Introduction II Thermodynamics I Andy Howard Biochemistry Lectures

Introduction II; Thermodynamics I Andy Howard Biochemistry Lectures, Spring 2019 17 January 2019, Illinois Tech

Water, catalysis, math, thermo l All of the above are critical to biochemistry; we’ll focus on those today 01/17/2019 Intro 2; Thermo 1 p. 2 of 89

Plans l l l Water Catalysis Energy Regulation Molecular Biology Mathematics 01/17/2019 l Thermodynamics l Laws l Realities l Enthalpy and entropy l Free energy l Le Chatelier’s principle l Protein folding Intro 2; Thermo 1 p. 3 of 89

Water: a complex substance (CF&M Chapter 2) Oxygen atom is covalently bonded to 2 hydrogens l Single bond character of these bonds means the H-O-H bond angle is close to 109. 5º = acos(-1/3): actually more like 104. 5º l This contrasts with O=C=O (angle=180º) or urea ((NH 2)2 -C=O) (angles=120º) l Two lone pairs available per oxygen: these are available as H-bond acceptors l 01/17/2019 Intro 2; Thermo 1 p. 4 of 89

Water is polar (CF&M § 2. 1) Charge is somewhat unequally shared l Small positive charge on H’s (d+); small negative charge on O (2 d-) (Why? ) l A water molecule will orient itself to align partial negative charge on one molecule close to partial positive charges on another. l Hydrogen bonds are involved in this. l 01/17/2019 Intro 2; Thermo 1 p. 5 of 89

Liquid water is mobile The hydrogen-bond networks created among water molecules change constantly on a sub-picosecond time scale l At any moment the H-bonds look like those in crystalline ice l Solutes disrupt the H-bond networks l 01/17/2019 Intro 2; Thermo 1 p. 6 of 89

Water is a good solvent l Polar molecules and ions are soluble in water l Non-polar molecules aren’t l Most, but not all, biologically important molecules are polar 01/17/2019 Intro 2; Thermo 1 p. 7 of 89

Water in reactions Water is a medium wherein reactions occur; l But it also participates in reactions. l Enzymes often make water oxygen atoms better nucleophiles or water H’s better electrophiles l So water can participate in reactions that wouldn’t work without enzymes! l 01/17/2019 Intro 2; Thermo 1 p. 8 of 89

Water’s physical properties High heat capacity: stabilizes temperature in living things l High surface tension l Nearly incompressible (density almost independent of pressure) l Density max at 3. 98ºC l 01/17/2019 Intro 2; Thermo 1 p. 9 of 89

Catalysis is the lowering of activation energy barrier between reactants & products l How? l – Physical surface on which reactants can be exposed to one another – Providing moieties that can temporarily participate in the reaction and be restored to their original state at the end 01/17/2019 Intro 2; Thermo 1 p. 10 of 89

Biological catalysts I 1890’s: Emil Fischer realized that there had to be catalysts in biological systems l 1920’s: James Sumner said they were proteins l It took another 10 years for the whole community to accept that l 01/17/2019 Intro 2; Thermo 1 p. 11 of 89

Biological catalysts II l It’s now known that RNA can be catalytic too: – Can catalyze modifications in itself – Catalyzes the key step in protein synthesis in the ribosome l But we’ll mostly discuss proteinaceous enzymes in this course 01/17/2019 Intro 2; Thermo 1 p. 12 of 89

Energy in biological systems We distinguish between thermodynamics and kinetics: l Thermodynamics characterizes the energy associated with equilibrium conditions in reactions l Kinetics describes the rate at which a reaction moves toward equilibrium l 01/17/2019 Intro 2; Thermo 1 p. 13 of 89

Thermodynamics Equilibrium constant is a measure of the ratio of product concentrations to reactant concentrations at equilibrium l Free energy is a measure of the available energy in the products and reactants l They’re related by Go = -RT ln Keq l 01/17/2019 Intro 2; Thermo 1 p. 14 of 89

Kinetics Rate of reaction is dependent on Kelvin Svante Arrhenius temperature T and on activation barrier G‡ preventing conversion from one site to the other ‡/RT ‡ - G l Rate = Qexp(- G /RT) = Qe l Job of an enzyme is to reduce G‡ l 01/17/2019 Intro 2; Thermo 1 p. 15 of 89
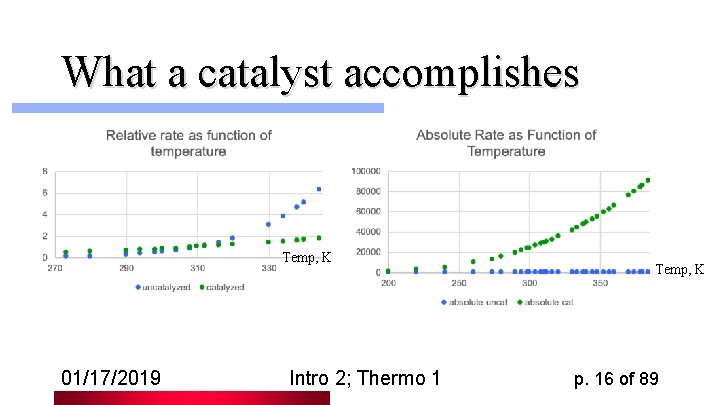
What a catalyst accomplishes Temp, K 01/17/2019 Intro 2; Thermo 1 Temp, K p. 16 of 89

Regulation Biological reactions are regulated in the sense that they’re catalyzed by enzymes, so the presence or absence of the enzyme determines whether the reaction will proceed l The enzymes themselves are subject to extensive regulation so that the right reactions occur in the right places and times l 01/17/2019 Intro 2; Thermo 1 p. 17 of 89

Typical enzymatic regulation l Suppose enzymes are involved in converting A to B, B to C, C to D, and D to F. E is the enzyme that converts A to B: (E) A B C D F 01/17/2019 Intro 2; Thermo 1 p. 18 of 89

Regulation, continued In many instances F will inhibit (interfere) with the reaction that converts A to B by binding to a site on enzyme E so that it can’t convert A to B as readily. l This feedback inhibition helps to prevent overproduction of F—homeostasis. l 01/17/2019 Intro 2; Thermo 1 p. 19 of 89

Molecular biology This phrase means something much more specific than biochemistry: l It’s the chemistry of replication, transcription, and translation, i. e. , the ways that genes are reproduced and expressed. l Many of you have taken biology 214 or 515 or their equivalents; we’ll review some of the contents of those courses here, mostly near the middle of the semester. l 01/17/2019 Intro 2; Thermo 1 p. 20 of 89

Molecules of molecular biology Deoxyribonucleic acid: polymer; backbone is deoxyribose-phosphate; side chains are nitrogenous ring compounds l RNA: polymer; backbone is ribose-phosphate; side chains as above l Protein: polymer: backbone is NH-(CHR)-CO; side chains (R-groups) are 20 ribosomally encoded styles l 01/17/2019 Intro 2; Thermo 1 p. 21 of 89

Steps in molecular biology: the Central Dogma DNA replication (makes accurate copy of double-stranded DNA prior to mitosis) l Transcription (RNA version of DNA message is created) l Translation (m. RNA copy of gene serves as template for making protein: 3 bases of RNA per amino acid of synthesized protein) l 01/17/2019 Intro 2; Thermo 1 p. 22 of 89

How viruses can be so simple Viruses co-opt the replicative systems of their hosts, so they don’t need to carry around all the tools of their own synthesis l Coding can be by RNA or DNA; RNA viruses code for a reverse transcriptase to make DNA from their RNA l 01/17/2019 Intro 2; Thermo 1 p. 23 of 89

Evolution and Taxonomy Traditional studies of interrelatedness of organisms focused on functional similarities l Enables production of phylogenetic trees l Molecular biology provides an alternative, possibly more quantitative, approach to phylogenetic tree-building l More rigorous hypothesis-testing possible l 01/17/2019 Intro 2; Thermo 1 p. 24 of 89

Quantitation • Biochemistry is a quantitative science. • Results in biochemistry are significant only if they can be couched in quantifiable terms. • Thermodynamic & kinetic behavior of systems must be described quantitatively. • Even descriptive biochemistry, e. g. compartmentalization of reactions & metabolites into cells and organelles, must be characterized numerically. 01/17/2019 Intro 2; Thermo 1 p. 25 of 89

Mathematics in biochemistry Ooo: I went into biology rather than physics because I don’t like math l Too bad. You need some math here: but not much. l Biggest problem in past years: exponentials and logarithms l 01/17/2019 Intro 2; Thermo 1 p. 26 of 89

Exponentials Many important biochemical equations are expressed in the form Y = ef(x) l … which can also be written Y = exp(f(x)) l The number e is the base of the natural logarithm system and is, very roughly, 2. 71828459045 l That is, it’s 2. 7 1828 45 90 45 l 01/17/2019 Intro 2; Thermo 1 p. 27 of 89

Algebra of exponentials l l l Recognize that (e. A)(e. B) = e(A+B), or exp(A) exp(B) = exp(A+B) Similarly e. A/e. B = e(A-B) This becomes particularly useful when calculating ratios of similar quantities: Arrhenius relationship says k = Qe- G‡/RT Therefore: ratio of k values at two different temperatures is k 1/k 2 = e( G‡/R)(1/T 2 -1/T 1) 01/17/2019 Intro 2; Thermo 1 p. 28 of 89

Logarithms First developed as computational tools: they convert multiplication problems into addition problems John Napier l They have a fundamental connection with raising a value to a power: l Y = xa logx(Y) = a l Thus Y = exp(a) = ea ln. Y = loge(Y) = a l 01/17/2019 Intro 2; Thermo 1 p. 29 of 89

Algebra of logarithms logv(A) = logu(A) / logu(v) l logu(A/B) = logu(A) - logu(B) l logu(AB) = Blogu(A) l log 10(A) = ln(A) / ln(10) = ln(A) / 2. 3026 = 0. 4343 * ln(A) l ln(A) = log 10(A) / log 10 e = log 10(A) / 0. 4343 = 2. 3026 * log 10(A) l 01/17/2019 Intro 2; Thermo 1 p. 30 of 89

first: i. Clicker quiz, question 1! • 1. Which of the following statements is true? – (a) All enzymes are proteins. – (b) All proteins are enzymes. – (c) All viruses use RNA as their transmittable genetic material. – (d) None of the above. 01/11/2017 Thermodynamics p. 31 of 83

i. Clicker quiz, question 2 l 2. Biopolymers are generally produced in reactions in which building blocks are added head to tail. Apart from the polymer, what is the most common product of these reactions? (a) Water (b) Ammonia (c) Carbon Dioxide (d) Glucose (e) None of the above. Polymerization doesn’t produce secondary products 01/11/2017 Thermodynamics p. 32 of 83

i. Clicker quiz, question 3 l 3. Which type of biopolymer is sometimes branched? (a) DNA (b) Protein (c) Polysaccharide (d) RNA (e) They’re all branched. 01/11/2017 Thermodynamics p. 33 of 83
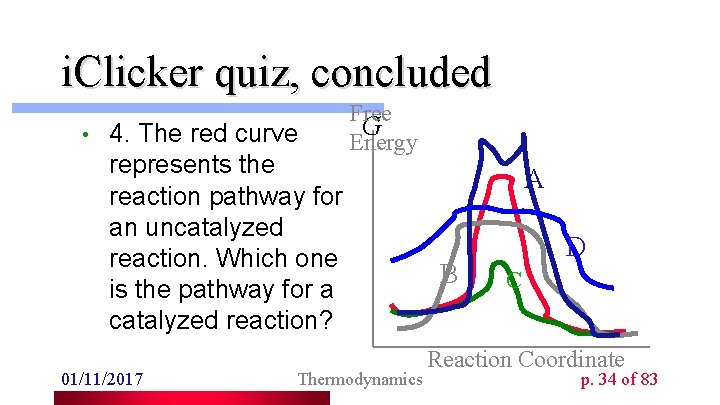
i. Clicker quiz, concluded • 4. The red curve represents the reaction pathway for an uncatalyzed reaction. Which one is the pathway for a catalyzed reaction? 01/11/2017 Free G Energy Thermodynamics A B D C Reaction Coordinate p. 34 of 83

Thermodynamics! CF&M § 1. 6 • Moran et al. (textbook we used previously in this course) put this subject in the middle of chapter 10; Other textbooks, including ours, are smart enough to put it in the beginning. • You can tell which I prefer! • 01/17/2019 Intro 2; Thermo 1 p. 35 of 89

Why we care G Reaction Coordinate Free energy is directly related to the equilibrium of a reaction l It doesn’t tell us how fast the system will come to equilibrium l Kinetics, and the way that enzymes influence kinetics, tell us about rates l Today we’ll focus on equilibrium energetics; we’ll call that thermodynamics l 01/17/2019 Intro 2; Thermo 1 p. 36 of 89

Laws of Thermodynamics Traditionally four (0, 1, 2, 3) • Can be articulated in various ways • First law: The energy of an isolated system is constant. • Second law: Entropy of an isolated system increases. • 01/17/2019 Intro 2; Thermo 1 p. 37 of 89

What do we mean by systems, closed, open, and isolated? A system is the portion of the universe with which we’re concerned (e. g. , an organism or a rock or an ecosystem) l If it doesn’t exchange energy or matter with the outside, it’s isolated. l If it exchanges energy but not matter, it’s closed l If it exchanges energy & matter, it’s open l 01/17/2019 Intro 2; Thermo 1 p. 38 of 89

That makes sense if… l l l 01/17/2019 Boltzmann Gibbs It makes sense provided that we understand the words! Energy. Hmm. Capacity to do work. Entropy: Disorder. (Boltzmann): S = k. Bln Isolated system: one in which energy and matter don’t enter or leave An organism is not an isolated system: so S can decrease within an organism! Intro 2; Thermo 1 p. 39 of 89

Thermodynamic properties Extensive properties: Thermodynamic properties that are directly related to the amount (e. g. mass, or # moles) of stuff present (e. g. E, H, S) • Intensive properties: not directly related to mass (e. g. P, T) • If you divide an extensive property by mass or # of moles, you get an intensive property • 01/17/2019 Intro 2; Thermo 1 p. 40 of 89

Units Energy unit: Joule (kg m 2 s-2) • 1 k. J/mol = 103 J/(6. 022*1023) = 1. 661*10 -21 J • 1 cal = 4. 184 J: so 1 kcal/mol = 6. 948 *10 -21 J • 1 e. V = 1 e * J/Coulomb = 1. 602*10 -19 C * 1 J/C = 1. 602*10 -19 J = 96. 4 k. J/mol = 23. 1 kcal/mol • 01/17/2019 Intro 2; Thermo 1 James Prescott Joule p. 41 of 89

Typical energies in biochemistry • Go for hydrolysis of high-energy phosphate bond in adenosine triphosphate: 33 k. J/mol = 7. 9 kcal/mol = 0. 34 e. V • Hydrogen bond: 4 k. J/mol=1 kcal/mol • van der Waals force: ~ 1 k. J/mol • See textbook for others 01/17/2019 Intro 2; Thermo 1 p. 42 of 89

Enthalpy, H Closely related to energy: H = E + PV • Therefore changes in H are: Kamerlingh Onnes H = E + P V + V P • Most, but not all, biochemical systems have constant V, P: H = E • Related to the heat content in a system • 01/17/2019 Intro 2; Thermo 1 p. 43 of 89

Entropy, S • • • 01/17/2019 Related to disorder: Boltzmann: S = k ln k. B=Boltzmann constant = 1. 38*10 -23 J K-1 Note that k. B = R / N 0 is # of degrees of freedom in the system Entropy in 1 mole = N 0 S = Rln Number of degrees of freedom can be calculated for simple molecules Intro 2; Thermo 1 p. 44 of 89

Components of entropy Liquid propane (as surrogate): Type of Entropy Translational Rotational Vibrational Electronic Total 01/17/2019 k. J (mol K)-1 36. 04 23. 38 1. 05 0 60. 47 Intro 2; Thermo 1 p. 45 of 89

Real biomolecules l l l 01/17/2019 Entropy is mostly translational & rotational Enthalpy is mostly electronic Translational entropy = (3/2) R ln Mr So when a molecule dimerizes, the total translational entropy decreases (there’s half as many molecules, but ln Mr only goes up by ln 2) Rigidity decreases entropy Intro 2; Thermo 1 p. 46 of 89

Entropy in solvation: solute l When molecules go into solution, their entropy increases because they’re freer to move around 01/17/2019 Intro 2; Thermo 1 p. 47 of 89

Entropy in solvation: Solvent entropy usually decreases because solvent molecules must become more ordered around solute • Overall effect: typically slightly tending toward solution • 01/17/2019 Intro 2; Thermo 1 p. 48 of 89

Entropy matters a lot! Most biochemical reactions involve small ( < 10 k. J/mol) changes in enthalpy • Driving force is often entropic • Increases in solute entropy often is at war with decreases in solvent entropy. • The winner tends to take the prize. • 01/17/2019 Intro 2; Thermo 1 p. 49 of 89

Apolar molecules in water Water molecules tend to form ordered structure surrounding apolar molecule l Entropy decreases because they’re so ordered l 01/17/2019 Intro 2; Thermo 1 p. 50 of 89

Binding to surfaces Happens a lot in biology, e. g. binding of small molecules to relatively immobile protein surfaces l Bound molecules suffer a decrease in entropy because they’re trapped l Solvent molecules are displaced and liberated from the protein surface l 01/17/2019 Intro 2; Thermo 1 p. 51 of 89

Free Energy Gibbs: Free Energy Equation Josiah Willard Gibbs G = H - TS • So if isothermal, G = H - T S • Gibbs showed that a reaction will be spontaneous (proceed to right) if and only if G < 0 • 01/17/2019 Intro 2; Thermo 1 p. 52 of 89

Standard free energy l 01/17/2019 We often want to know what the energetic differences are between reactants and products based on their inherent properties, rather than on the specific conditions of an experiment. We then define a standard free energy difference, ΔGo or ΔGo’, for the reaction Intro 2; Thermo 1 p. 53 of 89

Definition of ΔGo l 01/17/2019 For a reaction involving various reactants and products, we envision measuring the free energy differences if the initial concentrations of all reactants and products is 1 M. That measurement becomes the standard free energy ΔGo for the reaction. Intro 2; Thermo 1 p. 54 of 89

Special case: water Note that pure water has a density of about 1 g/m. L = 1000 g /L l It also has a molecular weight of 18 g/mole l Therefore the molarity of pure water is (1000 g/L)/(18 g/mole) = 55. 5 moles/L = 55. 5 M l This is a useful number by itself, but also… l 01/17/2019 Intro 2; Thermo 1 p. 55 of 89

Water as a reactant or product Often water does appear as a reactant or product in a reaction. l By convention: water concentration is taken to be 55. 5 M, not 1 M, in our definition of ΔGo. l In practice that’s a good assumption: even if solutes are present, the concentration of water will be close to 55. 5 M. l 01/17/2019 Intro 2; Thermo 1 p. 56 of 89

Definition for ΔGo’ Sometimes we want to be even more specific: we’ll envision measuring ΔGo at p. H 7 & 25ºC = 298. 16 K. l That measurement of ΔGo is called ΔGo’ l We’ll often be casual about the difference between ΔGo and ΔGo’ in this class. l But we will distinguish between ΔG and ΔGo! l 01/17/2019 Intro 2; Thermo 1 p. 57 of 89

Standard free energy of formation, Gof l Difference between compound’s free energy & sum of free energies of the elements from which it is made Substance Gof, k. J/mol Lactate -516 Pyruvate -474 Succinate -690 Glycerol -488 Acetate -369 Oxaloacetate -797 HCO 3 - -394 01/17/2019 Intro 2; Thermo 1 p. 58 of 89

Free energy and equilibrium Go = -RT ln keq, or keq = exp(- Go/RT) l keq is equilibrium constant; formula depends on reaction type l For a. A + b. B c. C + d. D, keq = ([C]c[D]d)/([A]a[B]b) l If all proportions are equal, keq = ([C][D])/([A][B]) l These values ([C], [D] …) denote the concentrations at equilibrium l 01/17/2019 Intro 2; Thermo 1 p. 59 of 89

Spontaneity and free energy Thus if reaction is just spontaneous, i. e. Go = 0, then keq = 1 l If Go < 0, then keq > 1: Exergonic l If Go > 0, then keq < 1: Endergonic l Distinguishable from exothermic and endothermic, which are concerned only with enthalpy, not the whole free-energy package. l 01/17/2019 Intro 2; Thermo 1 p. 60 of 89

Free energy as a source of work Change in free energy indicates that the reaction could be used to perform useful work • If Go < 0, we can do work • If Go > 0, we need to do work to make the reaction occur • 01/17/2019 Intro 2; Thermo 1 p. 61 of 89

What kind of work? Movement (flagella, muscles) • Chemical work: • – Transport molecules against concentration gradients – Transport ions against potential gradients • Driving otherwise endergonic reactions – by direct coupling of reactions – by depletion of products 01/17/2019 Intro 2; Thermo 1 p. 62 of 89

Coupled reactions • • • 01/17/2019 Often a single enzyme catalyzes 2 reactions, shoving them together: reaction 1, A B: Go 1 < 0 reaction 2, C D: Go 2 > 0 Coupled reaction: A + C B + D: Go. C = Go 1 + Go 2 If Go. C < 0, reaction 1 is driving reaction 2! Intro 2; Thermo 1 p. 63 of 89

How else can we win? Concentration of products & reactants may play a role l As we’ll discuss in a moment, the actual free energy depends on Go and on concentration of products and reactants l So if the first reaction withdraws product of reaction B away, that drives the equilibrium of reaction 2 to the right l 01/17/2019 Intro 2; Thermo 1 p. 64 of 89

Le Chatelier’s Principle & ΔGo Le Chatelier’s Principle says that a reaction can be driven to the right if one of the products is being removed from availability l This can happen in various ways: l – A gaseous product can evaporate – An insoluble product can precipitate – A reactive product can get converted to something else. 01/17/2019 Intro 2; Thermo 1 p. 65 of 89

Quantitation of Le Chatelier’s Principle Relationship between G (actual free energy experienced in a reaction under real conditions) & standard free energy Go : • G = Go + RT ln([products]/[reactants]) • So for a typical bimolecular reaction A + B C + D, • G = Go + RT ln{[C][D]/([A][B])} • 01/17/2019 Intro 2; Thermo 1 p. 66 of 89

Why does this embody Le Chatelier’s Principle? Suppose product D is being taken out of the reaction vessel (e. g. the mitochondrion) either by being converted to something else or by being transported away. • Then [D] will be small, so Q=[C][D]/([A][B]) will be small • 01/17/2019 Intro 2; Thermo 1 p. 67 of 89

Consequence of that! l Therefore ln. Q will be negative, and G = Go + RT ln. Q can become negative even if Go is positive (especially at high T) 01/17/2019 Intro 2; Thermo 1 p. 68 of 89

How hard is it to o measure G ? Not as bad as you might think. l It’s true that it’s hard to set up [C]=[D]=[A]=[B] = 1 M; but we don’t actually have to do that. l l 01/17/2019 Note that since G = Go +RTln{[C][D]/([A][B])}, we can get to G = Go simply by ensuring that [C][D]/([A][B]) = 1, so that ln{[C][D]/([A][B]) = 0. Intro 2; Thermo 1 p. 69 of 89

Adenosine Triphosphate ATP readily available in cells • Derived from catabolic reactions • Contains two high-energy phosphate bonds that can be hydrolyzed to release energy: • 01/17/2019 O O|| | (AMP)-O~P-O| || Intro 2; Thermo 1 O- O p. 70 of 89

Hydrolysis of ATP • • 01/17/2019 Hydrolysis at the rightmost high-energy bond: ATP + H 2 O ADP + Pi , Go = -33 k. J/mol Hydrolysis of middle bond: ATP + H 2 O AMP + PPi Go ~ -40 k. J/mol BUT PPi + H 2 O 2 Pi, Go = -31 k. J/mol So, appropriately coupled, we get roughly twice as much! Intro 2; Thermo 1 p. 71 of 89

ATP as energy currency Any time we wish to drive a reaction that has Go < +30 k. J/mol, we can couple it to ATP hydrolysis and come out ahead l If the reaction we want has Go < +60 k. J/mol, we can couple it to ATP AMP and come out ahead l So ATP is a convenient source of energy — an energy currency for the cell l 01/17/2019 Intro 2; Thermo 1 p. 72 of 89

Coin analogy Think of store of ATP as a roll of quarters l Vendors don’t give change l Use one quarter for some reactions, two for others l Inefficient for buying $0. 35 items l 01/17/2019 Intro 2; Thermo 1 p. 73 of 89

Other high-energy compounds l Creatine phosphate: ~ $0. 35 l Phosphoenolpyruvate: ~ $0. 40 l So for some reactions, they’re more efficient than ATP 01/17/2019 Intro 2; Thermo 1 p. 74 of 89

Why not use those always? There’s no such thing as a free lunch! l In order to store a compound, you have to create it in the first place l So an intermediate-energy currency is the most appropriate l 01/17/2019 Intro 2; Thermo 1 p. 75 of 89
![Le Chatelier’s principle in ATP-dependent reactions G = Go + RT ln{[C][D]/([A][B]) • In Le Chatelier’s principle in ATP-dependent reactions G = Go + RT ln{[C][D]/([A][B]) • In](http://slidetodoc.com/presentation_image_h/cacc4d08eddc7d1c8aea2ebf281539e7/image-76.jpg)
Le Chatelier’s principle in ATP-dependent reactions G = Go + RT ln{[C][D]/([A][B]) • In an ATP-coupled reaction, D=ADP, B=ATP; C and A are the other reactants • But note that G = Go + RT ln{[C][D]/([A][B]) = Go + RT { ln([C]/[A])+ ln([ADP]/[ATP])} • 01/17/2019 Intro 2; Thermo 1 p. 76 of 89
![Why that matters in cells… l 01/17/2019 The fact is that often [ADP]/[ATP] ~ Why that matters in cells… l 01/17/2019 The fact is that often [ADP]/[ATP] ~](http://slidetodoc.com/presentation_image_h/cacc4d08eddc7d1c8aea2ebf281539e7/image-77.jpg)
Why that matters in cells… l 01/17/2019 The fact is that often [ADP]/[ATP] ~ 0. 1, so at T=300. 6 K, RTln[ADP]/[ATP] = -5. 8 k. J /mol, so that provides even more available energy to drive the reaction! Intro 2; Thermo 1 p. 77 of 89

How else can Le Chatelier’s principle help? Often coupled reactions involve withdrawal of a product from availability • If that happens, [product] / [reactant] shrinks, the second term becomes negative, and G < 0 even if Go > 0 • 01/17/2019 Intro 2; Thermo 1 p. 78 of 89

Example: glycolysis Later this semester we’ll spend at least one lecture looking at glycolysis, one of the fundamental pathways l Some of the glycolytic reactions have Go’ or Go > 0 l But all have G values that are negative or zero because of this concentration effect l 01/17/2019 Intro 2; Thermo 1 p. 79 of 89

How to solve energy problems involving coupled equations l General principles: – If two equations are added, their energetics add – An item that appears on the left and right side of the combined equation can be cancelled – Reversing a reaction reverses the sign of G. l 01/17/2019 “Hydrolysis” means “reacting with water” Intro 2; Thermo 1 p. 80 of 89

A bit more detail Suppose we couple two equations: A + B C + D, Go’ = x C + F B + G, Go’ = y l The result is: A + B + C + F B + C + D + G, or A + F D + G, Go’ = x + y l … since B & C appear on both sides l 01/17/2019 Intro 2; Thermo 1 p. 81 of 89

Slightly more complex… Suppose we couple two equations: A + B C + D, Go’ = x H + A J + C, Go’ = z l Reverse the second equation: J + C A + H, Go’ = -z l Add this to 1 st eqn. & simplify: B + J D + H, Go’ = x - z l … since A & C appear on both sides l 01/17/2019 Intro 2; Thermo 1 p. 82 of 89

What do we mean by hydrolysis? It simply means a reaction with water • Typically involves cleaving a bond: • U + H 2 O V + W is described as hydrolysis of U to yield V and W • 01/17/2019 Intro 2; Thermo 1 p. 83 of 89

Protein Folding Proteins (about which we’ll say a lot soon) are typically folded into a definite conformation at room temp in solution l They unfold into a state where they have no definite conformation, at higher temperature l We describe this as melting, and it’s frequently a first-order phase transition l 01/17/2019 Intro 2; Thermo 1 p. 84 of 89

How does that work thermodynamically? Consider the reaction (Protein+Solvent)folded (Protein+Solvent)unfolded l This reaction has a free energy: G = H - T S l System includes both protein and solvent: G = Gprotein + Gsolvent = Hprotein + Hsolvent - T Sprotein - T Ssolvent l 01/17/2019 Intro 2; Thermo 1 p. 85 of 89

Unfolding: effect on enthalpy Typically Hprotein > 0 because we lose some hydrogen bonds and van der Waals interactions when the protein unfolds • Typically Hsolvent ~ 0 because we make extra protein-solvent H-bonds and break some protein-protein H-bonds • 01/17/2019 Intro 2; Thermo 1 p. 86 of 89

Unfolding: entropy Sprotein > 0 because the protein becomes more disordered • Therefore -T Sprotein < 0. • Ssolvent < 0 because some water molecules become aggregated around the protein; therefore -T Ssolvent > 0. • 01/17/2019 Intro 2; Thermo 1 p. 87 of 89

Putting the entropic terms together Overall entropic term in free energy is -T( Sprotein + Ssolvent) • These 2 terms have opposite signs but typically the protein term dominates, just barely • Therefore the overall term is slightly negative, and it becomes more negative at higher temperature • 01/17/2019 Intro 2; Thermo 1 p. 88 of 89
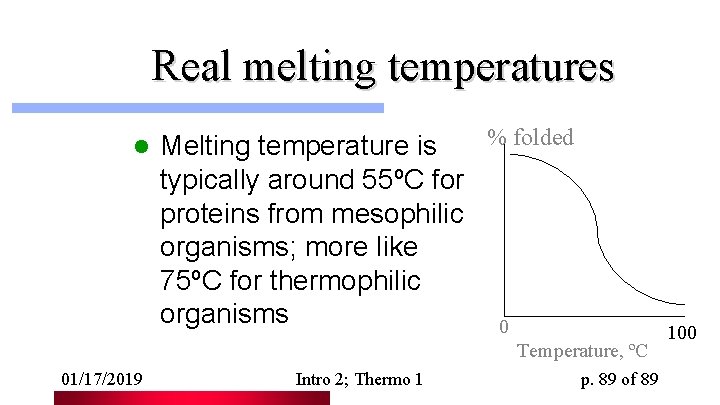
Real melting temperatures l % folded Melting temperature is typically around 55ºC for proteins from mesophilic organisms; more like 75ºC for thermophilic organisms 0 Temperature, ºC 01/17/2019 Intro 2; Thermo 1 p. 89 of 89 100
- Slides: 89