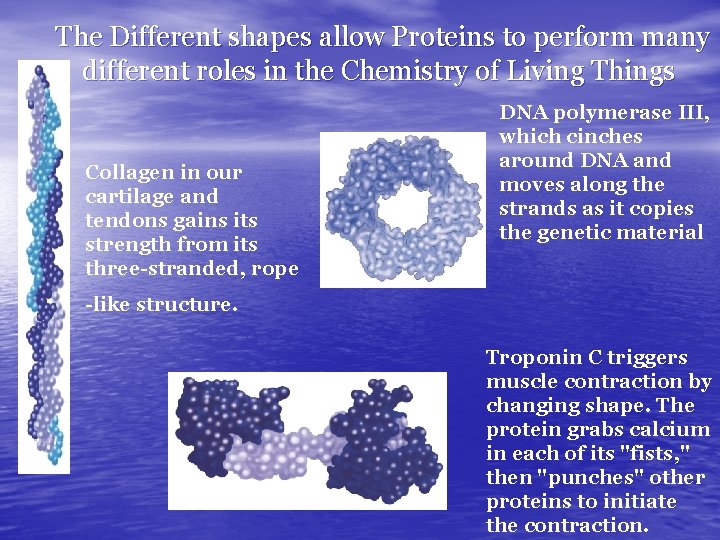
The Different shapes allow Proteins to perform many different roles in the Chemistry of Living Things Collagen in our cartilage and tendons gains its strength from its three-stranded, rope -like structure. DNA polymerase III, which cinches around DNA and moves along the strands as it copies the genetic material Troponin C triggers muscle contraction by changing shape. The protein grabs calcium in each of its "fists, " then "punches" other proteins to initiate the contraction.
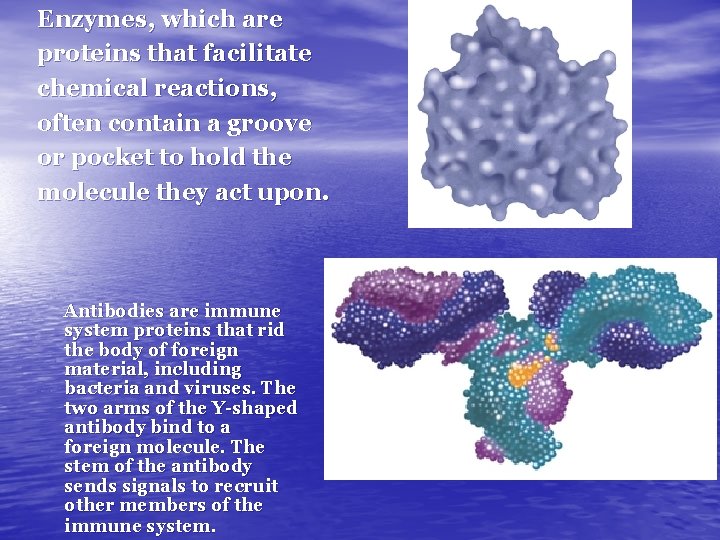
Enzymes, which are proteins that facilitate chemical reactions, often contain a groove or pocket to hold the molecule they act upon. Antibodies are immune system proteins that rid the body of foreign material, including bacteria and viruses. The two arms of the Y-shaped antibody bind to a foreign molecule. The stem of the antibody sends signals to recruit other members of the immune system.

The Problem of Protein Folding A given sequence of amino acids almost always folds into a characteristic, threedimensional structure. So scientists reason that the instructions for folding a protein must be encoded within the sequence. Researchers can easily determine a protein's amino acid sequence. But for 50 years they've tried— and failed —to crack the code that governs folding. • • Scientists call this the "protein folding problem, " and it remains one of the great challenges in structural biology. Although researchers have teased out some general rules and, in some cases, can make rough guesses of a protein's shape, they cannot accurately and reliably predict a final structure from an amino acid sequence. The medical incentives for cracking the folding code are great. Several diseases —including Alzheimer's, cystic fibrosis, and "mad cow" disease— are thought to result from misfolded proteins. Many scientists believe that if we could decipher the structures of proteins from their sequences, we could improve the treatment of these diseases. "If we could decipher the structures of proteins from their sequences, we could better understand all sorts of biological phenomena, from cancer to AIDS. Then we might be able to do more about these disorders. " James Cassatt Director, Division of Cell Biology and Biophysics National Institute of General Medical Sciences

ENZYMES • proteins • help control Chemical Reactions by acting • • as CATALYSTS, essential for the functioning of cells catalysts can speed up some reactions by more than a billion fold

How do enzymes work? • Enzyme Molecule and its SUBSTRATE, the • • reactant being catalyzed The fit of enzymes on a substrate weakens some chemical bonds, which reduces the activation energy for the chemical reaction to occur then the enzyme is released & it’s unchanged, so it can be used many times
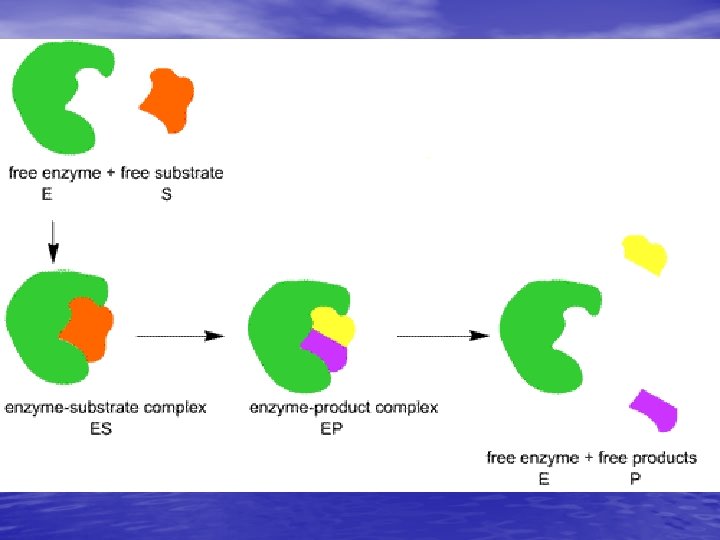

Lipids • Monomer— • • triglyceride/3 fatty acids + glycerol CHO High-energy; long-term storage!! • Fats • Saturated: single bonds between carbons (mostly animal fats) • Unsaturated: double or triple bonds between carbons (many plant fats)

The Good and the Bad? ? ?
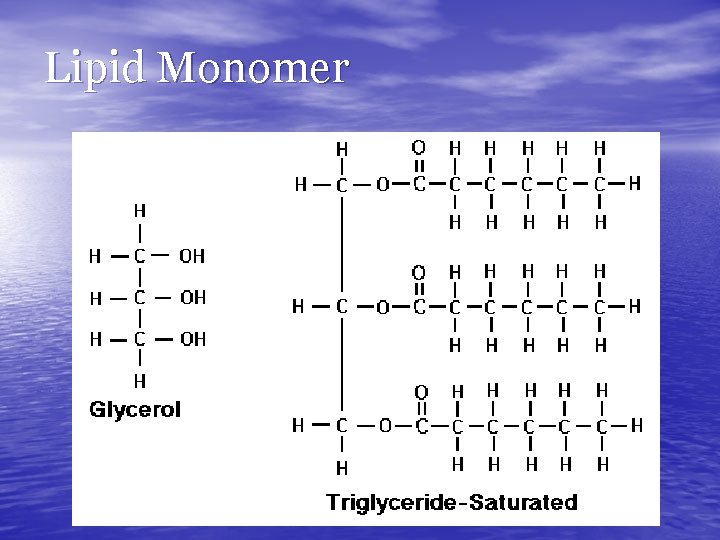
Lipid Monomer
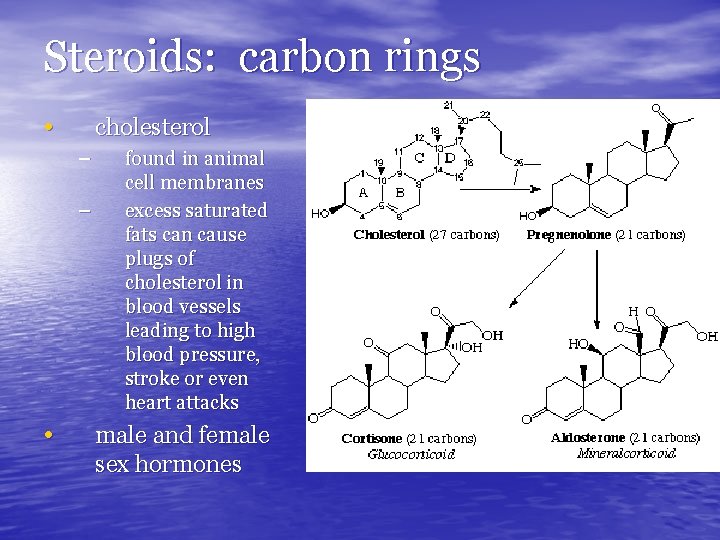
Steroids: carbon rings • cholesterol – – • found in animal cell membranes excess saturated fats can cause plugs of cholesterol in blood vessels leading to high blood pressure, stroke or even heart attacks male and female sex hormones

Anabolic steroids – used to “bulk up” – dangerous • liver damage • heart disease • psychological disorders • cancer • infertility (difficulty having children)

Other Important Lipids • • • rubber wax pigments (chlorophyll)
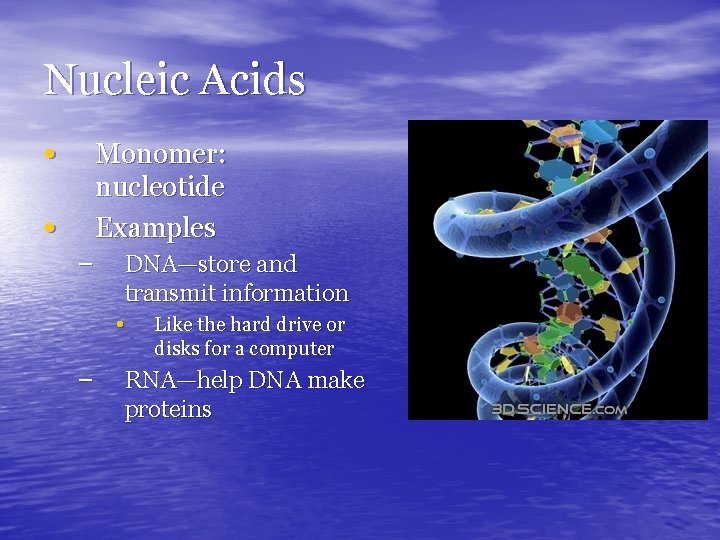
Nucleic Acids • Monomer: nucleotide Examples • – DNA—store and transmit information • Like the hard drive or disks for a computer – RNA—help DNA make proteins

DNA is a long chain of nucleotides • The order on the DNA specifies the • amino acids sequence The amino acids sequence folds into a certain protein
- Slides: 43