Matter I States of Matter Kinetic Molecular Theory

















- Slides: 18

Matter I. States of Matter Kinetic Molecular Theory States of Matter

A. Kinetic Molecular Theory �KMT �Particles of matter are always in motion. �The kinetic energy (speed) of these particles increases as temperature increases.

B. Four States of Matter �Solids �very low KE - particles vibrate but can’t move around �fixed shape �fixed volume

B. Four States of Matter �Liquids �low KE - particles can move around but are still close together �variable shape �fixed volume

B. Four States of Matter �Gases �high KE - particles can separate and move throughout container �variable shape �variable volume

B. Four States of Matter �Plasma �very high KE - particles collide with enough energy to break into charged particles (+/-) �What isn’t matter? �Light �Energy

Matter II. Classification of Matter Flowchart Pure Substances Mixtures

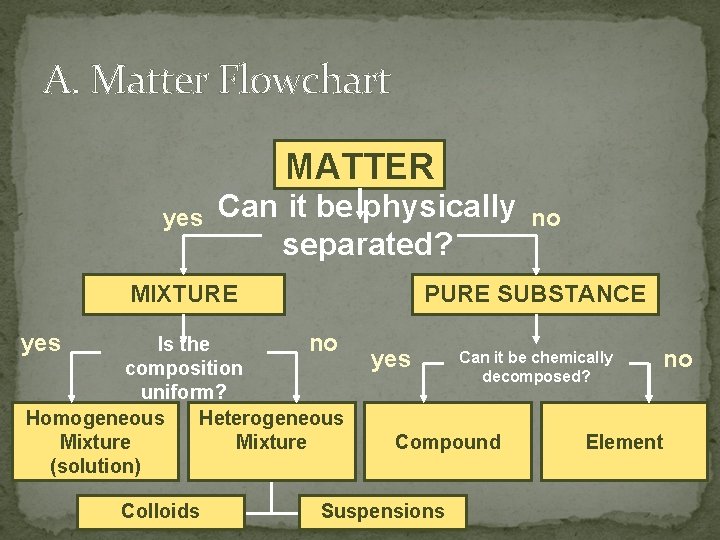
A. Matter Flowchart MATTER yes Can it be physically separated? MIXTURE yes PURE SUBSTANCE Is the no composition uniform? Homogeneous Heterogeneous Mixture (solution) Colloids no yes Can it be chemically decomposed? Compound Suspensions no Element

A. Matter Flowchart �Examples: �sodium element �pepper hetero. mixture �sugar (sucrose) �paint �soda compound hetero. mixture solution

B. Pure Substances �Pure substances may either be an element or a compound �Element �composed of identical atoms �EX: copper wire, aluminum foil

B. Pure Substances �Compound �composed of 2 or more elements in a fixed ratio �properties differ from those of individual elements �EX: table salt (Na. Cl)

B. Pure Substances �Law of Definite Composition �A given compound always contains the same, fixed ratio of elements. �Law of Multiple Proportions �Elements can combine in different ratios to form different compounds.

B. Pure Substances �For example… Ratio: 1: 1 Ratio: 1: 2 Two different compounds, each has a definite composition.

C. Mixtures �A mixture is a combination of 2 or more pure substances. Heterogeneous Homogeneous

C. Mixtures �Solution �homogeneous �very small particles �no Tyndall effect Tyndall Effect wparticles don’t settle w. EX: rubbing alcohol

C. Mixtures �Colloid �heterogeneous �medium-sized particles �Tyndall effect �particles don’t settle �EX: milk

C. Mixtures �Suspension �heterogeneous �large particles �Tyndall effect �particles settle �EX: fresh-squeezed lemonade

C. Mixtures �Examples: �mayonnaise colloid �muddy water suspension �fog colloid �saltwater �Italian salad dressing solution suspension