Continuum of care en cncer de colon metastsico










- Slides: 52

“Continuum of care” en cáncer de colon metastásico no curable Mauricio Lema Medina MD Clínica de Oncología Astorga – Clínica SOMA – Medicáncer Medellín, Colombia

Quimioterapia A qué llegamos?

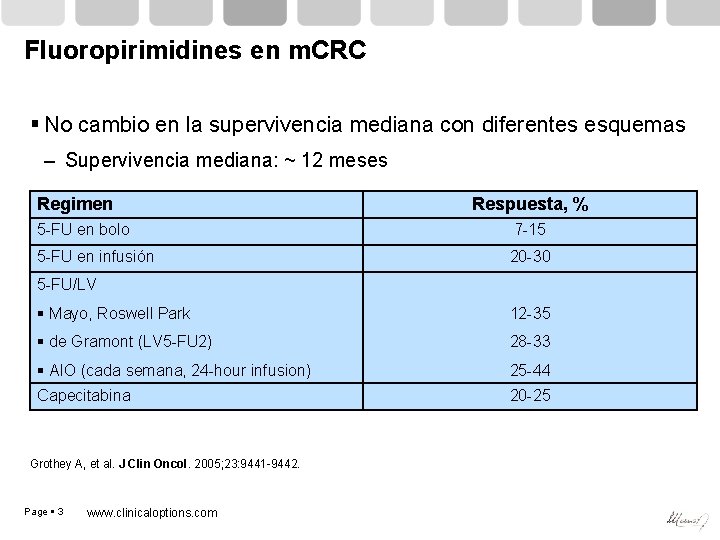
Fluoropirimidines en m. CRC No cambio en la supervivencia mediana con diferentes esquemas – Supervivencia mediana: ~ 12 meses Regimen Respuesta, % 5 -FU en bolo 7 -15 5 -FU en infusión 20 -30 5 -FU/LV Mayo, Roswell Park 12 -35 de Gramont (LV 5 -FU 2) 28 -33 AIO (cada semana, 24 -hour infusion) Capecitabina 25 -44 Grothey A, et al. J Clin Oncol. 2005; 23: 9441 -9442. Page 3 www. clinicaloptions. com 20 -25

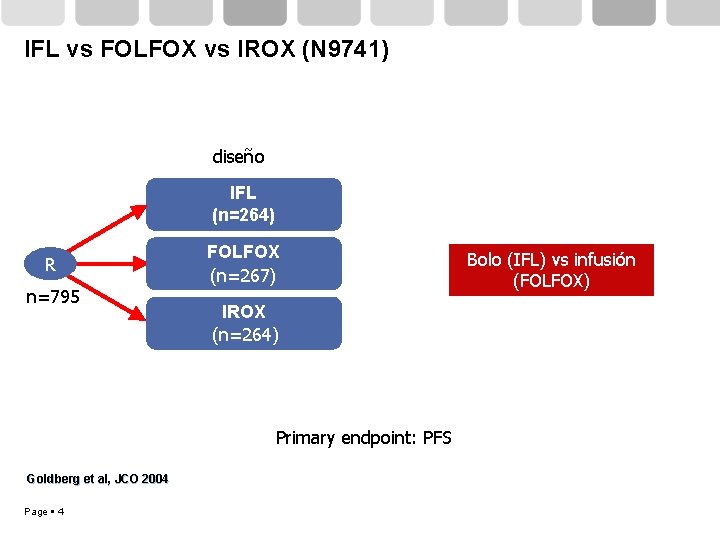
IFL vs FOLFOX vs IROX (N 9741) diseño IFL (n=264) R n=795 FOLFOX (n=267) IROX (n=264) Primary endpoint: PFS Goldberg et al, JCO 2004 Page 4 Bolo (IFL) vs infusión (FOLFOX)

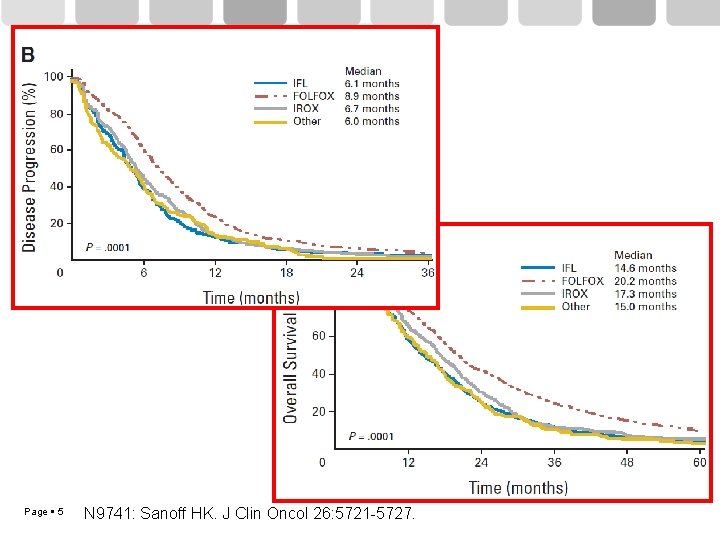
Page 5 N 9741: Sanoff HK. J Clin Oncol 26: 5721 -5727.

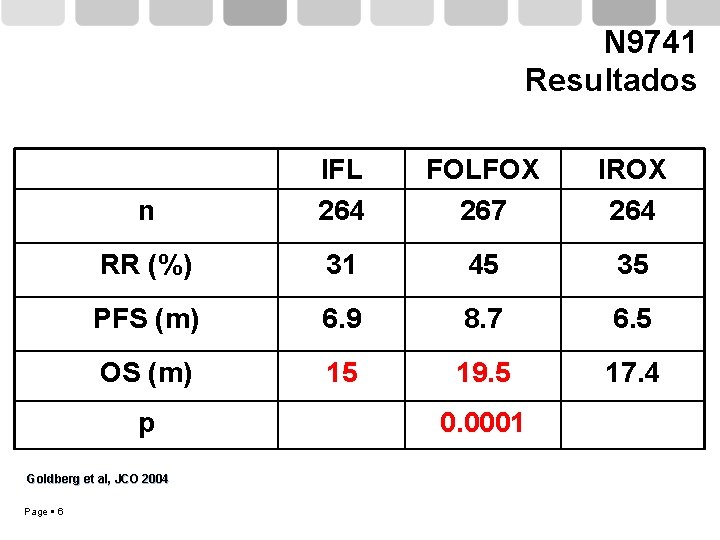
N 9741 Resultados n IFL 264 FOLFOX 267 IROX 264 RR (%) 31 45 35 PFS (m) 6. 9 8. 7 6. 5 OS (m) 15 19. 5 17. 4 p Goldberg et al, JCO 2004 Page 6 0. 0001

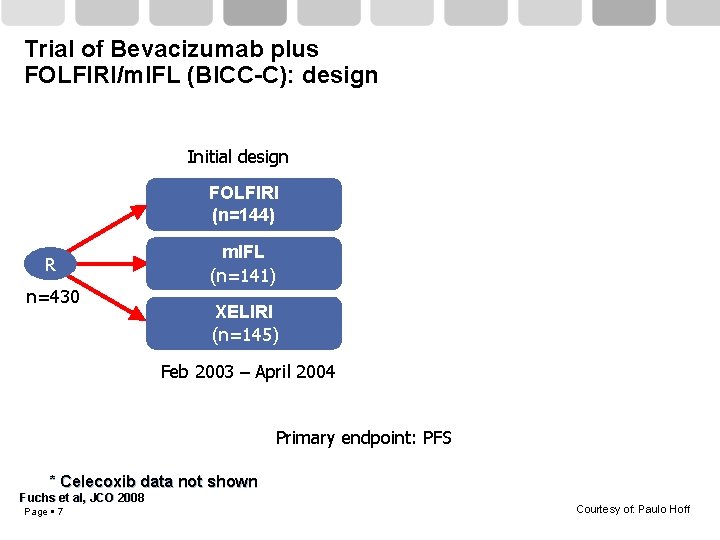
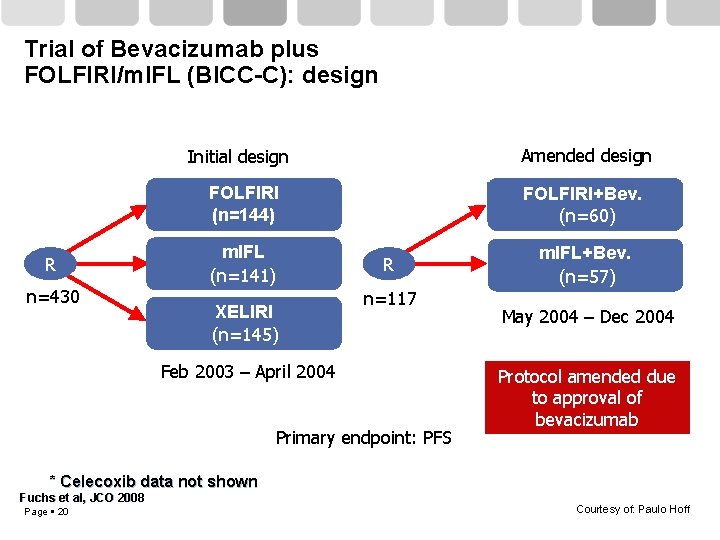
Trial of Bevacizumab plus FOLFIRI/m. IFL (BICC-C): design Initial design FOLFIRI (n=144) R n=430 m. IFL (n=141) XELIRI (n=145) Feb 2003 – April 2004 Primary endpoint: PFS * Celecoxib data not shown Fuchs et al, JCO 2008 Page 7 Courtesy of: Paulo Hoff

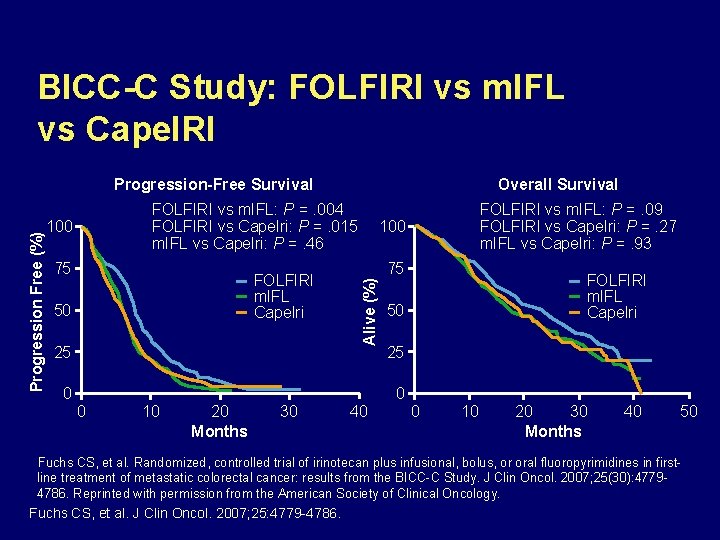
BICC-C Study: FOLFIRI vs m. IFL vs Cape. IRI FOLFIRI vs m. IFL: P =. 004 FOLFIRI vs Capelri: P =. 015 m. IFL vs Capelri: P =. 46 100 75 FOLFIRI m. IFL Capelri 50 25 0 Overall Survival 0 10 20 Months 30 FOLFIRI vs m. IFL: P =. 09 FOLFIRI vs Capelri: P =. 27 m. IFL vs Capelri: P =. 93 100 75 Alive (%) Progression Free (%) Progression-Free Survival 40 FOLFIRI m. IFL Capelri 50 25 0 0 10 20 30 Months 40 50 Fuchs CS, et al. Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in firstline treatment of metastatic colorectal cancer: results from the BICC-C Study. J Clin Oncol. 2007; 25(30): 47794786. Reprinted with permission from the American Society of Clinical Oncology. Fuchs CS, et al. J Clin Oncol. 2007; 25: 4779 -4786.

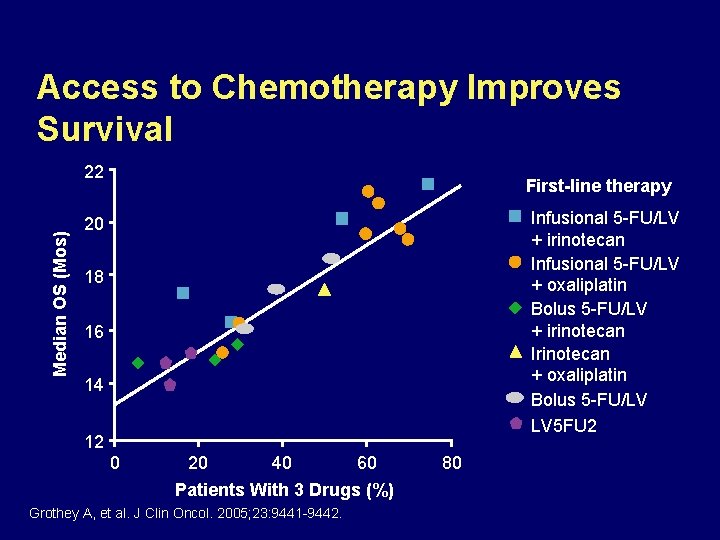
Access to Chemotherapy Improves Survival Median OS (Mos) 22 First-line therapy Infusional 5 -FU/LV + irinotecan Infusional 5 -FU/LV + oxaliplatin Bolus 5 -FU/LV + irinotecan Irinotecan + oxaliplatin Bolus 5 -FU/LV LV 5 FU 2 20 18 16 14 12 0 20 40 60 Patients With 3 Drugs (%) Grothey A, et al. J Clin Oncol. 2005; 23: 9441 -9442. 80

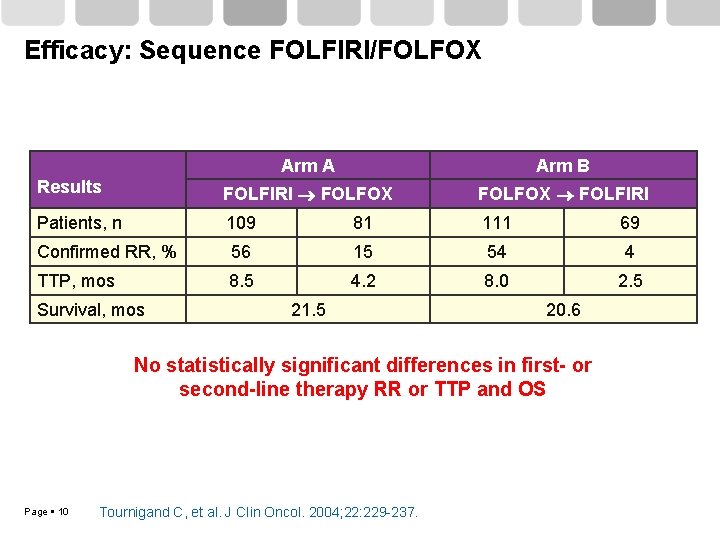
Efficacy: Sequence FOLFIRI/FOLFOX Arm A Arm B Results FOLFIRI FOLFOX FOLFIRI Patients, n 109 81 111 69 Confirmed RR, % 56 15 54 4 TTP, mos 8. 5 4. 2 8. 0 2. 5 Survival, mos 21. 5 20. 6 No statistically significant differences in first- or second-line therapy RR or TTP and OS Page 10 Tournigand C, et al. J Clin Oncol. 2004; 22: 229 -237.

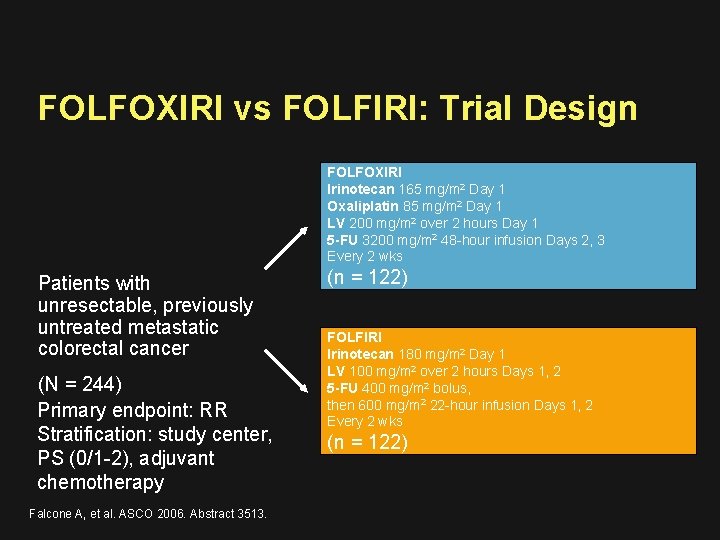
FOLFOXIRI vs FOLFIRI: Trial Design FOLFOXIRI Irinotecan 165 mg/m 2 Day 1 Oxaliplatin 85 mg/m 2 Day 1 LV 200 mg/m 2 over 2 hours Day 1 5 -FU 3200 mg/m 2 48 -hour infusion Days 2, 3 Every 2 wks Patients with unresectable, previously untreated metastatic colorectal cancer (N = 244) Primary endpoint: RR Stratification: study center, PS (0/1 -2), adjuvant chemotherapy Falcone A, et al. ASCO 2006. Abstract 3513. (n = 122) FOLFIRI Irinotecan 180 mg/m 2 Day 1 LV 100 mg/m 2 over 2 hours Days 1, 2 5 -FU 400 mg/m 2 bolus, then 600 mg/m 2 22 -hour infusion Days 1, 2 Every 2 wks (n = 122)

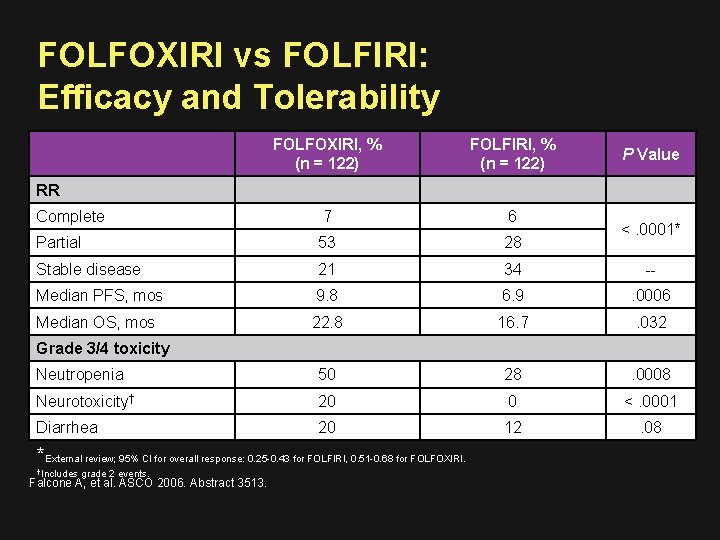
FOLFOXIRI vs FOLFIRI: Efficacy and Tolerability FOLFOXIRI, % (n = 122) FOLFIRI, % (n = 122) Complete 7 6 Partial 53 28 Stable disease 21 34 -- Median PFS, mos 9. 8 6. 9 . 0006 Median OS, mos 22. 8 16. 7 . 032 Neutropenia 50 28 . 0008 Neurotoxicity† 20 0 <. 0001 Diarrhea 20 12 . 08 P Value RR <. 0001* Grade 3/4 toxicity *External review; 95% CI for overall response: 0. 25 -0. 43 for FOLFIRI, 0. 51 -0. 68 for FOLFOXIRI. †Includes grade 2 events. Falcone A, et al. ASCO 2006. Abstract 3513.

Quimioterapia más Bevacizumab

VEGF es expresado durante toda la historia natural b. FGF TGFb-1 VEGF TGFb-1 PIGF b. FGF TGFb-1 VEGF PIGF PD-ECGF Pleiotrophin Evolución tumoral b. FGF = basic fibroblast growth factor TGFb-1 = transforming growth factor b-1 PIGF = placenta growth factor PD-ECGF = platelet-derived endothelial cell growth factor Page 14 www. clinicaloptions. com Adapted from Folkman. Cancer. Principles and practice of oncology 2005

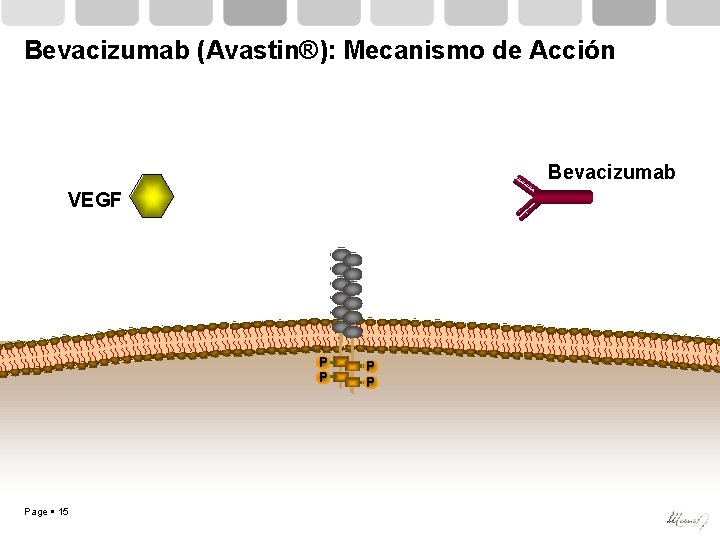
Bevacizumab (Avastin®): Mecanismo de Acción Bevacizumab VEGF P P Page 15 P P

Bevacizumab (Avastin®): Mecanismo de Acción Bevacizumab VEGF P P BLOQUEO de la activación del VEGFR Page 16

Phase III Trial With Bevacizumab Therapy in First-Line MCRC Untreated MCRC R A N D O M I Z E Bolus IFL + placebo (n = 412) Bolus IFL + BV (n = 403) 5 -FU/LV + BV (n = 110): Closed due to lack of efficacy Hurwitz. NEJM, 2004 Page 17 Courtesy of: Paulo Hoff

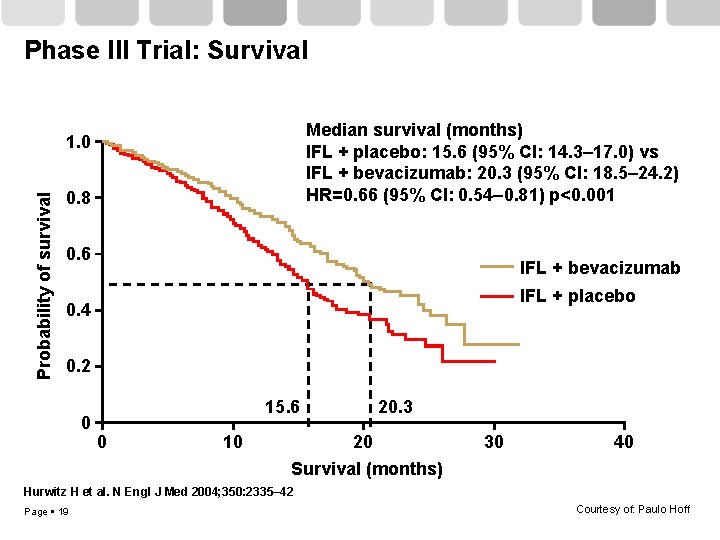
Probability of being progression-free Phase III Trial : PFS Median PFS (months) IFL + placebo: 6. 2 (95% CI: 5. 6– 7. 7) IFL + bevacizumab: 10. 6 (95% CI: 9. 0– 1. 0) HR=0. 54 (95% CI: 0. 45– 0. 66) p<0. 001 1. 0 0. 8 0. 6 IFL + bevacizumab 0. 4 IFL + placebo 0. 2 0 6. 2 0 10. 6 10 20 30 PFS (months) Hurwitz H et al. N Engl J Med 2004; 350: 2335– 42 Page 18 Courtesy of: Paulo Hoff

Phase III Trial: Survival Median survival (months) IFL + placebo: 15. 6 (95% CI: 14. 3– 17. 0) vs IFL + bevacizumab: 20. 3 (95% CI: 18. 5– 24. 2) HR=0. 66 (95% CI: 0. 54– 0. 81) p<0. 001 Probability of survival 1. 0 0. 8 0. 6 IFL + bevacizumab IFL + placebo 0. 4 0. 2 0 15. 6 0 10 20. 3 20 Survival (months) 30 40 Hurwitz H et al. N Engl J Med 2004; 350: 2335– 42 Page 19 Courtesy of: Paulo Hoff

Trial of Bevacizumab plus FOLFIRI/m. IFL (BICC-C): design R n=430 Initial design Amended design FOLFIRI (n=144) FOLFIRI+Bev. (n=60) m. IFL (n=141) XELIRI (n=145) R n=117 Feb 2003 – April 2004 Primary endpoint: PFS m. IFL+Bev. (n=57) May 2004 – Dec 2004 Protocol amended due to approval of bevacizumab * Celecoxib data not shown Fuchs et al, JCO 2008 Page 20 Courtesy of: Paulo Hoff

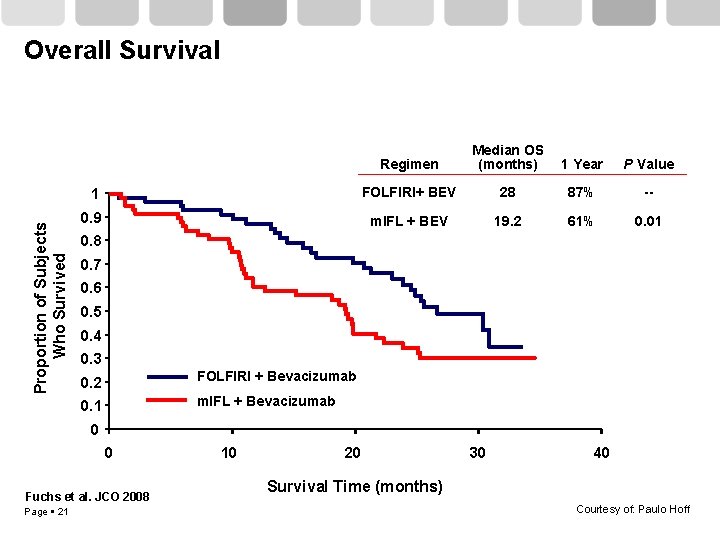
Proportion of Subjects Who Survived Overall Survival Regimen Median OS (months) 1 Year P Value 1 FOLFIRI+ BEV 28 87% -- 0. 9 m. IFL + BEV 19. 2 61% 0. 01 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 FOLFIRI + Bevacizumab 0. 1 m. IFL + Bevacizumab 0 0 Fuchs et al. JCO 2008 Page 21 10 20 30 40 Survival Time (months) Courtesy of: Paulo Hoff

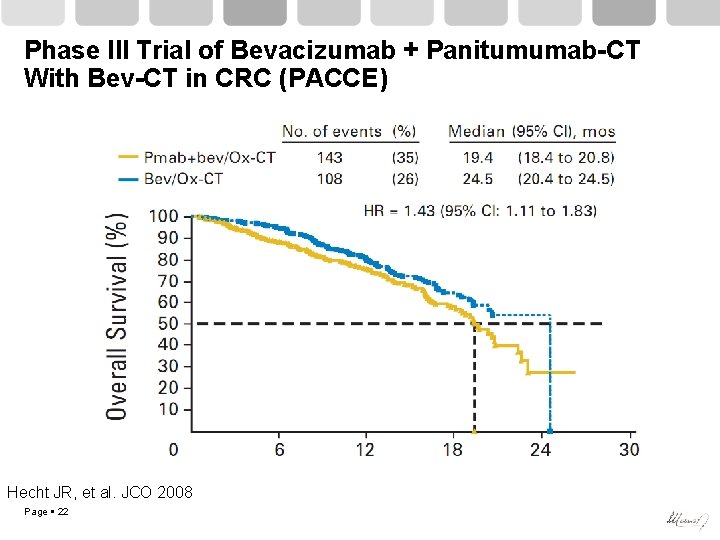
Phase III Trial of Bevacizumab + Panitumumab-CT With Bev-CT in CRC (PACCE) Hecht JR, et al. JCO 2008 Page 22

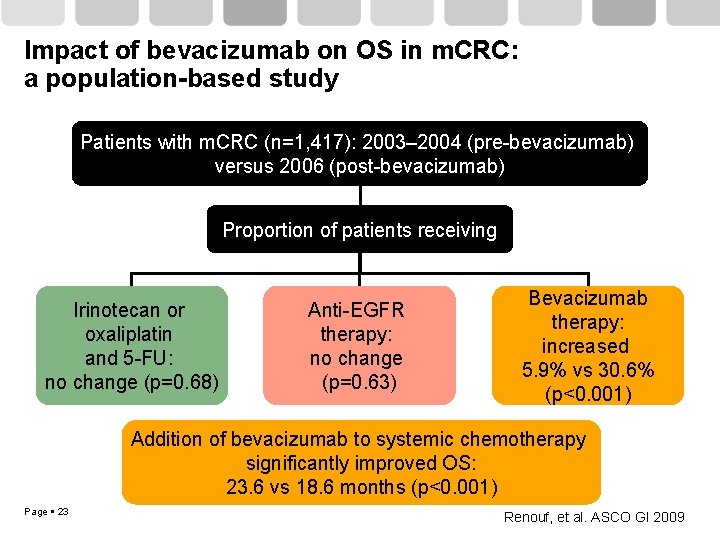
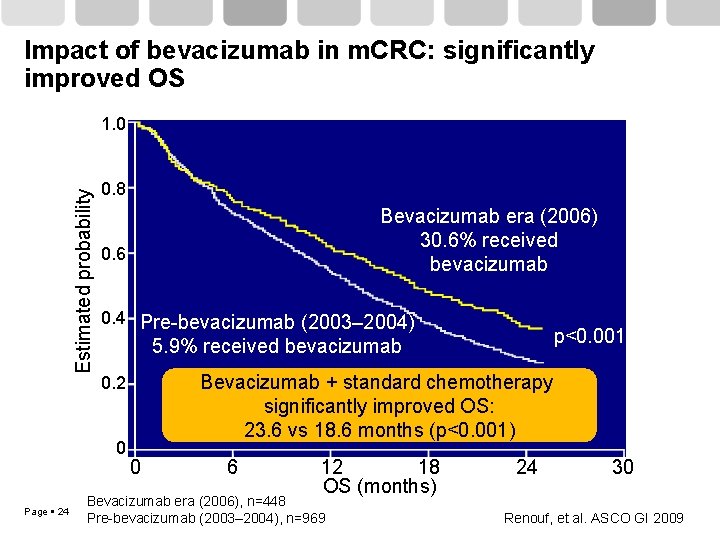
Impact of bevacizumab on OS in m. CRC: a population-based study Patients with m. CRC (n=1, 417): 2003– 2004 (pre-bevacizumab) versus 2006 (post-bevacizumab) Proportion of patients receiving Irinotecan or oxaliplatin and 5 -FU: no change (p=0. 68) Anti-EGFR therapy: no change (p=0. 63) Bevacizumab therapy: increased 5. 9% vs 30. 6% (p<0. 001) Addition of bevacizumab to systemic chemotherapy significantly improved OS: 23. 6 vs 18. 6 months (p<0. 001) Page 23 Renouf, et al. ASCO GI 2009

Impact of bevacizumab in m. CRC: significantly improved OS Estimated probability 1. 0 0. 8 0. 6 0. 4 Pre-bevacizumab (2003– 2004) p<0. 001 5. 9% received bevacizumab Bevacizumab + standard chemotherapy significantly improved OS: 23. 6 vs 18. 6 months (p<0. 001) 0. 2 0 Page 24 Bevacizumab era (2006) 30. 6% received bevacizumab 0 6 12 18 OS (months) Bevacizumab era (2006), n=448 Pre-bevacizumab (2003– 2004), n=969 24 30 Renouf, et al. ASCO GI 2009

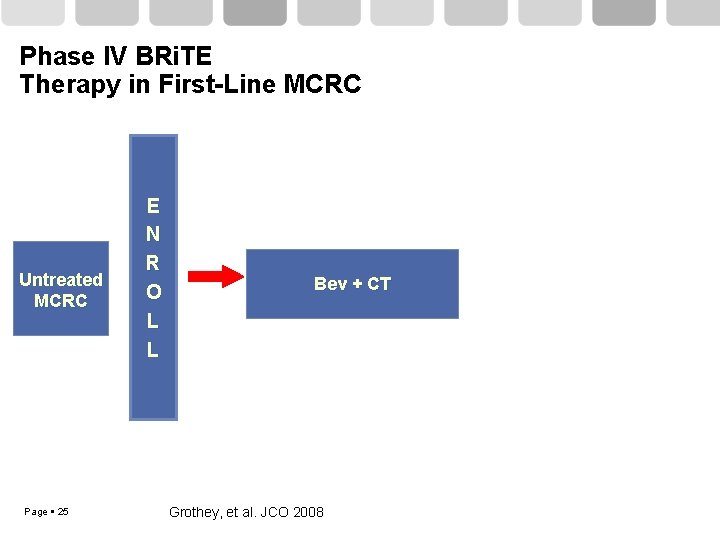
Phase IV BRi. TE Therapy in First-Line MCRC Untreated MCRC Page 25 E N R O L L Bev + CT Grothey, et al. JCO 2008

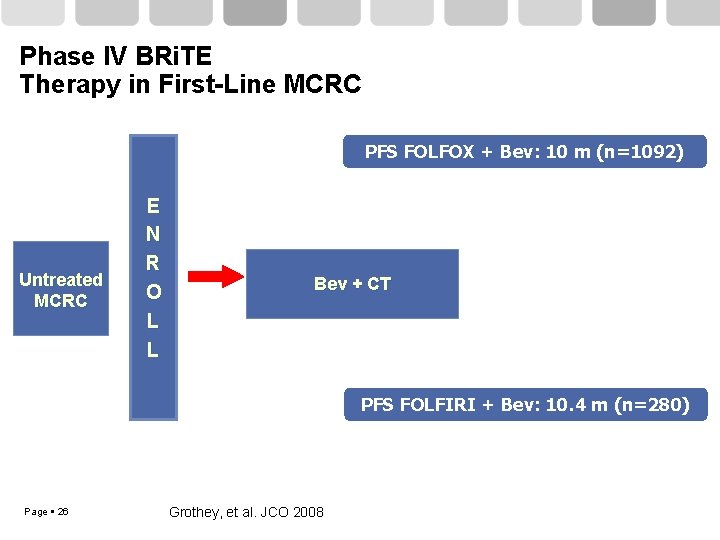
Phase IV BRi. TE Therapy in First-Line MCRC PFS FOLFOX + Bev: 10 m (n=1092) Untreated MCRC E N R O L L Bev + CT PFS FOLFIRI + Bev: 10. 4 m (n=280) Page 26 Grothey, et al. JCO 2008

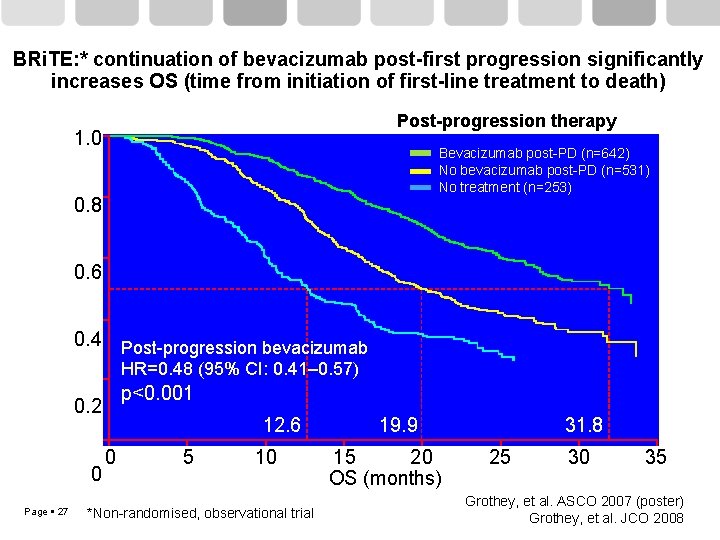
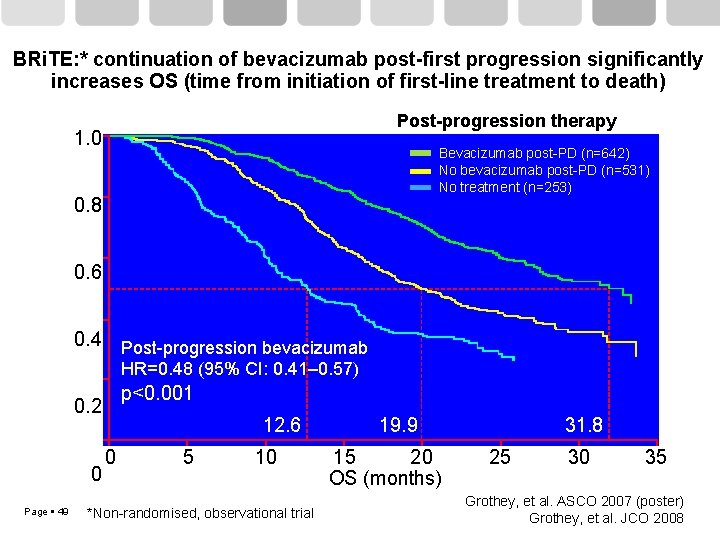
BRi. TE: * continuation of bevacizumab post-first progression significantly increases OS (time from initiation of first-line treatment to death) Post-progression therapy Estimated probability 1. 0 Bevacizumab post-PD (n=642) No bevacizumab post-PD (n=531) No treatment (n=253) 0. 8 0. 6 0. 4 Post-progression bevacizumab HR=0. 48 (95% CI: 0. 41– 0. 57) p<0. 001 0. 2 0 Page 27 12. 6 0 5 10 *Non-randomised, observational trial 19. 9 15 20 OS (months) 31. 8 25 30 35 Grothey, et al. ASCO 2007 (poster) Grothey, et al. JCO 2008

Quimioterapia más cetuximab

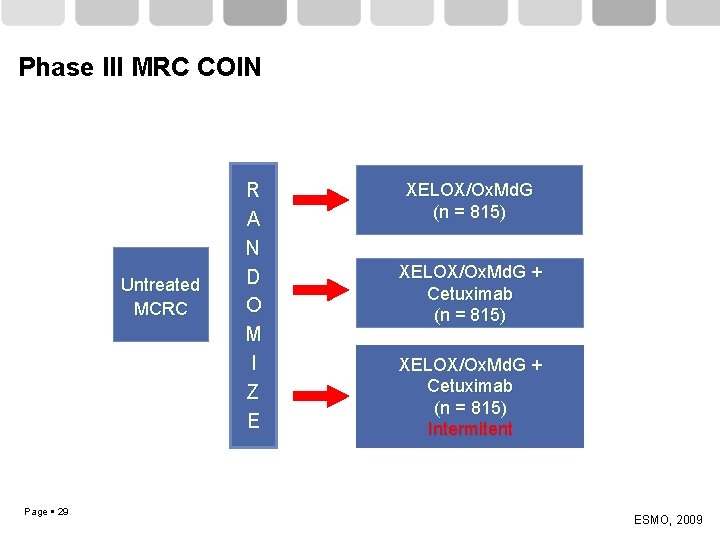
Phase III MRC COIN Untreated MCRC Page 29 R A N D O M I Z E XELOX/Ox. Md. G (n = 815) XELOX/Ox. Md. G + Cetuximab (n = 815) Intermitent ESMO, 2009

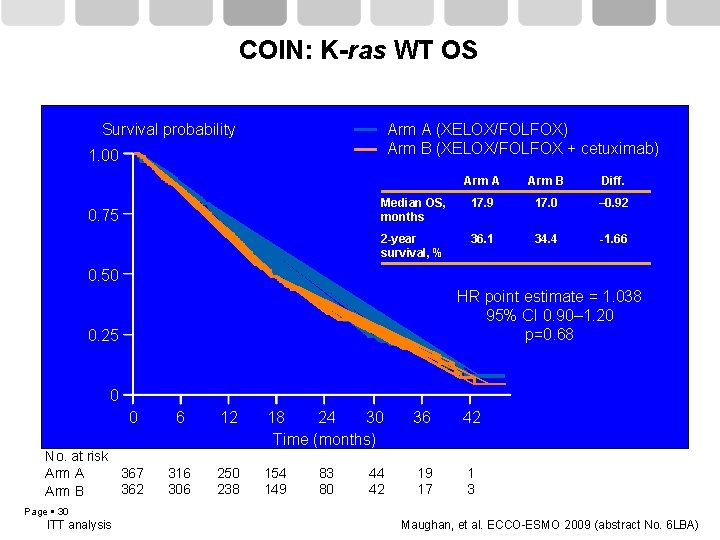
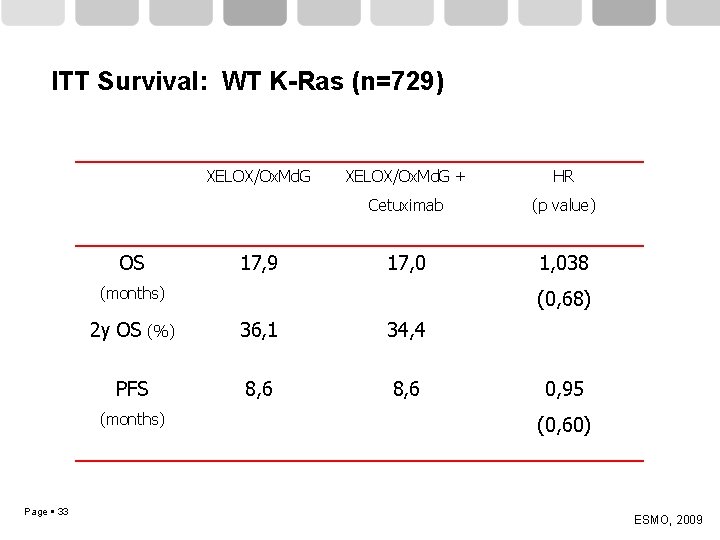
COIN: K-ras WT OS Survival probability Arm A (XELOX/FOLFOX) Arm B (XELOX/FOLFOX + cetuximab) 1. 00 0. 75 Arm A Arm B Diff. Median OS, months 17. 9 17. 0 – 0. 92 2 -year survival, % 36. 1 34. 4 -1. 66 0. 50 HR point estimate = 1. 038 95% CI 0. 90– 1. 20 p=0. 68 0. 25 0 0 No. at risk 367 Arm A 362 Arm B 6 12 18 24 30 Time (months) 36 42 316 306 250 238 154 149 19 17 1 3 83 80 44 42 Page 30 ITT analysis Maughan, et al. ECCO-ESMO 2009 (abstract No. 6 LBA)

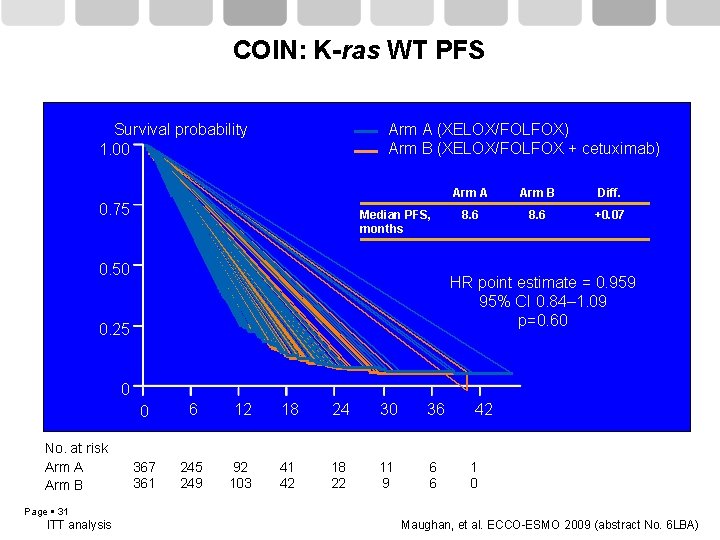
COIN: K-ras WT PFS Arm A (XELOX/FOLFOX) Arm B (XELOX/FOLFOX + cetuximab) Survival probability 1. 00 0. 75 Median PFS, months 0. 50 Arm A Arm B Diff. 8. 6 +0. 07 HR point estimate = 0. 959 95% CI 0. 84– 1. 09 p=0. 60 0. 25 0 No. at risk Arm A Arm B 0 6 12 18 24 30 36 367 361 245 249 92 103 41 42 18 22 11 9 6 6 42 1 0 Page 31 ITT analysis Maughan, et al. ECCO-ESMO 2009 (abstract No. 6 LBA)

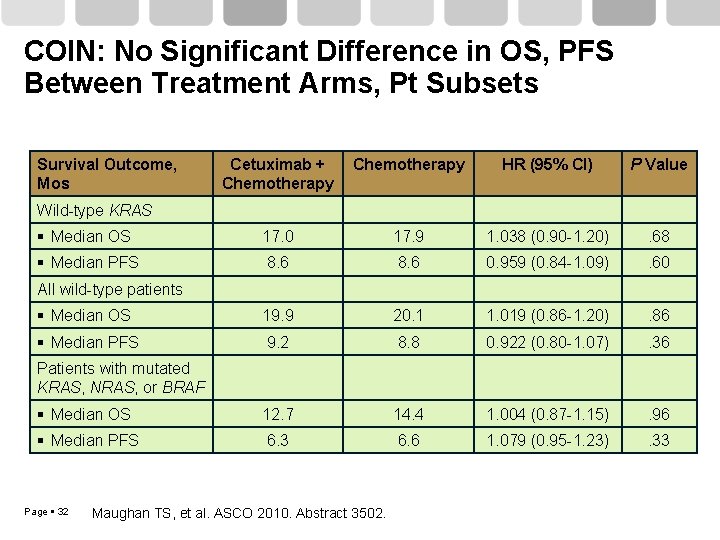
COIN: No Significant Difference in OS, PFS Between Treatment Arms, Pt Subsets Survival Outcome, Mos Cetuximab + Chemotherapy HR (95% CI) P Value Median OS 17. 0 17. 9 1. 038 (0. 90 -1. 20) . 68 Median PFS 8. 6 0. 959 (0. 84 -1. 09) . 60 Median OS 19. 9 20. 1 1. 019 (0. 86 -1. 20) . 86 Median PFS 9. 2 8. 8 0. 922 (0. 80 -1. 07) . 36 Median OS 12. 7 14. 4 1. 004 (0. 87 -1. 15) . 96 Median PFS 6. 3 6. 6 1. 079 (0. 95 -1. 23) . 33 Wild-type KRAS All wild-type patients Patients with mutated KRAS, NRAS, or BRAF Page 32 Maughan TS, et al. ASCO 2010. Abstract 3502.

ITT Survival: WT K-Ras (n=729) XELOX/Ox. Md. G OS 17, 9 XELOX/Ox. Md. G + HR Cetuximab (p value) 17, 0 1, 038 (months) 2 y OS (%) 36, 1 34, 4 PFS 8, 6 (months) Page 33 (0, 68) 0, 95 (0, 60) ESMO, 2009

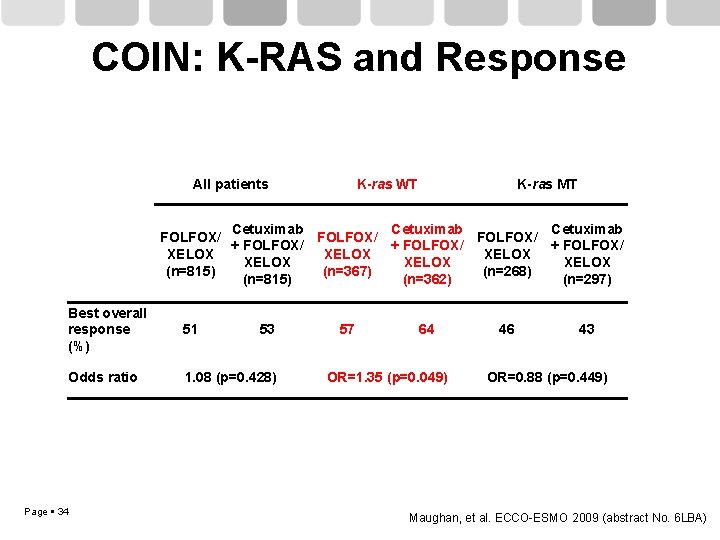
COIN: K-RAS and Response All patients K-ras WT K-ras MT Cetuximab FOLFOX/ + FOLFOX/ XELOX XELOX (n=815) (n=367) (n=268) (n=815) (n=362) (n=297) Best overall response (%) 51 Odds ratio 1. 08 (p=0. 428) Page 34 53 57 64 OR=1. 35 (p=0. 049) 46 43 OR=0. 88 (p=0. 449) Maughan, et al. ECCO-ESMO 2009 (abstract No. 6 LBA)

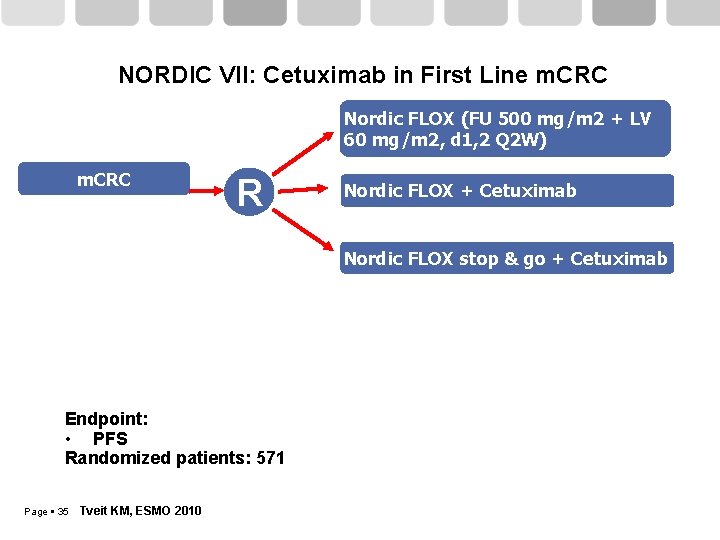
NORDIC VII: Cetuximab in First Line m. CRC Nordic FLOX (FU 500 mg/m 2 + LV 60 mg/m 2, d 1, 2 Q 2 W) m. CRC R Nordic FLOX + Cetuximab Nordic FLOX stop & go + Cetuximab Endpoint: • PFS Randomized patients: 571 Page 35 Tveit KM, ESMO 2010

NORDIC VII: Cetuximab in First Line m. CRC Nordic FLOX (FU 500 mg/m 2 + LV 60 mg/m 2, d 1, 2 Q 2 W) m. CRC R Nordic FLOX + Cetuximab Nordic FLOX stop & go + Cetuximab Outcome FLOX F+Cet Stop&Go-Cet PFS 7. 9 8. 3 7. 3 RR 41% 49% 47% OS 20. 4 19. 7 20. 3 Endpoint: • PFS Randomized patients: 571 Page 36 Tveit KM, ESMO 2010

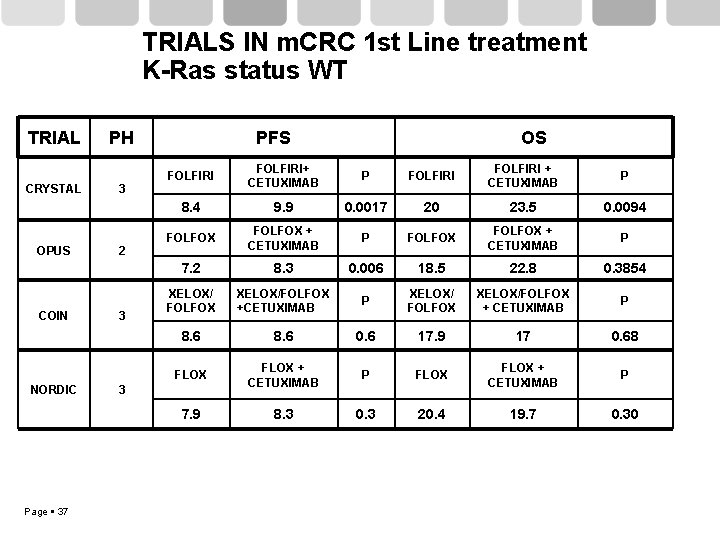
TRIALS IN m. CRC 1 st Line treatment K-Ras status WT TRIAL CRYSTAL OPUS COIN NORDIC Page 37 PH 3 2 3 PFS OS FOLFIRI+ CETUXIMAB P FOLFIRI + CETUXIMAB P 8. 4 9. 9 0. 0017 20 23. 5 0. 0094 FOLFOX + CETUXIMAB P 7. 2 8. 3 0. 006 18. 5 22. 8 0. 3854 XELOX/ FOLFOX XELOX/FOLFOX +CETUXIMAB P XELOX/ FOLFOX XELOX/FOLFOX + CETUXIMAB P 8. 6 0. 6 17. 9 17 0. 68 FLOX + CETUXIMAB P 7. 9 8. 3 0. 3 20. 4 19. 7 0. 30 3

EPIC: Cetuximab + Irinotecan after Fluoropyrimidine + Oxaliplatin failure Cetuximab 400 mg/m 2 initial dose m. CRC with progression after 1 st line fluoropyirimidine and Oxaliplatin (n=1298) cycle 1, wk 1, 250 mg/m 2 weekly Irinotecan 350 mg/m 2 (n=648) R Primary endpoint: Overall survival Page 38 Sobrero AF. J Clin Oncol. 26: 2311 -2319, 2008 Irinotecan (n=650)

Cetuximab + Irinotecan vs Irinotecan in 2 nd line - EPIC Page 39 Cet + Iri (n=648) (n=650) RR 16. 4% 4. 2% P<0. 05 PFS 4 months 2. 6 months P<0. 005 OS 10. 7 m 10 m P=0. 71 Sobrero AF. J Clin Oncol. 26: 2311 -2319, 2008 P value

Cetuximab versus BSC Metastatic colorectal cancer with prior 5 -FU, irinotecan and oxaliplatin (572 pts) R A N D O M I Z E Cetuximab + BSC (287) BSC (285) Jonker et al. NEJM 2007; 357: 2040 Page 40 Courtesy of: Paulo Hoff

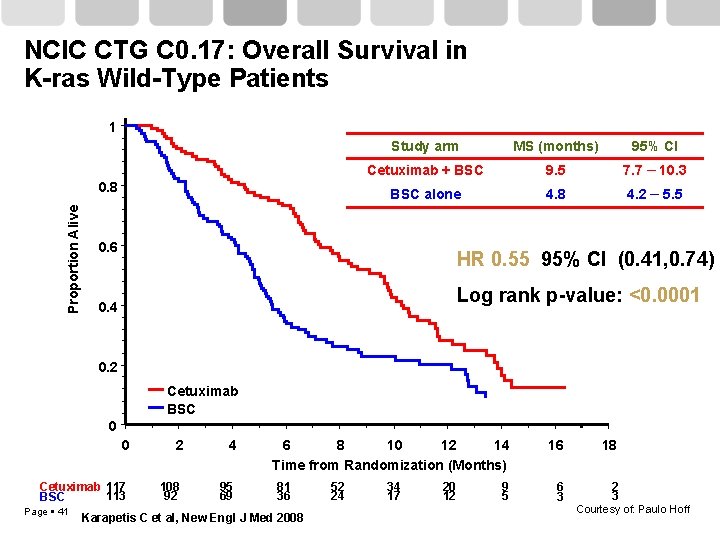
NCIC CTG C 0. 17: Overall Survival in K-ras Wild-Type Patients 1 Proportion Alive 0. 8 Study arm MS (months) 95% CI Cetuximab + BSC 9. 5 7. 7 – 10. 3 BSC alone 4. 8 4. 2 – 5. 5 0. 6 HR 0. 55 95% CI (0. 41, 0. 74) Log rank p-value: <0. 0001 0. 4 0. 2 Cetuximab BSC 0 0 Cetuximab 117 113 BSC Page 41 2 108 92 4 95 69 6 8 10 12 14 Time from Randomization (Months) 81 36 Karapetis C et al, New Engl J Med 2008 52 24 34 17 20 12 9 5 16 18 6 3 2 3 Courtesy of: Paulo Hoff

Continuum of care

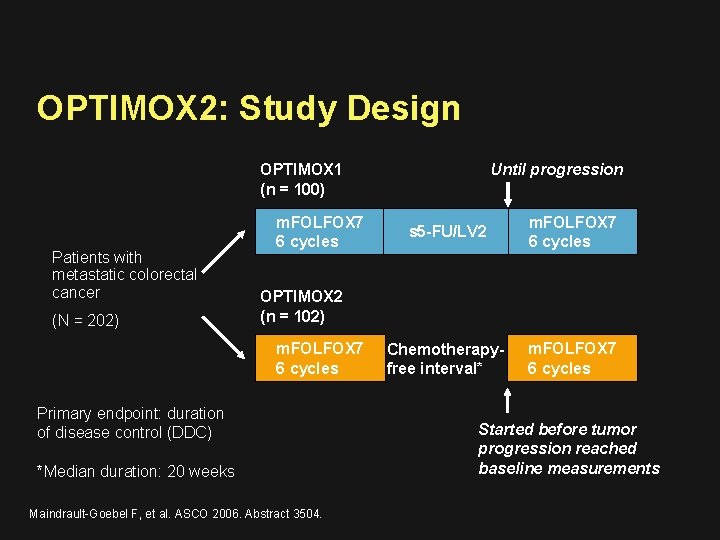
OPTIMOX 2: Study Design Until progression OPTIMOX 1 (n = 100) Patients with metastatic colorectal cancer (N = 202) m. FOLFOX 7 6 cycles s 5 -FU/LV 2 m. FOLFOX 7 6 cycles Chemotherapyfree interval* m. FOLFOX 7 6 cycles OPTIMOX 2 (n = 102) m. FOLFOX 7 6 cycles Primary endpoint: duration of disease control (DDC) *Median duration: 20 weeks Maindrault-Goebel F, et al. ASCO 2006. Abstract 3504. Started before tumor progression reached baseline measurements

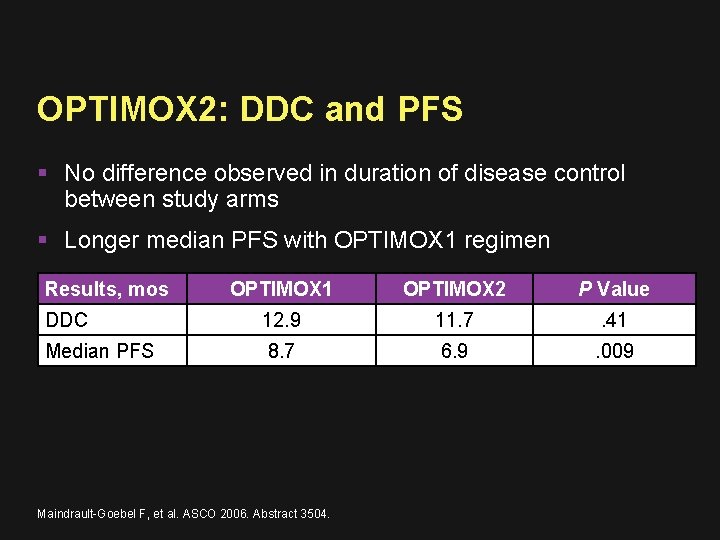
OPTIMOX 2: DDC and PFS No difference observed in duration of disease control between study arms Longer median PFS with OPTIMOX 1 regimen Results, mos OPTIMOX 1 OPTIMOX 2 P Value DDC 12. 9 11. 7 . 41 Median PFS 8. 7 6. 9 . 009 Maindrault-Goebel F, et al. ASCO 2006. Abstract 3504.

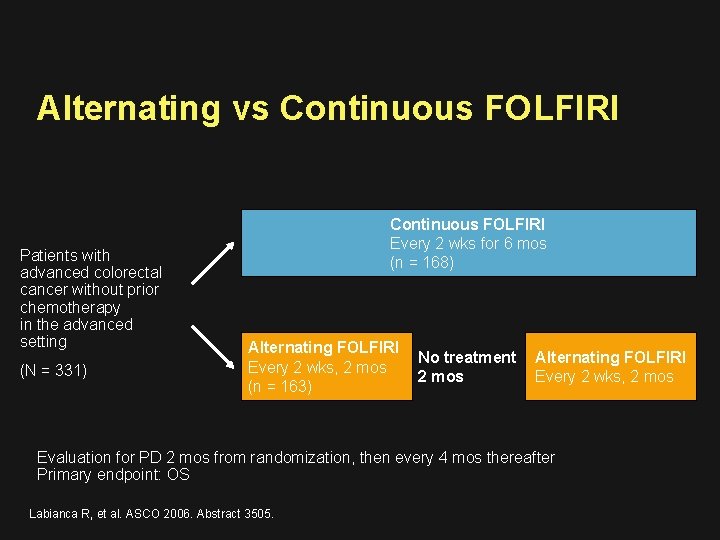
Alternating vs Continuous FOLFIRI Patients with advanced colorectal cancer without prior chemotherapy in the advanced setting (N = 331) Continuous FOLFIRI Every 2 wks for 6 mos (n = 168) Alternating FOLFIRI Every 2 wks, 2 mos (n = 163) No treatment 2 mos Alternating FOLFIRI Every 2 wks, 2 mos Evaluation for PD 2 mos from randomization, then every 4 mos thereafter Primary endpoint: OS Labianca R, et al. ASCO 2006. Abstract 3505.

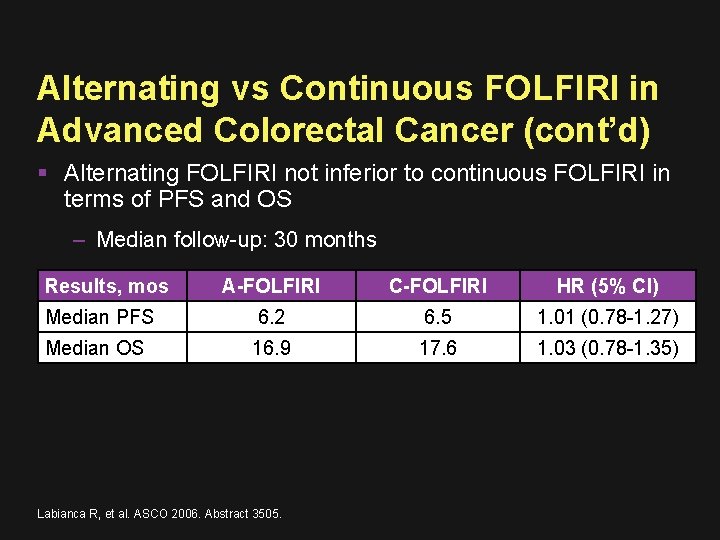
Alternating vs Continuous FOLFIRI in Advanced Colorectal Cancer (cont’d) Alternating FOLFIRI not inferior to continuous FOLFIRI in terms of PFS and OS – Median follow-up: 30 months Results, mos A-FOLFIRI C-FOLFIRI HR (5% CI) Median PFS 6. 2 6. 5 1. 01 (0. 78 -1. 27) Median OS 16. 9 17. 6 1. 03 (0. 78 -1. 35) Labianca R, et al. ASCO 2006. Abstract 3505.

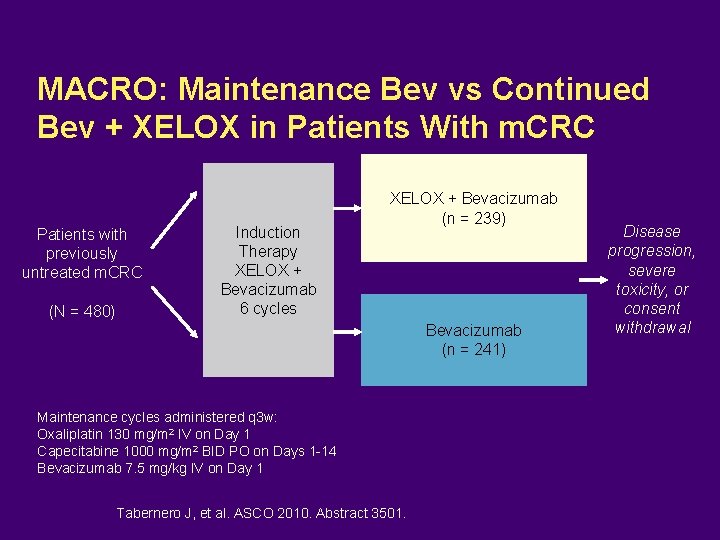
MACRO: Maintenance Bev vs Continued Bev + XELOX in Patients With m. CRC Patients with previously untreated m. CRC (N = 480) Induction Therapy XELOX + Bevacizumab 6 cycles XELOX + Bevacizumab (n = 239) Bevacizumab (n = 241) Maintenance cycles administered q 3 w: Oxaliplatin 130 mg/m 2 IV on Day 1 Capecitabine 1000 mg/m 2 BID PO on Days 1 -14 Bevacizumab 7. 5 mg/kg IV on Day 1 Tabernero J, et al. ASCO 2010. Abstract 3501. Disease progression, severe toxicity, or consent withdrawal

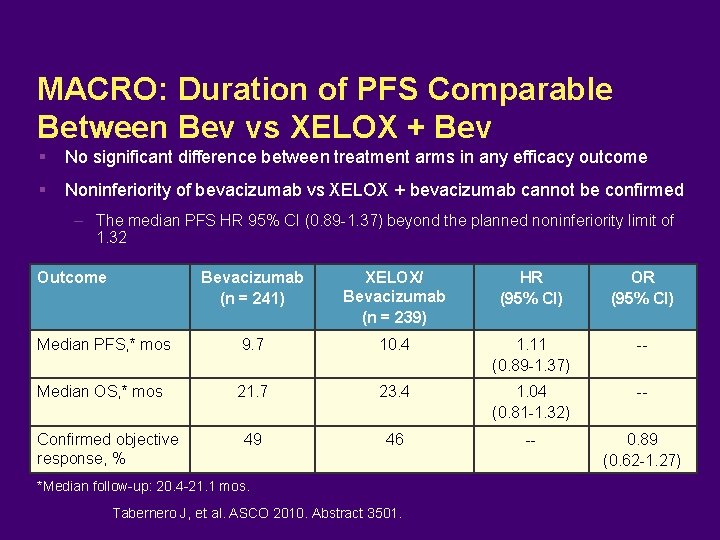
MACRO: Duration of PFS Comparable Between Bev vs XELOX + Bev No significant difference between treatment arms in any efficacy outcome Noninferiority of bevacizumab vs XELOX + bevacizumab cannot be confirmed – The median PFS HR 95% CI (0. 89 -1. 37) beyond the planned noninferiority limit of 1. 32 Outcome Bevacizumab (n = 241) XELOX/ Bevacizumab (n = 239) HR (95% CI) OR (95% CI) Median PFS, * mos 9. 7 10. 4 1. 11 (0. 89 -1. 37) -- Median OS, * mos 21. 7 23. 4 1. 04 (0. 81 -1. 32) -- 49 46 -- 0. 89 (0. 62 -1. 27) Confirmed objective response, % *Median follow-up: 20. 4 -21. 1 mos. Tabernero J, et al. ASCO 2010. Abstract 3501.

BRi. TE: * continuation of bevacizumab post-first progression significantly increases OS (time from initiation of first-line treatment to death) Post-progression therapy Estimated probability 1. 0 Bevacizumab post-PD (n=642) No bevacizumab post-PD (n=531) No treatment (n=253) 0. 8 0. 6 0. 4 Post-progression bevacizumab HR=0. 48 (95% CI: 0. 41– 0. 57) p<0. 001 0. 2 0 Page 49 12. 6 0 5 10 *Non-randomised, observational trial 19. 9 15 20 OS (months) 31. 8 25 30 35 Grothey, et al. ASCO 2007 (poster) Grothey, et al. JCO 2008

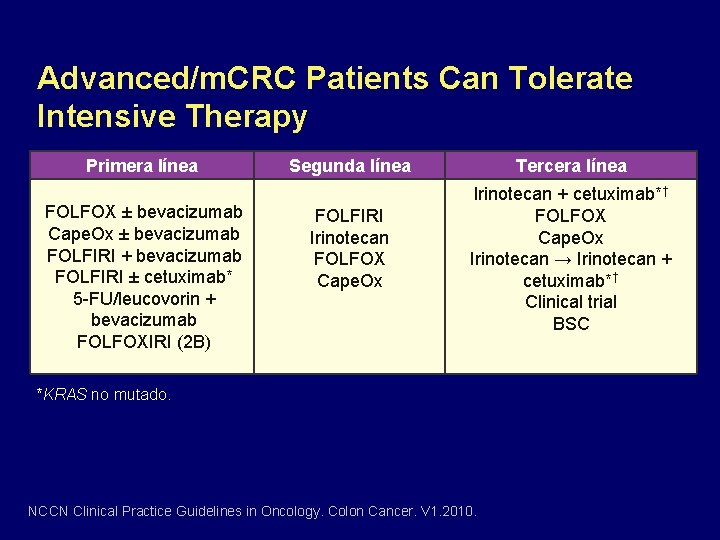
Advanced/m. CRC Patients Can Tolerate Intensive Therapy Primera línea FOLFOX ± bevacizumab Cape. Ox ± bevacizumab FOLFIRI + bevacizumab FOLFIRI ± cetuximab* 5 -FU/leucovorin + bevacizumab FOLFOXIRI (2 B) Segunda línea FOLFIRI Irinotecan FOLFOX Cape. Ox Tercera línea Irinotecan + cetuximab*† FOLFOX Cape. Ox Irinotecan → Irinotecan + cetuximab*† Clinical trial BSC *KRAS no mutado. NCCN Clinical Practice Guidelines in Oncology. Colon Cancer. V 1. 2010.

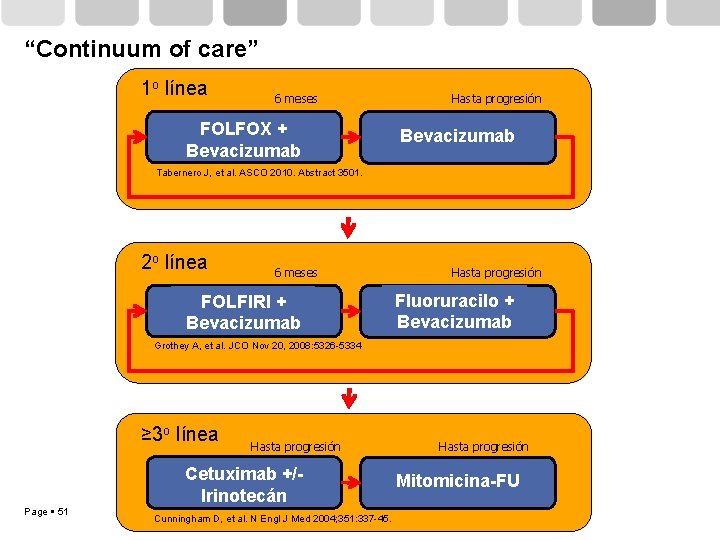
“Continuum of care” 1 o línea 6 meses FOLFOX + Bevacizumab Hasta progresión Bevacizumab Tabernero J, et al. ASCO 2010. Abstract 3501. 2 o línea 6 meses FOLFIRI + Bevacizumab Hasta progresión Fluoruracilo + Bevacizumab Grothey A, et al. JCO Nov 20, 2008: 5326 -5334 ≥ 3 o línea Page 51 Hasta progresión Cetuximab +/Irinotecán Cunningham D, et al. N Engl J Med 2004; 351: 337 -45. Hasta progresión Mitomicina-FU

Conclusiones Debe recibir Irinotecán, Oxaliplatino y Fluoruracilo… Bevacizumab + QT en primera línea metastásica Cetuximab +/- Irinotecán en última línea Disminución de intensidad (y toxicidad) es válida (sábados) Suspender el tratamiento: disminuye la supervivencia (domingos)