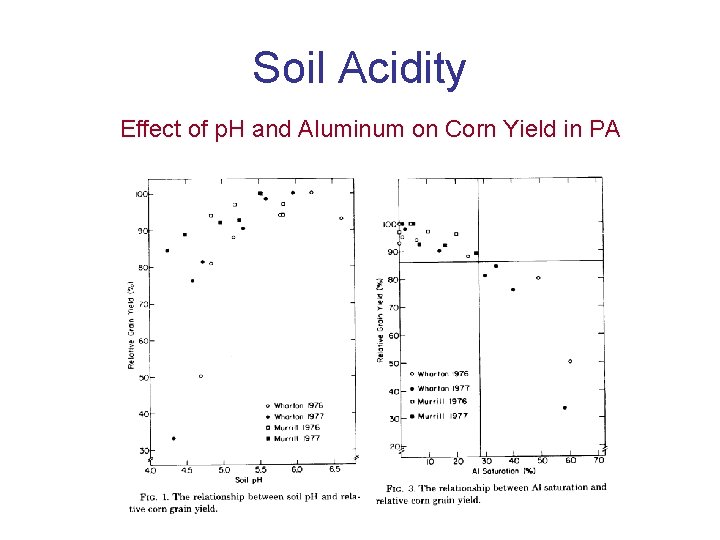
Soil Acidity Effect of p H and Aluminum






































- Slides: 57

Soil Acidity Effect of p. H and Aluminum on Corn Yield in PA

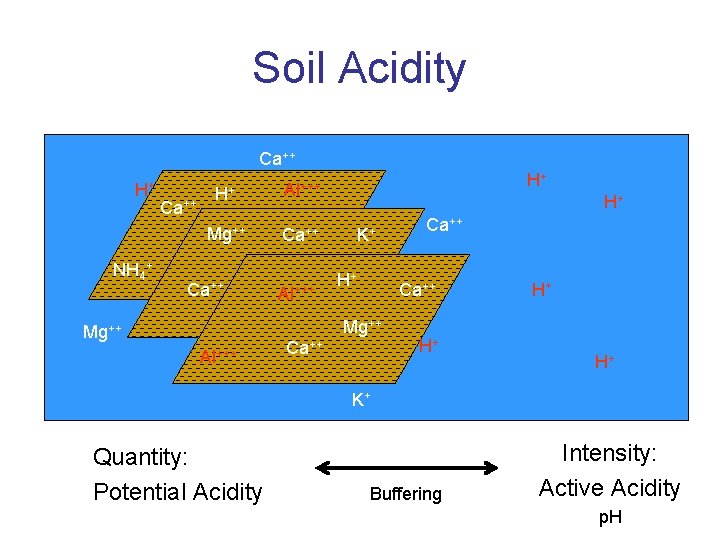
Soil Acidity • Active Acidity • Potential Acidity

Soil Acidity Ca++ H+ NH 4+ Ca++ H+ Al+++ Mg++ Ca++ Al+++ H+ H+ K+ H+ Ca++ Mg++ Al+++ Ca++ H+ H+ H+ K+ Quantity: Potential Acidity Buffering Intensity: Active Acidity p. H

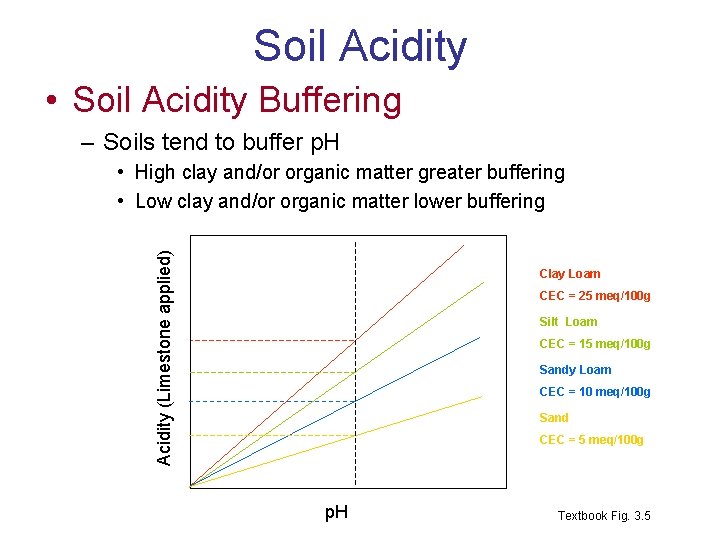
Soil Acidity • Soil Acidity Buffering – Soils tend to buffer p. H Acidity (Limestone applied) • High clay and/or organic matter greater buffering • Low clay and/or organic matter lower buffering Clay Loam CEC = 25 meq/100 g Silt Loam CEC = 15 meq/100 g Sandy Loam CEC = 10 meq/100 g Sand CEC = 5 meq/100 g p. H Textbook Fig. 3. 5

Soil Acidity and Liming

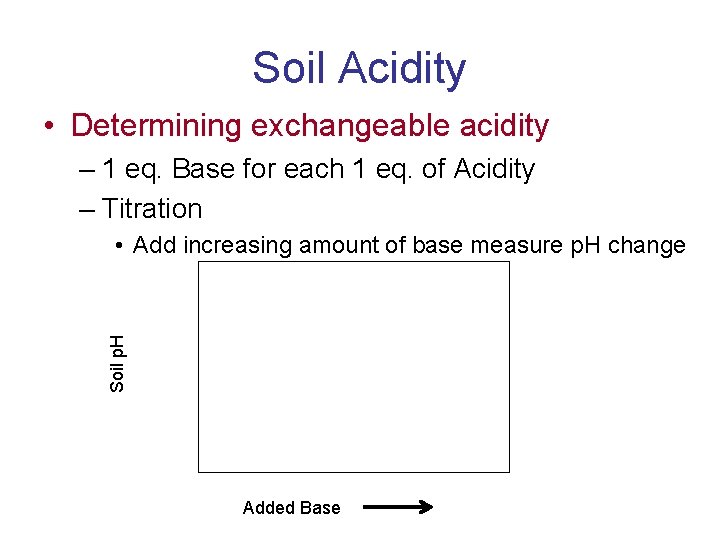
Soil Acidity • Determining exchangeable acidity – 1 eq. Base for each 1 eq. of Acidity – Titration Soil p. H • Add increasing amount of base measure p. H change Added Base

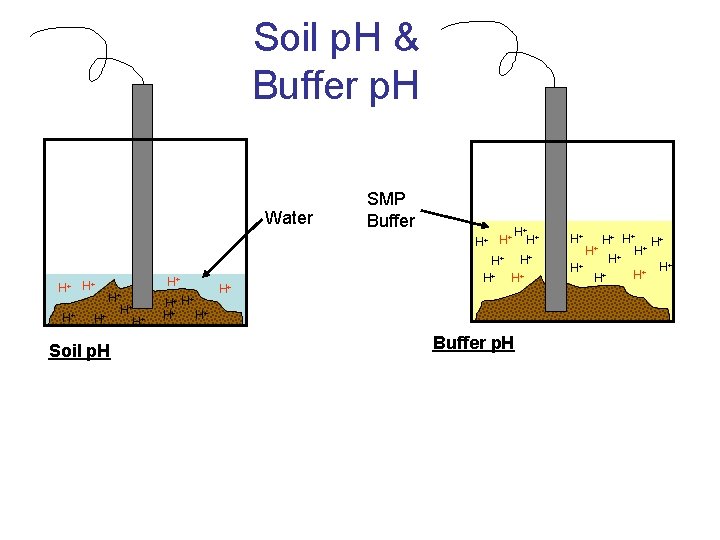
Soil Acidity • Determining exchangeable acidity – Buffer p. H • Add a p. H buffer solution and measure p. H change • Known relationship between p. H change and change in acidity • Common p. H buffers – SMP (used in PA) » Triethanolamine » Paranitrophenol » Potassium chromate » p. H 7. 5 – Adams-Evans – Mehlich – Woodruff

Soil Acidity NOTES • Determining exchangeable acidity – Buffer p. H • Add a p. H buffer solution and measure p. H change • Known relationship between p. H change and change in acidity • Common p. H buffers – SMP (used in PA) » Triethanolamine » Paranitrophenol » Potassium chromate » p. H 7. 5 – Adams-Evans – Mehlich - Many states including PA going to this – Woodruff

Soil p. H & Buffer p. H Water SMP Buffer H+ + H+ H+ + H H+ + H H H H+ H+ + + H H H+ H+ H+ Soil p. H H+ H+ H+ Buffer p. H

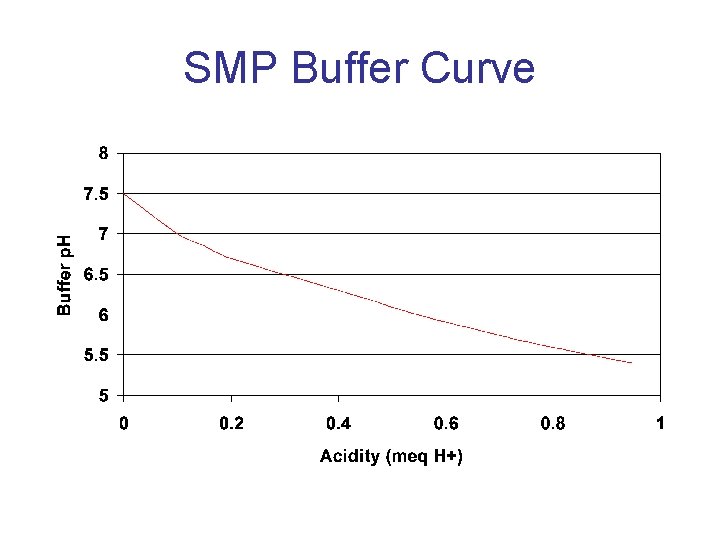
SMP Buffer Curve

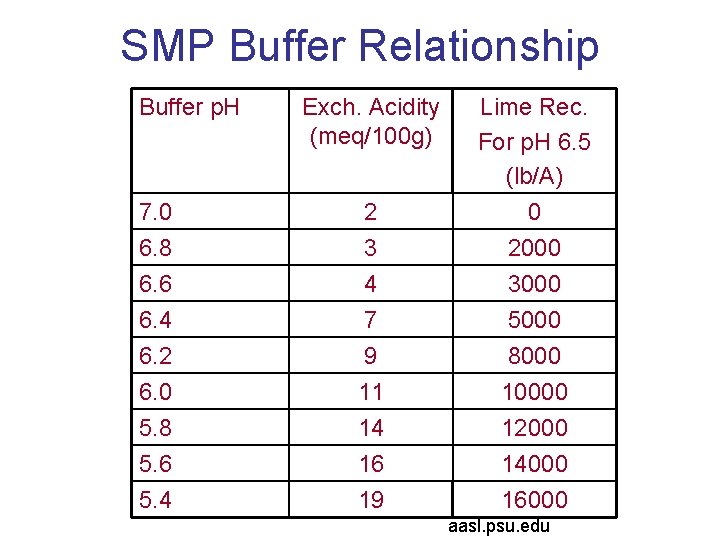
SMP Buffer Relationship Buffer p. H Exch. Acidity (meq/100 g) 7. 0 2 Lime Rec. For p. H 6. 5 (lb/A) 0 6. 8 6. 6 6. 4 6. 2 6. 0 5. 8 5. 6 5. 4 3 4 7 9 11 14 16 19 2000 3000 5000 8000 10000 12000 14000 16000 aasl. psu. edu

Soil Acidity and Liming • Determining Lime requirement – Exchangeable Acidity • 1 meq. Base/100 g for each 1 meq. of Acidity/100 g • How many pounds of lime do we need per acre?

Soil Acidity and Liming • Determining Lime requirement – Desired p. H • Acid sensitive crops p. H 7. 0 • Most crops and soils p. H 6. 5 • Highly weathered soils p. H 5. 5 • Acid loving crops 5. 5 - 6. 0

Soil Acidity and Liming • Determining Lime requirement – Based on target p. H and Exchangeable acidity Formulas used by PSU AASL • For p. H 7. 0 84500 – (12180 x SMP Buffer p. H) = Ca. CO 3/A • For p. H 6. 5 71240 – (10280 x SMP Buffer p. H) = Ca. CO 3/A • For p. H 6. 0 57840 – (8340 x SMP Buffer p. H) = Ca. CO 3/A

Soil Acidity and Liming • Determining Lime requirement – Rules of thumb • For p. H 7. 0 Exch. Acidity (meq/100 g) X 1000 = Ca. CO 3/A • For p. H 6. 5 Exch. Acidity (meq/100 g) X 840 = Ca. CO 3/A • For p. H 6. 0 Exch. Acidity (meq/100 g) X 750 = Ca. CO 3/A

Soil Acidity and Liming • Liming Materials – Material that will neutralize soil acidity • Calcium Oxide (Ca. O) – Lime, burnt lime, quick lime • Calcium Hydroxide (Ca(OH)2) – Hydrated lime, slaked lime • Calcium Carbonate (Ca. CO 3) – Calcitic limestone • Calcium/Magnesium Carbonate (Ca, Mg. CO 3) – Dolomitic limestone

Soil Acidity and Liming • For each equivalent of acidity we need to apply 1 equivalent of liming material • Chemical neutralizing ability of liming materials – Equivalents of OH- produced by the reaction of the liming material. • Ca(OH)2 → Ca 2+ + 2 OH • Ca. CO 3 + H 2 O → Ca 2+ + 2 OH- + CO 2 • Ca. O + H 2 O → Ca 2+ + 2 OH • Ca. Si. O 3 + H 2 O → Ca 2+ + HSi. O 3 - + OH • Ca. SO 4 → Ca 2+ + SO 42 -

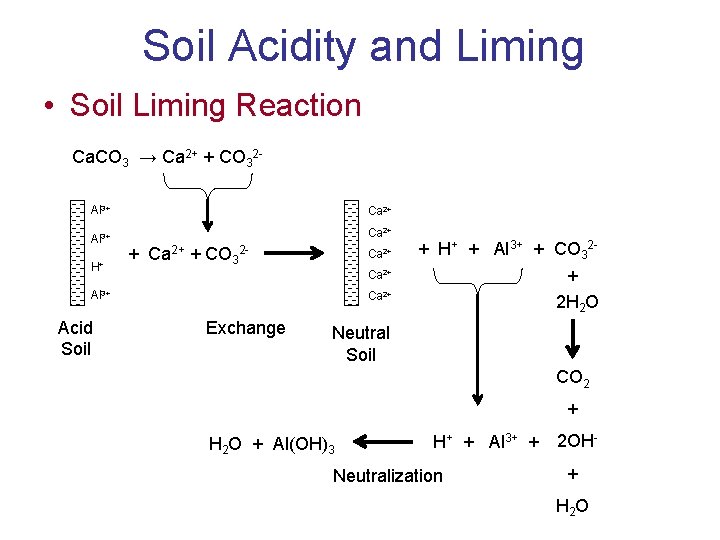
Soil Acidity and Liming • Soil Liming Reaction Ca. CO 3 → Ca 2+ + CO 32 Al 3+ Ca 2+ H+ + Ca 2+ + CO 32 - Ca 2+ + 2 H 2 O Ca 2+ Al 3+ Acid Soil + H+ + Al 3+ + CO 32 - Ca 2+ Exchange Neutral Soil CO 2 + H 2 O + Al(OH)3 H+ + Al 3+ + Neutralization 2 OH+ H 2 O

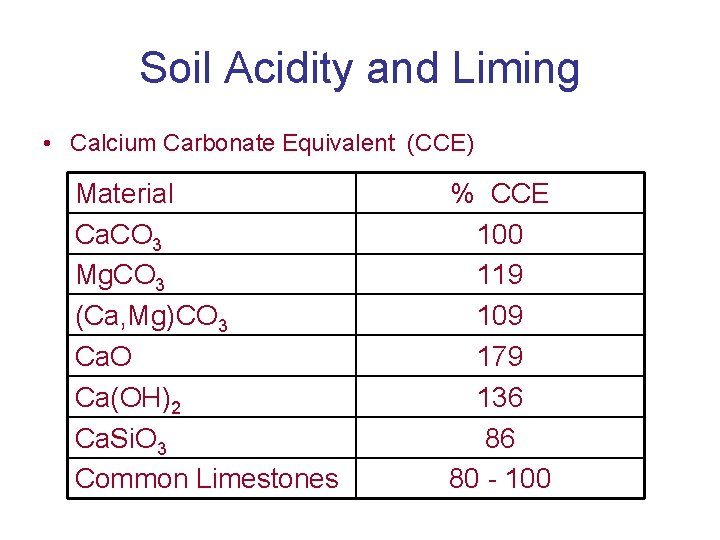
Soil Acidity and Liming • Liming material quality • Calcium Carbonate Equivalent (CCE) – Neutralizing value of any liming material compared to pure calcium carbonate

Soil Acidity and Liming • Calcium Carbonate Equivalent (CCE) – 1 Eq. of Ca. CO 3 = 1 Eq. of any liming material

Soil Acidity and Liming • Calcium Carbonate Equivalent – Example: Mg. CO 3 = 84 g/mole or 42 g/ eq. Ca. CO 3 = 100 g/mole or 50 g/ eq.

Soil Acidity and Liming • Calcium Carbonate Equivalent (CCE) Material Ca. CO 3 Mg. CO 3 (Ca, Mg)CO 3 Ca. O Ca(OH)2 Ca. Si. O 3 Common Limestones % CCE 100 119 109 179 136 86 80 - 100

Soil Acidity and Liming • Soil test recommendations are made as lb CCE/A • Adjust if CCE is different from 100% – Example: • Recommendation = 2000 lb CCE/A • Hydrated Lime Ca(OH)2 CCE = 136% • Required amount of liming material = 100 x Rec. CCE/A % CCE of Material

Soil Acidity and Liming • Real World – Not usually dealing with pure materials, so we can't always calculate the CCE – Measure CCE in lab – Required in most states • Titration with standard acid – CCE is provide on the label for all liming materials sold in PA – Be careful calculating CCE on unknown materials

Estimating Neutralizing Value of Liming Materials - Be very careful • Ca x 2. 5 = Ca. CO 3 • Mg x 3. 5 = Mg. CO 3 • Mg. CO 3 x 1. 19 = Ca. CO 3 • Mg. O x 2. 5 = Ca. CO 3 You have to know the form of the Ca or Mg to use these conversions! • Ca. O x 1. 79 = Ca. CO 3 Unknown material analysis given as 23% Ca

Estimating Neutralizing Value of Liming Materials - Be very careful • If you don’t know what the material is made of then you need to measure the CCE directly! • Cost ~ $10 -15

Soil Acidity and Liming • Calcium Oxide Equivalent (COE) – Neutralizing value of any liming material compared to pure calcium Oxide – 1 Eq. of Ca. O = 1 Eq. of any liming material – 1 Eq. wt. of Ca. O = 1 Eq. wt. of any liming material COE = eq. wt. Ca. O/ eq. wt. Liming material COE = 28/ eq. wt. Liming material

Soil Acidity and Liming • Soil test recommendations may be made as lb COE/A • Adjust if COE is different from 100% • Required amount of liming material = 100 x Rec. COE/A % COE of Material

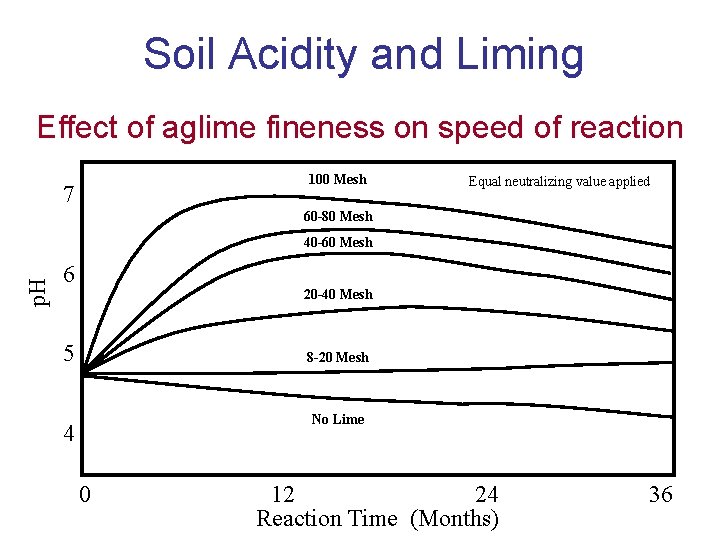
Soil Acidity and Liming • Physical fineness of liming materials – Fineness determines speed of reaction – The finer limestone is ground the faster it will react – Fineness given as % passing specific screen mesh sizes

Soil Acidity and Liming Effect of aglime fineness on speed of reaction 100 Mesh 7 Equal neutralizing value applied 60 -80 Mesh p. H 40 -60 Mesh 6 20 -40 Mesh 5 8 -20 Mesh No Lime 4 0 12 24 Reaction Time (Months) 36

Soil Acidity and Liming • Physical fineness of liming materials – Practical Limits to fineness – Larger than 20 mesh – not effective – Smaller than 100 mesh – little added benefit

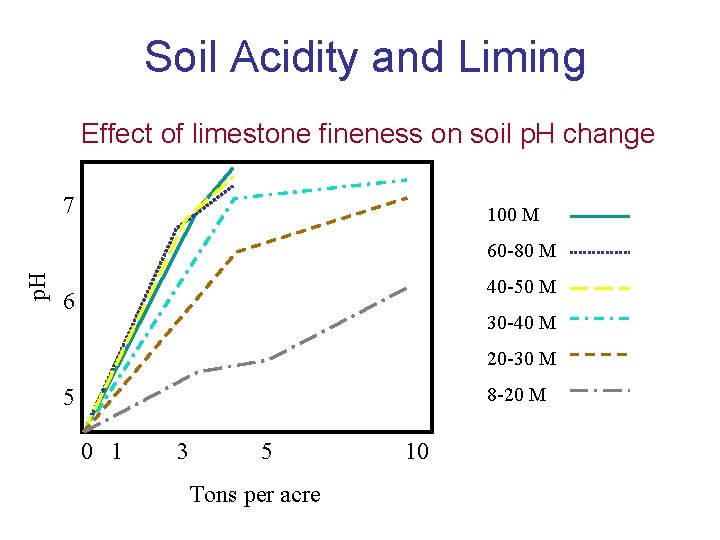
Soil Acidity and Liming Effect of limestone fineness on soil p. H change 7 100 M p. H 60 -80 M 40 -50 M 6 30 -40 M 20 -30 M 8 -20 M 5 0 1 3 5 Tons per acre 10

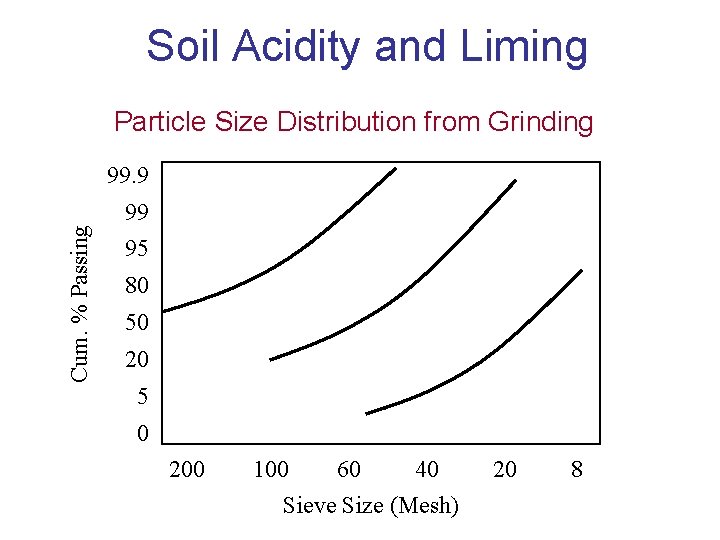
Soil Acidity and Liming Particle Size Distribution from Grinding 99. 9 Cum. % Passing 99 95 80 50 20 5 0 200 100 60 40 Sieve Size (Mesh) 20 8

Soil Acidity and Liming • Aglime Fineness – Pulverized Limestone • Normal ground Limestone – Granular Limestone • Coarser ground limestone • Easy spreading for homeowners • Slower to react

Aglime Fineness definitions for PA Fine Sized Materials Medium Sized Materials 95% - 20 mesh sieve 90% - 20 mesh sieve 60% - 60 mesh sieve 50% - 100 mesh sieve 30% - 100 mesh sieve Coarse sized materials - all liming materials failing to meet the above fineness criteria

Aglime Quality • Acid-Base Chemistry and Fineness • Ca. CO 3 + H 2 O Ca 2+ + H 2 CO 3 + 2 OH- + 2 H+ 2 H 2 O For a given amount of acidity an equivalent amount of base (liming material) will be required regardless if it is ground very fine.

Aglime Quality • Acid-Base Chemistry in soils 1 meq/100 g acidity in the soil will require 1 meq/100 g of Ca. CO 3 to neutralize it. 1 meq Ca. CO 3/100 g soil = 1000 lb Ca. CO 3/A

Soil Acidity and Liming • Aglime Fineness – No matter how fine you grind limestone you cannot increase the chemical neutralizing ability – Finer the better, because it will react faster, but there are practical limitations – Distribution in “Fine Size” Limestone – usually adequate for practical liming for field crops – Little difference between calcite and dolomite

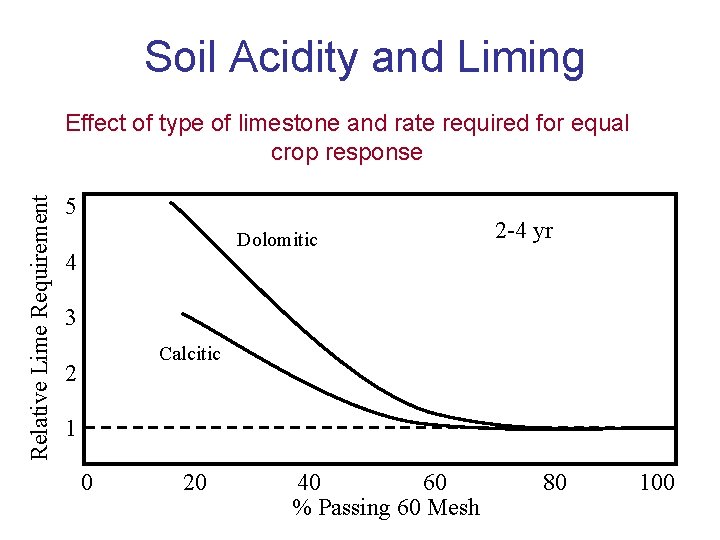
Soil Acidity and Liming Relative Lime Requirement Effect of type of limestone and rate required for equal crop response 5 Dolomitic 4 2 -4 yr 3 Calcitic 2 1 0 20 40 60 % Passing 60 Mesh 80 100

Soil Acidity and Liming • Pellet Lime – Finely ground limestone – Glued together into water soluble pellets – Advantage: Handling, spreading, speed of reaction – Disadvantage: Cost 100 -200 mesh

Soil Acidity and Liming • Fluid Lime – Finely ground limestone – Suspended in water with clay – Approx. 1000 lb CCE/ton material – Advantage: Spreading, speed of reaction – Disadvantage: Cost

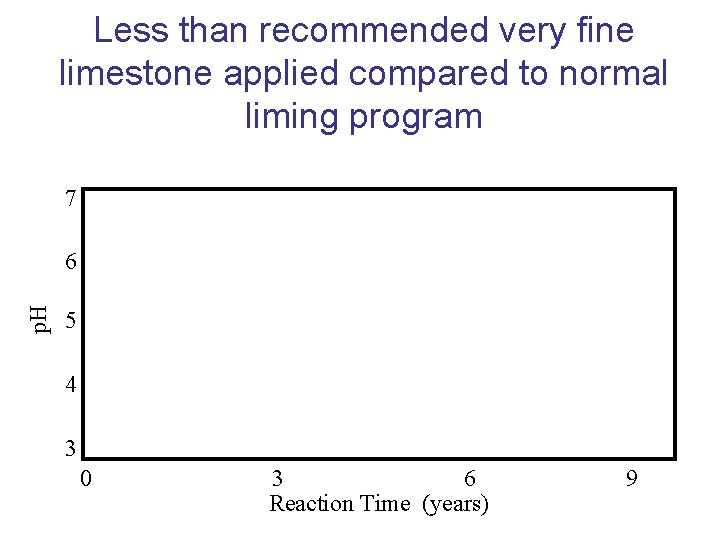
Less than recommended very fine limestone applied compared to normal liming program 7 p. H 6 5 4 3 0 3 6 Reaction Time (years) 9

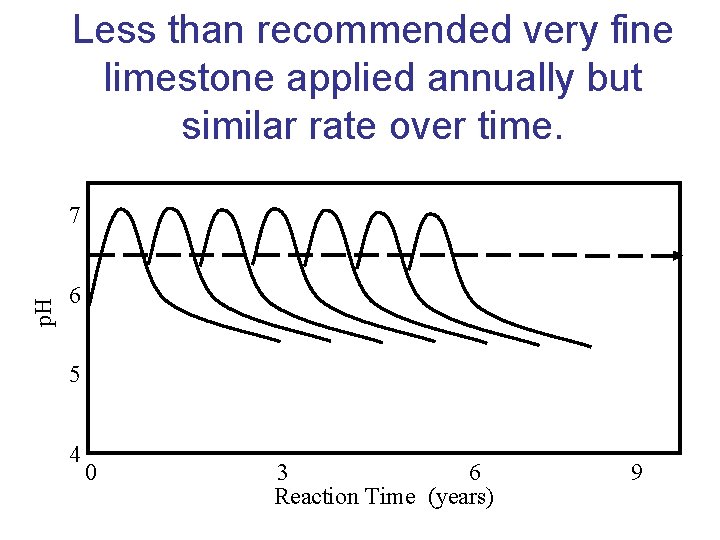
Less than recommended very fine limestone applied annually but similar rate over time. p. H 7 6 5 4 0 3 6 Reaction Time (years) 9

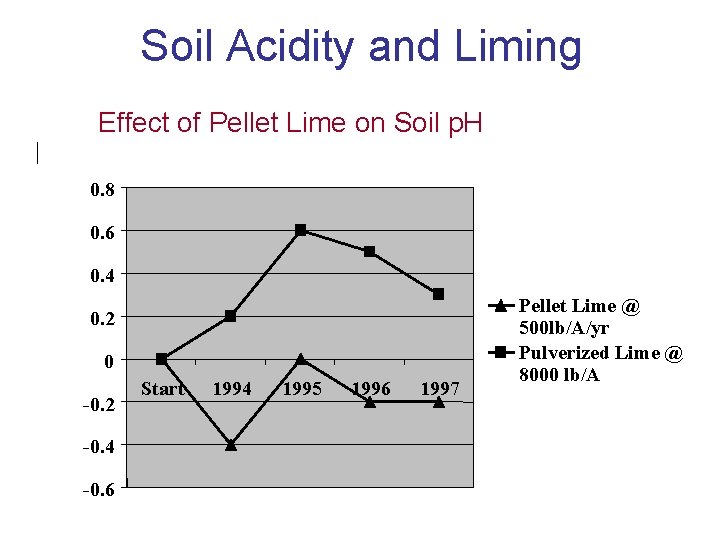
Soil Acidity and Liming Effect of Pellet Lime on Soil p. H 0. 8 Change in p. H 0. 6 0. 4 0. 2 0 -0. 2 -0. 4 -0. 6 Start 1994 1995 1996 1997 Pellet Lime @ 500 lb/A/yr Pulverized Lime @ 8000 lb/A

Soil Acidity and Liming • Calcium and Magesium – Normal liming practices will also supply required calcium and magnesium • At normal rates usually adequate Ca will be supplied for most crops • Magnesium will depend on the type of limestone used – If Mg is required use a Mg containing limestone (dolomitic) – Mg recommendations » pounds Mg/A » % Mg in recommended limestone

Calcium and Magnesium • Management Recommendations – Maintain optimum p. H – Maintain at least optimum levels of Mg (60 ppm) and K (100 ppm) – Don’t worry about ratios until they are way out of balance • eg. Mg: Ca >1, K: Mg>1 • Rare

By-product Liming Materials • Quality –Liming value –Undesirable components

Example: Liming Value Material sold in western PA for $2/ton CCE = 5. 38% For 1 ton of neutralizing value: 2000 X 100 ÷ 5. 38 = 37, 174 lbs. or 18. 6 tons ($37. 20) Fineness: Through 20 Mesh = 98. 8% (90%) Through 60 Mesh = 78. 2 % (60%) Through 100 Mesh = 63. 2% (50%)

Gypsum • Ca. SO 4 • Excellent source of Ca and S – 33% Ca & 27% S • No neutralizing value • Not a liming material

Limestone vs Gypsum • Limestone changes p. H Gypsum doesn’t • Both are good sources of Ca – Lime to recommended p. H. . . no need for additional Ca for the soil or for agronomic crops in PA – High p. H but low calcium soils (Sodic Soils) use gypsum as a source of Ca for the soil and the crop (Western US) • Improves physical properties Relieves chemical compaction – Low p. H, acid loving crops or highly weathered soils, use gypsum as a source of Ca for the soil and the crop (Tropics) • Minimal lime to reduce Al toxicity

Byproduct Materials Undesirable components • Should be registered (PDA) • Must be approved for land application by DEP – May limit lifetime application • Problem with unregistered materials and materials from out of state – May be little or no checking of quality • Determine the source – Dust, screenings – Stainless steel slag • Ask what contaminants might be present • If in doubt. . . get it analyzed OR don’t use it

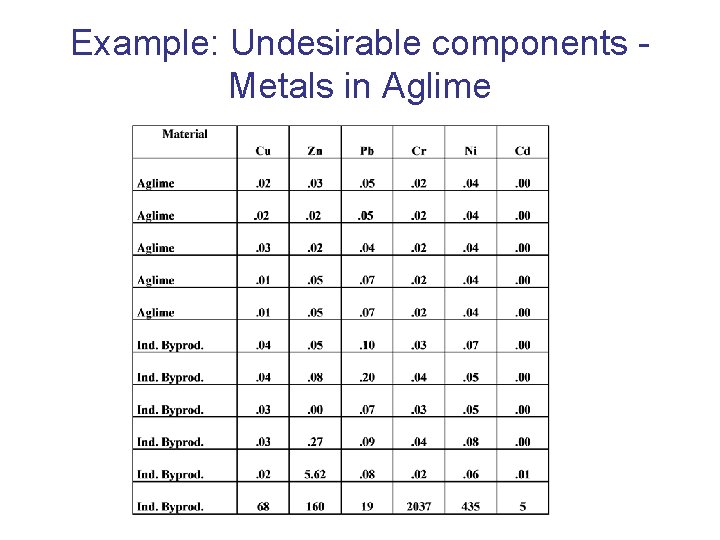
Example: Undesirable components Metals in Aglime

Other Materials • Biosolids and Water Treatment Sludges – Often have significant neutralizing value – Must be clean – Must be registered if sold as liming materials

Other Materials Organic Calcium Compounds • Promesol 30, Liquid Lime – Trihydroxy glutaric acid 25% Ca – 1 gallon = 500 -750 lb CCE? • Liqui-Til – Trihydroxy glutaric acid – Neutralizes p. H in alkaline soil? • KK Organic Soil Builder – Neutralizes both acidic and alkaline soils? • Liqui-Cal – 8% Ca – 1 gallon = 500 lb Ca. CO 3? • Liquid Calcium – 1 gallon = Ca in 500 lb Ca. CO 3? – Suggest it is a lime substitute • Golden Cal – Glucoheptomic acid – 1 gallon = 500 lb CCE? • p. H Plus – 1 gallon = 500 -750 lb CCE?

Other Materials • Remember: 1 Eq. of base is required to neutralize 1 Eq. of acid • Watch out for materials that contain Ca with unwritten or suggestive claims for liming value

Soil Acidity and Liming • Limestone Application – Apply limestone far enough ahead of time to be effective – Spread limestone uniformly • • Spinner spreaders Boom spreaders Damp lime Dust – Spit high rates of limestone (>4 ton/A) – Time of year is not too critical • Consider soil quality issues – compaction – Mix limestone as much as practical • Adjust for depth of mixing - 6 2/3 in. standard depth • No till – Correct p. H before going to no-till

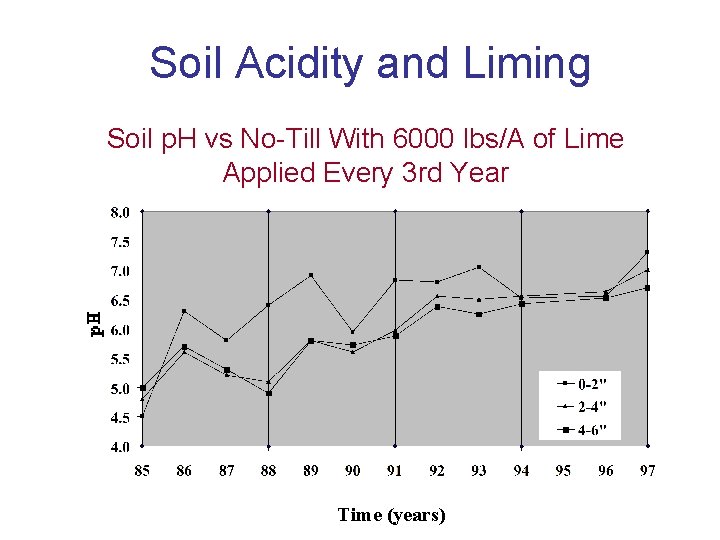
Soil Acidity and Liming Soil p. H vs No-Till With 6000 lbs/A of Lime Applied Every 3 rd Year Time (years)