ACCP Adult Medicine PRN Journal Club Announcements Thank

ACCP Adult Medicine PRN Journal Club

Announcements • Thank you for attending the ACCP Adult Medicine PRN Journal Club! • If there any suggestions, please let us know

Bezlotoxumab for Prevention of Recurrent Clostridium difficile Infection (MODIFY I and MODIFY II) Samarth P. Shah, Pharm. D, BCPS PGY 2 Internal Medicine Resident Methodist University Hospital /University of Tennessee Health Sciences Center Memphis, TN February 28, 2017

Disclosure I have no conflicts to report related to financial or personal relationships with commercial entities (or their competitors) that may be referenced in this presentation.

Clostridium difficile • Gram positive, spore forming anaerobe • Resides in the gastrointestinal tract • Endogenous and exogenous toxin producer Leading Cause of Healthcare-Associated infectious diarrhea Clin Infect Dis 1998; 26: 1027 -36
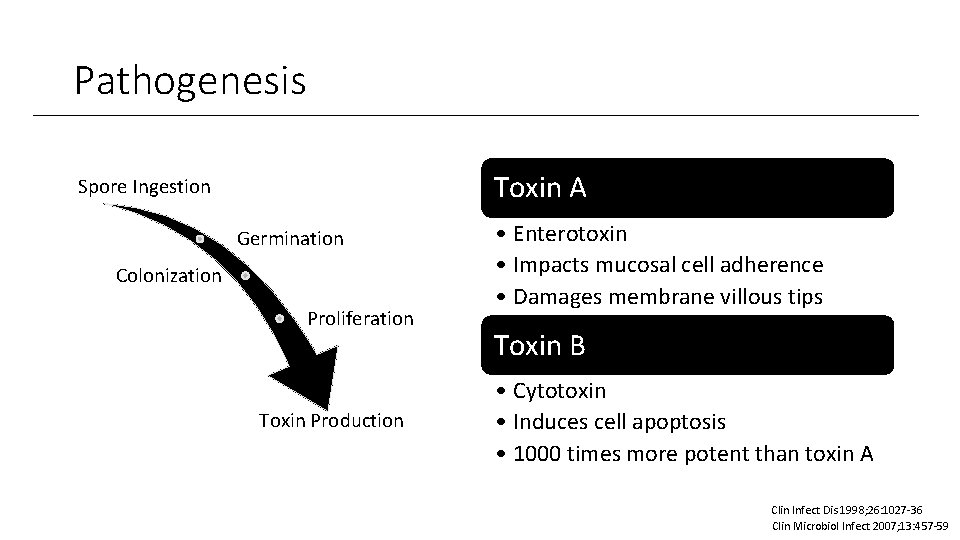
Pathogenesis Toxin A Spore Ingestion Germination Colonization Proliferation Toxin Production • Enterotoxin • Impacts mucosal cell adherence • Damages membrane villous tips Toxin B • Cytotoxin • Induces cell apoptosis • 1000 times more potent than toxin A Clin Infect Dis 1998; 26: 1027 -36 Clin Microbiol Infect 2007; 13: 457 -59

Recurrent Clostridium difficile Infection • Extremely common • Up to 20% recurrence after initial episodes • Up to 60% recurrence after multiple episodes Risk Factors • • • Antibiotic usage Age > 65 Prolonged hospitalization Prior clostridium difficile NAP 1 strain infection Inflammatory bowel disease Immunodeficiency Increased hospitalizations, healthcare cost, morbidity and mortality Clin Infect Dis 2005; 41: 1254 -60 N Engl J Med 2015; 372: 1539 -48

Recurrent Clostridium difficile infection Literature Review Rates of Recurrent CDI Metronidazole vs. vancomycin 1 Recurrence rates • Recurrent CDI (22. 8%) • Rates similar between vancomycin and metronidazole Fidaxomicin vs. vancomycin 2, 3 Recurrence rates (P=0. 005) • Prior recurrence 3 – fidaxomicin 19% vs. vancomycin 35% • Recurrence significantly less in the fidaxomicin group Fecal Transplantation vs. vancomycin ± bowel lavage 4 Sustained cure rates (P < 0. 001) • Fecal transplant 94%; vancomycin 31%; vanc + lavage 23% Time to recurrence • Fecal transplant 35 days; vancomycin 23 days; vanc + lavage 25 days Probiotics vs. placebo 5 Antibiotic-associated diarrhea (P=0. 71) • Probiotics 10. 8% vs. placebo 10. 4% CDAD (P=0. 35) • Probiotics 0. 8% vs. placebo 1. 2% Intravenous Immune Globulin (IVIG)6 No clinical or statistical significant difference in recurrence rates between patients that received IVIG vs. those that did not 1. JAMA Intern Med. doi: 10. 1001/jamainternmed. 2016. 9045 2. N Engl J Med 2011; 364: 422 -31 3. Clin Infect Dis 2012; 55: S 154 -61 4. N Eng J Med 2013; 368: 407 -15 5. Lancet 2013; 382: 1249 -57 6. Am J Infect Control 2007; 35: 131

Current Treatment for Recurrence of Clostridium difficile infection • Metronidazole • Oral vancomycin pulse therapy • Fidaxomicin • Oral fecal transplant Overall lack of great options to treat and have sustained cure rate!

Passive Immunity with Monoclonal Antibodies Actoxumab • Blocks binding site of C. diff toxin A • 10 mg/kg infusion over 60 minutes Bezlotoxumab (Zinplava®) • Blocks binding site of C. diff toxin B • 10 mg/kg infusion over 60 minutes • FDA approved for prevention of CDI Zinplava®[package insert]. Merck 2017


Purpose and Design • Purpose – To determine the efficacy of Bezlotoxumab in prevention of recurrent Clostridium difficile infection • Design – • MODIFY I - Randomized, double-blinded, placebo controlled • MODIFY II - Randomized, double-blinded, placebo controlled N Engl J Med 2017; 376: 305 -17

Inclusion Criteria • Age ≥ 18 years old • Primary or recurrent Clostridium difficile infection receiving oral standard-of-care antibiotics • Metronidazole • Vancomycin • Fidaxomicin Duration of 10 -14 days N Engl J Med 2017; 376: 305 -17

Exclusion Criteria • Inflammatory bowel disease • Pregnant or breast feeding • IVIG within previous 6 months • Cholestyramine or rifaximin within previous 14 days N Engl J Med 2017; 376: 305 -17

Patient Selection • Patients on oral standard-of-care antibiotics (metronidazole, vancomycin, fidaxomicin) with C. difficile infection • C. difficile defined as diarrhea (≥ 3 unformed bowel movements in 24 hours), with a stool test result that was positive for C. difficile • • Cytotoxicity assays Culture with toxin detection Strain typing Commercial assays that detect toxin B or its gene N Engl J Med 2017; 376: 305 -17
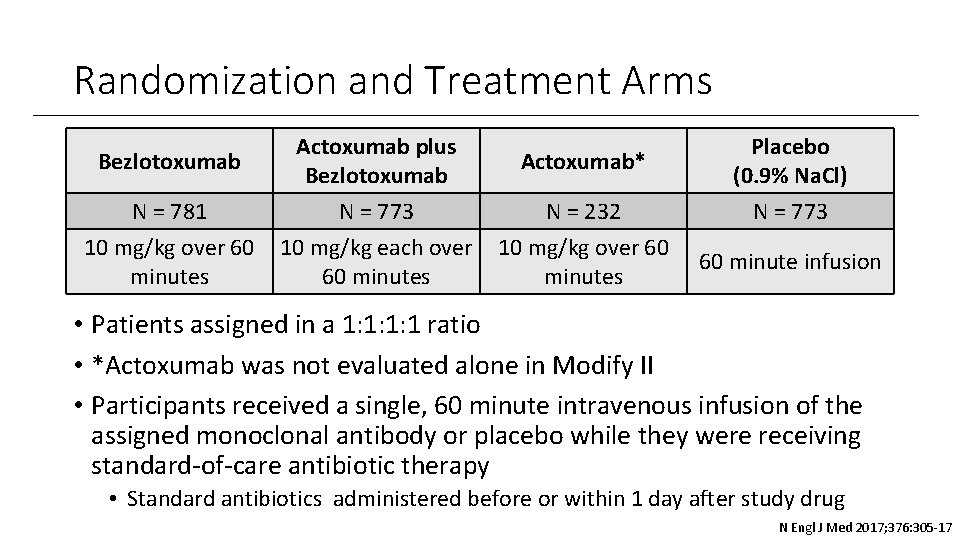
Randomization and Treatment Arms Bezlotoxumab Actoxumab plus Bezlotoxumab Actoxumab* N = 781 10 mg/kg over 60 minutes N = 773 10 mg/kg each over 60 minutes N = 232 10 mg/kg over 60 minutes Placebo (0. 9% Na. Cl) N = 773 60 minute infusion • Patients assigned in a 1: 1: 1: 1 ratio • *Actoxumab was not evaluated alone in Modify II • Participants received a single, 60 minute intravenous infusion of the assigned monoclonal antibody or placebo while they were receiving standard-of-care antibiotic therapy • Standard antibiotics administered before or within 1 day after study drug N Engl J Med 2017; 376: 305 -17

Procedures and Follow Up • Patients recorded unformed bowel movements daily until day 80 to 90 after the infusion • New episodes of diarrhea were monitored through telephone contact N Engl J Med 2017; 376: 305 -17
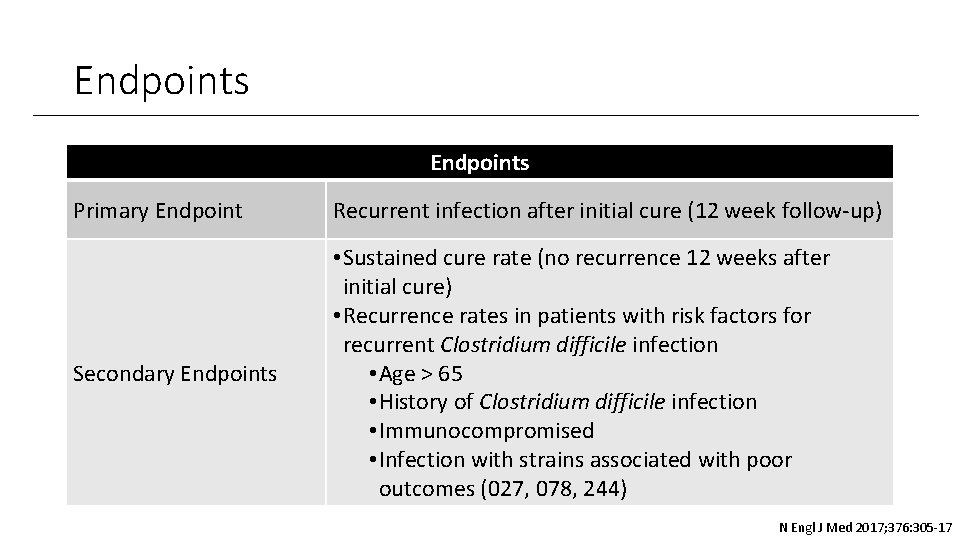
Endpoints Primary Endpoint Recurrent infection after initial cure (12 week follow-up) Secondary Endpoints • Sustained cure rate (no recurrence 12 weeks after initial cure) • Recurrence rates in patients with risk factors for recurrent Clostridium difficile infection • Age > 65 • History of Clostridium difficile infection • Immunocompromised • Infection with strains associated with poor outcomes (027, 078, 244) N Engl J Med 2017; 376: 305 -17

Statistical Analysis • Both powered to determine the efficacy of bezlotoxumab alone or in combination with actoxumab for prevention of recurrent C. difficile infection • MODIFY I – adaptive design • Comparisons between placebo and treatment with regard to primary endpoint were performed at a two-sided alpha level of 0. 025 (MODIFY I) and 0. 05 (MODIFY II) • Both trials had a 90% power to determine a difference of 9 to 10 percentage points in recurrent infection • Planned pooling of MODIFY I and II data for subgroup analysis N Engl J Med 2017; 376: 305 -17
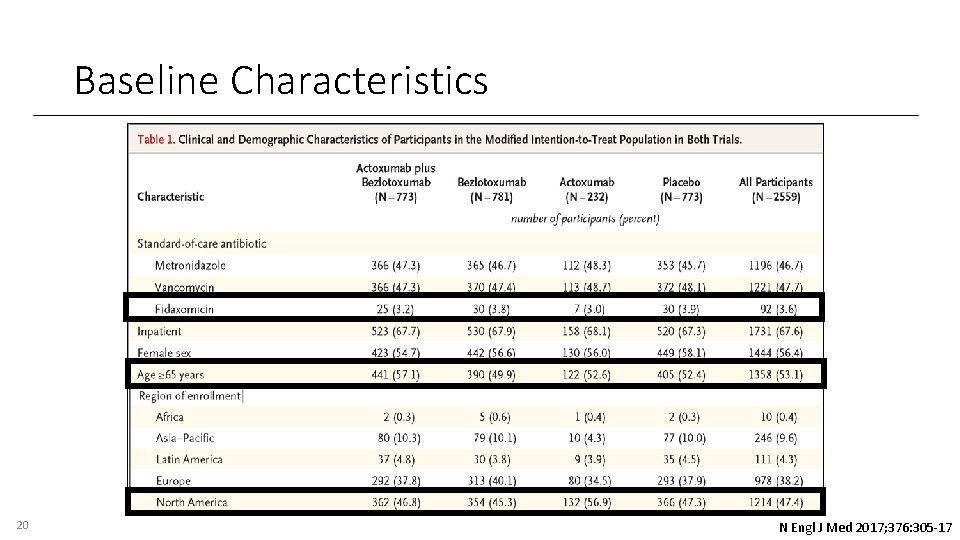
Baseline Characteristics 20 N Engl J Med 2017; 376: 305 -17
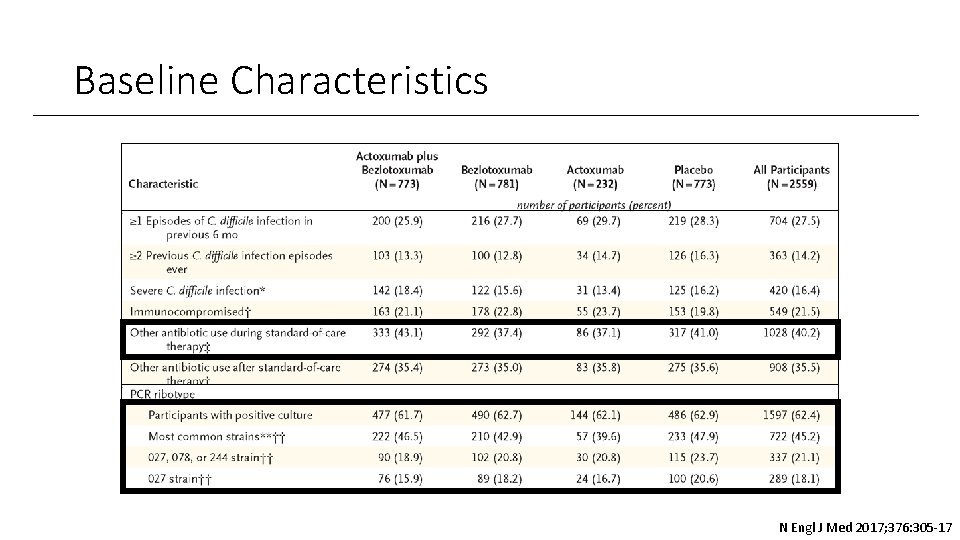
Baseline Characteristics N Engl J Med 2017; 376: 305 -17
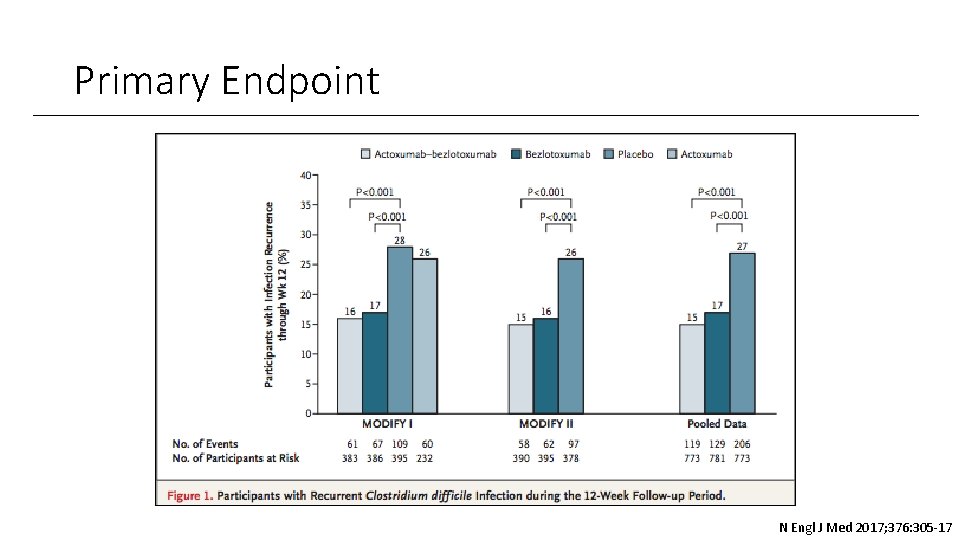
Primary Endpoint N Engl J Med 2017; 376: 305 -17
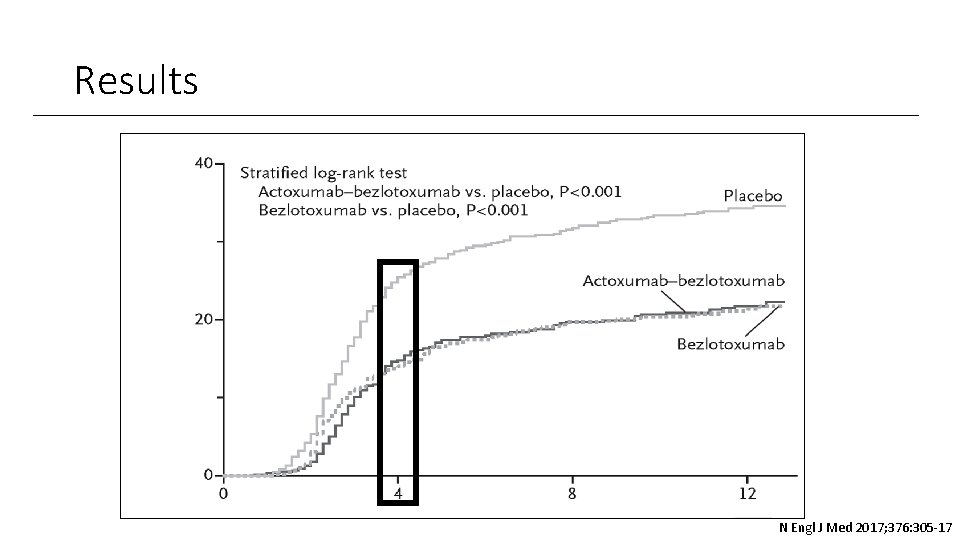
Results N Engl J Med 2017; 376: 305 -17
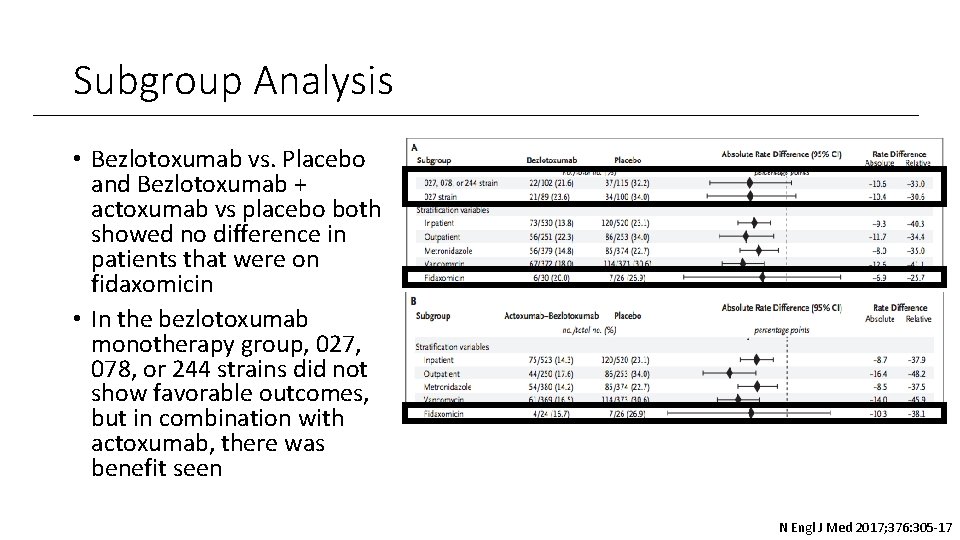
Subgroup Analysis • Bezlotoxumab vs. Placebo and Bezlotoxumab + actoxumab vs placebo both showed no difference in patients that were on fidaxomicin • In the bezlotoxumab monotherapy group, 027, 078, or 244 strains did not show favorable outcomes, but in combination with actoxumab, there was benefit seen N Engl J Med 2017; 376: 305 -17

Sustained Cure MODIFY I • Bezlotoxumab • 60% (232 of 386) • Actoxumab plus Bezlotoxumab • 59% (225 of 383) • Placebo • 55% (218 of 395) MODIFY II • Bezlotoxumab • 67% (264 of 395) • Actoxumab plus Bezlotoxumab • 57% (224 of 390) • Placebo • 52% (197 of 398) N Engl J Med 2017; 376: 305 -17

Study Conclusion • Single intravenous dose of bezlotoxumab against Clostridium difficile toxin B, when given with standard-of-care antibiotics, provided protection against recurrent Clostridium difficile infection for up to 12 weeks • Of note, otherapies used for prevention of current Clostridium difficile recurrence were not allowed • Impact of combination of bezlotoxumab with otherapies unknown N Engl J Med 2017; 376: 305 -17
Study Critique Strengths • Patient selection well defined • Mention of PCR ribotypes to determine which strains therapy would be useful in • Patients randomized appropriately • • • Limitations Antibiotic choice physician’s discretion Limited assessment with fidaxomicin Relative risks and benefits not fully understood Pooled data analysis Not studied with other approaches Adaptive design in MODIFY I could have impacted results N Engl J Med 2017; 376: 305 -17

Practical Implications • May be an option in certain patients in prevention of Clostridium difficile recurrence • Clinical utility and strain typing • Will be cost prohibitive • Inpatient utilization • Outpatient utilization • Less effective against NAP 1 strains • Alternative treatments cost effective and highly successful N Engl J Med 2017; 376: 305 -17

Bezlotoxumab for Prevention of Recurrent Clostridium difficile Infection (MODIFY I and MODIFY II) Samarth P. Shah, Pharm. D, BCPS PGY 2 Internal Medicine Resident Methodist University Hospital /University of Tennessee Health Sciences Center Memphis, TN February 28, 2017

Acknowledgements • Jared Heiles, Pharm. D; PGY 1 Pharmacy Resident; Methodist University Hospital, Memphis, TN • Journal Club Mentor – Winter J. Smith, Pharm. D, BCPS; Texas Tech University Health Sciences Center School of Pharmacy, Dallas, TX • ACCP Adult Medicine PRN Journal Club Coordinator – Ryan Owens, Pharm. D, BCPS; The University of Oklahoma College of Pharmacy, OKC, OK
Thank you for attending! • If you would like to have your resident present, would like to serve as a mentor, or have questions or comments please e-mail us at accpamedprn@gmail. com or ryan-owens@ouhsc. edu • Look for our announcement of our March Journal Club soon!
- Slides: 31