ACCP Cardiology PRN Journal Club Announcements Thank you

ACCP Cardiology PRN Journal Club

Announcements § Thank you attending the ACCP Cardiology PRN Journal Club – Thank you if you attended last time or have been attending § I have created a PB Works Site that will house our recorded calls, handouts, and Summary/Q&A documents. The link is https: //accpcardsprnjournalclub. pbworks. com/ § If there any suggestions, please let us know.

Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism Cardiology PRN Journal Club April 2, 2015 Mentor: Evan Nix, Pharm. D Leo Buckley, Pharm. D BCPS Presenter: Leo Buckley, PGY-2 Cardiovascular Pharmacy Resident Department of Pharmacy Disclosures: none Services
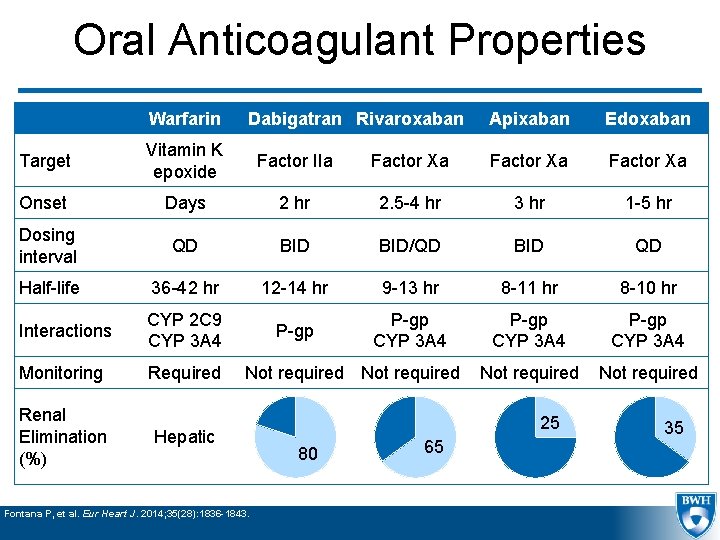
Oral Anticoagulant Properties Warfarin Dabigatran Rivaroxaban Apixaban Edoxaban Target Vitamin K epoxide Factor IIa Factor Xa Onset Days 2 hr 2. 5 -4 hr 3 hr 1 -5 hr Dosing interval QD BID/QD BID QD Half-life 36 -42 hr 12 -14 hr 9 -13 hr 8 -11 hr 8 -10 hr Interactions CYP 2 C 9 CYP 3 A 4 P-gp CYP 3 A 4 Monitoring Required Not required Renal Elimination (%) Hepatic Not required Fontana P, et al. Eur Heart J. 2014; 35(28): 1836 -1843. 25 80 65 35
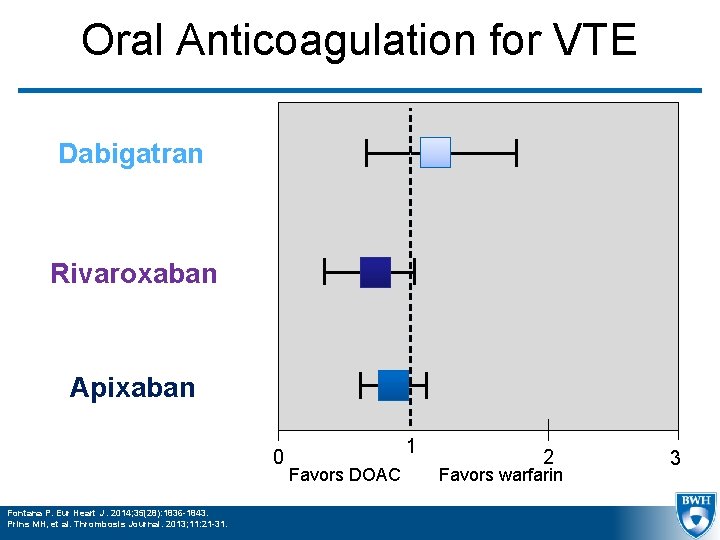
Oral Anticoagulation for VTE Dabigatran Rivaroxaban Apixaban 0 Fontana P. Eur Heart J. 2014; 35(28): 1836 -1843. Prins MH, et al. Thrombosis Journal. 2013; 11: 21 -31. 1 Favors DOAC 2 Favors warfarin 3
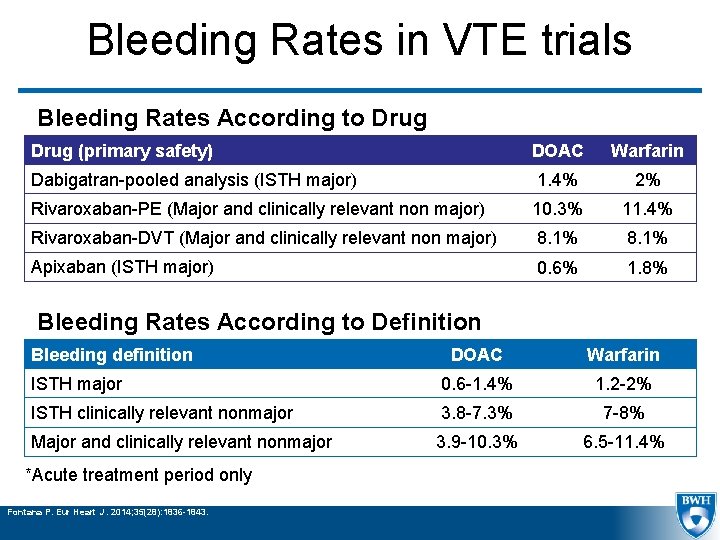
Bleeding Rates in VTE trials Bleeding Rates According to Drug (primary safety) DOAC Warfarin Dabigatran-pooled analysis (ISTH major) 1. 4% 2% Rivaroxaban-PE (Major and clinically relevant non major) 10. 3% 11. 4% Rivaroxaban-DVT (Major and clinically relevant non major) 8. 1% Apixaban (ISTH major) 0. 6% 1. 8% Bleeding Rates According to Definition Bleeding definition DOAC Warfarin ISTH major 0. 6 -1. 4% 1. 2 -2% ISTH clinically relevant nonmajor 3. 8 -7. 3% 7 -8% Major and clinically relevant nonmajor 3. 9 -10. 3% 6. 5 -11. 4% *Acute treatment period only Fontana P. Eur Heart J. 2014; 35(28): 1836 -1843.
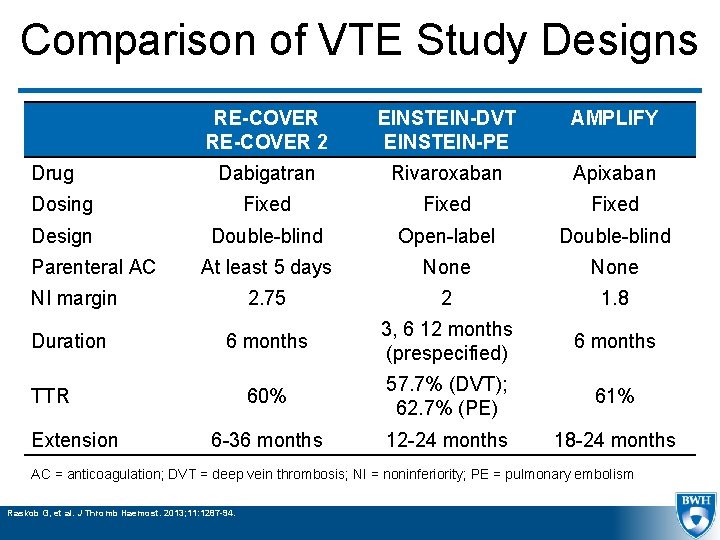
Comparison of VTE Study Designs RE-COVER 2 EINSTEIN-DVT EINSTEIN-PE AMPLIFY Dabigatran Rivaroxaban Apixaban Dosing Fixed Design Double-blind Open-label Double-blind At least 5 days None NI margin 2. 75 2 1. 8 Duration 6 months 3, 6 12 months (prespecified) 6 months 60% 57. 7% (DVT); 62. 7% (PE) 61% 6 -36 months 12 -24 months 18 -24 months Drug Parenteral AC TTR Extension AC = anticoagulation; DVT = deep vein thrombosis; NI = noninferiority; PE = pulmonary embolism Raskob G, et al. J Thromb Haemost. 2013; 11: 1287 -94.


Study Design § Randomized, double-blind, double dummy, active control § Objective: evaluate whether initial heparin followed by edoxaban is noninferior to heparin, overlapped with and followed by warfarin for the prevention of symptomatic recurrent VTE Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.
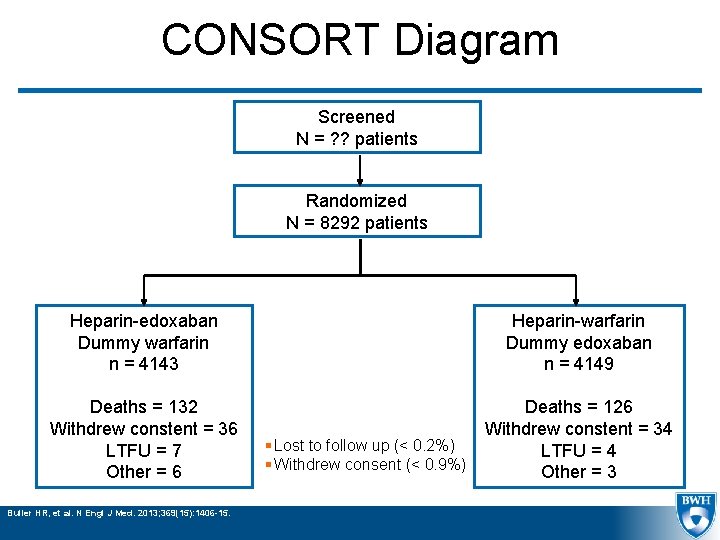
CONSORT Diagram Screened N = ? ? patients Randomized N = 8292 patients Heparin-edoxaban Dummy warfarin n = 4143 Heparin-warfarin Dummy edoxaban n = 4149 Deaths = 132 Withdrew constent = 36 LTFU = 7 Other = 6 Deaths = 126 Withdrew constent = 34 LTFU = 4 Other = 3 Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. § Lost to follow up (< 0. 2%) § Withdrew consent (< 0. 9%)
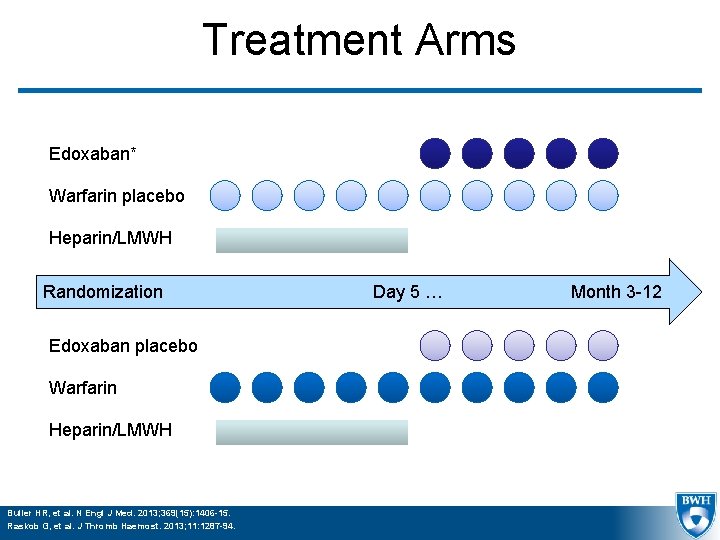
Treatment Arms Edoxaban* Warfarin placebo Heparin/LMWH Randomization Edoxaban placebo Warfarin Heparin/LMWH Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. Raskob G, et al. J Thromb Haemost. 2013; 11: 1287 -94. Day 5 … Month 3 -12

Treatment § Edoxaban: – 60 mg QD – 30 mg QD if: weight < 60 kg, Cr. Cl 30 -50, P-gp inhibitors (verapamil, quinidine azithromycin, clarithromycin, erythromycin, oral itraconazole or oral ketoconazole) § Warfarin INR 2 -3 – TTR calculated by Rosendaal method – Center TTR monitoring, dose adjustment protocol provided Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. Raskob G, et al. J Thromb Haemost. 2013; 11: 1287 -94.

Inclusion/Exclusion Criteria Inclusion § DVT (acute, symptomatic) – – – Compression ultrasound Venography with contrast CT-scan MR-venography Popliteal, femoral, iliac vein § PE – Spiral CT – Pulmonary angiogram – Ventilation/perfusion scan Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. Raskob G, et al. J Thromb Haemost. 2013; 11: 1287 -94. Exclusion § Contraindications to warfarin or heparin § > 48 hr therapeutic UFH § > 1 dose VKA § Cancer + long-term LMWH § Other indication for VKA § > 100 mg ASA or DAPT § Cr. Cl < 30 ml/min § Thrombectomy/olysis § Mechanical valve § Recent or current bleeding

Endpoints § Primary efficacy: – Symptomatic, objectively verified recurrent DVT or fatal or nonfatal PE § Primary safety: – Clinically relevant bleeding • Composite of ISTH major bleeding and clinically relevant nonmajor bleeding § Follow-up: – 12 months from randomization, regardless of treatment duration Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. Raskob G, et al. J Thromb Haemost. 2013; 11: 1287 -94.
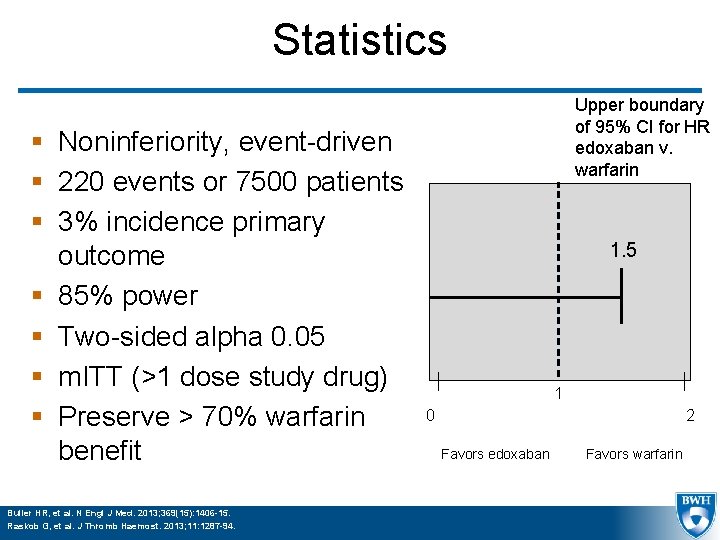
Statistics § Noninferiority, event-driven § 220 events or 7500 patients § 3% incidence primary outcome § 85% power § Two-sided alpha 0. 05 § m. ITT (>1 dose study drug) § Preserve > 70% warfarin benefit Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. Raskob G, et al. J Thromb Haemost. 2013; 11: 1287 -94. Upper boundary of 95% CI for HR edoxaban v. warfarin 1. 5 1 0 2 Favors edoxaban Favors warfarin
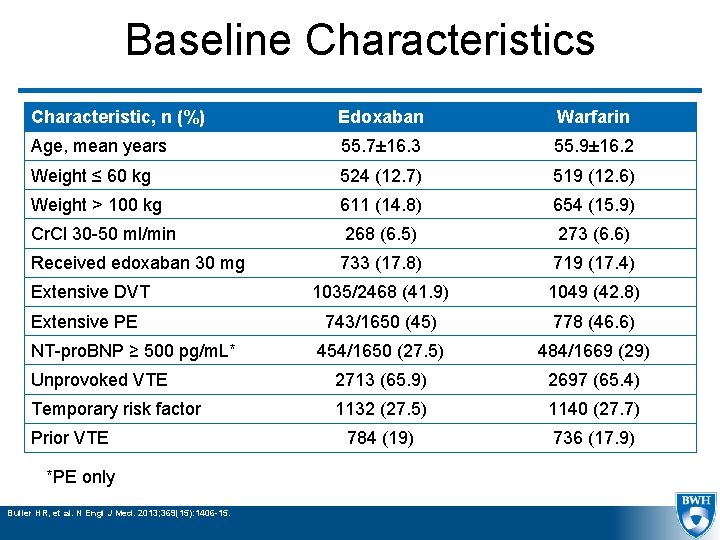
Baseline Characteristics Characteristic, n (%) Edoxaban Warfarin Age, mean years 55. 7± 16. 3 55. 9± 16. 2 Weight ≤ 60 kg 524 (12. 7) 519 (12. 6) Weight > 100 kg 611 (14. 8) 654 (15. 9) Cr. Cl 30 -50 ml/min 268 (6. 5) 273 (6. 6) Received edoxaban 30 mg 733 (17. 8) 719 (17. 4) 1035/2468 (41. 9) 1049 (42. 8) 743/1650 (45) 778 (46. 6) 454/1650 (27. 5) 484/1669 (29) Unprovoked VTE 2713 (65. 9) 2697 (65. 4) Temporary risk factor 1132 (27. 5) 1140 (27. 7) 784 (19) 736 (17. 9) Extensive DVT Extensive PE NT-pro. BNP ≥ 500 pg/m. L* Prior VTE *PE only Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.
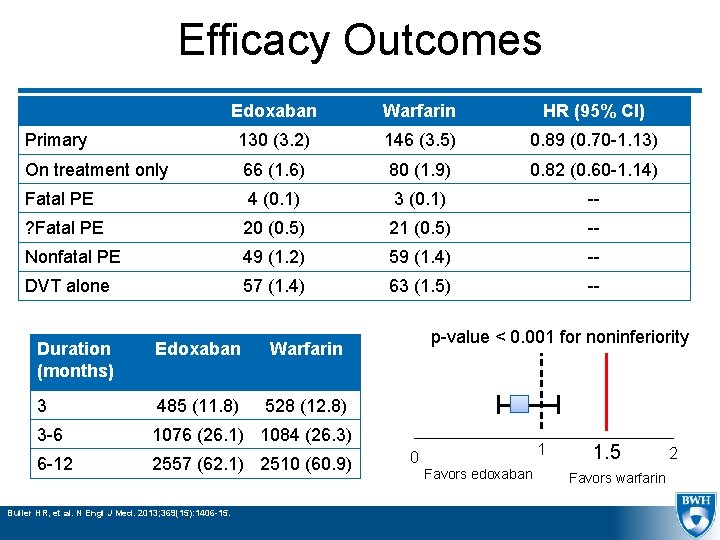
Efficacy Outcomes Edoxaban Warfarin HR (95% CI) Primary 130 (3. 2) 146 (3. 5) 0. 89 (0. 70 -1. 13) On treatment only 66 (1. 6) 80 (1. 9) 0. 82 (0. 60 -1. 14) Fatal PE 4 (0. 1) 3 (0. 1) -- ? Fatal PE 20 (0. 5) 21 (0. 5) -- Nonfatal PE 49 (1. 2) 59 (1. 4) -- DVT alone 57 (1. 4) 63 (1. 5) -- Duration (months) Edoxaban Warfarin 3 485 (11. 8) 528 (12. 8) 3 -6 1076 (26. 1) 1084 (26. 3) 6 -12 2557 (62. 1) 2510 (60. 9) Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15. p-value < 0. 001 for noninferiority 0 1 Favors edoxaban 1. 5 Favors warfarin 2
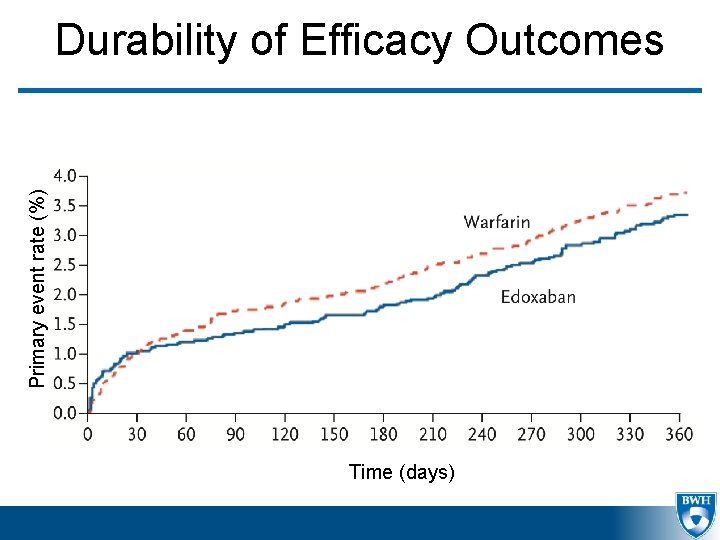
Primary event rate (%) Durability of Efficacy Outcomes Time (days)
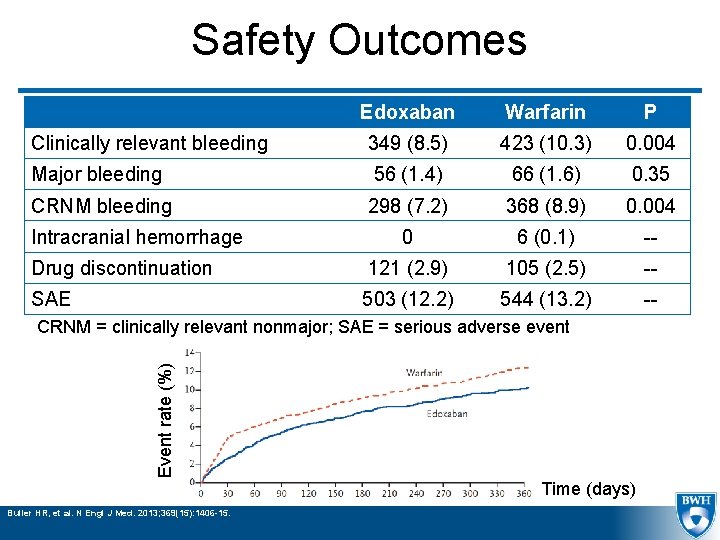
Safety Outcomes Edoxaban Warfarin P Clinically relevant bleeding 349 (8. 5) 423 (10. 3) 0. 004 Major bleeding 56 (1. 4) 66 (1. 6) 0. 35 CRNM bleeding 298 (7. 2) 368 (8. 9) 0. 004 0 6 (0. 1) -- Drug discontinuation 121 (2. 9) 105 (2. 5) -- SAE 503 (12. 2) 544 (13. 2) -- Intracranial hemorrhage Event rate (%) CRNM = clinically relevant nonmajor; SAE = serious adverse event Time (days) Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.
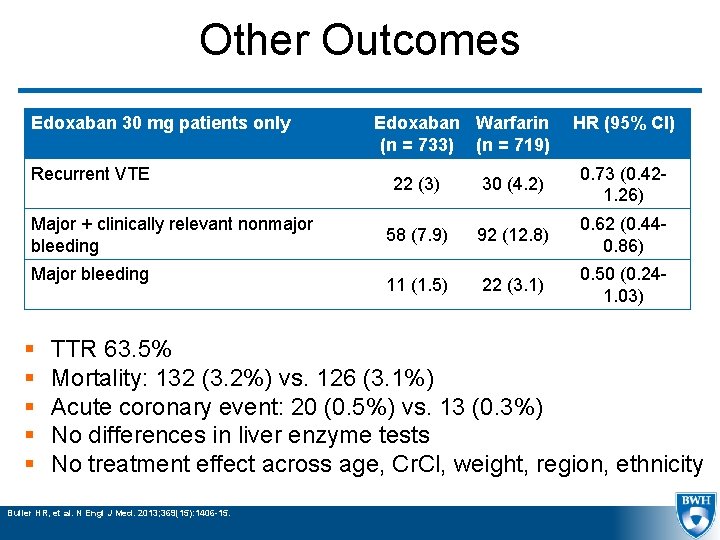
Other Outcomes Edoxaban 30 mg patients only Recurrent VTE Major + clinically relevant nonmajor bleeding Major bleeding § § § Edoxaban Warfarin (n = 733) (n = 719) HR (95% CI) 22 (3) 30 (4. 2) 0. 73 (0. 421. 26) 58 (7. 9) 92 (12. 8) 0. 62 (0. 440. 86) 11 (1. 5) 22 (3. 1) 0. 50 (0. 241. 03) TTR 63. 5% Mortality: 132 (3. 2%) vs. 126 (3. 1%) Acute coronary event: 20 (0. 5%) vs. 13 (0. 3%) No differences in liver enzyme tests No treatment effect across age, Cr. Cl, weight, region, ethnicity Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.

Discussion: Patients § Significant disease burden – ~40% per study definitions of extensive DVT and PE – Approximately 1/3 RV dysfunction on CT, elevated BNP § Subgroups addressed: – Advanced age (~13% were ≥ 75 years) – Renal impairment (~6% had Cr. Cl 30 -50) – Weight (~12% were ≤ 60 kg and ~15% were >100 kg) Baroletti S, Szumita P. Crit Pathways in Cardiol. 2004; 3: 205 -8. Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.

Discussion: Intervention § 60 mg dose tested for SPAF and VTE prophylaxis after orthopedic surgery § Dose adjustment for ~16% of patients with similar efficacy/safety as 60 mg dose § Use of parenteral anticoagulation? Baroletti S, Szumita P. Crit Pathways in Cardiol. 2004; 3: 205 -8. Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.

Discussion: Intervention § Flexible (ad hoc) treatment duration – Dabigatran = 6 months – Rivaroxaban = prespecified 3, 6 or 12 months – Apixaban = 6 months – Extension studies not available for edoxaban § Center TTR monitoring

Discussion: Endpoints § Standard definitions with central adjudication § Composite bleeding endpoint § Would ICH and fatal bleeding have been significantly lower if tested? Results summary: Recurrent VTE: edoxaban noninferior Clinically relevant bleeding: edoxaban noninferior Major bleeding: edoxaban noninferior CRNM bleeding: edoxaban superior Baroletti S, Szumita P. Crit Pathways in Cardiol. 2004; 3: 205 -8. Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.

Discussion: Statistics § Stricter noninferiority margin than prior studies § Double blind design to reduce suspicion bias § Met power requirements (276 events vs. 220 events) Baroletti S, Szumita P. Crit Pathways in Cardiol. 2004; 3: 205 -8. Buller HR, et al. N Engl J Med. 2013; 369(15): 1406 -15.

Conclusions § Edoxaban provides a convenient, safe and effective alternative to warfarin. § Edoxaban adds valuable experience on the use of FXa inhibitors in patients with: – – Submassive PE and extensive DVT Weight extremes Advanced age Renal impairment § Additional data is required for direct comparisons of DOACs, DAPT, ASA > 100 mg, reversal, treatment beyond 12 months, cancer, systemic thrombolysis…

References Bauersachs R, Berkowitz SD, Brenner B, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010; 363(26): 2499 -510. Buller HR, Decousus H, Grosso MA, et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N Engl J Med. 2013; 369(15): 1406 -15. Buller HR, Prins MH, Lensing AWA, et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012; 366(14): 1287 -97. Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009; 361(12): 1139 -51. Fontana P, Goldhaber SZ, Bounameaux H. Direct oral anticoagulants in the treatment and long-term prevention of venous thrombo-embolism. Eur Heart J. 2014; 35(28): 1836 -1843. Labropoulos N, Spentzouris G, Gasparis AP, Meissner M. Impact and clinical significance of recurrent venous thromboembolism. Br J Surg. 2010; 97: 989 -99. Mahmoodi BK, Gansevoort RT, Naess IA, et al. Association of mild to moderate chronic kidney disease with venous thromboembolism: pooled analysis of five prospective general population cohorts. Circulation. 2012; 126(16): 1964 -71. Martinez C, Cohen AT, Rietbrock S. Epidemiology of first and recurrent venous thromboembolism: a population-based cohort study in patients without active cancer. Thromb Haemost. 2014; 112: [epub ahead of print].

References Prins MH, Lensing AWA, Bauersachs R, et al. Oral rivaroxaban versus standard therapy for the treatment of symptomatic venous thromboembolism: a pooled analysis of the EINSTEIN-DVT and PE randomized studies. Thrombosis Journal. 2013; 11: 21 -31. Raskob G, Buller H, Prins M, et al. Edoxaban for the long-term treatment of venous thromboembolism: rationale and design of the Hokusai-venous thromboembolism study – methodological implications for clinical trials. J Thromb Haemost. 2013; 11: 1287 -94. Ridout G, de la Motte S, Niemczyk S, et al. Effect of renal function on edoxaban pharmacokinetics (PK) and on population PK/PK-PD model. J Clin Pharmacol. 2009; 49: 1124. Schulman S, Kakkar AK, Goldhaber SZ, et al. Treatment of acute venous thromboembolism with dabigatran or wafarin and pooled analysis. Circulation. 2014; 129: 764 -72. Schulman S, Kearon C. Definition of major bleeding in clinical investigations of antihemostatic medicinal products in non-surgical patients. J Thromb Haemost. 2005; 3: 692 -4. Schulman S, Kearon C, Kakkar AK, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009; 361(24): 2342 -52. Schulman S, Kearon C, Kakkar AK, et al. Extended use of dabigatran, warfarin, or placebo in venous thromboembolism. N Engl J Med. 2013; 368(8): 709 -18. Weitz JI, Connolly SJ, Patel I, et al. Randomised, parallel-group, multicentre, multinational phase 2 study comparing edoxaban, an oral factor Xa inhibitor, with warfarin for stroke prevention in patients with atrial fibrillation. Thromb Haemost. 2010; 104: 633 -41.

Questions? Thank you to Dr. Evan Nix, Pharm. D and Dr. Craig Beavers, Pharm. D, from ACCP Cardiology PRN. Special acknowledgement for Ahmed Aldemerdash, Pharm. D.

Thank you for attending! § If you would like to have your resident present, would like to be a mentor, or have questions or comments please e-mail the journal club at accpcardsprnjournalclub@gmail. com or craig. beaverspharmd@gmail. com § Our next Journal Club will be in late April – Laura Halder from Abbott Northwestern will be presenting on OSLER Trial from ACC. 15
- Slides: 30


















































