Aluminum Anodize By Randall Marks Aluminum Oxide Amorphous

Aluminum Anodize By: Randall Marks

Aluminum Oxide • Amorphous aluminum oxide (alumina, Al 2 O 3) forms in a layer 2 -3 nm thick when bare aluminum is exposed to oxygen • Protects from further oxidation • Stress causes fractures in the surface, but not peeling • How can this natural coating be improved upon?
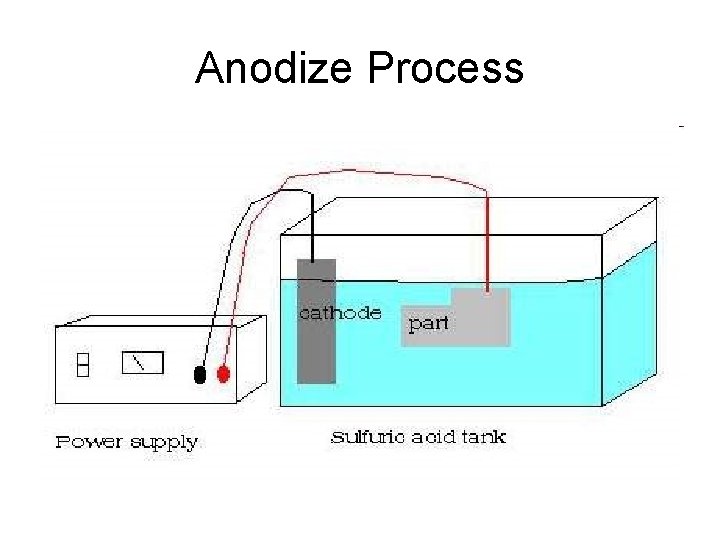
Anodize Process

Types of Anodize • Barrier Anodize – Near neutral electrolyte solution – Max 1 um thick • Porous Anodize – Acidic electrolyte solution – Can be several hundred um thick
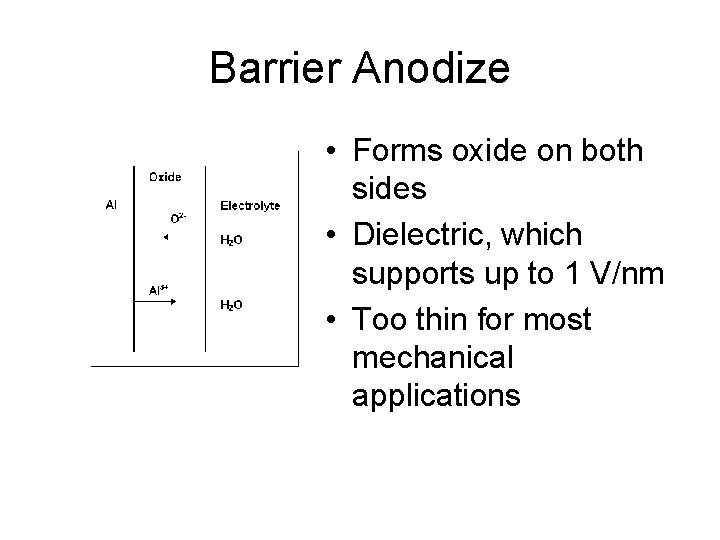
Barrier Anodize • Forms oxide on both sides • Dielectric, which supports up to 1 V/nm • Too thin for most mechanical applications
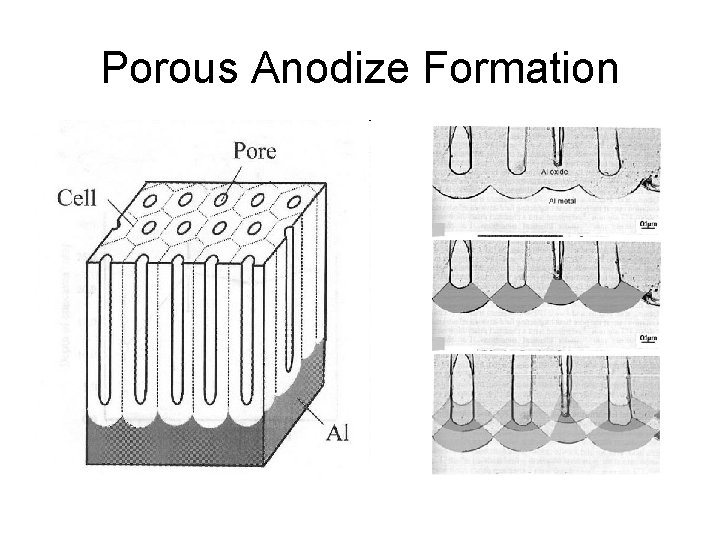
Porous Anodize Formation
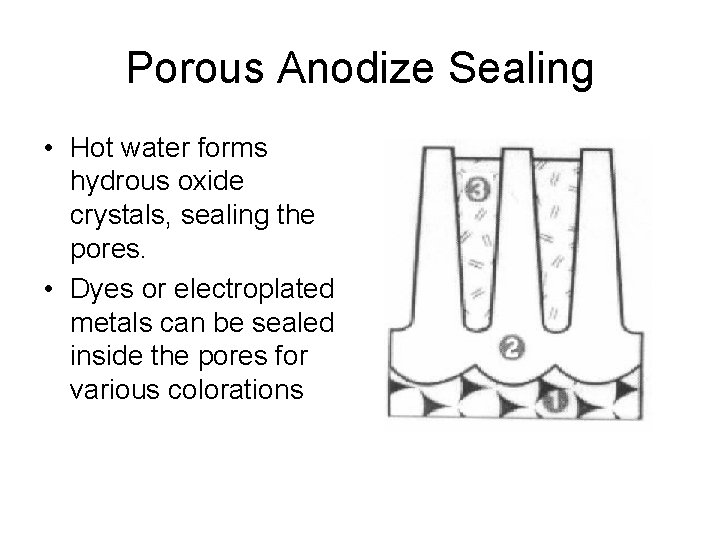
Porous Anodize Sealing • Hot water forms hydrous oxide crystals, sealing the pores. • Dyes or electroplated metals can be sealed inside the pores for various colorations

Hard Anodize • Hard Anodize Formation – Low temperature – Low acidity • Applications – Durable protective layer – Bearings • Not sealed to hold lubricants in the pores • Surface is second hardest material known

Optics Applications • Reduction of stray light/spectral reflections – Bead blasted black anodize – No outgassing – Black anodize not good for bearing type surfaces • Adhesive preparation – Phosphoric acid preparation – Left unsealed • Corrosion resistance

Important Application Considerations • AMS governing specs: – AMS 2469 -AMS 2474 • Part grows by approximately half the oxide thickness • Surface Young’s Modulus around 300 MPa. • Does not protect against all chemicals – Highly susceptible to strong acids and bases

References • Case Western Reserve University Electrochemistry Encyclopedia – http: //electrochem. cwru. edu/ed/encycl/art-a 02 anodizing. htm • AACOA Inc. Anodizing Presentation – http: //www. aacoa. com/presentations/anodizin g_presentation-c. pdf
- Slides: 11