Naming Ionic Compounds The Big Idea in Naming

Naming Ionic Compounds

The Big Idea in Naming Ionic Compounds Every compound needs to have a unique name. This is accomplished by naming the two ions that make up the compound. By identifying both the metal ion and the non-metal ion, the compound is identified.
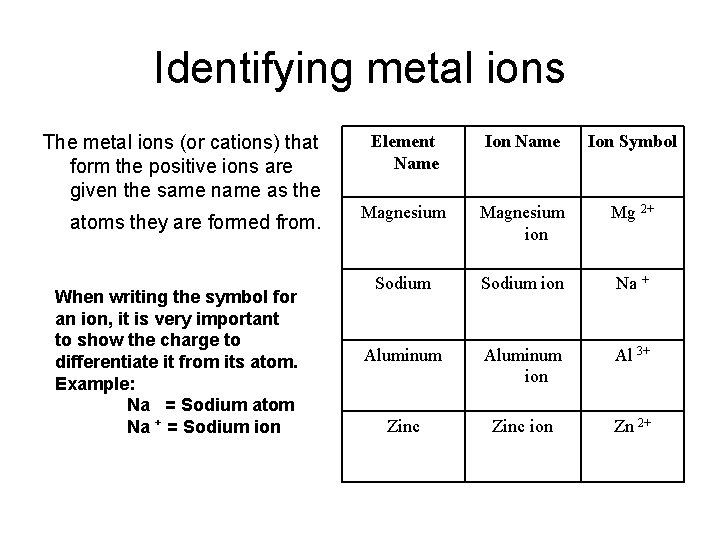
Identifying metal ions The metal ions (or cations) that form the positive ions are given the same name as the atoms they are formed from. When writing the symbol for an ion, it is very important to show the charge to differentiate it from its atom. Example: Na = Sodium atom Na + = Sodium ion Element Name Ion Symbol Magnesium ion Mg 2+ Sodium ion Na + Aluminum ion Al 3+ Zinc ion Zn 2+

Identifying non-metal ions The non-metal ions (or anions) that are formed when an atom accepts electrons are distinguished from their atoms by the “ide” ending. Element Name Ion Symbol Chlorine Chloride ion Cl - When writing the symbol for an ion, it is very important to show the charge to differentiate it from its atom. Oxygen Oxide ion O 2 - Nitrogen Nitride ion N 3 - Sulfur Sulfide ion S 2 - Example: Cl = Chlorine atom Cl - = Chloride ion
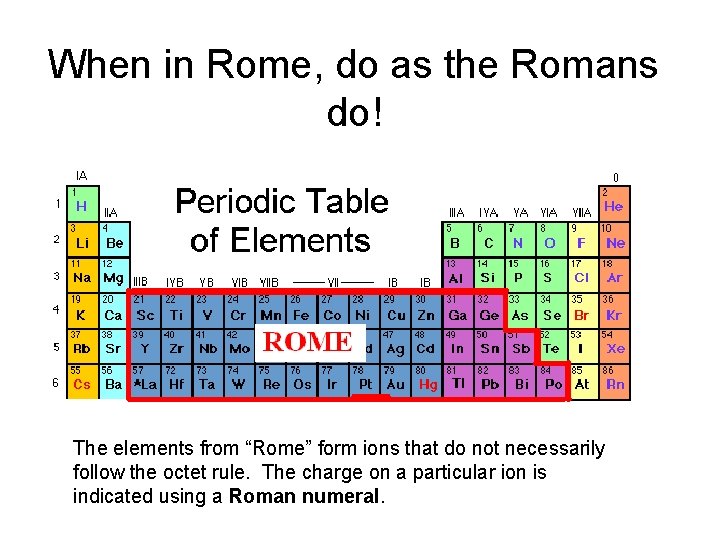
When in Rome, do as the Romans do! The elements from “Rome” form ions that do not necessarily follow the octet rule. The charge on a particular ion is indicated using a Roman numeral.

Examples from “Rome” Atoms from Rome can form more than one ion. This requires a way of telling the ions apart. This is done by using Roman numerals to tell the different ions apart. The Roman numeral indicates the charge on the ion. This method of using Roman numerals to indicate the charge is called the Stock system and is used only for atoms that form more than one ion. Element Name Ion Symbol Copper (I) ion Cu + Copper (II) ion Cu 2+ Iron (II) ion Fe 2+ Iron (III) ion Fe 3+ Lead (II) ion Pb 2+ Lead (IV) ion Pb 4+ Iron Lead
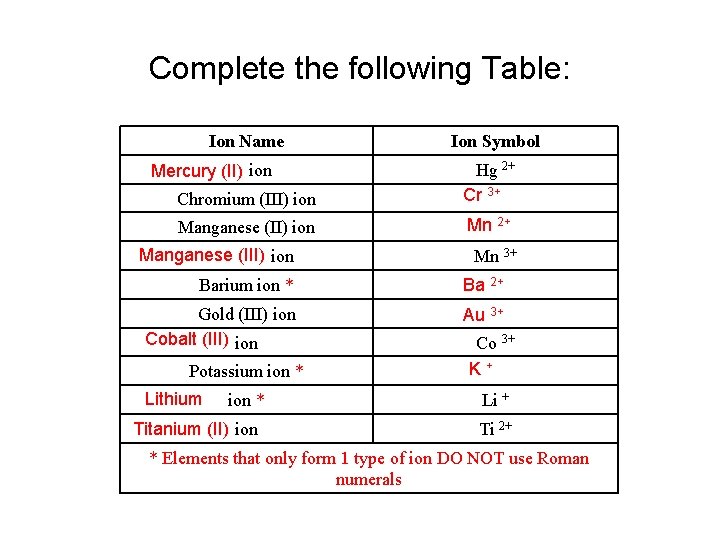
Complete the following Table: Ion Name Mercury (II) ion Ion Symbol Chromium (III) ion Hg 2+ Cr 3+ Manganese (II) ion Mn 2+ Manganese (III) ion Mn 3+ Barium ion * Ba 2+ Gold (III) ion Cobalt (III) ion Au 3+ Potassium ion * Lithium ion * Titanium (II) ion Co 3+ K+ Li + Ti 2+ * Elements that only form 1 type of ion DO NOT use Roman numerals

You Try! Write the formulas for the following: 1. Chromium (III) chloride 2. Barium bromide 3. Iron (III) sulfide 4. Copper (I) oxide 5. Potassium oxide Note: atoms that are not from “Rome” do not use Roman numerals.

Finding the charge on the metal ion • In order to use the Stock system we must know the charge on the metal ion. This can be determined by using the charge on the negative ion and remembering that the sum of the charges on all the ions in a compound must be zero.

Naming compounds using the Stock System The Stock system is important in naming compounds formed from elements that form more than one type of ion. Fe. Cl 2 – Iron (II) Cloride Fe. Cl 3 – Iron (III) Chloride Fe. O – Iron (II) oxide Fe 2 O 3 – Iron (III) oxide Cu 2 O – Copper (I) oxide Cu. O – Copper (II) oxide Pb. O - Lead (II) oxide Pb. O 2 – Lead (IV) oxide

You Try! Write the names for the following Manganese (II) oxide compounds: Nickel (II) Bromide 1. Mn. O Copper (I) sulfide 2. Ni. Br 2 Tin (IV) chloride 3. Cu 2 S Cr. Cl 4. Sn. Cl 4 Cu I 5. Ba. Cl 2 3 2 Hg. Cl 2
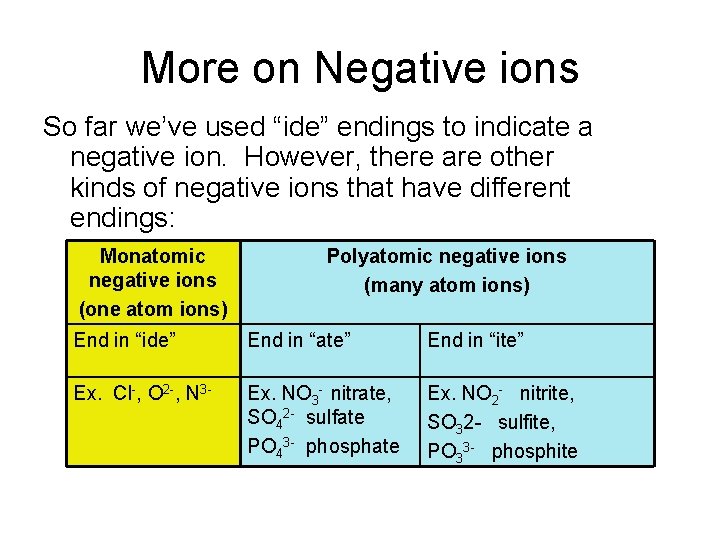
More on Negative ions So far we’ve used “ide” endings to indicate a negative ion. However, there are other kinds of negative ions that have different endings: Monatomic negative ions (one atom ions) Polyatomic negative ions (many atom ions) End in “ide” End in “ate” End in “ite” Ex. Cl-, O 2 -, N 3 - Ex. NO 3 - nitrate, SO 42 - sulfate PO 43 - phosphate Ex. NO 2 - nitrite, SO 32 - sulfite, PO 33 - phosphite
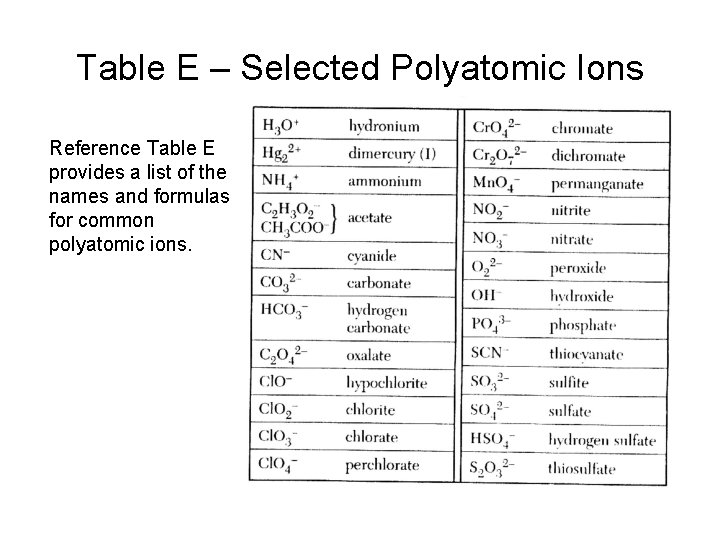
Table E – Selected Polyatomic Ions Reference Table E provides a list of the names and formulas for common polyatomic ions.
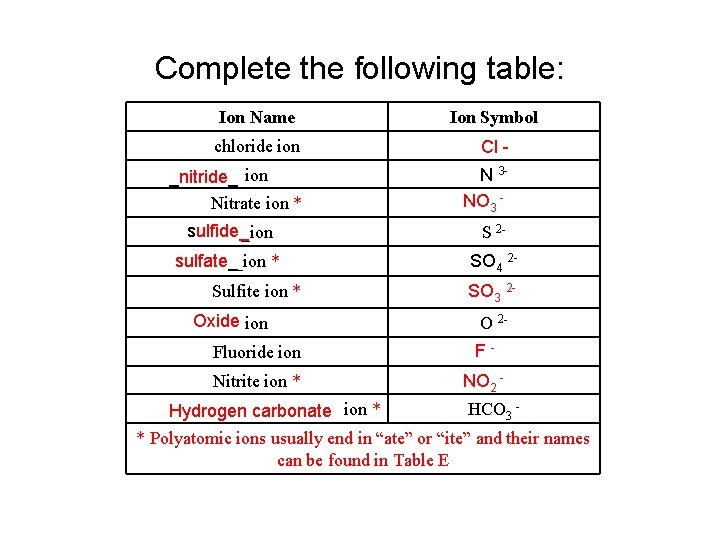
Complete the following table: Ion Name Ion Symbol chloride ion Cl - _nitride_ ion Nitrate ion * N 3 NO 3 - sulfide__ion S 2 - sulfate__ion * SO 4 2 - Sulfite ion * Oxide ion SO 3 2 O 2 - Fluoride ion F- Nitrite ion * NO 2 - Hydrogen carbonate ion * HCO 3 - * Polyatomic ions usually end in “ate” or “ite” and their names can be found in Table E

Use Table E to identify the following polyatomic ions 1. 2. 3. 4. 5. 6. 7. 8. Nitrate Sulfate Phosphate chlorate Nitrite Sulfite Phosphite chlorite

Let’s review! • The names of the metal ions are the same as their atoms. • When a metal atom forms more than one ion, a Roman numeral is used to indicate the charge on the ion. • Most monatomic negative ions end in “ide”. • Polyatomic negative ions end in “ate” or “ite”. The names and formulas of these ions can be found in Table E.

The Crossover Rule re-visited Once the ions making up a compound have been identified, the formula for the compound can be easily determined using the crossover rule. For instance: copper (II) acetate is made up of Cu 2+ ions and acetate ions (C 2 H 3 O 2 – ions). Since the atoms in the polyatomic ion are bonded together they will act as a single unit.

Reality check! Write the formulas for the following compounds: • barium nitrate ____ • tin (II) chloride ____ • sodium sulfate ____ • copper (II) oxide ____ • copper (I) oxide ____ • iron (III) sulfide ____ • potassium chlorate ____ • silver (I) nitrate ____ • lead (IV) bromide ____ • sodium phosphate ____

Complete the following: Compound Formula Potassium chloride KCl Calcium chloride Manganese (II) chloride Ca. Cl 2 Mn. Cl 2 Manganese (III) chloride Mn. Cl 3 Barium oxide Ba. O Sodium sulfate Na 2 SO 4 Copper (II) sulfate Copper (I) sulfate Cu. SO 4 Cu 2 SO 4 Magnesium nitrate Mg(NO 3)2 Calcium acetate Ca(C 2 H 3 O 2)2

Things to think about 1. Why is KCl called potassium chloride when Cu. Cl is called copper K only forms a 1+ ion, while Cu can form a 1+ or a 2+ ion. (I) chloride? Therefore you need the Roman numeral to identify which ion is in the compound 2. 3. Why is sodium nitrate written as Na. NO 3 (no parenthesis) while magnesium nitrate is written as Mg(NO 3)2 (with parenthesis)? Parentheses are required when there is more than one polyatomic ion to indicate that are multiple numbers of the whole ion. Why would it be incorrect to refer to Fe. O as simply iron oxide? Fe 2 O 3 and Fe. O are both iron oxide. Need a unique name. 4. Most polyatomic ions are negative, but some positive ions exist. See if you can find the positive ions in Table E and list them. Ammonium NH 4+ Hydronium H 3 O+
- Slides: 20