ACCP Cardiology PRN Journal Club Announcements Thank you

ACCP Cardiology PRN Journal Club
Announcements � Thank you attending the ACCP Cardiology PRN Journal Club � Thank you if you attended last time � I have created a PB Works Site that will house our recorded calls, handouts, and Summary/Q&A documents. I will be e-mailing the link to this in the next 1 -2 weeks. � Still trying to explore option of getting CE. � If there any suggestions, please let us know.

Angiotensin-Neprilysin Inhibition versus Enalapril in Heart Failure (PARADIGMHF) Prepared By: Robert K. Tunney, Pharm. D, BCPS PGY 2 Cardiology Resident, Vanderbilt University Medical Center, Nashville, TN Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.

Disclosures �I have no financial interest or affiliation with the manufacturer of any marketed product discussed herein

Background � Heart Failure (HF) � Constellation of symptoms � 5. 1 million Americans (and rising) with HF � ~20% lifetime risk for the development of HF in patients > 40 years old � Lack of consistency in mortality benefit with angiotensin-receptor blockers (ARBs) � CHARM (2004) and Val-He. FT (2001) � 2013 ACC/AHA Guidelines: ARBs recommended in patients intolerant to ACE-I therapy (IA) Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004. Yancy CW, et al. JACC. 2013; 62(16): e 147 -e 239.
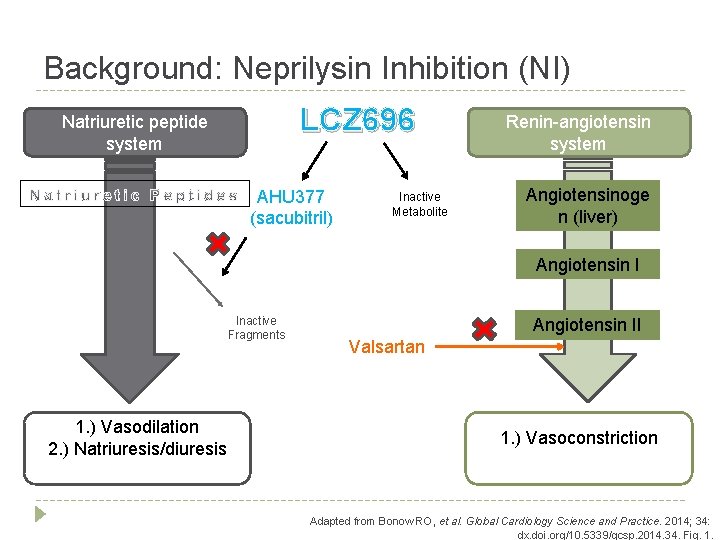
Background: Neprilysin Inhibition (NI) LCZ 696 Natriuretic peptide system AHU 377 (sacubitril) Inactive Metabolite Renin-angiotensin system Angiotensinoge n (liver) Angiotensin I Inactive Fragments 1. ) Vasodilation 2. ) Natriuresis/diuresis Angiotensin II Valsartan 1. ) Vasoconstriction Adapted from Bonow RO, et al. Global Cardiology Science and Practice. 2014; 34: dx. doi. org/10. 5339/gcsp. 2014. 34. Fig. 1.

Background and Purpose � OCTAVE trial (2004): Neprilysin (omapatrilat) and ACE-I combination intolerance secondary to angioedema � LCZ 696: sacubitril + valsartan (fixed molecular complex) � Purpose � Assess the relative superiority of morbidity and mortality benefits between LCZ 696 200 mg BID and ACE inhibition (enalapril 10 mg BID) in patients with HFr. EF Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
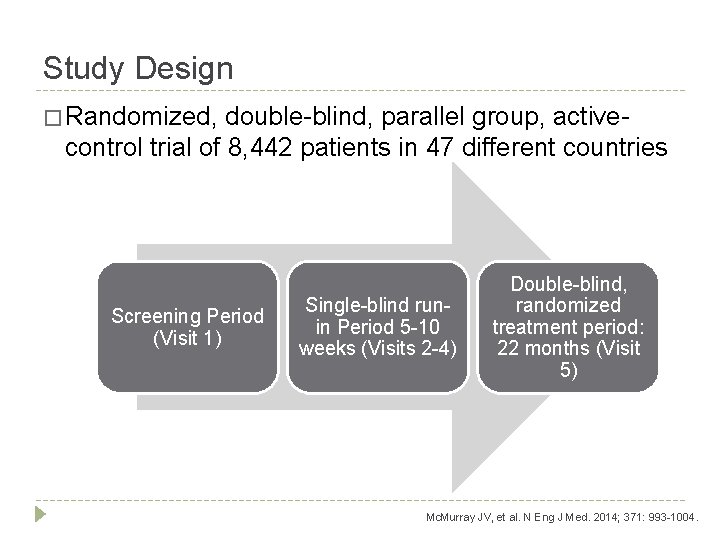
Study Design � Randomized, double-blind, parallel group, active- control trial of 8, 442 patients in 47 different countries Screening Period (Visit 1) Single-blind runin Period 5 -10 weeks (Visits 2 -4) Double-blind, randomized treatment period: 22 months (Visit 5) Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
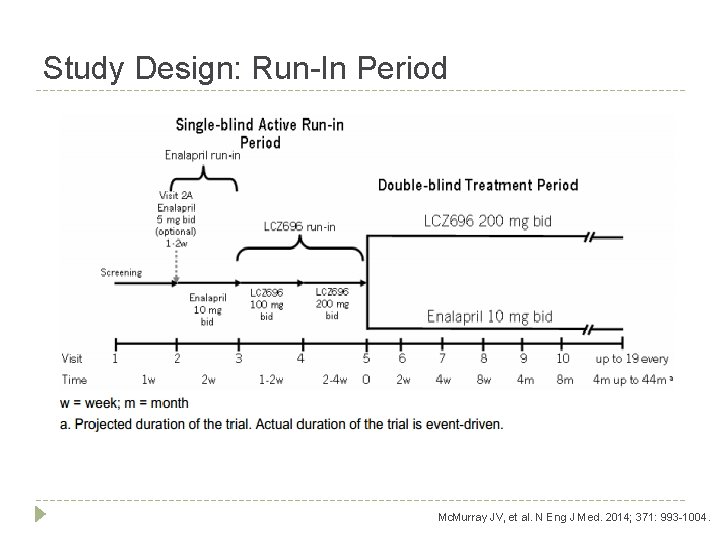
Study Design: Run-In Period Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
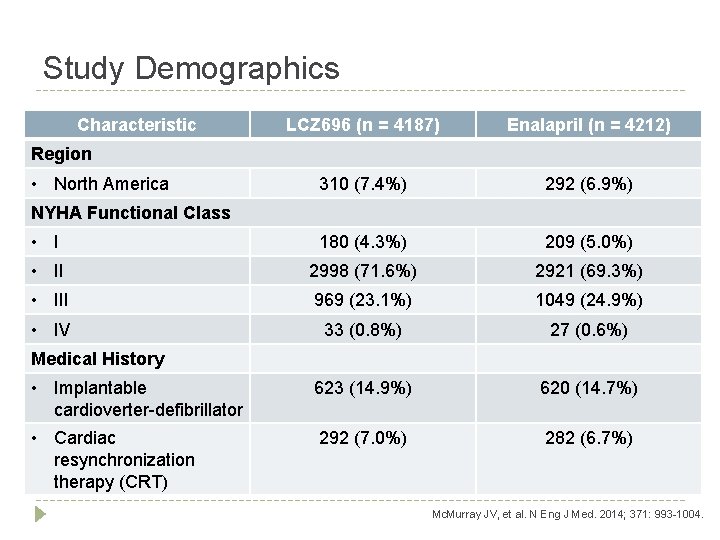
Study Demographics Characteristic LCZ 696 (n = 4187) Enalapril (n = 4212) 310 (7. 4%) 292 (6. 9%) • I 180 (4. 3%) 209 (5. 0%) • II 2998 (71. 6%) 2921 (69. 3%) • III 969 (23. 1%) 1049 (24. 9%) • IV 33 (0. 8%) 27 (0. 6%) • Implantable cardioverter-defibrillator 623 (14. 9%) 620 (14. 7%) • Cardiac resynchronization therapy (CRT) 292 (7. 0%) 282 (6. 7%) Region • North America NYHA Functional Class Medical History Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
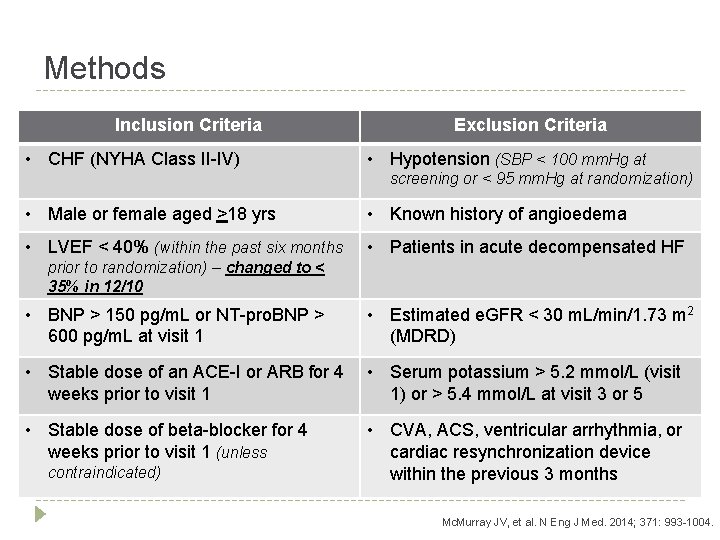
Methods Inclusion Criteria • CHF (NYHA Class II-IV) Exclusion Criteria • Hypotension (SBP < 100 mm. Hg at screening or < 95 mm. Hg at randomization) • Male or female aged >18 yrs • Known history of angioedema • LVEF < 40% (within the past six months • Patients in acute decompensated HF prior to randomization) – changed to < 35% in 12/10 • BNP > 150 pg/m. L or NT-pro. BNP > 600 pg/m. L at visit 1 • Estimated e. GFR < 30 m. L/min/1. 73 m 2 (MDRD) • Stable dose of an ACE-I or ARB for 4 • Serum potassium > 5. 2 mmol/L (visit weeks prior to visit 1 1) or > 5. 4 mmol/L at visit 3 or 5 • Stable dose of beta-blocker for 4 weeks prior to visit 1 (unless contraindicated) • CVA, ACS, ventricular arrhythmia, or cardiac resynchronization device within the previous 3 months Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.

Outcomes � Primary: Time to first occurrence of composite endpoint: CV death or HF hospitalization � Secondary: Improvement in the Kansas City Cardiomyopathy Questionaire (KCCQ) at 8 months All-cause mortality Decline in renal function New-onset atrial fibrillation Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
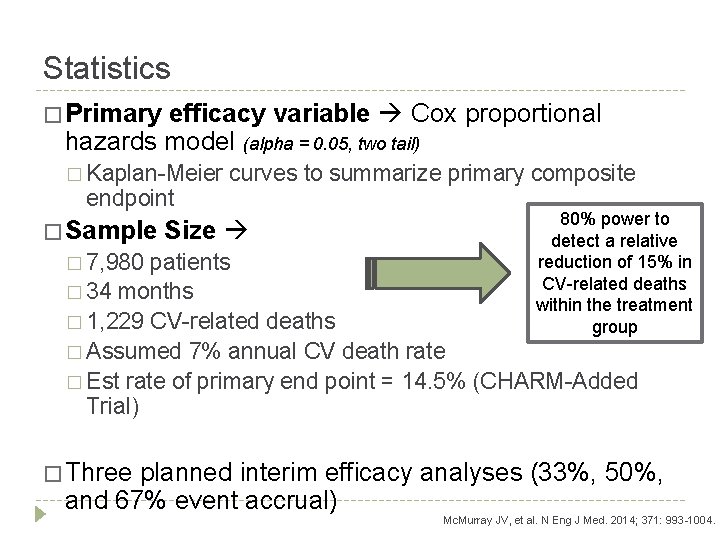
Statistics � Primary efficacy variable Cox proportional hazards model (alpha = 0. 05, two tail) � Kaplan-Meier curves to summarize primary composite endpoint � Sample 80% power to detect a relative reduction of 15% in CV-related deaths within the treatment group Size � 7, 980 patients � 34 months � 1, 229 CV-related deaths � Assumed 7% annual CV death rate � Est rate of primary end point = 14. 5% (CHARM-Added Trial) � Three planned interim efficacy analyses (33%, 50%, and 67% event accrual) Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
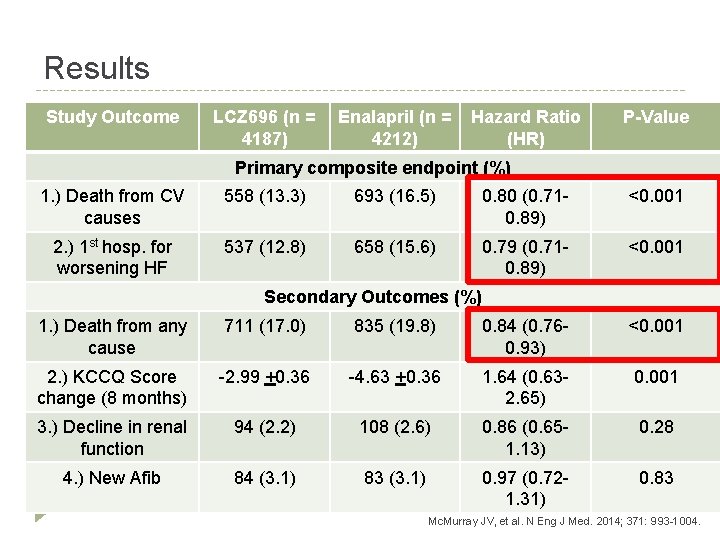
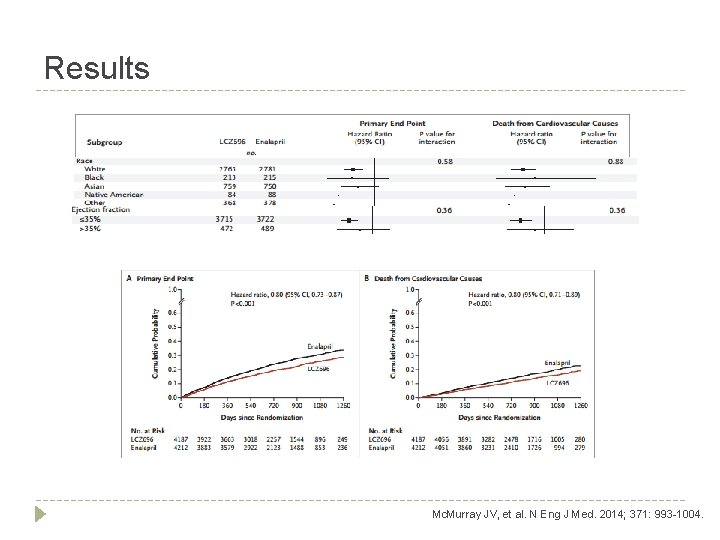
Results Study Outcome LCZ 696 (n = 4187) Enalapril (n = 4212) Hazard Ratio (HR) P-Value Primary composite endpoint (%) 1. ) Death from CV causes 558 (13. 3) 693 (16. 5) 0. 80 (0. 710. 89) <0. 001 2. ) 1 st hosp. for worsening HF 537 (12. 8) 658 (15. 6) 0. 79 (0. 710. 89) <0. 001 Secondary Outcomes (%) 1. ) Death from any cause 711 (17. 0) 835 (19. 8) 0. 84 (0. 760. 93) <0. 001 2. ) KCCQ Score change (8 months) -2. 99 +0. 36 -4. 63 +0. 36 1. 64 (0. 632. 65) 0. 001 3. ) Decline in renal function 94 (2. 2) 108 (2. 6) 0. 86 (0. 651. 13) 0. 28 4. ) New Afib 84 (3. 1) 83 (3. 1) 0. 97 (0. 721. 31) 0. 83 Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
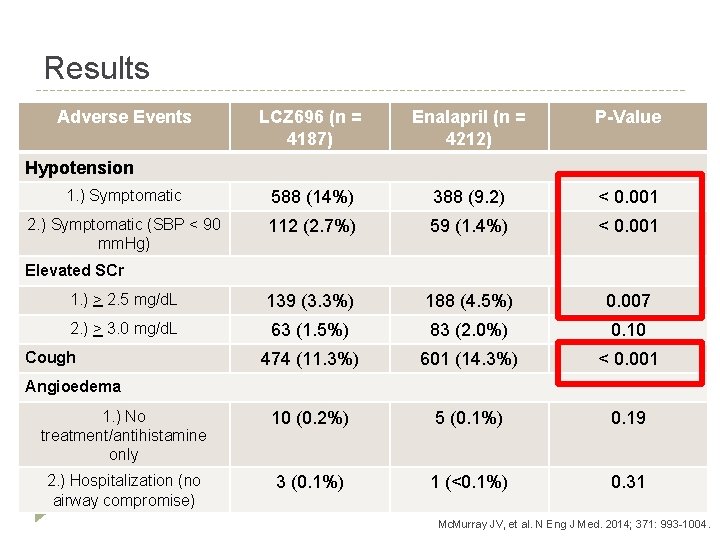
Results Adverse Events LCZ 696 (n = 4187) Enalapril (n = 4212) P-Value 1. ) Symptomatic 588 (14%) 388 (9. 2) < 0. 001 2. ) Symptomatic (SBP < 90 mm. Hg) 112 (2. 7%) 59 (1. 4%) < 0. 001 1. ) > 2. 5 mg/d. L 139 (3. 3%) 188 (4. 5%) 0. 007 2. ) > 3. 0 mg/d. L 63 (1. 5%) 83 (2. 0%) 0. 10 474 (11. 3%) 601 (14. 3%) < 0. 001 1. ) No treatment/antihistamine only 10 (0. 2%) 5 (0. 1%) 0. 19 2. ) Hospitalization (no airway compromise) 3 (0. 1%) 1 (<0. 1%) 0. 31 Hypotension Elevated SCr Cough Angioedema Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
Results Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.

Discussion � Inhibition of both angiotensin II receptors and neprilysin was more effective in reducing morbidity and mortality relative to the control therapy � Mean enalapril dose utilized = 18. 9 mg daily � CONSENSUS (1987): 18. 4 mg – 20 mg BID target dose � SOLVD (1991): 16. 6 mg – 10 mg BID target dose � LCZ 696 resulted in greater incidence of hypotension but was not associated with significant increases in serious angioedema Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.

Author’s Conclusion � Angiotensin-receptor-neprilysin inhibition with LCZ 696 proves superior to ACE inhibition alone in reducing the risk of death and re-hospitalization due to HF Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
Critique Strengths Study Design Robust statistical power High statistical significance Potential degree of clinical significance Weaknesses Were the “sickest” patients studied? Reproducibility to U. S. patients Under-representation of ICD/CRT patients and those of African descent Thought-Provoking Reduction in atrial fibrillation Comparative efficacy of valsartan 320 mg versus enalapril 20 mg Previous use of ACE/ARB Drug run-in period Cost-prohibitive therapy Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004. Yancy, Clyde W. "Commentary on PARADIGM HF Study Design. " ESC 2014 Science News (31 Aug. 2014).

Impact on Clinical Practice � Potential exists for a profound impact on HF pharmacotherapy � Considerations for utilization in specific patients: Patients with past history of angioedema are excluded Lack of reproducible safety data in patients of African descent (OCTAVE) Insurance status Previous use and tolerance of an ACE-I/ARB Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004. Kostis JB, et al. Am J Hypertens. 2004; 17: 103 -111.

Acknowledgements � Journal Club Mentors � 1. ) Elizabeth Mc. Neely, Pharm. D, BCPS � Tri. Star Centennial Medical Center, Nashville, TN � 2. ) Herb Patterson, Pharm. D, FCCP � UNC Eshelman School of Pharmacy, Chapel Hill, NC � Program Director � Daniel Johnson, Pharm. D, BCPS(AQ-Cardiology) � Vanderbilt University Medical Center, Nashville, TN � ACC PRN Journal Club Coordinator � Craig Beavers; Pharm. D, BCPS (AQ-Cardiology) � Tri. Star Centennial Medical Center, Nashville, TN

Angiotensin-Neprilysin Inhibition versus Enalapril in Heart Failure (PARADIGMHF) Prepared By: Robert K. Tunney, Pharm. D, BCPS PGY 2 Cardiology Resident, Vanderbilt University Medical Center, Nashville, TN Mc. Murray JV, et al. N Eng J Med. 2014; 371: 993 -1004.
Thank you for attending! � If you would like to have your resident present, would like to be a mentor, or have questions or comments please e-mail the journal club at accpcardsprnjournalclub@gmail. com or craig. beaverspharmd@gmail. com � Our next Journal Club will be in early January with the date to be determined. � Nicole Gasbarro from University of Chicago-Illinois will be presenting DAPT trial.
- Slides: 23









































