Redox potentials Electrochemistry ICS Redox Reactions Oxidation loss























![EXAMPLE: What is the cell potential for the Daniel's cell when the [Zn+2] = EXAMPLE: What is the cell potential for the Daniel's cell when the [Zn+2] =](https://slidetodoc.com/presentation_image_h/12d7301410ae3c5b6393f427f221f116/image-36.jpg)
![Nernst Equation [H + ] acid side ® [H + ] base side RT Nernst Equation [H + ] acid side ® [H + ] base side RT](https://slidetodoc.com/presentation_image_h/12d7301410ae3c5b6393f427f221f116/image-37.jpg)
- Slides: 37

Redox potentials Electrochemistry ICS

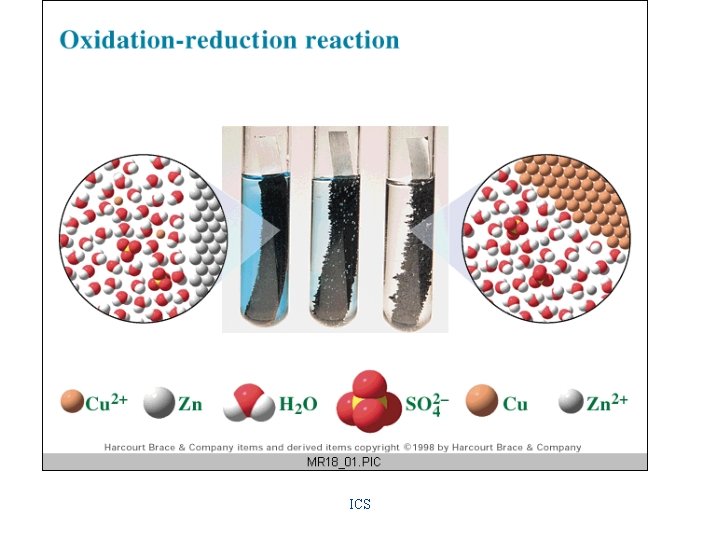
Redox Reactions Oxidation • loss of electrons Reduction • gain of electrons oxidizing agent • substance that cause oxidation by being reduced reducing agent • substance that cause oxidation by being reduced ICS

Electrochemistry In the broadest sense, electrochemistry is the study of chemical reactions that produce electrical effects and of the chemical phenomena that are caused by the action of currents or voltages.

ICS

Voltaic Cells • harnessed chemical reaction which produces an electric current

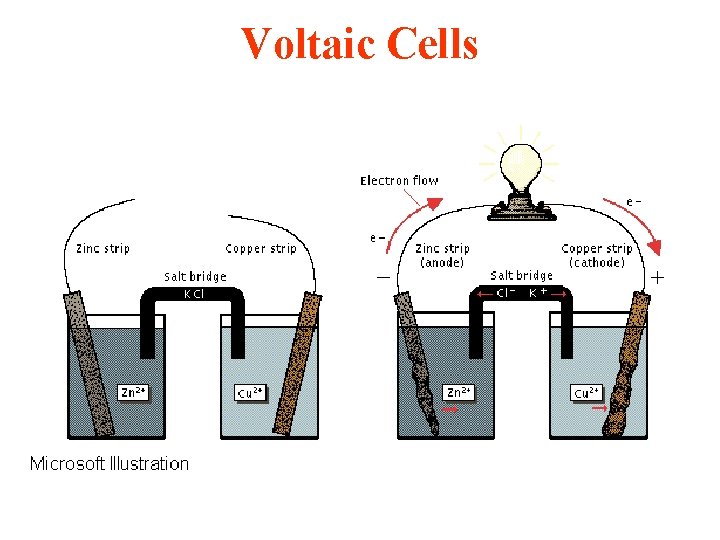
Voltaic Cells and Cell Reactions Daniel's Cell Zn(s) + Cu+2(aq) ---> Zn+2(aq) + Cu(s) oxidation half reaction anode Zn(s) ---> Zn+2(aq) + 2 ereduction half reaction cathode Cu+2(aq) + 2 e- ---> Cu(s)

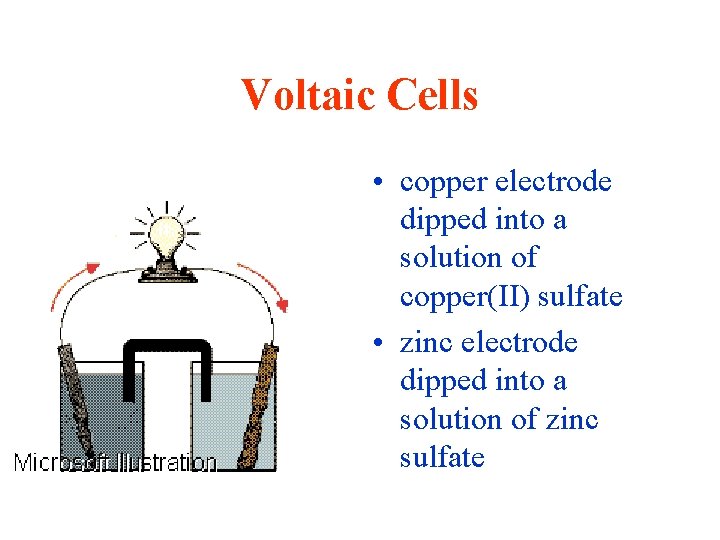
Voltaic Cells • copper electrode dipped into a solution of copper(II) sulfate • zinc electrode dipped into a solution of zinc sulfate

Voltaic Cells

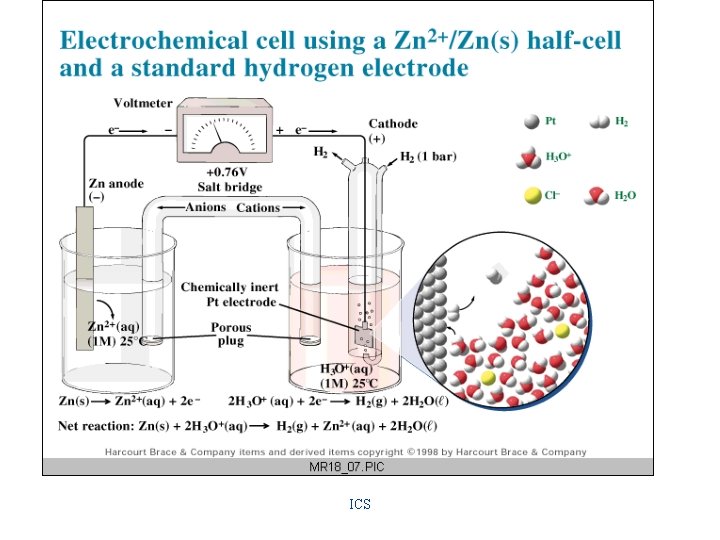
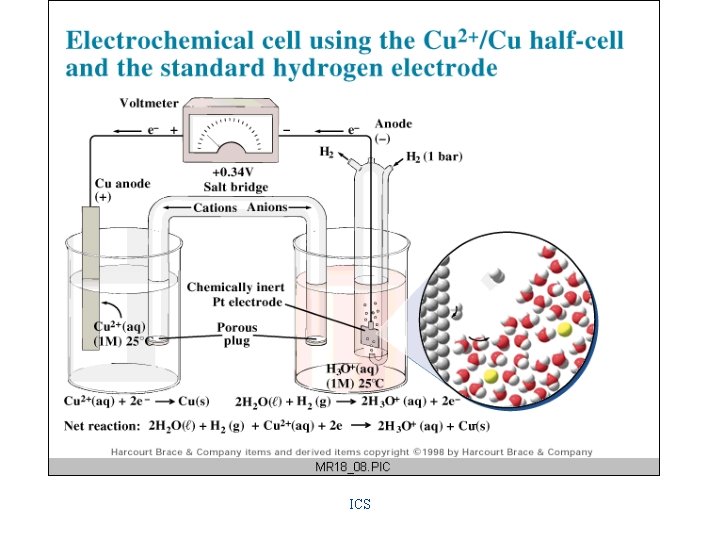
Hydrogen Electrode • consists of a platinum electrode covered with a fine powder of platinum around which H 2(g) is bubbled. Its potential is defined as zero volts. Hydrogen Half-Cell H 2(g) = 2 H+(aq) + 2 ereversible reaction

ICS

ICS

Standard Reduction Potentials • the potential under standard conditions (25 o. C with all ions at 1 M concentrations and all gases at 1 atm pressure) of a halfreaction in which reduction is occurring

Some Standard Reduction Potentials Table 18 -1, pg 837 Li+ + e- ---> Li Zn+2 + 2 e- ---> Zn Fe+2 + 2 e- ---> Fe 2 H+(aq) + 2 e- ---> H 2(g) Cu+2 + 2 e- ---> Cu O 2(g) + 4 H+(aq) + 4 e- ---> 2 H 2 O(l) F 2 + 2 e- ---> 2 F- -3. 045 v -0. 763 v -0. 44 v 0. 00 v +0. 337 v +1. 229 v +2. 87 v

If the reduction of mercury (I) in a voltaic cell is desired, the half reaction is: Which of the following reactions could be used as the anode (oxidation)? A, B ICS

Cell Potential • the potential difference, in volts, between the electrodes of an electrochemical cell • Direction of Oxidation-Reduction Reactions • positive value indicates a spontaneous reaction

Standard Cell Potential • the potential difference, in volts, between the electrodes of an electrochemical cell when the all concentrations of all solutes is 1 molar, all the partial pressures of any gases are 1 atm, and the temperature at 25 o. C

Cell Diagram • the shorthand representation of an electrochemical cell showing the two halfcells connected by a salt bridge or porous barrier, such as: Zn(s)/Zn. SO 4(aq)//Cu. SO 4(aq)/Cu(s) anode cathode

Metal Displacement Reactions • solid of more reactive metals will displace ions of a less reactive metal from solution • relative reactivity based on potentials of half reactions • metals with very different potentials react most vigorously

Ag+ + e- --->Ag E°= 0. 80 V Cu 2+ + 2 e- ---> Cu E°= 0. 34 V Will Ag react with Cu 2+? yes, no Will Cu react with Ag+? yes, no ICS

Gibbs Free Energy and Cell Potential DG = - n. FE where n => number of electrons changed F => Faraday’s constant E => cell potential

Applications of Electrochemical Cells Batteries – device that converts chemical energy into electricity Primary Cells – non-reversible electrochemical cell – non-rechargeable cell Secondary Cells – reversible electrochemical cell – rechargeable cell

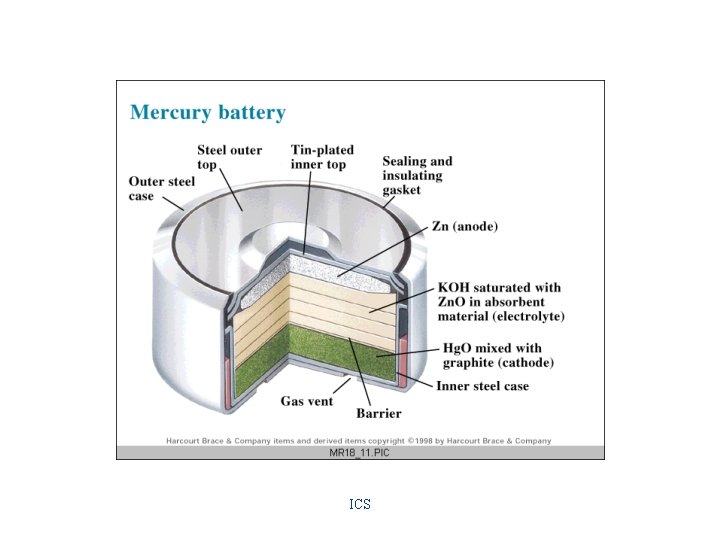
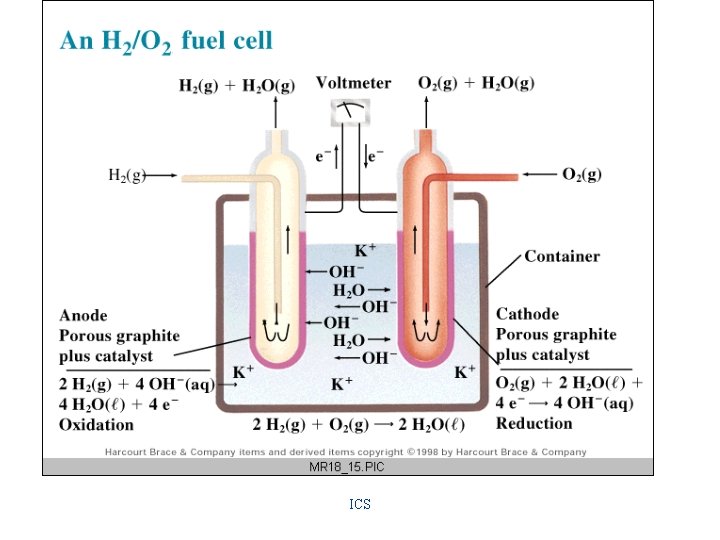
Applications of Electrochemical Cells Batteries Primary Cells "dry" cell & alkaline cell 1. 5 v/cell mercury cell 1. 34 v/cell fuel cell 1. 23 v/cell Secondary Cells lead-acid (automobile battery) 2 v/cell Ni. Cad 1. 25 v/cell

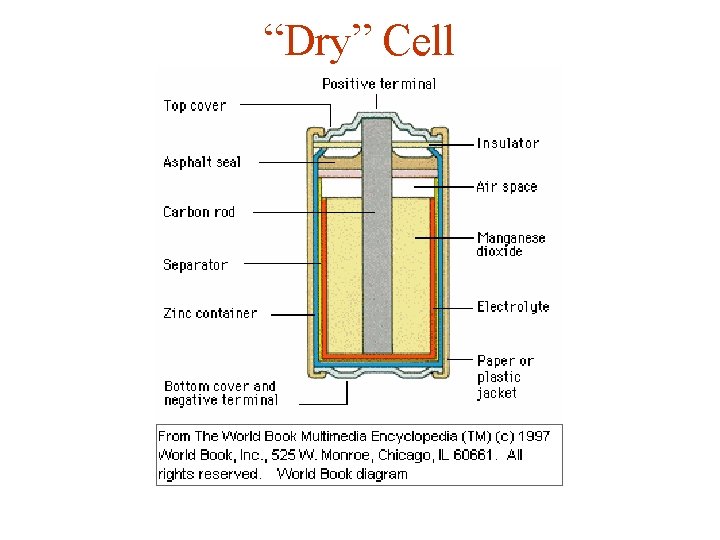
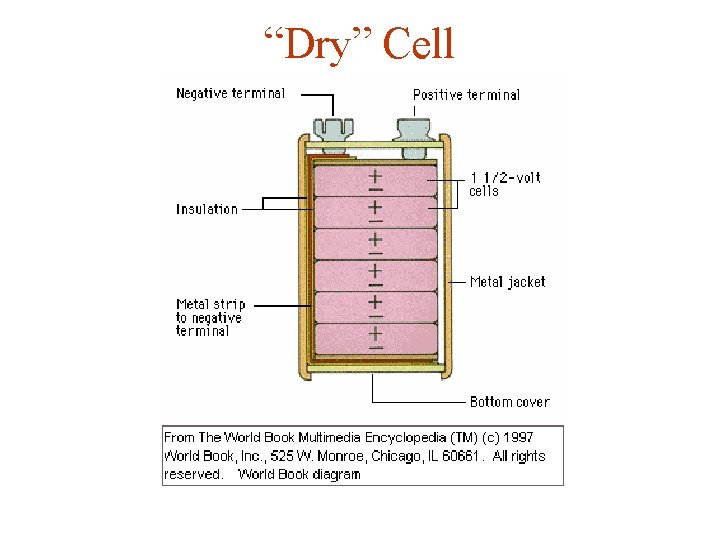
“Dry” Cell

“Dry” Cell

“Flash Light” Batteries "Dry" Cell Zn(s) + 2 Mn. O 2(s) + 2 NH 4+ -----> Zn+2(aq) + 2 Mn. O(OH)(s) + 2 NH 3 Alkaline Cell Zn(s) + 2 Mn. O 2(s) ---> Zn. O(s) + Mn 2 O 3(s)

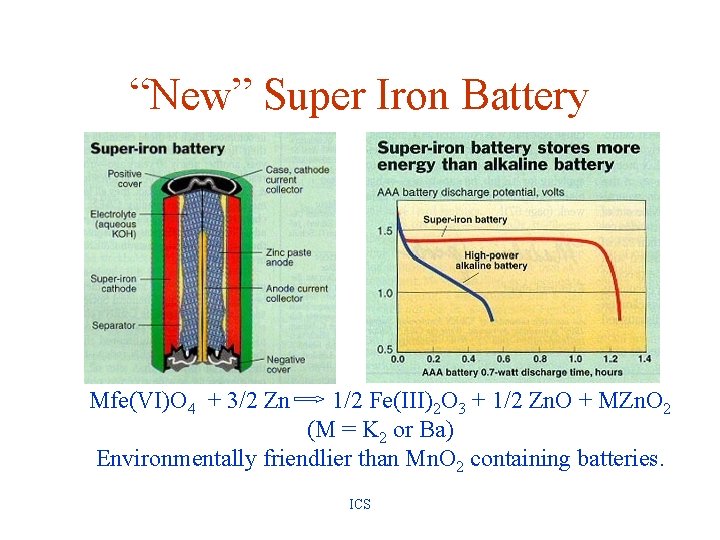
“New” Super Iron Battery Mfe(VI)O 4 + 3/2 Zn 1/2 Fe(III)2 O 3 + 1/2 Zn. O + MZn. O 2 (M = K 2 or Ba) Environmentally friendlier than Mn. O 2 containing batteries. ICS

ICS

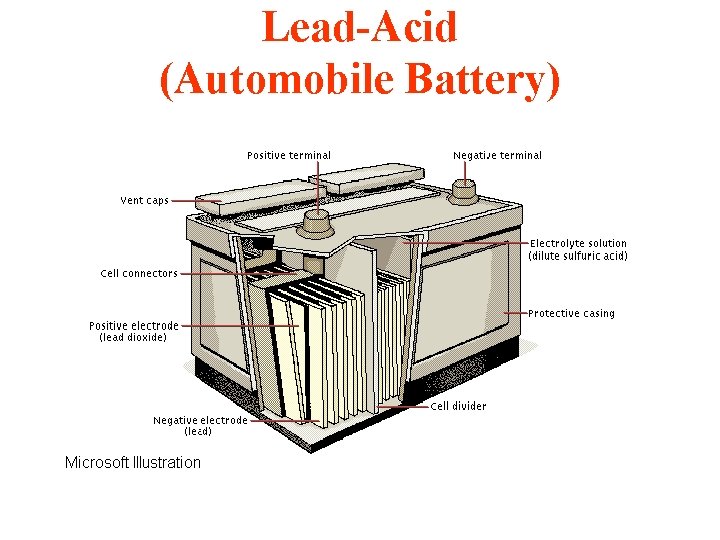
Lead-Acid (Automobile Battery)

Lead-Acid (Automobile Battery) Pb(s) + Pb. O 2(s) + 2 H 2 SO 4 = 2 Pb. SO 4(s) + 2 H 2 O 2 v/cell

Nickel-Cadmium (Ni-Cad) Cd(s) + 2 Ni(OH)3(s) = Cd(OH)2(s) + 2 Ni(OH)2(s) Ni. Cad 1. 25 v/cell

ICS

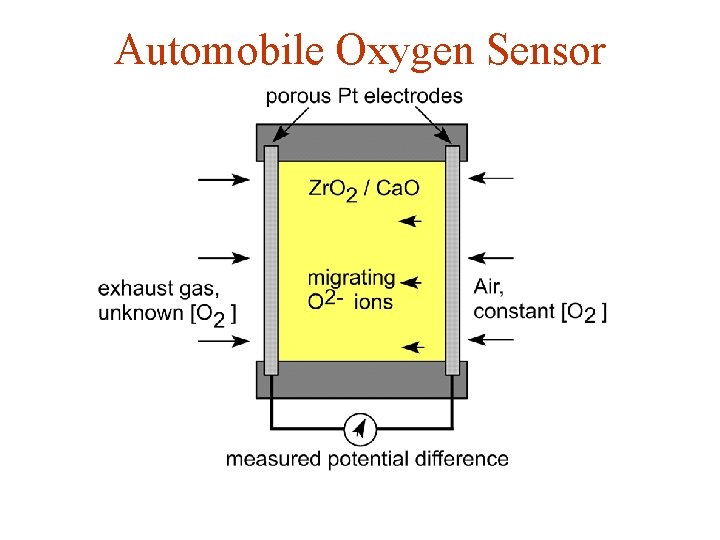
Automobile Oxygen Sensor

Automobile Oxygen Sensor • see Oxygen Sensor Movie from Solid-State Resources CD-ROM

p. H Meter p. H = (Eglass electrode - constant)/0. 0592

Effect of Concentration on Cell Voltage: The Nernst Equation Ecell = Eocell - (RT/n. F)ln Q Ecell = Eocell - (0. 0592/n)log Q where Q => reaction quotient Q = [products]/[reactants]
![EXAMPLE What is the cell potential for the Daniels cell when the Zn2 EXAMPLE: What is the cell potential for the Daniel's cell when the [Zn+2] =](https://slidetodoc.com/presentation_image_h/12d7301410ae3c5b6393f427f221f116/image-36.jpg)
EXAMPLE: What is the cell potential for the Daniel's cell when the [Zn+2] = 10 [Cu+2] ? Q = ([Zn+2]/[Cu+2] = (10 [Cu+2])/[Cu+2] = 10 Eo = (0. 34 V)Cu couple + (-(-0. 76 V)Zn couple n = 2, 2 electron change Ecell = Eocell - (0. 0257/n)ln Q thus Ecell = (1. 10 - (0. 0257/2)ln 10) V Ecell = (1. 10 - (0. 0257/2)2. 303) V Ecell = (1. 10 - 0. 0296) V = 1. 07 V
![Nernst Equation H acid side H base side RT Nernst Equation [H + ] acid side ® [H + ] base side RT](https://slidetodoc.com/presentation_image_h/12d7301410ae3c5b6393f427f221f116/image-37.jpg)
Nernst Equation [H + ] acid side ® [H + ] base side RT – 2. 3 RT log[H + ] n. F ln Q = F acid side E = E o – [h + ] p-type side ® [h + ] n-type side E (in volts) = [h + ] n-type side – 2. 3 RT log F [h + ] p-type side