Principles of Corrosion Professor Grace Burke Director Materials

Principles of Corrosion Professor Grace Burke Director, Materials Performance Centre Room E 3 Phone 64858 m. g. burke@manchester. ac. uk With acknowledgement to Emeritus Prof. Bob Cottis

Learning Objectives…. After this module, you will be able to: • Describe Corrosion (and its various forms) • Identify Anodic and Cathodic reactions • Describe the Corrosion Potential for a reaction • Define the Nernst Equation • Derive thermodynamic stability of H 2 O • Understand p. H effects • Describe Pourbaix Diagrams and their applications • Discuss Passivity

Corrosion! q A major materials issue - everywhere! q From the Latin “corrodere” – to gnaw into pieces! q Definition: A chemical or electrochemical reaction between a material (generally a metal) and the environment that leads to a degradation/deterioration in the material and its properties (ASM International) q Economic Cost of Corrosion Worldwide: • 2010 - >$2. 2 TRILLION Annually! Corrosion Organization) (World

Corrosion: some historical examples! q Speculated that corrosion may have contributed to the downfall of Roman Empire! • Wine was stored in Pb-lined vessels by Roman heirarchy • Pb corrodes in the acetic acid-containing solutions (like red wine!) • Pb+2 in red wine would be routinely ingested by these affluent Romans, leading to Pbpoisoning/insanity!

More Corrosion Effects q In the 1700’s, Pb coils were used in condensers in making brandy • Subsequently outlawed because it became recognised that Pb was causing illness! q In mid-1700’s, Pewter (Sn-up to 50% Pb) was recognised as a serious health hazard, so Pb was significantly reduced and other alloying elements were added to Sn. (no more Pb+2 into drinks)

Examples of Corrosion q Bridges q Steel-reinforcements in concrete q Some Implants q Microbial corrosion/marine environments q Rusty metal/rails, beams, girders q Some cars; exhaust systems q Pipelines q Oil rigs & storage q Aircraft

Forms of Corrosion q Uniform or general corrosion q Galvanic corrosion (2 dissimilar metals) q Pitting corrosion (localised corrosion on flat surface) q Crevice corrosion (geometry; occluded region) q Intergranular corrosion (preferential corrosion) q De-alloying (selective corrosion of an element) q Erosion corrosion (wear + corrosion) q Flow-Assisted Corrosion q Microbial-Induced Corrosion (MIC) -- Environmentally-Assisted Cracking (not pure corrosion)

Corrosion: How to control it? q Select a corrosion-resistant material q Use a coating (paint) (barrier layer!) q Use an inhibitor (added to solution or surface) q Cathodic protection (external applied potential) q Design to avoid corrosion

Corrosion of Metals in Acid q Zinc dissolves rapidly with hydrogen evolution Zn + 2 HCl Zn. Cl 2 + H 2 q Zinc is the base or active metal

Corrosion of Platinum in Acid q Platinum does not react with acids q Platinum is known as a noble metal
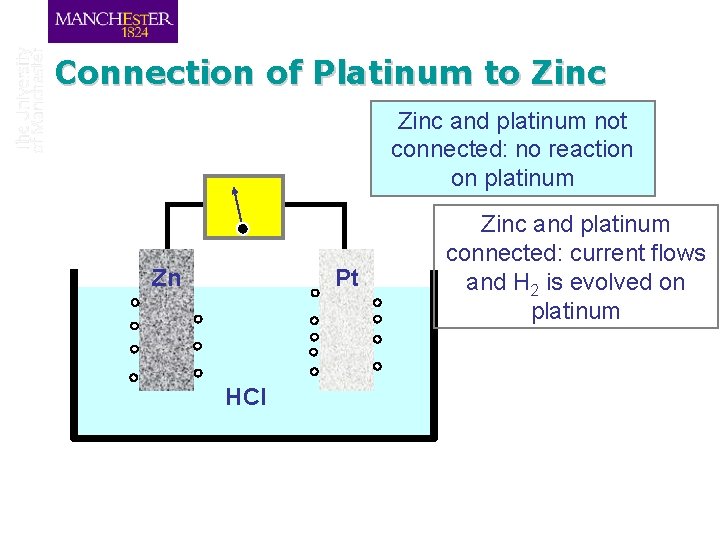
Connection of Platinum to Zinc and platinum not connected: no reaction on platinum Zn Pt HCl Zinc and platinum connected: current flows and H 2 is evolved on platinum

Connection of Platinum to Zinc Zn + 2 HCl Zn. Cl 2 + H 2 q But we can separate metal dissolution and hydrogen evolution Zn 2+ + 2 e 2 H+ + 2 e- H 2 q These are known as electrochemical reactions Reactions that involve both chemical change and the transfer of charge

External Current Applied to Pt q Hydrogen evolution at one electrode 2 H+ + 2 e- H 2 A piece of metal or 2 H 2 O + 2 e- H 2 + 2 OHin the solution q Oxygen evolution at the other electrode 4 OH- O 2 + 2 H 2 O + 4 eor 2 H 2 O O 2 + 4 H+ + 4 efor Platinum – because it is a noble metal (for other metals (active) the reaction will be written in reverse!)

Electrodes q Electrodes are pieces of metal on which an electrochemical reaction is occurring q An anode is an electrode on which an anodic or oxidation reaction is occurring q A cathode is an electrode on which a cathodic or reduction reaction is occurring

Corrosion of Metals in Acid q Zinc dissolves rapidly with hydrogen evolution Zn + 2 HCl Zn. Cl 2 + H 2 q Zinc is the base or active metal q Fe dissolves with H 2 evolution Fe + 2 HCl Fe. Cl 2 + H 2 q Iron is the base or active metal
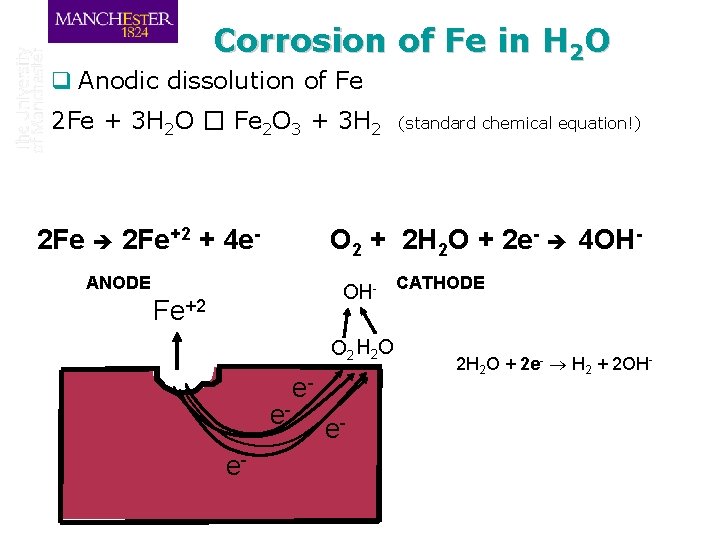
Corrosion of Fe in H 2 O q Anodic dissolution of Fe 2 Fe + 3 H 2 O � Fe 2 O 3 + 3 H 2 2 Fe+2 + 4 e- O 2 + 2 H 2 O + 2 e- 4 OH- ANODE OH- Fe+2 O 2 H 2 O ee- (standard chemical equation!) ee- CATHODE 2 H 2 O + 2 e- H 2 + 2 OH-

Corrosion q 2 Fe + 3 H 2 O Fe 2 O 3 + 3 H 2 (standard chemical equation!) DGreaction = DGproducts – DGreactants DGreaction = (DGFe 2 O 3+ 3 DGH 2) – (2 DG 0 Fe+ 3 DGH 2 O) Remember, there are other reactions included: Anodic reaction Cathodic reaction 2 Fe+2 + 4 e 2 H 2 O + O 2 + 4 e- 4 OH- q What is the driving force? Lowering the free energy of the system!

Some Thermodynamics…. q Standard Gibbs Free Energy Change of the Reaction: DGo = -RT ln Kp (Kp is Equilib. Constant at 1 atm. ) q Also: DGo = -n. FE 0 • where n= number of electrons transferred • F is Faraday’s Constant (~96500 Coulombs/equivalent) • E 0 is the standard electrode potential UNITS: n (equivalents/mole) F (Coulombs/equivalent) E 0 (Volts) (Volts Coul)/mole = Joules/mole (DGo )

Some Thermodynamics…. q Standard Gibbs Free Energy Change of the Reaction: DGo = -RT ln Kp (Kp is Equilib. Constant at 1 atm. ) DG = -RT ln K q Also: DGo = -n. FE 0 (and DG = -n. FE) q DGo = -RT ln Kp = - n. FE 0

Some Thermodynamics…. q Standard Gibbs Free Energy Change of the Reaction: DGo = -RT ln Kp (Kp is Equilib. Constant at 1 atm. ) q Chemical Potential, m, denotes the change of DG of a substance (as a f (composition of a given species) at constant T and P and # of moles of other reactants m 1 = mo 1 + RT ln a 1 where a 1 = activity of component 1 Example: Me + O 2 Me. O 2 q So, DG = moprod – moreact 1 – moreact 2 + RT ln (1/a. O 2) (since activities of the metal and metal oxide = 1) DG = mo. Me. O 2 – mo. Me – mo. O 2 – RT ln a. O 2 DG = DGo – RT ln p. O 2

Electrodes q Electrodes are pieces of metal on which an electrochemical reaction is occurring q An anode is an electrode on which an anodic or oxidation reaction is occurring q A cathode is an electrode on which a cathodic or reduction reaction is occurring

Anodic Reactions q Examples Zn 2+ + 2 e. Fe 2+ + 2 e. Al 3+ + 3 e. Fe 2+ Fe 3+ + 3 e. H 2 2 H+ + 2 e 2 H 2 O O 2 + 4 H+ + 4 e- q Oxidation reactions produce electrons LEO: Losing Electrons is Oxidation!

Cathodic Reactions q Examples O 2 + 2 H 2 O + 4 e- 4 OH 2 H 2 O + 2 e- H 2 + 2 OHCu 2+ + 2 e- Cu Fe 3+ + e- Fe 2+ q Reduction reactions consume electrons GER: Gaining Electrons is Reduction

Anodic and Cathodic Reactions LEO – GER! Losing Electrons is Oxidation – Gaining Electrons is Reduction

Effect of Potential q The voltage of the metal with respect to the solution will affect electrochemical reactions • Voltage will affect the concentration of e- and H+ q Voltage of metal with respect to solution is known as the electrochemical potential

Corrosion of zinc in acid q When zinc is placed in acid the metal will start to dissolve and hydrogen will start to be liberated according to the potential of the metal q Consider the anodic zinc dissolution reaction Zn 2+ + 2 e-
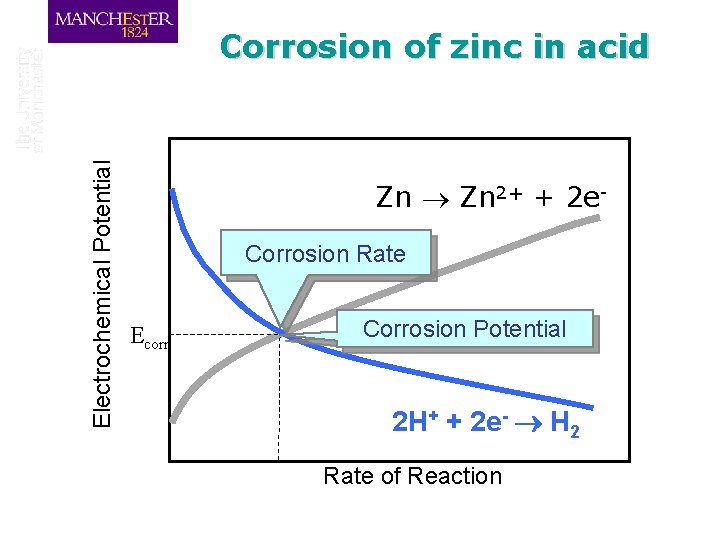
Electrochemical Potential Corrosion of zinc in acid Zn 2+ + 2 e. Corrosion Rate Ecorr Corrosion Potential 2 H+ + 2 e- H 2 Rate of Reaction
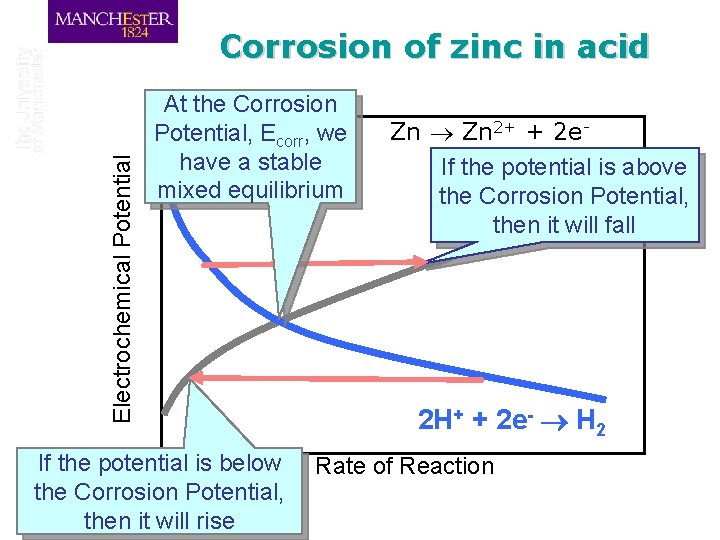
Electrochemical Potential Corrosion of zinc in acid At the Corrosion Potential, Ecorr, we have a stable mixed equilibrium If the potential is below the Corrosion Potential, then it will rise Zn 2+ + 2 e. If the potential is above the Corrosion Potential, then it will fall 2 H+ + 2 e- H 2 Rate of Reaction
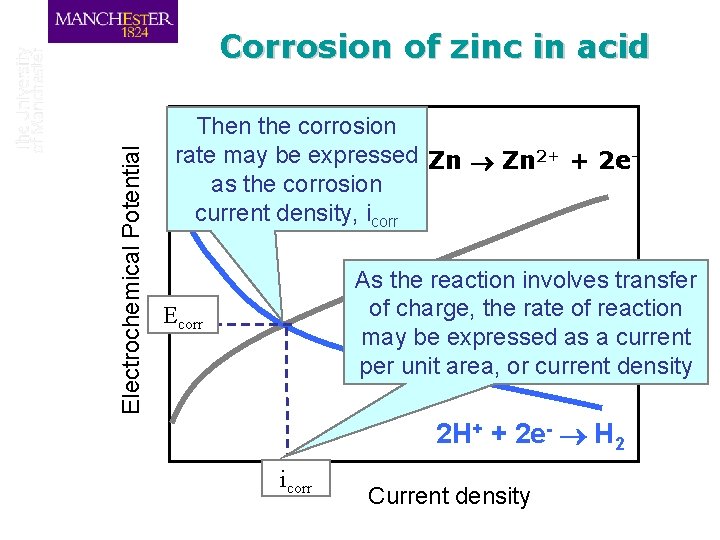
Electrochemical Potential Corrosion of zinc in acid Then the corrosion rate may be expressed Zn 2+ + 2 eas the corrosion current density, icorr As the reaction involves transfer of charge, the rate of reaction may be expressed as a current per unit area, or current density Ecorr 2 H+ + 2 e- H 2 icorr Current density

When can corrosion occur? 2 H+ + 2 e- H 2 q The concentration of hydrogen ions will influence the rate of the reaction q As the hydrogen ion concentration is increased, so the rate of the reaction increases

When can corrosion occur? q On platinum (Pt) no metal dissolution will occur, but to balance the charge a reaction, which creates electrons, must occur q One such reaction is the reverse of the hydrogen evolution reaction: H 2 2 H+ + 2 e-

Thermodynamic Equilibrium 2 H+ + 2 e- H 2 q The potential at which this reaction occurs is known as the equilibrium potential q The concentrations of reactants controls the rates of the forward and reverse reactions, and hence the equilibrium potential

Corrosion q 2 Fe + 3 H 2 O Fe 2 O 3 + 3 H 2 (standard chemical equation!) DGreaction = DGproducts – DGreactants DGreaction = (DGFe 2 O 3+ 3 DGH 2) – (2 DG 0 Fe+ 3 DGH 2 O) Remember, there are other reactions included: Anodic reaction Cathodic reaction 2 Fe+2 + 4 e 2 H 2 O + O 2 + 4 e- 4 OH- q What is the driving force? Lowering the free energy of the system!

Some Thermodynamics…. q Standard Gibbs Free Energy Change of the Reaction: DGo = -RT ln Kp (Kp is Equilib. Constant at 1 atm= activities of the products/activities of the reactants) q Also: DGo = -n. FE 0 • where n= number of electrons transferred • F is Faraday’s Constant (~96500 Coulombs/equivalent) • E 0 is the standard electrode potential UNITS: n (equivalents/mole) F (Coulombs/equivalent) E 0 (Volts) (Volts Coul)/mole = Joules/mole (DGo )

Some Thermodynamics…. q Standard Gibbs Free Energy Change of the Reaction: DGo = -RT ln Kp (Kp is Equilib. Constant at 1 atm. ) DG = -RT ln K q Also: DGo = -n. FE 0 DG = -n. FE q DGo = -RT ln Kp = - n. FE 0 DG = DGo - RT ln K/Kp

Some Thermodynamics…. q Chemical Potential, m, denotes the change of DG of a substance (as a f(composition of a given species) at constant T and P and # of moles of other reactants m 1 = mo 1 + RT ln a 1 where a 1 = activity of component 1 Example: Consider Oxidation of Metal Me: Me + O 2 Me. O 2 q So, DG = moprod – moreact 1 – moreact 2 + RT ln (1/a. O 2) (since activities of the metal and metal oxide = 1) DG = mo. Me. O 2 – mo. Me – mo. O 2 – RT ln a. O 2 (a. O 2= p. O 2) DG = DGo – RT ln p. O 2

Nernst Equation (1) E = Eo -(RT/n. F)ln(aprod/areact) Eo = True thermodynamic equilibrium potential, NOT the free corrosion potential, Ecorr Eo obtained by calculation; standard conditions: 1 atm, and 25 C (298 K). The Nernst Equation relates the effective concentration (activities) of the components of the cell reaction to the standard cell potential.

Nernst Equation (2) H 2 = 2 H+ + 2 e- q The Nernst equation gives q For 1 atm. hydrogen gas F is Faraday’s Constant: 96487 coul/mol R is the Gas Constant: 8. 314 Joule/K/mole

Nernst Equation (3) H 2 = 2 H+ + 2 e. So, substituting for R and F, and T=298 K (25°C) E = Eo + (RT/n. F) ln[(a. H+)2/(a. H 2)] n=2; a. H 2 = 1 = Eo + (RT/2 F) 2 ln [H+] = Eo + (RT/F) 2. 303 log [H+] = Eo - 0. 0591 p. H = -log[H+]

Thermodynamic Stability of H 2 O From Marcel Pourbaix: (using cal, K, R=1. 985 cal/K) Dissociation of Water: H 2 O = H+ + OHlog [H+] x [OH-] = log K (which is the equilib. const) [H 2 O] Also: Log K = - S (activities or fugacities) 2. 303 RT = - S (activities or fugacities) 1363 (T=298 K)

Thermodynamic Stability of H 2 O Dissociation of Water: H 2 O = H+ + OHlog([H+] x [OH-]} = log K (which is the equilib. const) At 25°C: log K = (m 0 H+ + m 0 OH- )- (m 0 H O) 2 = 0 - 37595 + 56690 = -14 1363 [H+] x [OH-] = 10 -14 log[H+] + log[OH-] = -14. 00 Remember, p. H = -log[H+] So, for p. H = 7. 00, [H+] = [OH-] p. H < 7. 00, [H+] > [OH-] p. H > 7. 00, [H+] < [OH-] 1363
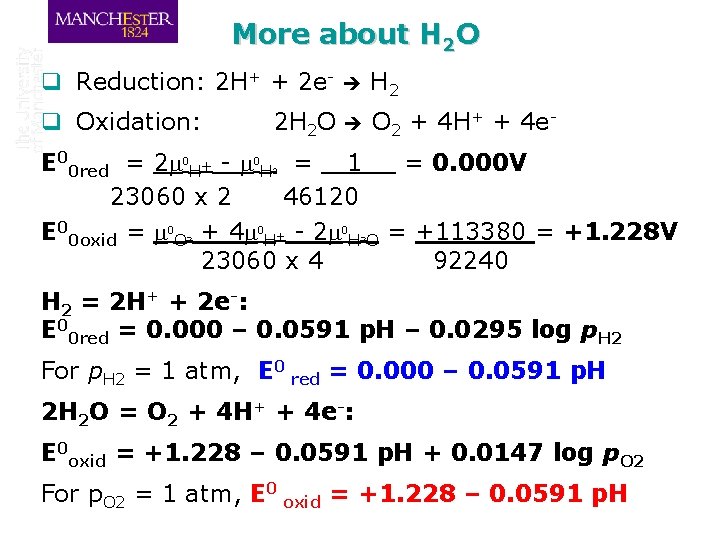
More about H 2 O q Reduction: 2 H+ + 2 e- H 2 q Oxidation: O 2 + 4 H+ + 4 e- 2 H 2 O E 00 red = 2 m 0 H+ - m 0 H = 1 = 0. 000 V 23060 x 2 46120 E 00 oxid = m 0 O + 4 m 0 H+ - 2 m 0 H O = +113380 = +1. 228 V 23060 x 4 92240 2 2 2 H 2 = 2 H+ + 2 e-: E 00 red = 0. 000 – 0. 0591 p. H – 0. 0295 log p. H 2 For p. H 2 = 1 atm, E 0 red = 0. 000 – 0. 0591 p. H 2 H 2 O = O 2 + 4 H+ + 4 e-: E 0 oxid = +1. 228 – 0. 0591 p. H + 0. 0147 log p. O 2 For p. O 2 = 1 atm, E 0 oxid = +1. 228 – 0. 0591 p. H

Marcel Pourbaix (1904 -1998) q Belgian Chemist (born in Russia while his father was working there as an engineer) q Professor at the University of Brussels q Derived Potential-p. H diagrams, known as “Pourbaix Diagrams” • Constructed using the Nernst Equation • Provide a visual depiction of the relationship between thermodynamically possible phases of a system, bounded by lines representing the reactions between phases • Based on thermodynamics!
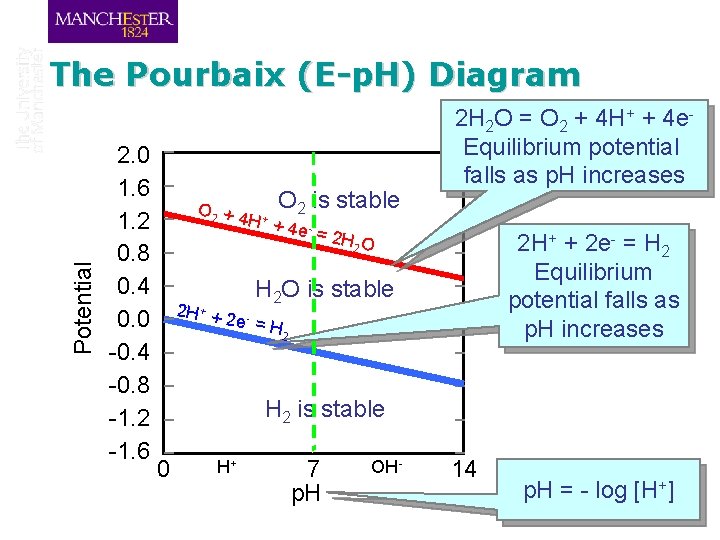
Potential The Pourbaix (E-p. H) Diagram 2. 0 1. 6 1. 2 0. 8 0. 4 0. 0 -0. 4 -0. 8 -1. 2 -1. 6 O 2 + 4 + O 2 is stable H +4 e =2 H 2 O 2 H 2 O = O 2 + 4 H+ + 4 e. Equilibrium potential falls as p. H increases 2 H+ + 2 e- = H 2 Equilibrium potential falls as p. H increases H 2 O is stable 2 H + + 2 e- = H 2 is stable 0 H+ 7 p. H OH- 14 p. H = - log [H+]
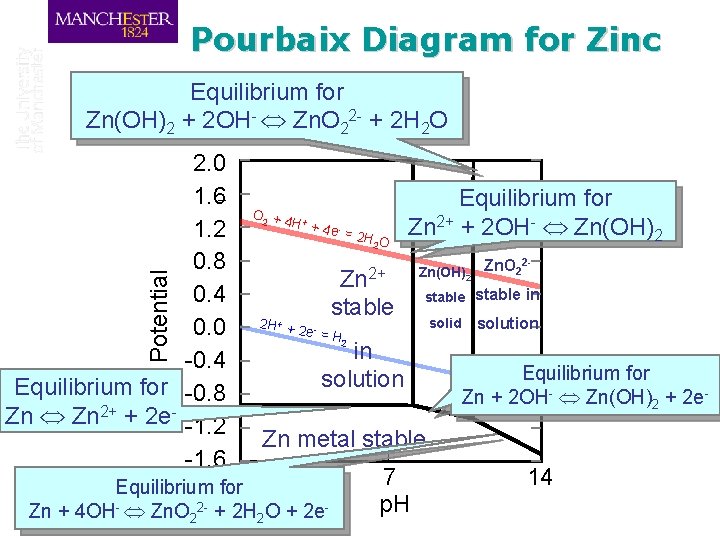
Pourbaix Diagram for Zinc Equilibrium for Zn(OH)2 + 2 OH- Zn. O 22 - + 2 H 2 O Potential 2. 0 1. 6 1. 2 0. 8 0. 4 0. 0 -0. 4 Equilibrium for -0. 8 Zn 2+ + 2 e- -1. 2 -1. 6 O 2 + 4 + H + 4 e - = 2 H O 2 Equilibrium for Zn 2+ + 2 OH- Zn(OH)2 Zn 2+ 2 H + + 2 e - = stable 2 Zn(OH)2 Zn. O 2 stable in H 2 in solution solid solution Equilibrium for Zn + 2 OH- Zn(OH)2 + 2 e- Zn metal stable 0 Equilibrium for Zn + 4 OH- Zn. O 22 - + 2 H 2 O + 2 e- 7 p. H 14
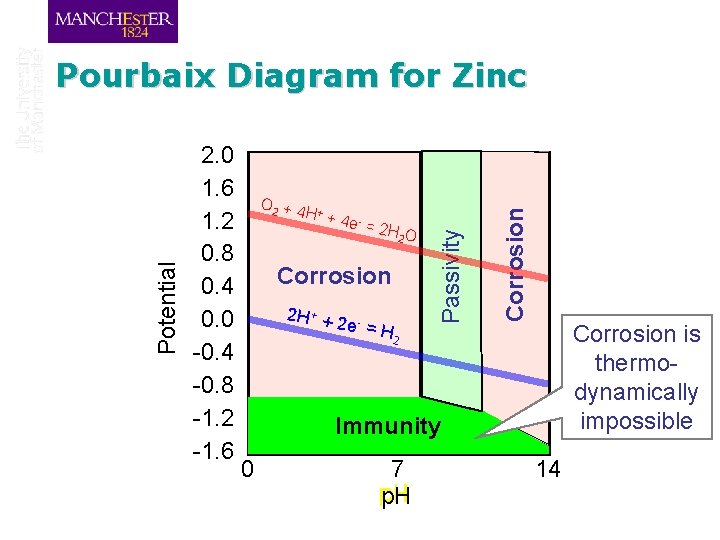
O 2 + 4 + H + 4 e - = 2 H O 2 Zn(OH)2 Corrosion 22+ H+ + 2 Zn stable e- = H stable solid 2 Corrosion 2. 0 1. 6 1. 2 0. 8 0. 4 0. 0 -0. 4 -0. 8 -1. 2 -1. 6 Passivity Potential Pourbaix Diagram for Zinc Zn. O 22 stable in solution Immunity Zn metal stable 0 7 p. H 14 Corrosion is thermodynamically impossible

What is Passivity? q Formation of an oxide/compound on a metal or alloy surface that is stable in the electrolyte, so that the metal is rendered “passive” in the environment (i. e. , the material is “passivated”) q Passivation limits corrosion q Generally, strong oxidising conditions are required for passivation q Note: Passive films have some electrical conduction

More on Passivity. . . q Many alloys exhibit passivity, such as Fe-Cr alloys • Fe-Cr alloys exhibit an increasing tendency to passivate as the Cr content increases! • The critical current density required for passivity in deaerated neutral solutions decreases as the Cr content is increased to 12 wt. %, beyond which it is constant • Fe-12+%Cr alloys and stainless steels are selfpassivating (no externally applied current nor strongly oxidizing conditions are required for passivation)

Breakdown of Passivity q Cl- ions can break down an existing passive film q It is generally difficult/impossible to passivate an alloy or metal in the presence of aggressive ions such as Cl-. • Aggressive ions affect the critical current density for passivity and may become incorporated into the oxide (and cause defects) q Local breakdown of a passive film can lead to. . Pitting Corrosion!

Immunity versus Passivity q Immunity means that the metal or alloy will not corrode – Period! It is thermodynamically impossible. q Passivity means that the alloy will not corrode under specific conditions, but it is not permanent. It is still thermodynamically possible for the alloy to corrode.
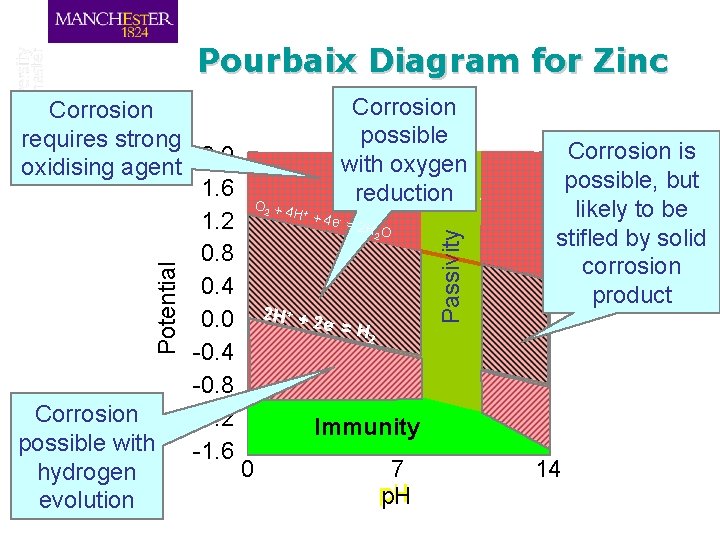
Corrosion possible requires strong 2. 0 with oxygen oxidising agent 1. 6 reduction O +4 H +4 e =2 1. 2 HO 0. 8 Zn(OH) Corrosion stable 0. 4 solid H + 2+ + 2 stable 0. 0 2 Zn e- = H 2 -0. 4 in solution -0. 8 Corrosion -1. 2 Immunity Zn metal stable possible with -1. 6 0 7 hydrogen p. H evolution + - Potential 2 Passivity 2 2 Corrosion Pourbaix Diagram for Zinc Zn. O 22 - Corrosion is possible, but likely to be stifled by solid corrosion product stable in solution 14
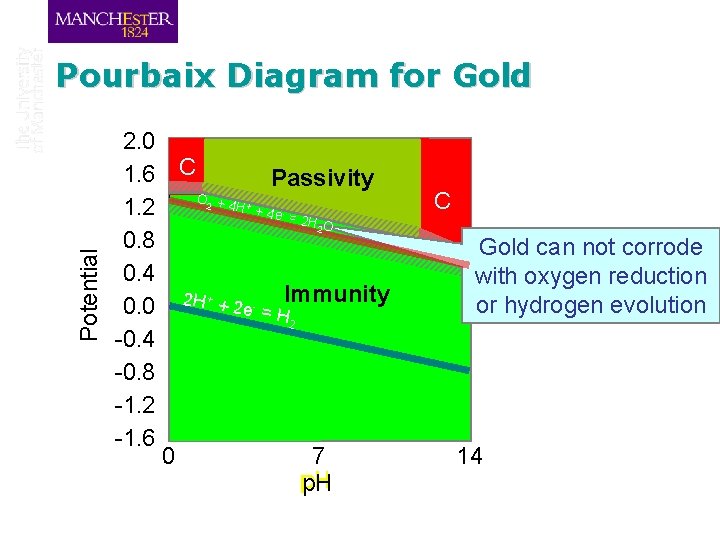
Pourbaix Diagram for Gold 2. 0 1. 6 C Passivity O +4 C H +4 1. 2 e =2 HO 0. 8 Gold can not corrode 0. 4 with oxygen reduction Gold metal stable or hydrogen evolution 0. 0 2 H + + 2 e - = HImmunity 2 -0. 4 -0. 8 -1. 2 -1. 6 0 7 14 p. H 2 + - Potential 2
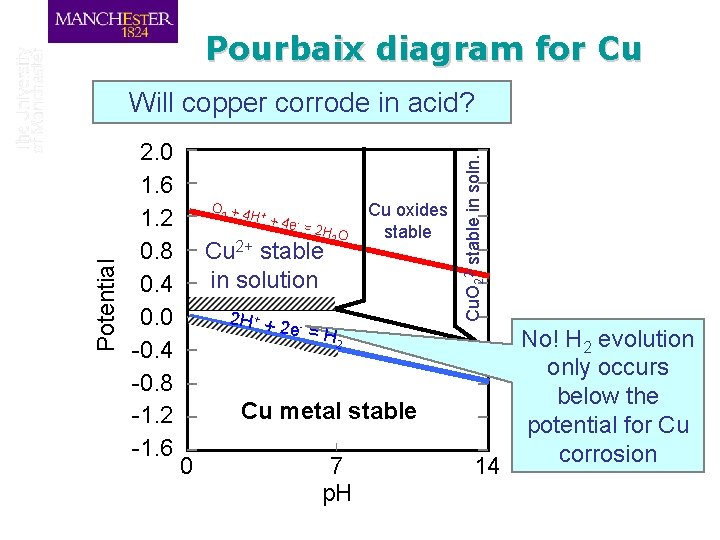
Pourbaix diagram for Cu 2. 0 1. 6 1. 2 0. 8 0. 4 0. 0 -0. 4 -0. 8 -1. 2 -1. 6 O 2 + 4 + H +4 e =2 H 2 O Cu 2+ stable in solution 2 H + + 2 Cu oxides stable e- = H Cu. O 22 - stable in soln. Potential Will copper corrode in acid? 2 Cu metal stable 0 7 p. H 14 No! H 2 evolution only occurs below the potential for Cu corrosion
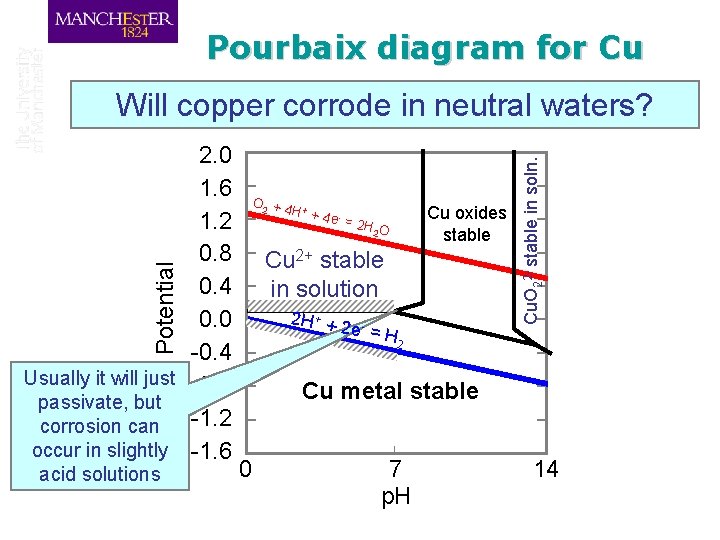
Pourbaix diagram for Cu O 2 + 4 + H + acid solutions 4 e - = Cu oxides stable 2 H O 2 Cu 2+ stable in solution Potential 2. 0 1. 6 1. 2 0. 8 0. 4 0. 0 -0. 4 Usually it will just -0. 8 passivate, but -1. 2 corrosion can occur in slightly -1. 6 2 H + + 2 e- = H Cu. O 22 - stable in soln. Will copper corrode in neutral waters? 2 Cu metal stable 0 7 p. H 14
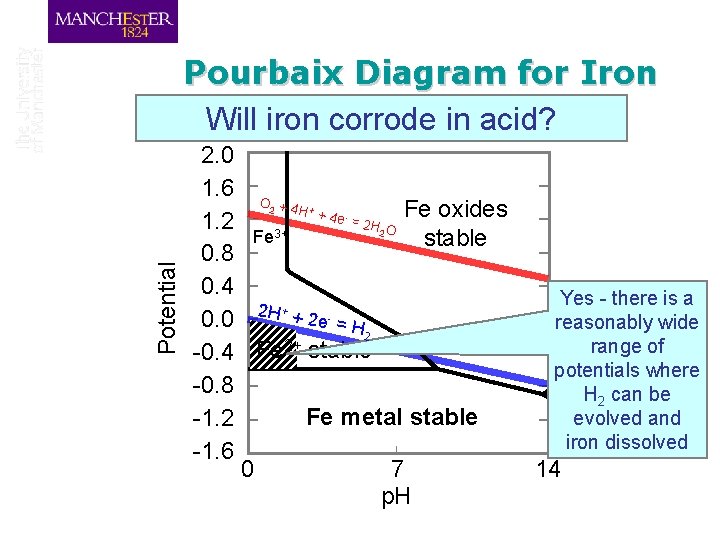
Pourbaix Diagram for Iron Will iron corrode in acid? 2. 0 1. 6 O + 4 H Fe oxides + 4 e = 2 H 1. 2 O Fe 3+ stable 0. 8 0. 4 + 0. 0 2 H + 2 e - = H 2 2+ -0. 4 Fe stable -0. 8 Fe metal stable -1. 2 -1. 6 0 7 p. H 2 + - Potential 2 Yes - there is a reasonably wide range of potentials where H 2 can be evolved and iron dissolved 14
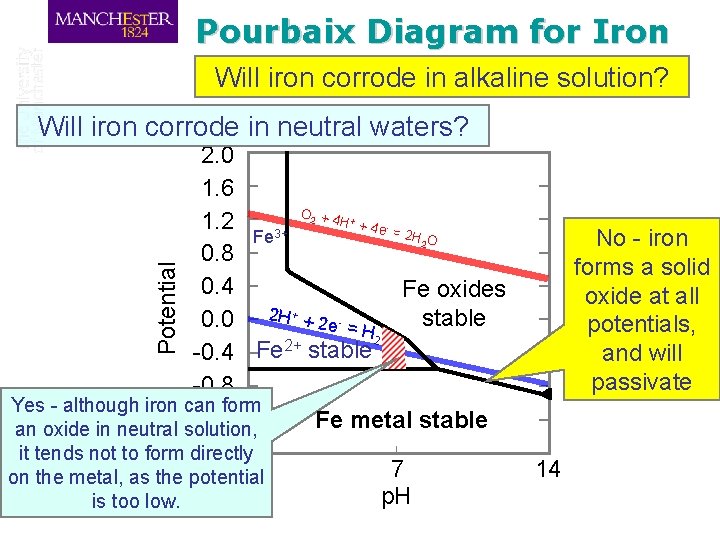
Pourbaix Diagram for Iron Will iron corrode in alkaline solution? Will iron corrode in neutral waters? 2. 0 1. 6 O +4 H +4 1. 2 e =2 3+ HO Fe 0. 8 0. 4 Fe oxides stable 0. 0 2 H + + 2 e - = H 2 2+ -0. 4 Fe stable -0. 8 Yes - although iron can form -1. 2 Fe metal stable an oxide in neutral solution, -1. 6 it tends not to form directly 0 7 on the metal, as the potential p. H is too low. 2 + - No - iron forms a solid oxide at all potentials, and will passivate Potential 2 14
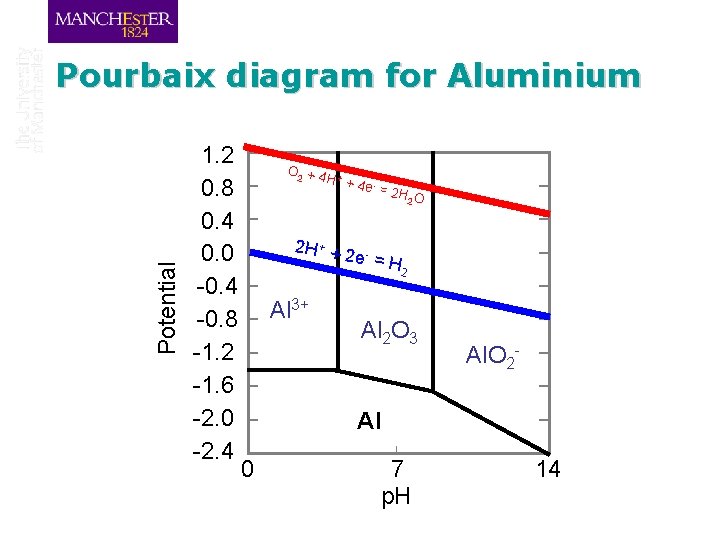
Potential Pourbaix diagram for Aluminium 1. 2 0. 8 0. 4 0. 0 -0. 4 -0. 8 -1. 2 -1. 6 -2. 0 -2. 4 O 2 + 4 + H + 4 e - = 2 H + + 2 2 H O 2 e- = H Al 3+ 2 Al 2 O 3 Al. O 2 - Al 0 7 p. H 14

How Fast will Corrosion Occur? q Corrosion kinetics • Concerned with the rates of corrosion reactions q Mixed potential theory: • The corrosion potential will be that potential at which the sum of all anodic (positive) and cathodic (negative) current on the electrode is zero q Polarization • The change in potential that is caused by the passage of a current

Types of Polarization q Activation Polarization q Concentration Polarization q Resistance Polarization

Types of Polarization q Activation Polarization • The polarization necessary for the electrochemical reaction to go at the given rate • Given by Tafel’s Law: E = potential at current i Eo = potential at current io b = Tafel slope (m. V/decade)
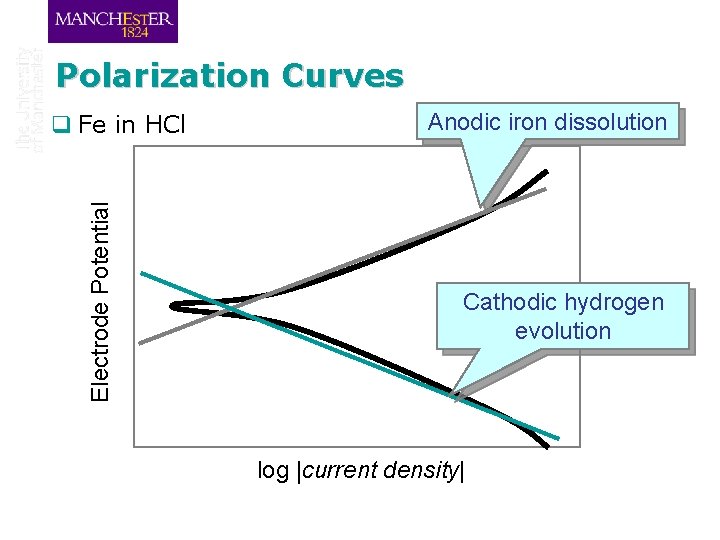
Polarization Curves Electrode Potential q Fe in HCl Anodic iron dissolution Cathodic hydrogen evolution log |current density|
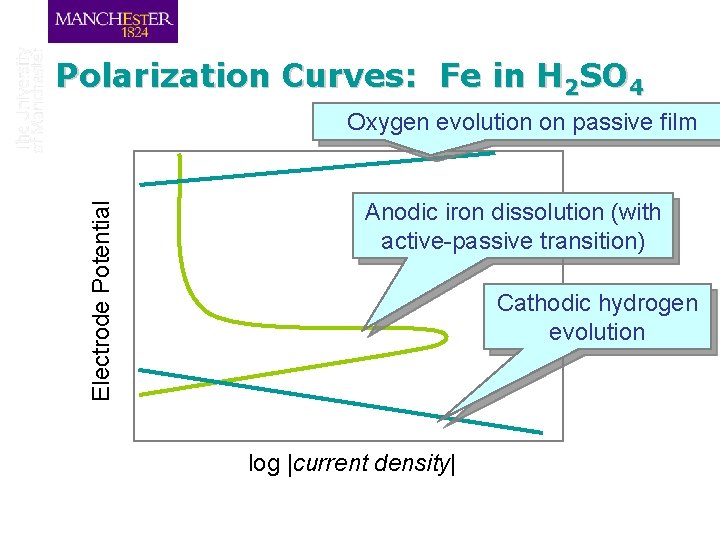
Polarization Curves: Fe in H 2 SO 4 Electrode Potential Oxygen evolution on passive film Anodic iron dissolution (with active-passive transition) Cathodic hydrogen evolution log |current density|
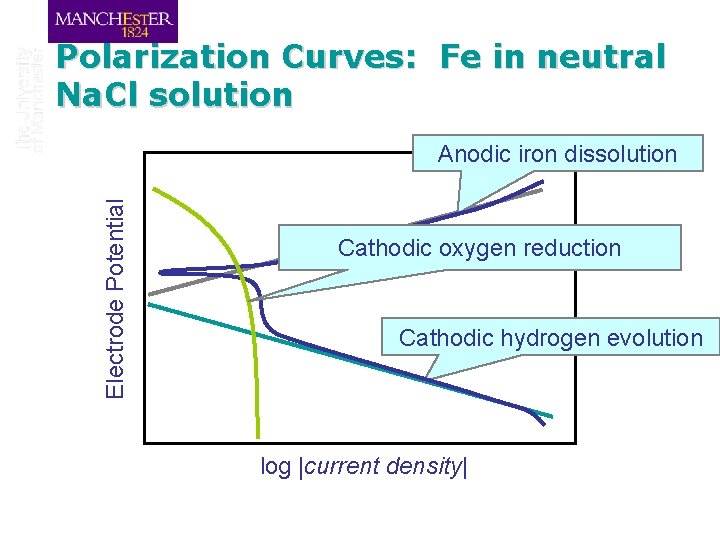
Polarization Curves: Fe in neutral Na. Cl solution Electrode Potential Anodic iron dissolution Cathodic oxygen reduction Cathodic hydrogen evolution log |current density|
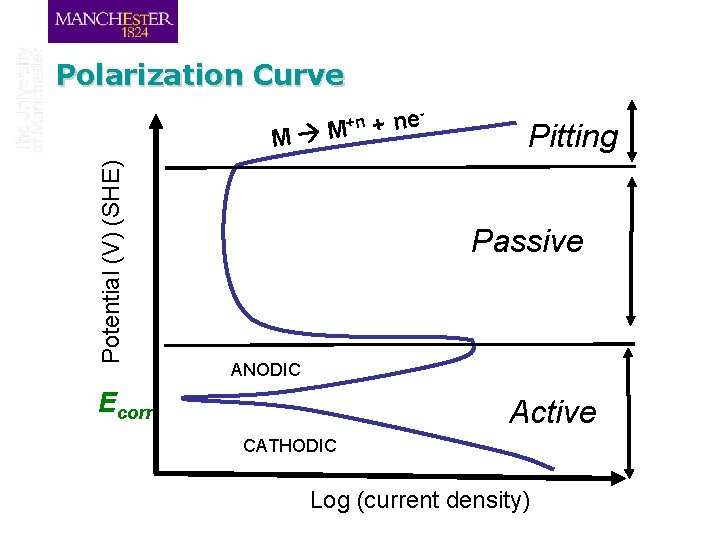
Polarization Curve +n Potential (V) (SHE) M M e n + Pitting Passive ANODIC Ecorr Active CATHODIC Log (current density)
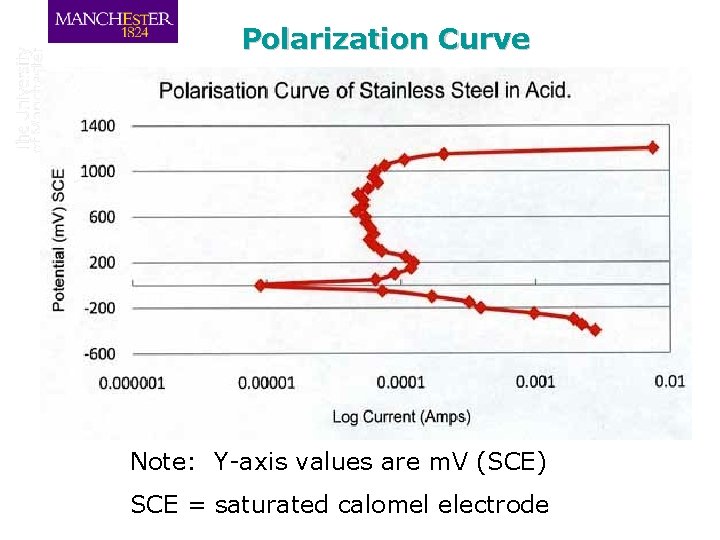
Polarization Curve Note: Y-axis values are m. V (SCE) SCE = saturated calomel electrode
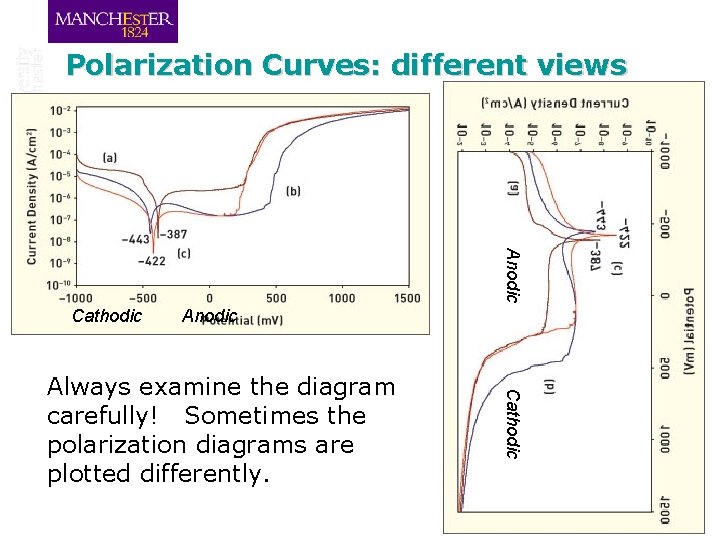
Polarization Curves: different views Anodic Cathodic Always examine the diagram carefully! Sometimes the polarization diagrams are plotted differently.

Reference Electrodes (1) • Provides a means to accurately measure the potential applied to the working electrode • Reference electrode is based upon a reaction which is generally at equilibrium and is not influenced by changes at the working electrode • The reaction must be reversible and non-polarizable (high exchange current density and low polarization resistance; that is, a large change in current will result in a very small change in potential) • Appropriate choice for Reference Electrode depends upon the application and environment • SCE (saturated calomel electrode); SHE (standard hydrogen electrode); Ag/Ag. Cl (for high T)

Reference Electrodes (2) q SHE: 2 H+ + 2 e- H 2 (gas) E = 0. 000 VSHE • Electrode potential is defined as 0 when all reactants and products are in their standard state (298 K, 1 bar pressure and 1 molar concentration) q SCE: Hg 2 Cl 2 + 2 e- 2 Hg + 2 Cl- Eo = 0. 268 VSHE q Ag/Ag. Cl: Ag. Cl + e- Ag + Cl- Eo = 0. 222 VSHE

Review q Corrosion involves ? reactions. q What is an Oxidation Reaction? LEO q What is a Reduction Reaction? GER q What will control the rate of a corrosion reaction? (2) q What is the driving force for corrosion reactions? q What does the Nernst Equation tell us? q What is a Pourbaix Diagram? What is the basis for a Pourbaix Diagram? q What are the 2 diagonal lines on the Pourbaix Diagram? What is stable in the region between those line?
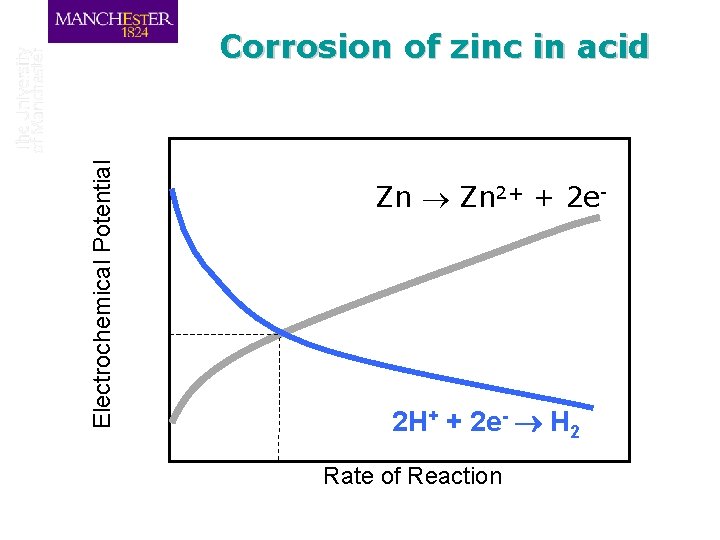
Electrochemical Potential Corrosion of zinc in acid Zn 2+ + 2 e- 2 H+ + 2 e- H 2 Rate of Reaction

Review (2) q What do we mean by “passivity”? q How can we passivate an alloy or metal? q What is special about a Fe-12 Cr alloy with respect to passivity? q What is a polarization curve? q What are the various regions on a polarization curve? q What does SHE mean? SCE?

Aqueous Corrosion (a few additional items)
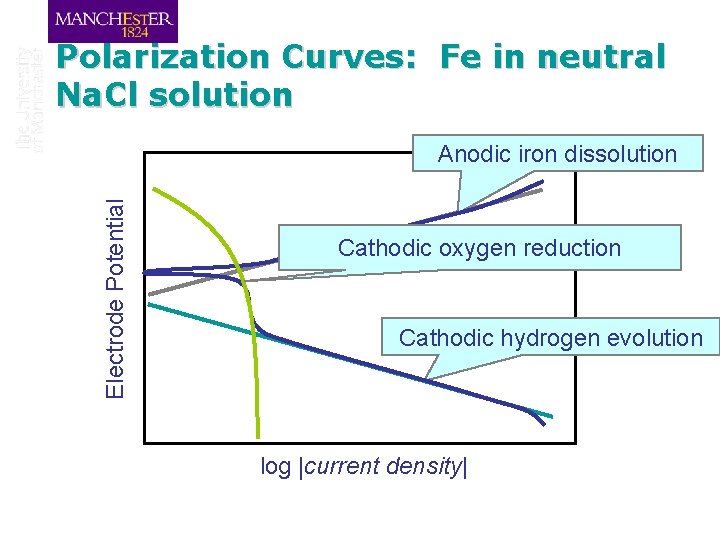
Polarization Curves: Fe in neutral Na. Cl solution Electrode Potential Anodic iron dissolution Cathodic oxygen reduction Cathodic hydrogen evolution log |current density|
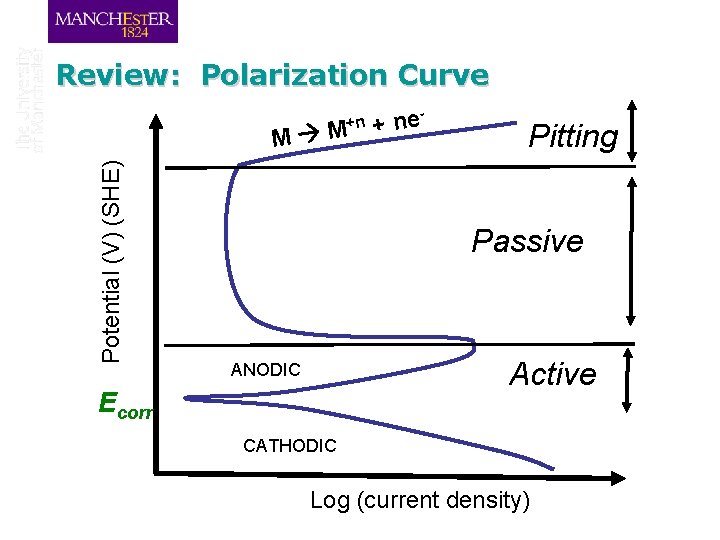
Review: Polarization Curve +n Potential (V) (SHE) M M e n + Pitting Passive Active ANODIC Ecorr CATHODIC Log (current density)

Concentration Polarization q Additional polarization caused by drop in concentration of a reactant at the electrode surface q As concentration falls, more polarization is needed to make the current flow q Eventually, no more current can flow because no more reactant can reach the metal, and a limiting current is reached

Concentration Polarization q Oxygen reduction is often affected by concentration polarization Electrode Potential Rate of cathodic oxygen reduction without concentration polarization Rate of cathodic oxygen reduction with concentration polarization Limiting current density log |current density|

Resistance Polarization q If there is a resistance between the anode and the cathode in a cell, then the current flowing through that resistance will cause a potential drop given by Ohm’s Law: V = IR q This is important for paint films and for high resistance solutions
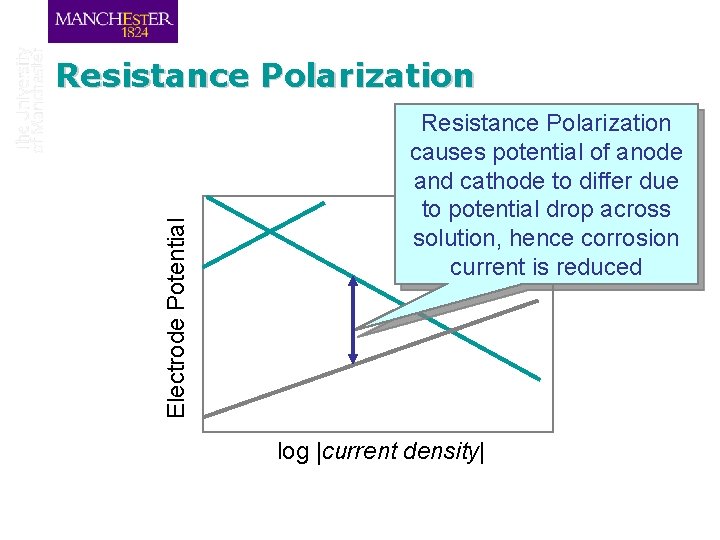
Electrode Potential Resistance Polarization causes potential of anode and cathode to differ due to potential drop across solution, hence corrosion current is reduced log |current density|

Passivation – from the Polarization Perspective q When a passive film is formed, this causes a marked drop in current density due to the resistance of the film and its effect as a barrier to diffusion q This effect is seen on the anodic curve
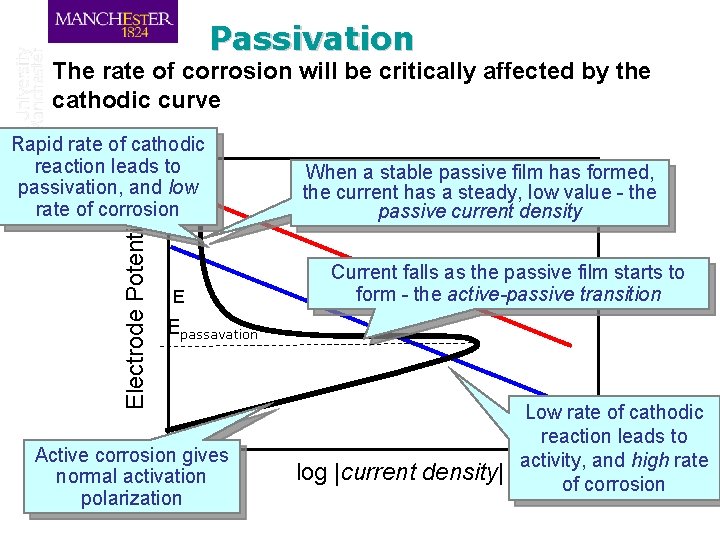
Passivation The rate of corrosion will be critically affected by the cathodic curve Electrode Potential Rapid rate of cathodic reaction leads to passivation, and low rate of corrosion E When a stable passive film has formed, the current has a steady, low value - the passive current density Current falls as the passive film starts to form - the active-passive transition Epassavation Active corrosion gives normal activation polarization log |current density| Low rate of cathodic reaction leads to activity, and high rate of corrosion
- Slides: 80