Chemistry This Powerpoint is hosted on www worldofteaching



























- Slides: 35

Chemistry This Powerpoint is hosted on www. worldofteaching. com Please 100’s more free More visit freefor powerpoints atpowerpoints www. worldofteaching. com

Matter What is Matter? Where is Matter made? Matter = any material substance with Mass, Density & Volume

Matter comes in 3 phases Solid Gas Liquid

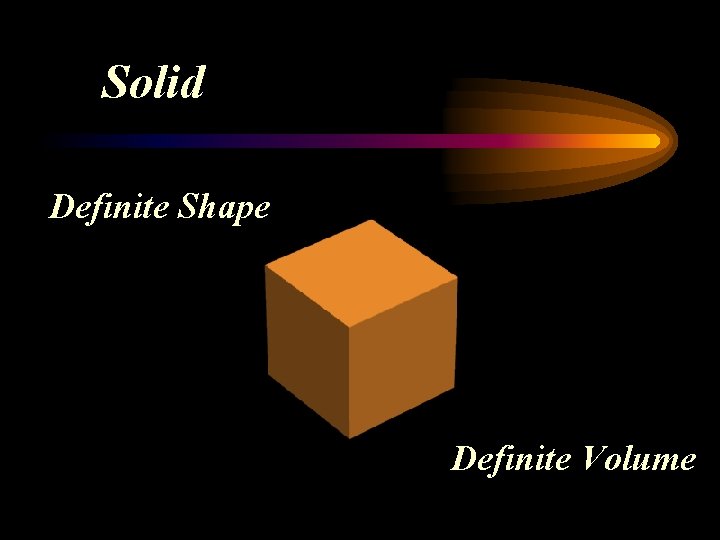
Solid Definite Shape Definite Volume

Liquid Indefinite Shape – takes the shape of the container Definite Volume

Gas Indefinite Shape – takes the shape of the container Indefinite Volume – can expand be compressed

Elements one of the 100+ pure substances that make up everything in the universe

Examples of Elements C = Carbon Na = Sodium O = Oxygen Ca = Calcium H = Hydrogen K = Potassium N = Nitrogen I = Iodine Cl = Chlorine S = Sulfur P = Phosphorus

C. Elements 1. All matter is composed of elements 2. Substances that cannot be broken into simple substances by chemical means Most Common Elements in Earth’s Crust 46. 6% Oxygen (O) 27. 7% Silicon (Si) 8. 1% Aluminum (Al) 5. 0% Iron (Fe) 3. 6% Calcium (Ca) 2. 8% Sodium (Na) 2. 6% Potassium (K) 2. 1% Magnesium (Mg)

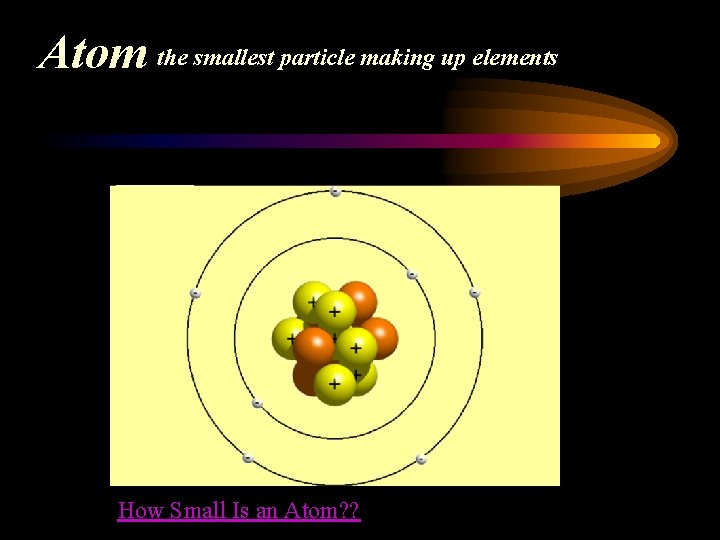
Atom the smallest particle making up elements How Small Is an Atom? ?

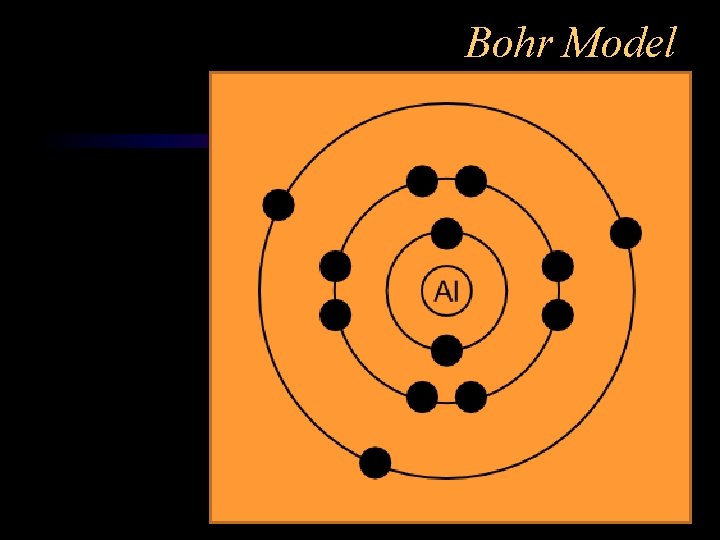
Bohr Model

Sub-atomic Particles Protons p+ - positive charge, in nucleus Neutrons n 0 – no charge, in nucleus Electrons - e- negative charge, orbiting nucleus

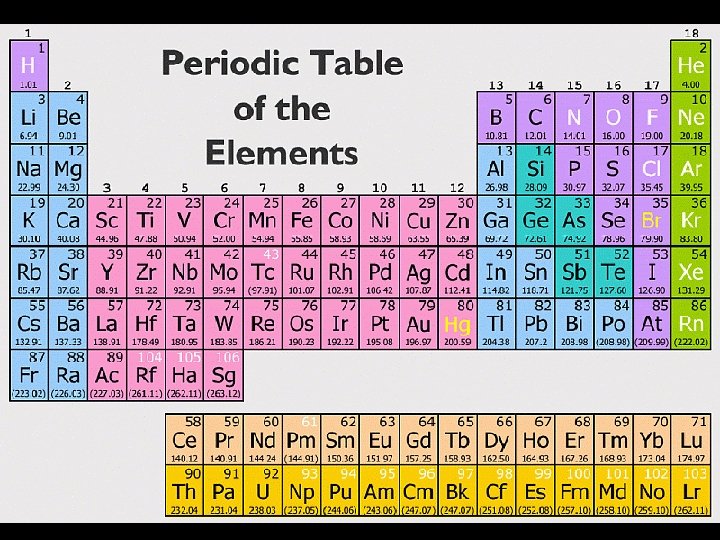
Periodic Table Cheat Sheet: • Atomic Number = # of protons • Atomic mass= # of protons & neutrons • Electrons= same # of protons (for stable elements)

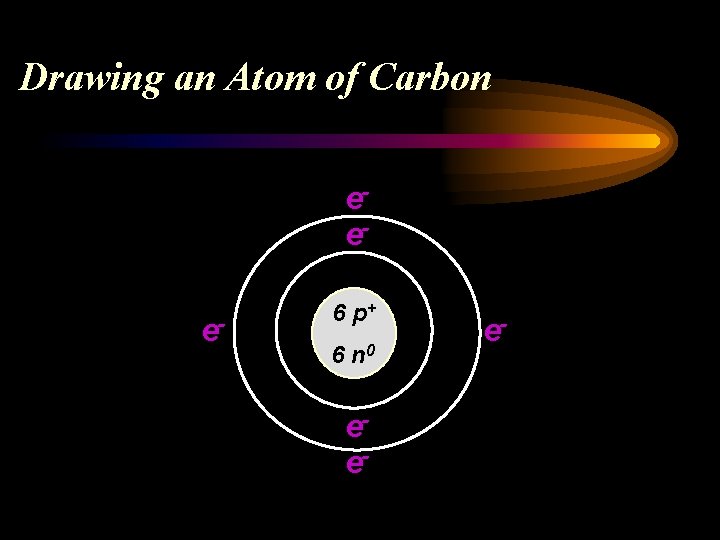
Drawing an Atom of Carbon 6 C 12. 011 Atomic # = # of p+ and # of e. Carbon has 6 p+ and 6 e- Atomic Mass minus Atomic # = # of n 0 Carbon has 6 n 0

Drawing an Atom of Carbon eee- 6 p+ 6 n 0 ee- e-

E. Compounds - A substance made of atoms combined in a fixed proportion. Ex. H – O = water Ex. H 2 O + Na = Saltwater

Reading Chemical Compounds How many different elements? H 2 O How many total atoms? Na. Cl H 2 SO 4 Be(OH)2

Compounds - 2 or more elements chemically combined to form a new substance with new properties Properties – The way a chemical substance looks and behaves

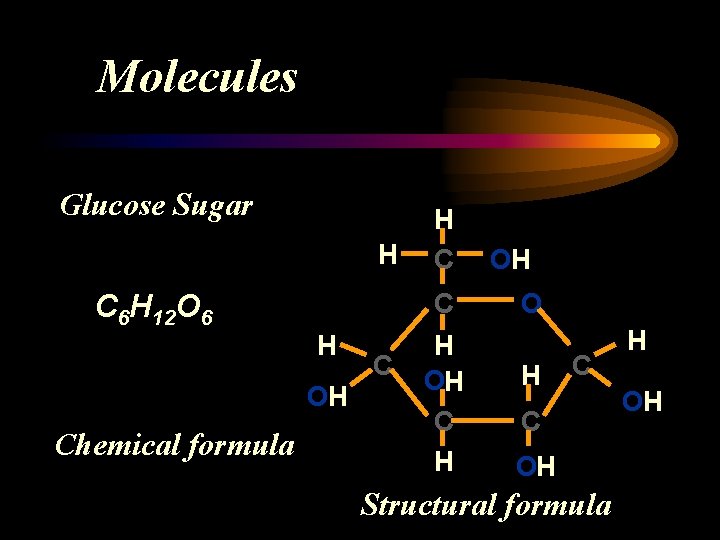
Compounds – are made of 2 or more different atoms combined to form Molecules O H+O H 2 O = H H Chemical formula lists the number of different atoms in a single molecule Structural formula shows the arrangement of the atoms in a single molecule

Molecules Glucose Sugar H C C 6 H 12 O 6 H OH Chemical formula H C C H OH O H C C OH Structural formula H OH

Compounds Inorganic Compounds • usually don’t contain Carbon • generally come from the earth • generally simple molecules or Organic Compounds • always contain C & H and usually O, N, sometimes S&P • originate in organisms • generally complex molecules

Examples of Inorganic Compounds H + O = H 2 O = Water H + Cl = Hydrochloric Acid C + O = CO 2 = Carbon Dioxide Na + Cl = Na. Cl = Common Table Salt

Examples of Organic Compounds C, H + O Carbohydrates = Sugars, starches & cellulose Lipids = Fats & Oils C, H, O, N, & sometimes P + S Proteins C, H, O, N, + P Nucleic Acids – DNA & RNA


PT Interactive

D. Isotopes 1. Definition: Atoms of the same number of protons, different number of neutrons Ex. Carbon-12 & Carbon-14 2. Useful because they decay at a specific rate. -tells us the age of a rock or fossil

1. Atom attraction a. Atoms in their normal state have an equal # of protons & electrons. b. Said to be “stable” 2. Ion a. An atom that gains or loses one or more electrons becoming either more positively or negatively charged

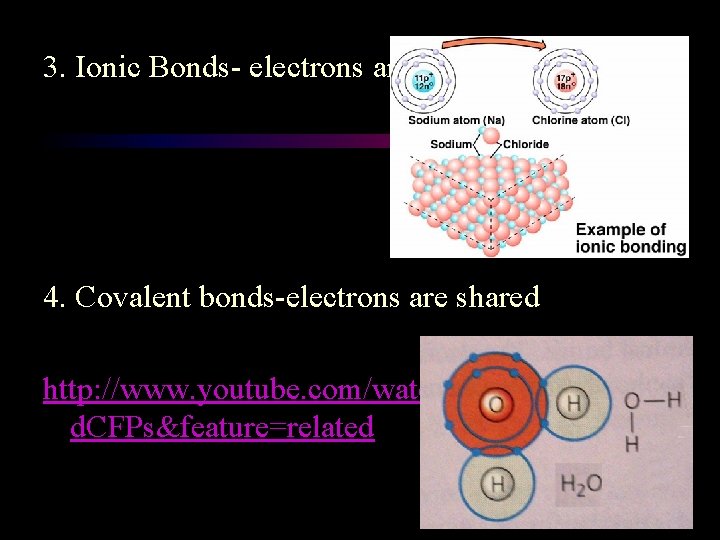
3. Ionic Bonds- electrons are transferred 4. Covalent bonds-electrons are shared http: //www. youtube. com/watch? v=yjge 1 W d. CFPs&feature=related

1. When an atom gains or loses an electron it is now called an ____. Ion

2. How many different elements are in the following compound? (CH 3)2 CO Answer- 3

3. How many total atoms are in the following compound? (CH 3)2 CO Answer- 10

4. The atomic number tells how many ______ are in the element. protons

5. What type of chemically bonding involves the sharing of electrons? Covalent

What is your stone?

Chemistry Diga, diga, that’s all folks!