ACCP Cardiology PRN Journal Club September 2018 Mentor

ACCP Cardiology PRN Journal Club September 2018

Mentor: Dr. Ian Hollis is a clinical specialist at the University of North Carolina Hospitals and Clinics. He received his Doctor of Pharmacy from the Ohio State University and went on to complete his PGY 1 pharmacy practice residency at the University of Michigan and a PGY 2 cardiology pharmacy residency at UNC. Dr. Hollis covers the cardiac surgery and advanced heart failure services and serves as the director of the PGY 2 cardiology pharmacy residency.

Presenter: Dr. Helen Leung is the PGY 2 Cardiology Pharmacy Resident at Michigan Medicine. A native of California, she received her Doctor of Pharmacy from Touro University California and completed her PGY 1 pharmacy residency at Memorial Hermann – Texas Medical Center in Houston, TX. Dr. Leung’s practice areas of interest include anticoagulation, advanced heart failure/heart transplantation, and general cardiology.

PARAMEDIC 2 Trial Helen Leung, Pharm. D PGY-2 Cardiology Pharmacy Resident University of Michigan Mentor: Ian Hollis, Pharm. D, BCPS-AQ Cardiology

Disclosure • The collaborators of this presentation have no disclosures.

Background • Role of epinephrine in cardiac arrest – Alpha-adrenergic receptor mediated arteriole constriction – Increased coronary blood flow; possible return of spontaneous circulation • Pitfalls of epinephrine – Dysrhythmias and increased myocardial oxygen demand – Worse neurologic outcomes Perkins GD, et al. N Engl J Med 2018; 379: 711 -721

Methods Design Multicenter, randomized, double-blind, placebo-controlled Population 8014 patients in the United Kingdom with out-of-hospital cardiac arrest Intervention Parenteral epinephrine 1 mg vs 0. 9% saline placebo Standard care Study Period December 2014 to October 2017 Follow up 30 days Inclusion Received advanced life support by trial-trained paramedics Exclusion Pregnancy, age <16 years old, cardiac arrest from anaphylaxis or asthma, administration of epinephrine prior to arrival of trialtrained paramedics Perkins GD, et al. N Engl J Med 2018; 379: 711 -721

Objectives • Primary Outcome: – Rate of survival at 30 days • Secondary Outcome: – Rate of survival until hospital admission – Length of stay in the ICU – Rates of survival at hospital discharge and at 3 months – Neurologic outcomes at hospital discharge and at 3 months Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
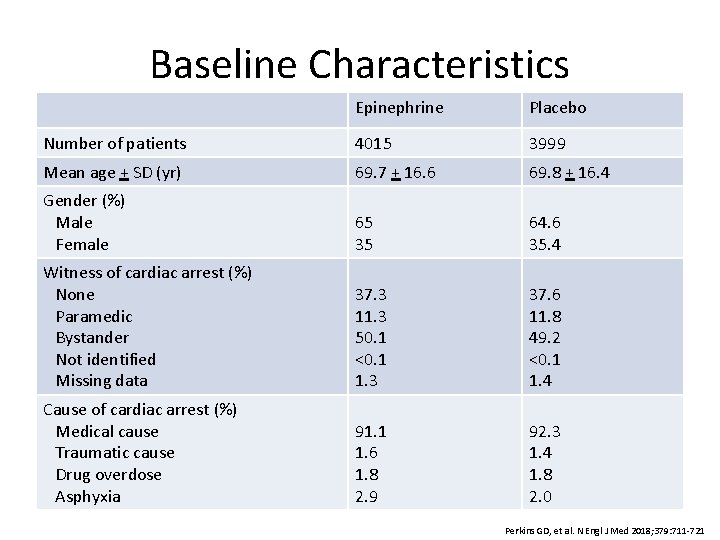
Baseline Characteristics Epinephrine Placebo Number of patients 4015 3999 Mean age + SD (yr) 69. 7 + 16. 6 69. 8 + 16. 4 Gender (%) Male Female 65 35 64. 6 35. 4 Witness of cardiac arrest (%) None Paramedic Bystander Not identified Missing data 37. 3 11. 3 50. 1 <0. 1 1. 3 37. 6 11. 8 49. 2 <0. 1 1. 4 Cause of cardiac arrest (%) Medical cause Traumatic cause Drug overdose Asphyxia 91. 1 1. 6 1. 8 2. 9 92. 3 1. 4 1. 8 2. 0 Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
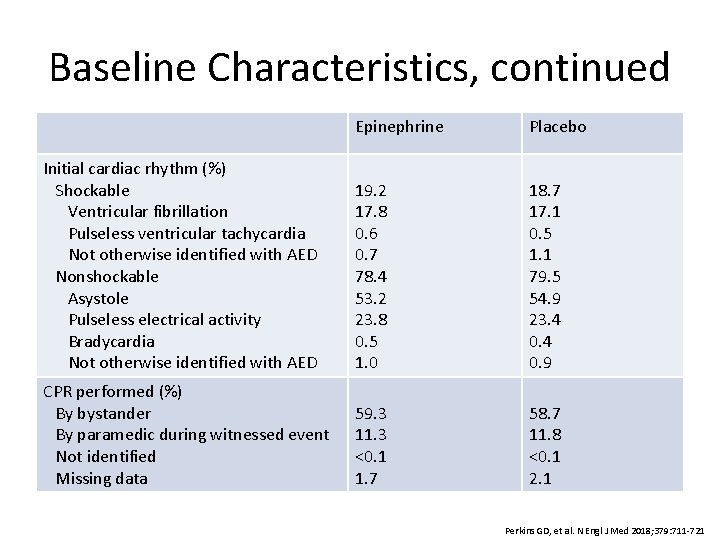
Baseline Characteristics, continued Epinephrine Placebo Initial cardiac rhythm (%) Shockable Ventricular fibrillation Pulseless ventricular tachycardia Not otherwise identified with AED Nonshockable Asystole Pulseless electrical activity Bradycardia Not otherwise identified with AED 19. 2 17. 8 0. 6 0. 7 78. 4 53. 2 23. 8 0. 5 1. 0 18. 7 17. 1 0. 5 1. 1 79. 5 54. 9 23. 4 0. 9 CPR performed (%) By bystander By paramedic during witnessed event Not identified Missing data 59. 3 11. 3 <0. 1 1. 7 58. 7 11. 8 <0. 1 2. 1 Perkins GD, et al. N Engl J Med 2018; 379: 711 -721

Statistics • Anticipated target enrollment: 8000 patients • Intention-to-treat • Survival outcomes – Fixed-effect regression models • Bayesian analysis – Primary outcome and for survival with favorable neurologic outcome Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
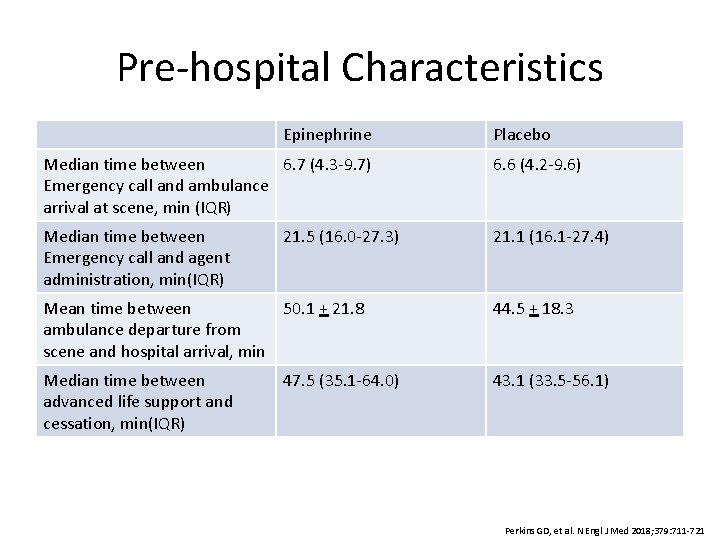
Pre-hospital Characteristics Epinephrine Placebo Median time between 6. 7 (4. 3 -9. 7) Emergency call and ambulance arrival at scene, min (IQR) 6. 6 (4. 2 -9. 6) Median time between Emergency call and agent administration, min(IQR) 21. 1 (16. 1 -27. 4) 21. 5 (16. 0 -27. 3) Mean time between 50. 1 + 21. 8 ambulance departure from scene and hospital arrival, min 44. 5 + 18. 3 Median time between advanced life support and cessation, min(IQR) 43. 1 (33. 5 -56. 1) 47. 5 (35. 1 -64. 0) Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
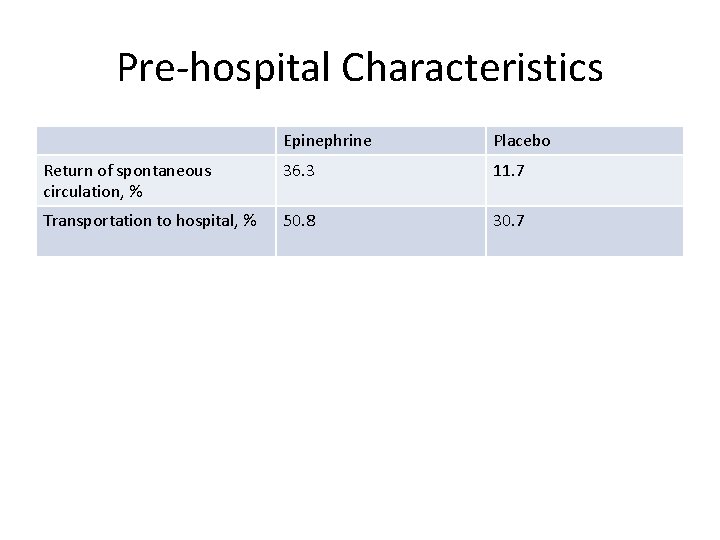
Pre-hospital Characteristics Epinephrine Placebo Return of spontaneous circulation, % 36. 3 11. 7 Transportation to hospital, % 50. 8 30. 7
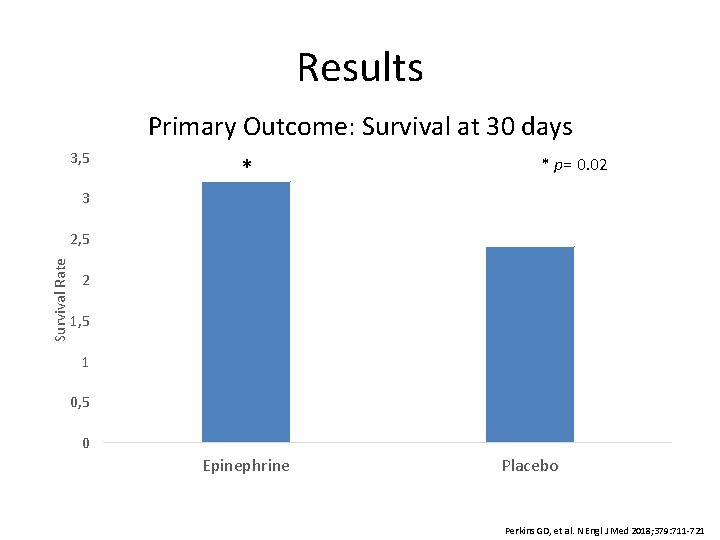
Results Primary Outcome: Survival at 30 days 3, 5 * * p= 0. 02 3 Survival Rate 2, 5 2 1, 5 1 0, 5 0 Epinephrine Placebo Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
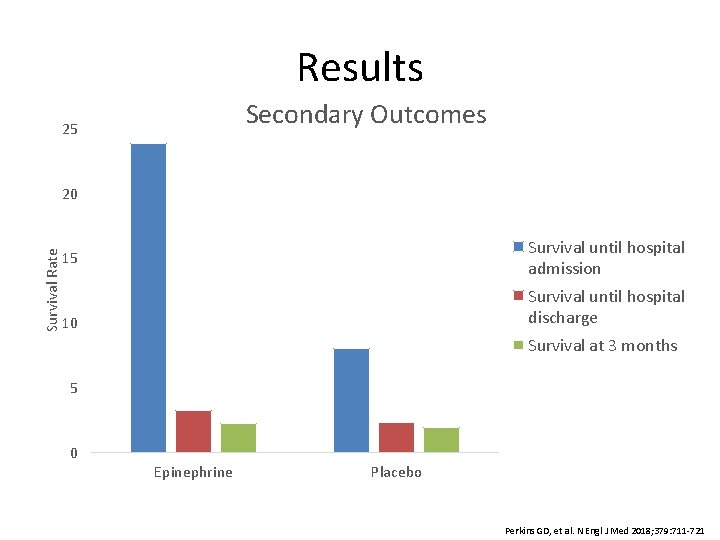
Results Secondary Outcomes 25 Survival Rate 20 Survival until hospital admission 15 Survival until hospital discharge 10 Survival at 3 months 5 0 Epinephrine Placebo Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
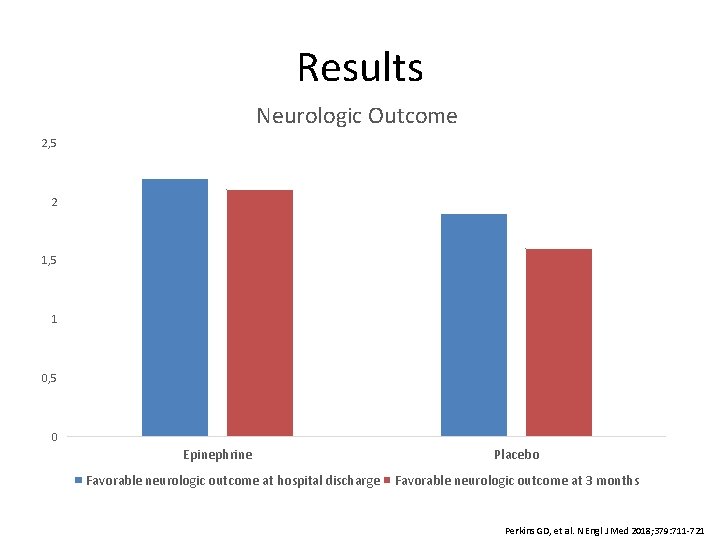
Results Neurologic Outcome 2, 5 2 1, 5 1 0, 5 0 Epinephrine Favorable neurologic outcome at hospital discharge Placebo Favorable neurologic outcome at 3 months Perkins GD, et al. N Engl J Med 2018; 379: 711 -721
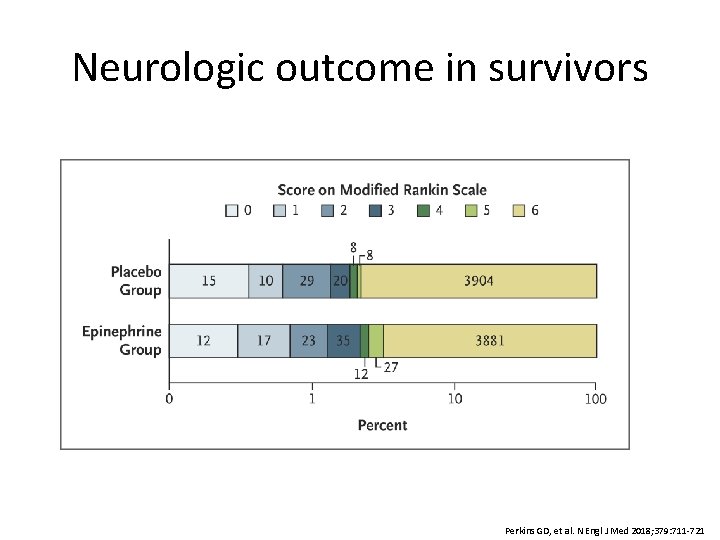
Neurologic outcome in survivors Perkins GD, et al. N Engl J Med 2018; 379: 711 -721

Conclusions • Epinephrine resulted in a significantly higher 30 -day survival rate compared to placebo – Number needed to treat: 112 • There was no difference in the rate of favorable neurologic outcome in survivors between epinephrine and placebo groups • More patients survived with severe neurologic impairment in the epinephrine group compared to placebo

Critique Strengths Weaknesses • Largest set of randomized • Protocol did not control or data on epinephrine use in measure in-hospital out-of-hospital cardiac arrest treatment • Baseline characteristics were • Administration of trial agents well-balanced between both took longer than other groups studies • Good external validity • 21 minutes • Excluded patients with early ROSC

Clinical Implications • For out-of-hospital cardiac arrest, epinephrine may improve 30 -day survival rates • Although more patients survive, many suffered poor neurologic outcomes and decreased quality of life – Mortality vs poor neurologic outcome • Early administration of epinephrine vs higher doses

Acknowledgements • Ian Hollis, Pharm. D, BCPS (AQ-Cardiology) • Thomas Szymanski, Pharm. D • Zachary Noel, Pharm. D, BCPS

PARAMEDIC 2 Trial Helen Leung, Pharm. D PGY-2 Cardiology Pharmacy Resident University of Michigan Mentor: Ian Hollis, Pharm. D, BCPS-AQ Cardiology

Questions?

Presenter: Dr. Michelle Palmer is the PGY 2 Cardiology Pharmacy Resident at the University of Chicago Medicine. She earned her Pharm. D from the University of Florida. She completed her PGY 1 residency at Inova Fairfax Medical Campus in Falls Church, VA. Her practice interests include heart failure, acute coronary syndromes, and mechanical circulatory support.

Study Comparing the Efficacy and Tolerability of Epinephrine and Norepinephrine in Cardiogenic Shock (Optima. CC) Michelle Palmer, Pharm. D PGY-2 Cardiology Pharmacy Resident University of Chicago Medicine

Disclosure Statement • I have no financial relationships with commercial interests that pertain to the content presented in this program

Acute Myocardial Infarction and Cardiogenic Shock • Acute myocardial infarction (AMI) accounts for 80% of cardiogenic shock cases • High mortality rates of 30 -40% • SHOCK (NEJM 1999) • Established shock criteria • Thirty day mortality was reduced by 17% with revascularization, but not significantly (p = 0. 11) • Significantly reduction in mortality at 30 days (p = 0. 01) and 60 days (p = 0. 03) in those younger than 75 years of age Thiele H et al. Eur Heart J. 2015; 36(20): 1223 -30. van Diepen S. Circulation. 2017; 136: e 232–e 268 Hochman JS et al. N Engl J Med 1999; 341: 625 -634
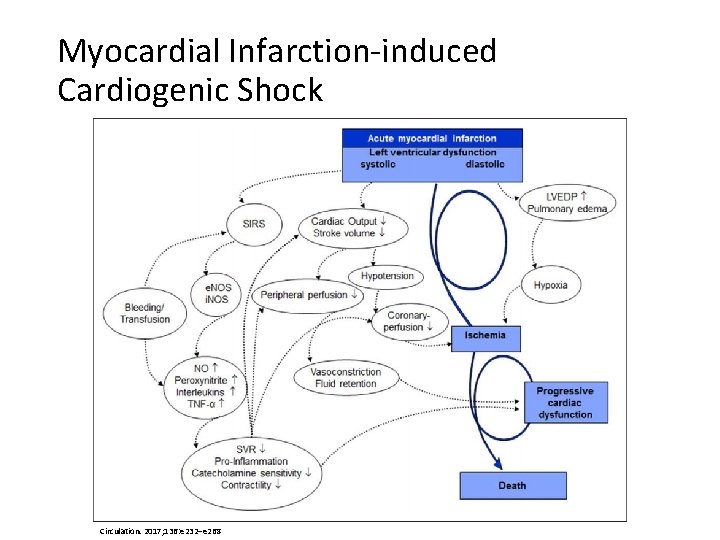
Myocardial Infarction-induced Cardiogenic Shock Circulation. 2017; 136: e 232–e 268
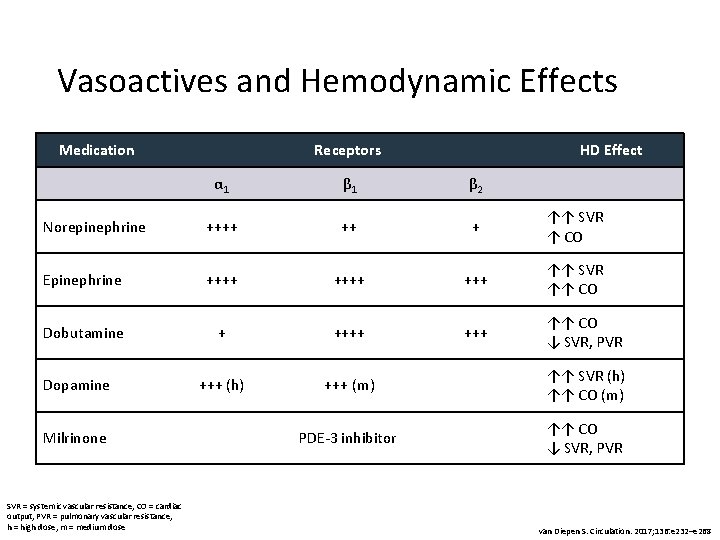
Vasoactives and Hemodynamic Effects Medication Receptors HD Effect α 1 β 2 Norepinephrine ++++ ++ + ↑↑ SVR ↑ CO Epinephrine ++++ +++ ↑↑ SVR ↑↑ CO Dobutamine + +++ ↑↑ CO ↓ SVR, PVR +++ (h) +++ (m) ↑↑ SVR (h) ↑↑ CO (m) PDE-3 inhibitor ↑↑ CO ↓ SVR, PVR Dopamine Milrinone SVR = systemic vascular resistance, CO = cardiac output, PVR = pulmonary vascular resistance, h = high dose, m = medium dose van Diepen S. Circulation. 2017; 136: e 232–e 268
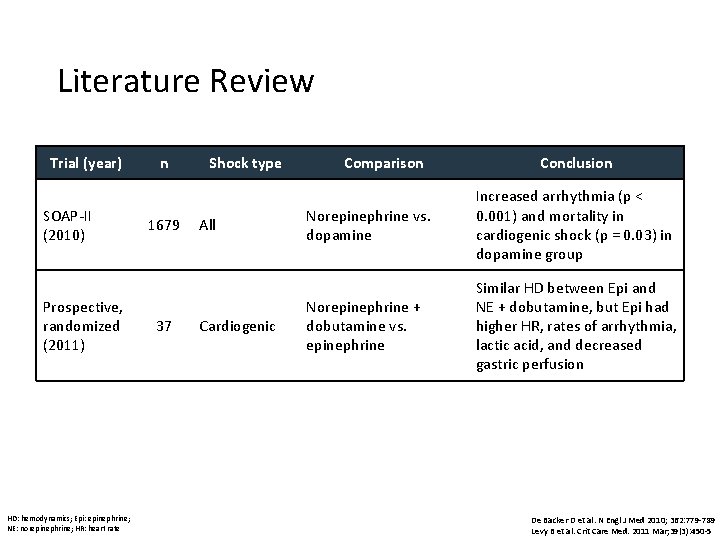
Literature Review Trial (year) SOAP-II (2010) Prospective, randomized (2011) HD: hemodynamics; Epi: epinephrine; NE: norepinephrine; HR: heart rate n 1679 37 Shock type All Cardiogenic Comparison Conclusion Norepinephrine vs. dopamine Increased arrhythmia (p < 0. 001) and mortality in cardiogenic shock (p = 0. 03) in dopamine group Norepinephrine + dobutamine vs. epinephrine Similar HD between Epi and NE + dobutamine, but Epi had higher HR, rates of arrhythmia, lactic acid, and decreased gastric perfusion De Backer D et al. N Engl J Med 2010; 362: 779 -789 Levy B et al. Crit Care Med. 2011 Mar; 39(3): 450 -5

Guideline Recommendations • Limited to management reviews and statements • European Heart Journal (2015): – Norepinephrine should be first choice as vasopressor – Use of catecholamines should be limited due to increase in oxygen consumption and tissue perfusion impairment – Dobutamine may be given simultaneously to norepinephrine to improve cardiac contractility • American Heart Association Scientific Statement (2017): – Classic wet and cold: norepinephrine or dopamine – Euvolemic cold and dry: norepinephrine or dopamine – Vasodilatory/mixed shock: norepinephrine Thiele H et al. Eur Heart J. 2015; 36(20): 1223 -30. Circulation. 2017; 136: e 232–e 268
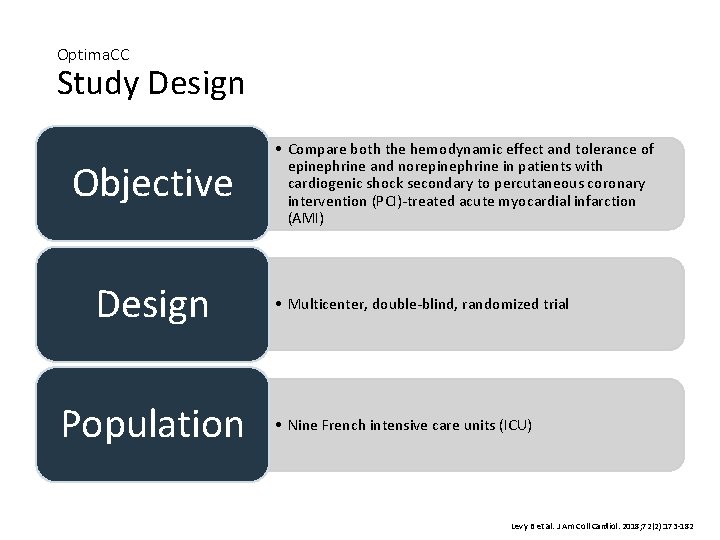
Optima. CC Study Design Objective Design Population • Compare both the hemodynamic effect and tolerance of epinephrine and norepinephrine in patients with cardiogenic shock secondary to percutaneous coronary intervention (PCI)-treated acute myocardial infarction (AMI) • Multicenter, double-blind, randomized trial • Nine French intensive care units (ICU) Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
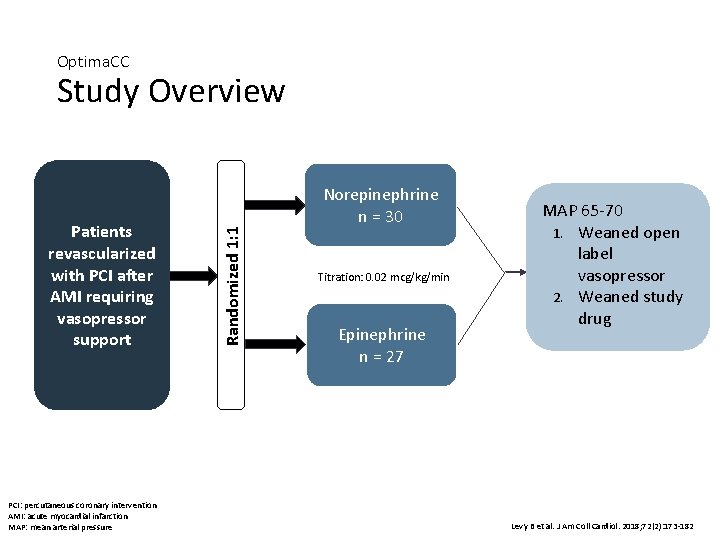
Optima. CC Patients revascularized with PCI after AMI requiring vasopressor support PCI: percutaneous coronary intervention AMI: acute myocardial infarction MAP: mean arterial pressure Randomized 1: 1 Study Overview Norepinephrine n = 30 Titration: 0. 02 mcg/kg/min Epinephrine n = 27 MAP 65 -70 1. Weaned open label vasopressor 2. Weaned study drug Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182

Optima. CC Study Population Inclusion • • ≥ 18 years old Cardiogenic shock (CS) due to AMI with successful revascularization via PCI SBP < 90 mm. Hg or MAP < 65 mm. Hg without vasopressor CI < 2. 2 L/min/m 2 without inotropes or vasopressors PCWP > 15 mm. Hg or ECHO evidence of high pressure EF < 40% without inotropes Evidence of tissue hypoperfusion PA catheter insertion CS: cardiogenic shock; AMI: acute myocardial infarction, SBP: systolic blood pressure; CI: cardiac index; PCWP: pulmonary capillary wedge pressure; ECHO: echocardiogram; EF: ejection fraction; PA: pulmonary artery; ECLS: extracorporeal life support Exclusion • • Shock of other origin Immediate indication for ECLS Age < 18 years Cardiac arrest with early signs of cerebral anoxia Septic, toxic, obstructive cardiomyopathy No medical insurance Under legal protection Considered moribund Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182

Optima. CC Methods Measured at hours 0, 2, 4, 6, 12, 24, 48, and 72: • • Vital signs, heart rate, MAP Systolic and diastolic arterial pressure Pulmonary artery pressures, cardiac index, LVEF Mixed venous oxygen saturation, arterial lactate Recorded daily for 7 days, then on days 14, 21, and 28 • Biological and microbiological data • Antibiotic therapy Measured at hours 0, 24, 48, and 72 • Troponin T • NT-pro. BNP Calculated variables • Cardiac index • Oxygen delivery and consumption indexes • Systemic vascular resistance, stroke volume MAP: mean arterial pressure LVEF: left ventricular ejection fraction Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
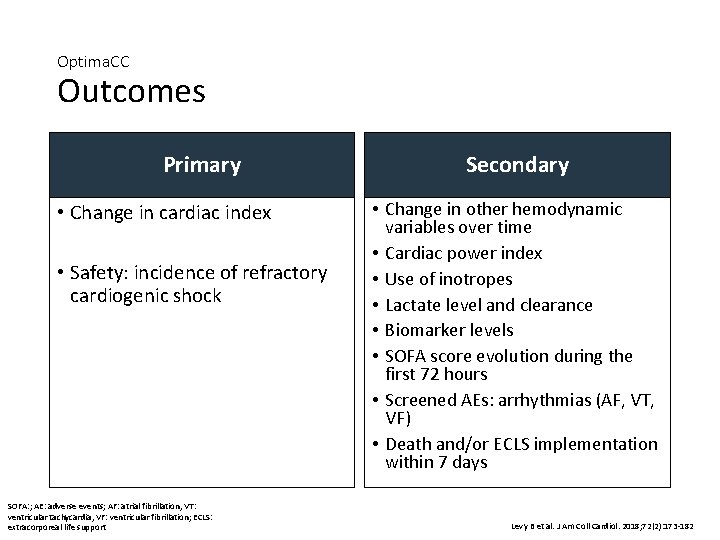
Optima. CC Outcomes Primary • Change in cardiac index • Safety: incidence of refractory cardiogenic shock SOFA: ; AE: adverse events; AF: atrial fibrillation, VT: ventricular tachycardia, VF: ventricular fibrillation; ECLS: extracorporeal life support Secondary • Change in other hemodynamic variables over time • Cardiac power index • Use of inotropes • Lactate level and clearance • Biomarker levels • SOFA score evolution during the first 72 hours • Screened AEs: arrhythmias (AF, VT, VF) • Death and/or ECLS implementation within 7 days Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182

Optima. CC Statistical Analysis • Patients needed for 80% power: 40 per group – Two-tailed significance: p < 0. 05 • Continuous variables – Described as median (interquartile range) – Wilcoxon test • Categorical variables – Described as frequencies (percentages) – Fisher exact test • Survival rates calculated using Kaplan-Meier analyses • Associations between treatment and adverse events assessed with the logistic regression model • Comparison of hemodynamics between groups with repeated-measures analysis Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
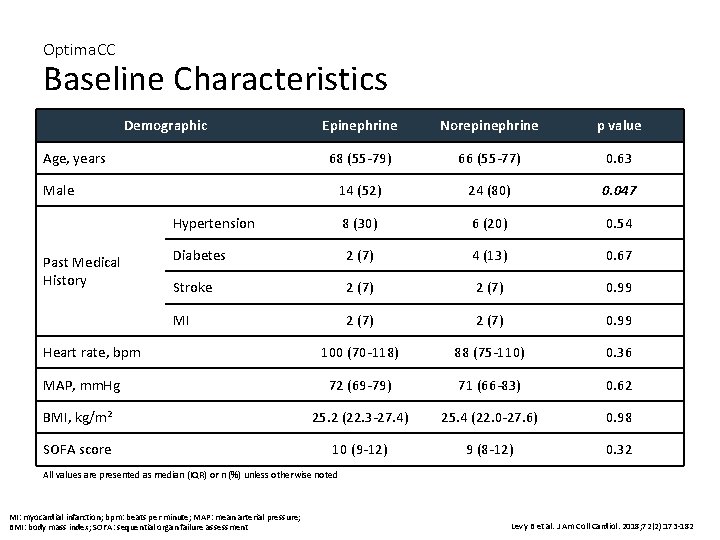
Optima. CC Baseline Characteristics Demographic Epinephrine Norepinephrine p value 68 (55 -79) 66 (55 -77) 0. 63 14 (52) 24 (80) 0. 047 Hypertension 8 (30) 6 (20) 0. 54 Diabetes 2 (7) 4 (13) 0. 67 Stroke 2 (7) 0. 99 MI 2 (7) 0. 99 100 (70 -118) 88 (75 -110) 0. 36 MAP, mm. Hg 72 (69 -79) 71 (66 -83) 0. 62 BMI, kg/m 2 25. 2 (22. 3 -27. 4) 25. 4 (22. 0 -27. 6) 0. 98 SOFA score 10 (9 -12) 9 (8 -12) 0. 32 Age, years Male Past Medical History Heart rate, bpm All values are presented as median (IQR) or n (%) unless otherwise noted MI: myocardial infarction; bpm: beats per minute; MAP: mean arterial pressure; BMI: body mass index; SOFA: sequential organ failure assessment Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
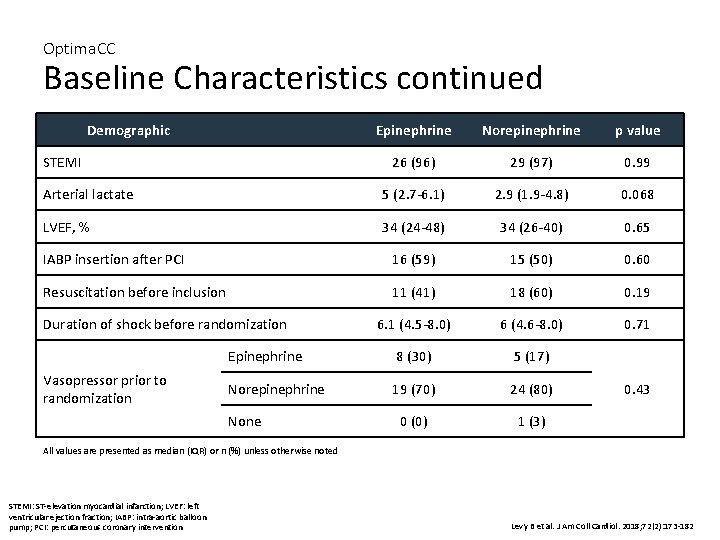
Optima. CC Baseline Characteristics continued Demographic Epinephrine Norepinephrine p value 26 (96) 29 (97) 0. 99 Arterial lactate 5 (2. 7 -6. 1) 2. 9 (1. 9 -4. 8) 0. 068 LVEF, % 34 (24 -48) 34 (26 -40) 0. 65 IABP insertion after PCI 16 (59) 15 (50) 0. 60 Resuscitation before inclusion 11 (41) 18 (60) 0. 19 6. 1 (4. 5 -8. 0) 6 (4. 6 -8. 0) 0. 71 Epinephrine 8 (30) 5 (17) Norepinephrine 19 (70) 24 (80) 0 (0) 1 (3) STEMI Duration of shock before randomization Vasopressor prior to randomization None 0. 43 All values are presented as median (IQR) or n (%) unless otherwise noted STEMI: ST-elevation myocardial infarction; LVEF: left ventricular ejection fraction; IABP: intra-aortic balloon pump; PCI: percutaneous coronary intervention Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
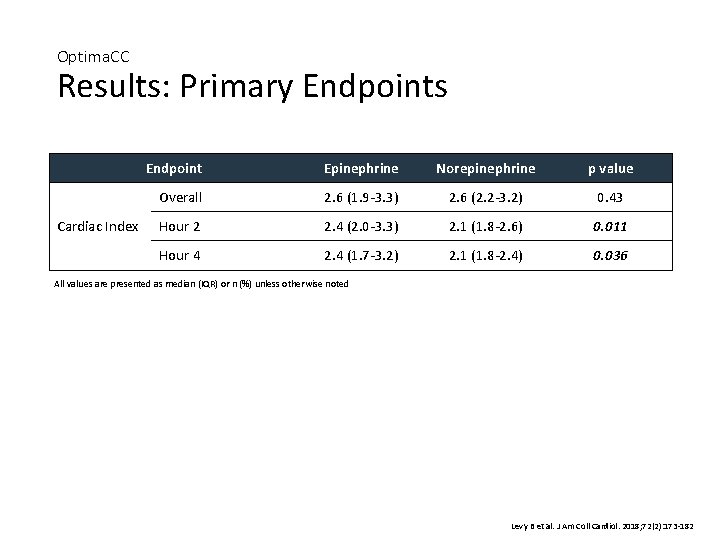
Optima. CC Results: Primary Endpoints Cardiac Index Endpoint Epinephrine Norepinephrine p value Overall 2. 6 (1. 9 -3. 3) 2. 6 (2. 2 -3. 2) 0. 43 Hour 2 2. 4 (2. 0 -3. 3) 2. 1 (1. 8 -2. 6) 0. 011 Hour 4 2. 4 (1. 7 -3. 2) 2. 1 (1. 8 -2. 4) 0. 036 All values are presented as median (IQR) or n (%) unless otherwise noted Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
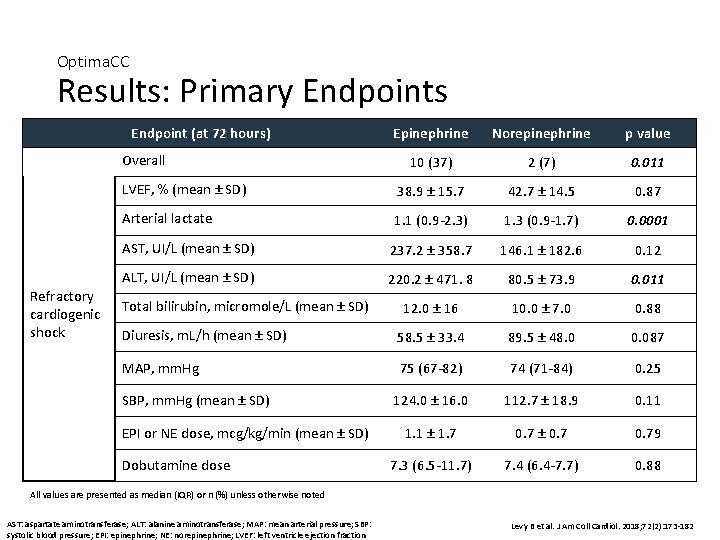
Optima. CC Results: Primary Endpoints Endpoint (at 72 hours) Epinephrine Norepinephrine p value 10 (37) 2 (7) 0. 011 LVEF, % (mean ± SD) 38. 9 ± 15. 7 42. 7 ± 14. 5 0. 87 Arterial lactate 1. 1 (0. 9 -2. 3) 1. 3 (0. 9 -1. 7) 0. 0001 AST, UI/L (mean ± SD) 237. 2 ± 358. 7 146. 1 ± 182. 6 0. 12 ALT, UI/L (mean ± SD) 220. 2 ± 471. 8 80. 5 ± 73. 9 0. 011 12. 0 ± 16 10. 0 ± 7. 0 0. 88 Diuresis, m. L/h (mean ± SD) 58. 5 ± 33. 4 89. 5 ± 48. 0 0. 087 MAP, mm. Hg 75 (67 -82) 74 (71 -84) 0. 25 124. 0 ± 16. 0 112. 7 ± 18. 9 0. 11 1. 1 ± 1. 7 0. 7 ± 0. 79 7. 3 (6. 5 -11. 7) 7. 4 (6. 4 -7. 7) 0. 88 Overall Refractory cardiogenic shock Total bilirubin, micromole/L (mean ± SD) SBP, mm. Hg (mean ± SD) EPI or NE dose, mcg/kg/min (mean ± SD) Dobutamine dose All values are presented as median (IQR) or n (%) unless otherwise noted AST: aspartate aminotransferase; ALT: alanine aminotransferase; MAP: mean arterial pressure; SBP: systolic blood pressure; EPI: epinephrine; NE: norepinephrine; LVEF: left ventricle ejection fraction Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
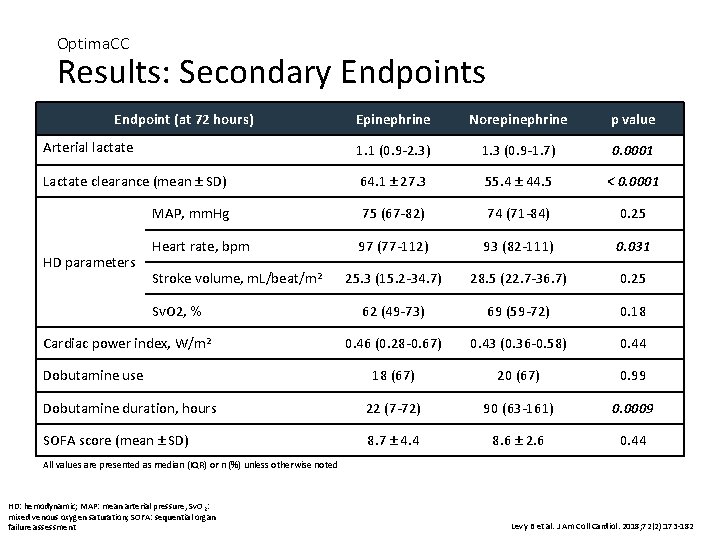
Optima. CC Results: Secondary Endpoints Endpoint (at 72 hours) Epinephrine Norepinephrine p value Arterial lactate 1. 1 (0. 9 -2. 3) 1. 3 (0. 9 -1. 7) 0. 0001 Lactate clearance (mean ± SD) 64. 1 ± 27. 3 55. 4 ± 44. 5 < 0. 0001 MAP, mm. Hg 75 (67 -82) 74 (71 -84) 0. 25 Heart rate, bpm 97 (77 -112) 93 (82 -111) 0. 031 25. 3 (15. 2 -34. 7) 28. 5 (22. 7 -36. 7) 0. 25 62 (49 -73) 69 (59 -72) 0. 18 0. 46 (0. 28 -0. 67) 0. 43 (0. 36 -0. 58) 0. 44 18 (67) 20 (67) 0. 99 Dobutamine duration, hours 22 (7 -72) 90 (63 -161) 0. 0009 SOFA score (mean ± SD) 8. 7 ± 4. 4 8. 6 ± 2. 6 0. 44 HD parameters Stroke volume, m. L/beat/m 2 Sv. O 2, % Cardiac power index, W/m 2 Dobutamine use All values are presented as median (IQR) or n (%) unless otherwise noted HD: hemodynamic; MAP: mean arterial pressure, Sv. O 2: mixed venous oxygen saturation; SOFA: sequential organ failure assessment Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
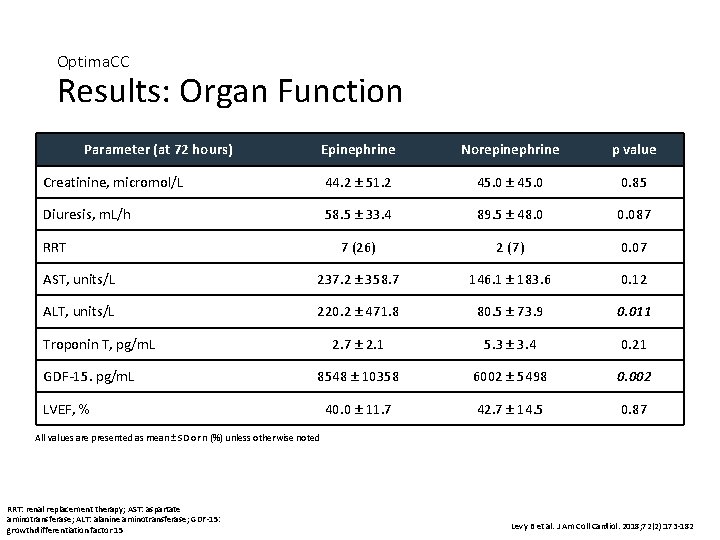
Optima. CC Results: Organ Function Parameter (at 72 hours) Epinephrine Norepinephrine p value Creatinine, micromol/L 44. 2 ± 51. 2 45. 0 ± 45. 0 0. 85 Diuresis, m. L/h 58. 5 ± 33. 4 89. 5 ± 48. 0 0. 087 7 (26) 2 (7) 0. 07 AST, units/L 237. 2 ± 358. 7 146. 1 ± 183. 6 0. 12 ALT, units/L 220. 2 ± 471. 8 80. 5 ± 73. 9 0. 011 2. 7 ± 2. 1 5. 3 ± 3. 4 0. 21 8548 ± 10358 6002 ± 5498 0. 002 40. 0 ± 11. 7 42. 7 ± 14. 5 0. 87 RRT Troponin T, pg/m. L GDF-15. pg/m. L LVEF, % All values are presented as mean ± SD or n (%) unless otherwise noted RRT: renal replacement therapy; AST: aspartate aminotransferase; ALT: alanine aminotransferase; GDF-15: growth differentiation factor 15 Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182
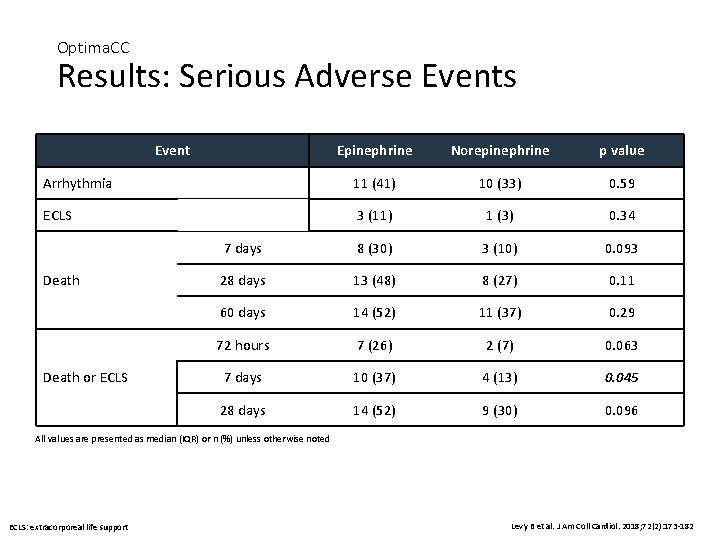
Optima. CC Results: Serious Adverse Events Event Epinephrine Norepinephrine p value Arrhythmia 11 (41) 10 (33) 0. 59 ECLS 3 (11) 1 (3) 0. 34 7 days 8 (30) 3 (10) 0. 093 28 days 13 (48) 8 (27) 0. 11 60 days 14 (52) 11 (37) 0. 29 72 hours 7 (26) 2 (7) 0. 063 7 days 10 (37) 4 (13) 0. 045 28 days 14 (52) 9 (30) 0. 096 Death or ECLS All values are presented as median (IQR) or n (%) unless otherwise noted ECLS: extracorporeal life support Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182

Optima. CC Authors’ conclusions • Epinephrine is associated with transient increases in cardiac index compared to norepinephrine, but with increases in: – – Refractory shock Prolonged acidosis Lactatemia Heart rate • Main aim of resuscitation during cardiogenic shock is to improve global perfusion, not obtain a predefined cardiac index • To obtain the same degree of tissue perfusion, the effects of epinephrine may mimic adrenergic overstimulation and a catecholamine-induced cardiomyopathy Levy B et al. J Am Coll Cardiol. 2018; 72(2): 173 -182

Optima. CC Critique Strengths • Randomized, controlled trial • Required revascularization • Inclusion criteria indicative of primary cardiogenic shock state that would benefit from vasopressors ECLS: extracorporeal life support Limitations • Refractory shock not a pre-specified outcome • Refractory shock seemingly driven by difference in arterial lactate • Higher baseline lactate in the EPI group • Significantly longer duration of dobutamine in the norepinephrine group • No criteria or protocol for escalation of therapy (e. g. , ECLS) • High doses of vasoactives • Small study size limited to French intensive care units

Practical implications • May strengthen suggestions from EHJ and AHA statements regarding vasopressors therapy in cardiogenic shock – Norepinephrine until hemodynamics are stabilized – Inotrope (such as dobutamine) after perfusion maintained • However, based on the several limitations of the study, epinephrine use cannot be excluded in cardiogenic shock • A larger, powered RCT is needed before drawing any concrete practical implications from this study

Acknowledgements • Ian Hollis, Pharm. D, BCPS (AQ-Cardiology) • Thomas Szymanski, Pharm. D • Zachary Noel, Pharm. D, BCPS

Study Comparing the Efficacy and Tolerability of Epinephrine and Norepinephrine in Cardiogenic Shock (Optima. CC) Michelle Palmer, Pharm. D PGY-2 Cardiology Pharmacy Resident University of Chicago Medicine

October Journal Club • Presenters • Nathan Wayne, University of Kentucky • Sarah Schaidle, University of Kentucky • Mentor • Mary Parker, clinical specialist at the VAMC in Durham, NC • Articles • ASCEND (fish oil data) • ARRIVE (aspirin data from ASCEND) • Date • Wednesday, October 31 st at 3 PM EST/2 PM CST
- Slides: 50