ACCP Cardiology PRN Club 28 March 2019 Journal

ACCP Cardiology PRN Club 28 March 2019 Journal

Mentor Bio Dr. Toby Trujillo is an associate professor at the University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences and a clinical cardiology pharmacist at the University of Colorado Hospital. He completed his Pharm. D and PGY 1 Pharmacy Practice residency at the University of California, San Francisco. Additionally, he completed a Fellowship in Cardiovascular Pharmacotherapy at The University of Arizona.

Presenter Bio Dr. Jaclyn Sullivan is a PGY 2 Cardiology Resident at The Johns Hopkins Hospital. She completed her PGY 1 Pharmacy Practice Residency at Rush University Medical Center. Jaclyn received her Pharm. D from Midwestern University Chicago College of Pharmacy.

AVERT Trial: Apixaban to Prevent Venous Thromboembolism in Patients with Cancer Jaclyn Sullivan, Pharm. D PGY 2 Cardiology Pharmacy Resident The Johns Hopkins Hospital 4

Background • • Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), is a major complication of cancer Occurs in 20% of patients with cancer and is one of the leading causes of death among cancer patients Treatment of VTE is associated with worsened quality of life, increased bleeding risk, and high rates of recurrent VTE Thromboprophylaxis with low molecular weight heparin (LMWH) therapy is not routinely recommended by practice guidelines due to a modest absolute risk reduction of VTE Lyman DH. Cancer. 2011 5

Current Guideline Recommendations Organization Recommendation 2019 National Comprehensive Cancer Network (NCCN) Guidelines • Surgical oncology patients - Recommend VTE prophylaxis for up to four weeks postoperation for high-risk abdominal or pelvic cancer surgery patients (Grade 2 A) • Medical oncology patients - Recommend VTE prophylaxis for high-risk multiple myeloma patients receiving thalidomide, lenalidomide, or pomalidomide (Grade 2 A) • Other outpatient settings - No routine VTE prophylaxis recommended Streiff MA, et al. NCCN Guidelines. 2019 6
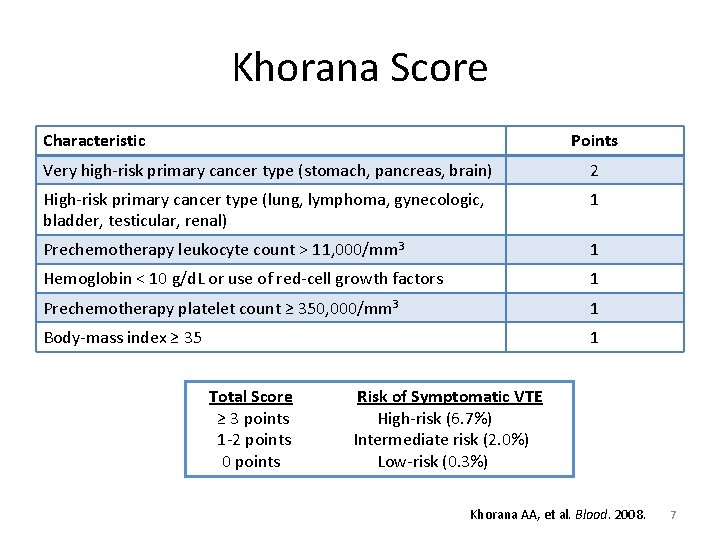
Khorana Score Characteristic Points Very high-risk primary cancer type (stomach, pancreas, brain) 2 High-risk primary cancer type (lung, lymphoma, gynecologic, bladder, testicular, renal) 1 Prechemotherapy leukocyte count > 11, 000/mm 3 1 Hemoglobin < 10 g/d. L or use of red-cell growth factors 1 Prechemotherapy platelet count ≥ 350, 000/mm 3 1 Body-mass index ≥ 35 1 Total Score ≥ 3 points 1 -2 points 0 points Risk of Symptomatic VTE High-risk (6. 7%) Intermediate risk (2. 0%) Low-risk (0. 3%) Khorana AA, et al. Blood. 2008. 7

AVERT Study Design Objective To assess the efficacy of apixaban thromboprophylaxis in ambulatory patients with cancer at intermediate-to-high risk for VTE Design • Randomized, placebo-controlled, double-blind trial • 13 participating centers in Canada • Patients randomized 1: 1 1. Apixaban 2. 5 mg PO BID 2. Placebo PO BID • Treatment period 180 days Carrier M, et al. NEJM. 2019. 8

Patient Selection Inclusion Criteria • 18 years of age or older with Khorana score of 2 or higher • Newly diagnosed cancer or progression of known cancer after complete or partial remission • Initiating a new course of chemotherapy with a minimum treatment intent of 3 months Exclusion Criteria • Conditions with increased risk of clinically significant bleeding - Hepatic disease - Basal-cell or squamous-cell skin carcinoma - Acute leukemia - Myeloproliferative neoplasm • Planned stem-cell transplantation • Life expectancy less than 6 months • Renal insufficiency (e. GFR < 30 m. L/min/1. 73 m 2) • Platelet count < 50, 000/mm 3 • Pregnancy, potential pregnancy, breast-feeding • Use of continuous anticoagulation • Weight < 40 kg Carrier M, et al. NEJM. 2019. 9

Endpoints Primary Efficacy Outcome First episode of objectively documented major VTE (proximal DVT or PE) • Patients who presented with symptoms of VTE during the follow-up period underwent diagnostic imaging • Routine ultrasonographic testing was not performed Primary Safety Outcome Major bleeding • Defined by ISTH criteria • The severity of major bleeding was defined as follows: 1. Not considered to be a clinical emergency 2. Led to some treatment but was not considered to be a clinical emergency 3. Considered to be a clinical emergency 4. Led to death before or almost immediately after the patient presented to the hospital ISTH: International Society on Thrombosis and Hemostasis Carrier M, et al. NEJM. 2019. 10

Statistical Analysis • • • Estimated that inclusion of 574 patients with Khorana score of 2 or higher would be sufficient to show relative difference of 64% in the primary outcome Two-sided alpha level of 0. 05 with power of 80% Expected event rate of 10% in placebo group over 6 months Primary analysis performed in the modified-intention to treat population – Included all patients who had undergone randomization and received at least one dose of apixaban Performed a time-to-event analysis on the primary efficacy outcome and safety outcome Carrier M, et al. NEJM. 2019. 11
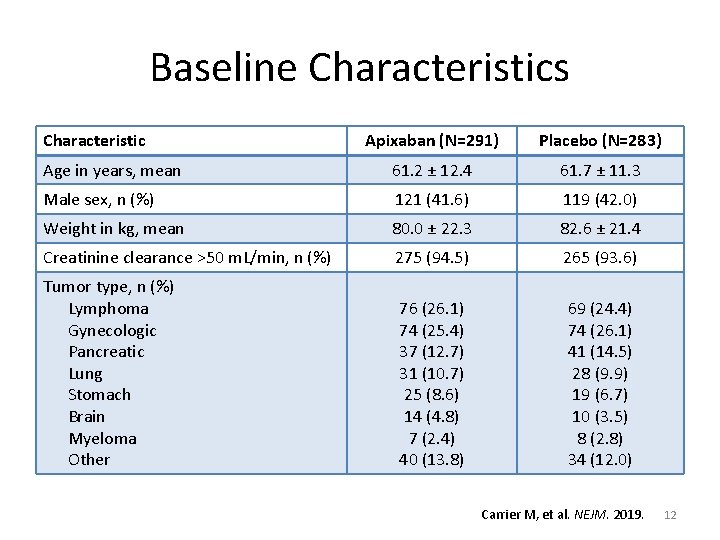
Baseline Characteristics Characteristic Apixaban (N=291) Placebo (N=283) Age in years, mean 61. 2 ± 12. 4 61. 7 ± 11. 3 Male sex, n (%) 121 (41. 6) 119 (42. 0) Weight in kg, mean 80. 0 ± 22. 3 82. 6 ± 21. 4 Creatinine clearance >50 m. L/min, n (%) 275 (94. 5) 265 (93. 6) Tumor type, n (%) Lymphoma Gynecologic Pancreatic Lung Stomach Brain Myeloma Other 76 (26. 1) 74 (25. 4) 37 (12. 7) 31 (10. 7) 25 (8. 6) 14 (4. 8) 7 (2. 4) 40 (13. 8) 69 (24. 4) 74 (26. 1) 41 (14. 5) 28 (9. 9) 19 (6. 7) 10 (3. 5) 8 (2. 8) 34 (12. 0) Carrier M, et al. NEJM. 2019. 12
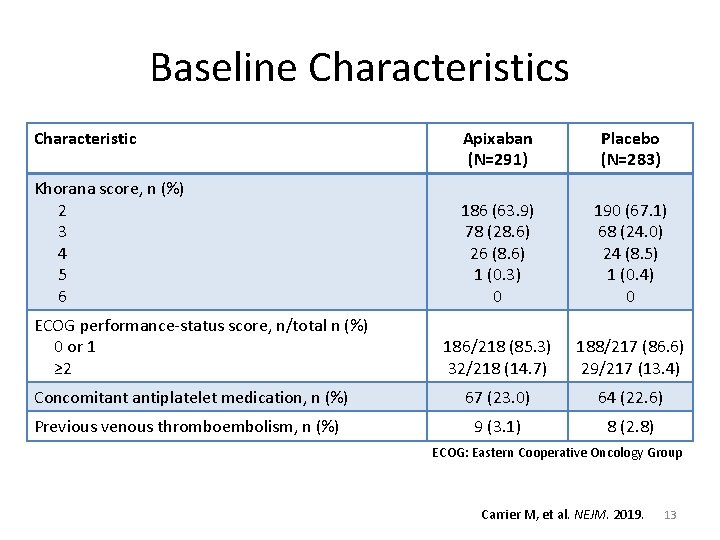
Baseline Characteristics Characteristic Apixaban (N=291) Placebo (N=283) 186 (63. 9) 78 (28. 6) 26 (8. 6) 1 (0. 3) 0 190 (67. 1) 68 (24. 0) 24 (8. 5) 1 (0. 4) 0 186/218 (85. 3) 32/218 (14. 7) 188/217 (86. 6) 29/217 (13. 4) Concomitant antiplatelet medication, n (%) 67 (23. 0) 64 (22. 6) Previous venous thromboembolism, n (%) 9 (3. 1) 8 (2. 8) Khorana score, n (%) 2 3 4 5 6 ECOG performance-status score, n/total n (%) 0 or 1 ≥ 2 ECOG: Eastern Cooperative Oncology Group Carrier M, et al. NEJM. 2019. 13
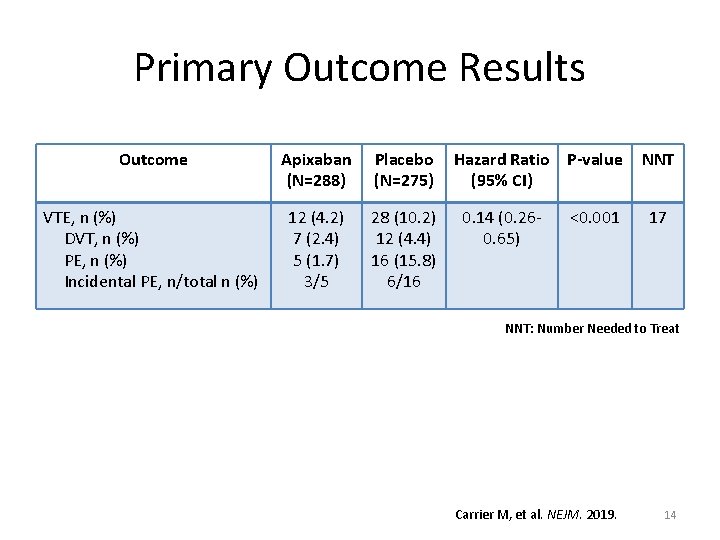
Primary Outcome Results Outcome Apixaban (N=288) Placebo (N=275) VTE, n (%) DVT, n (%) PE, n (%) Incidental PE, n/total n (%) 12 (4. 2) 7 (2. 4) 5 (1. 7) 3/5 28 (10. 2) 12 (4. 4) 16 (15. 8) 6/16 Hazard Ratio P-value (95% CI) 0. 14 (0. 260. 65) <0. 001 NNT 17 NNT: Number Needed to Treat Carrier M, et al. NEJM. 2019. 14
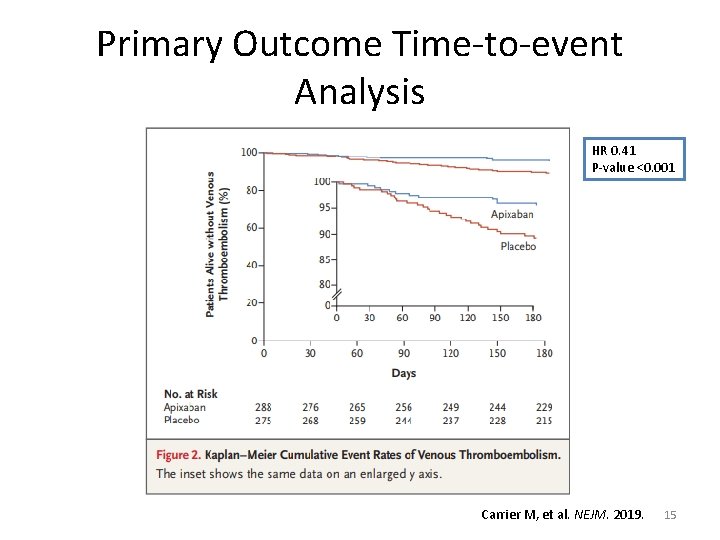
Primary Outcome Time-to-event Analysis HR 0. 41 P-value <0. 001 Carrier M, et al. NEJM. 2019. 15
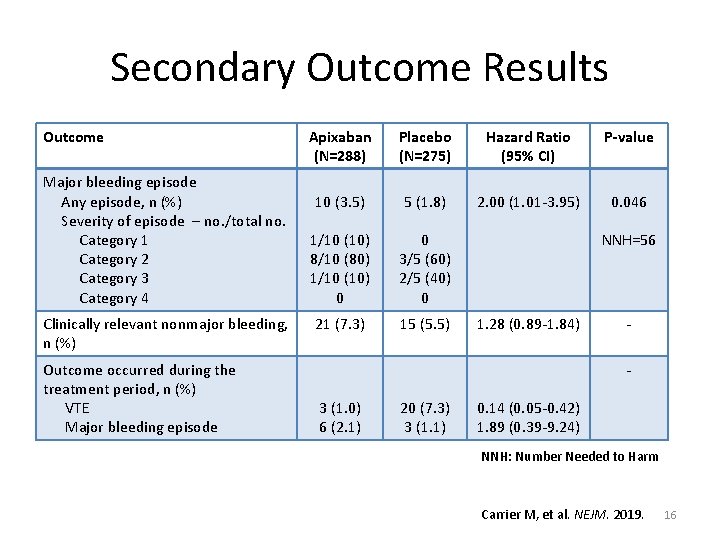
Secondary Outcome Results Outcome Major bleeding episode Any episode, n (%) Severity of episode – no. /total no. Category 1 Category 2 Category 3 Category 4 Clinically relevant nonmajor bleeding, n (%) Outcome occurred during the treatment period, n (%) VTE Major bleeding episode Apixaban (N=288) Placebo (N=275) Hazard Ratio (95% CI) P-value 10 (3. 5) 5 (1. 8) 2. 00 (1. 01 -3. 95) 0. 046 1/10 (10) 8/10 (80) 1/10 (10) 0 0 3/5 (60) 2/5 (40) 0 21 (7. 3) 15 (5. 5) NNH=56 1. 28 (0. 89 -1. 84) - 3 (1. 0) 6 (2. 1) 20 (7. 3) 3 (1. 1) 0. 14 (0. 05 -0. 42) 1. 89 (0. 39 -9. 24) NNH: Number Needed to Harm Carrier M, et al. NEJM. 2019. 16
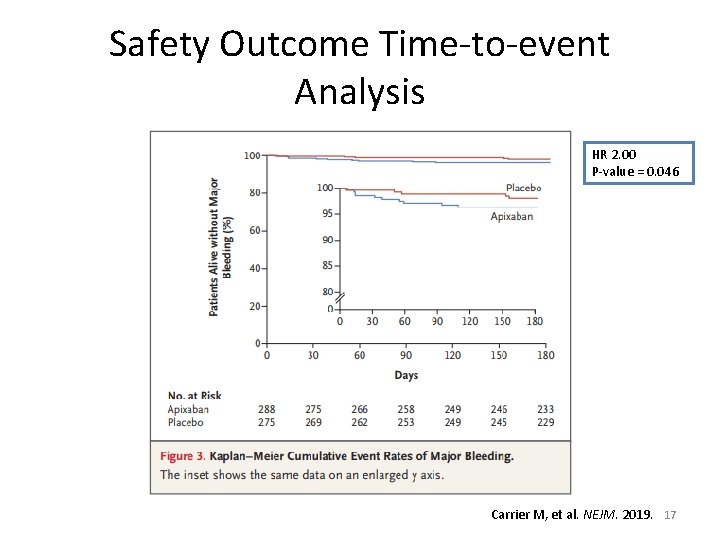
Safety Outcome Time-to-event Analysis HR 2. 00 P-value = 0. 046 Carrier M, et al. NEJM. 2019. 17
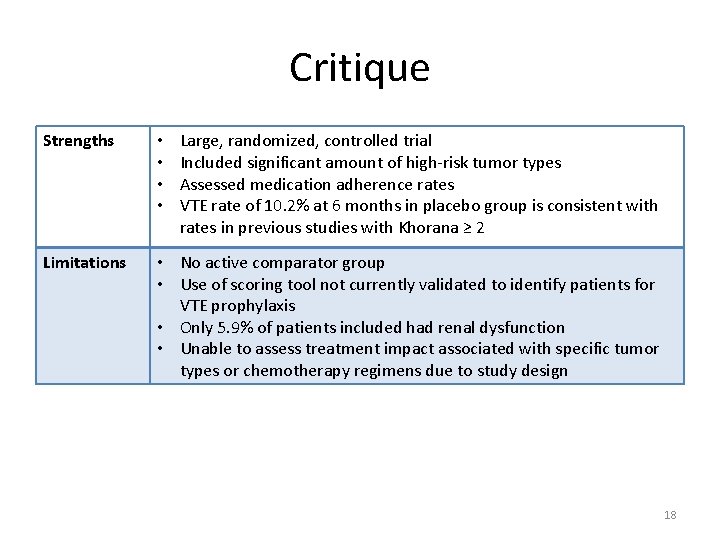
Critique Strengths • • Large, randomized, controlled trial Included significant amount of high-risk tumor types Assessed medication adherence rates VTE rate of 10. 2% at 6 months in placebo group is consistent with rates in previous studies with Khorana ≥ 2 Limitations • No active comparator group • Use of scoring tool not currently validated to identify patients for VTE prophylaxis • Only 5. 9% of patients included had renal dysfunction • Unable to assess treatment impact associated with specific tumor types or chemotherapy regimens due to study design 18

CASSINI Trial Methods Rivaroxaban 10 mg daily vs. placebo for preventing VTE in high-risk ambulatory patients with cancer Primary Efficacy Outcome Intention-to-treat analysis • 6% in the rivaroxaban vs. 8. 8% in the placebo group experienced the primary outcome during the observation period of 180 days (HR 0. 66, 95% CI 0. 40 -1. 09, p=0. 10) On-treatment analysis • 2. 6% with rivaroxaban vs. 6. 4% for placebo (HR 0. 40, 95% CI 0. 2 -0. 8) Safety Outcome Similar major bleeding rates observed • 2% with rivaroxaban vs. 1% with placebo (HR 1. 96, 95% CI 0. 596. 49) Khorana AA, et al. NEJM. 2019. 19
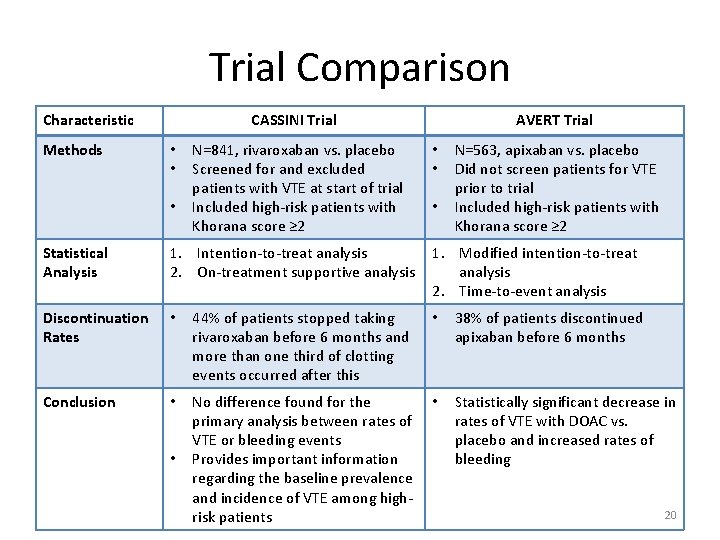
Trial Comparison Characteristic Methods CASSINI Trial • • • N=841, rivaroxaban vs. placebo Screened for and excluded patients with VTE at start of trial Included high-risk patients with Khorana score ≥ 2 AVERT Trial • • • N=563, apixaban vs. placebo Did not screen patients for VTE prior to trial Included high-risk patients with Khorana score ≥ 2 Statistical Analysis 1. Intention-to-treat analysis 2. On-treatment supportive analysis Discontinuation Rates • 44% of patients stopped taking rivaroxaban before 6 months and more than one third of clotting events occurred after this • 38% of patients discontinued apixaban before 6 months Conclusion • No difference found for the primary analysis between rates of VTE or bleeding events Provides important information regarding the baseline prevalence and incidence of VTE among highrisk patients • Statistically significant decrease in rates of VTE with DOAC vs. placebo and increased rates of bleeding • 1. Modified intention-to-treat analysis 2. Time-to-event analysis 20

AVERT Trial Conclusions • • Thromboprophylaxis with apixaban resulted in significantly lower rates of VTE compared to placebo among intermediate-to-high-risk ambulatory patients with cancer who were starting chemotherapy The rate of major bleeding episodes was higher was apixaban than with placebo NNT 17 vs. NNH 56 Overall, reasonable to consider VTE prophylaxis with rivaroxaban or apixaban over LMWH – Consider thrombotic risk associated with chemotherapy regimen – Use cautiously in patients at increased bleeding risk – Consider CYP 3 A 4 and p-glycoprotein substrate drug-drug interactions between DOACs and chemotherapy 21

AVERT Trial: Apixaban to Prevent Venous Thromboembolism in Patients with Cancer Jaclyn Sullivan, Pharm. D PGY 2 Cardiology Pharmacy Resident The Johns Hopkins Hospital 22

Presenter Bio Dr. Jennifer Cook is a PGY 2 Cardiology Resident at Duke University Hospital. She completed her PGY 1 Pharmacy Practice Residency at The Ohio State University Wexner Medical Center. Jennifer received her Pharm. D from the University of North Carolina Eshelman School of Pharmacy.

Andexanet Alfa for Bleeding Associated with Factor Xa Inhibitors Jennifer C. Cook, Pharm. D PGY 2 Cardiology Pharmacy Resident Duke University Hospital

Disclosures Dr. Cook does not have any relevant disclosures concerning possible financial or personal relationships with commercial entities that may have a direct or indirect interest in the subject matter of this presentation. 25

Background – Andexanet Alfa • Andexanet alfa is a recombinant human Factor Xa protein – Inhibits Factor Xa inhibitors and tissue factor pathway inhibitor – Approved for the reversal of apixaban and rivaroxaban in patients with life-threatening or uncontrolled bleeding • Warnings and Precautions: – Thromboembolic events, ischemic events, and sudden death – Re-elevation or complete reversal of anticoagulant activity • Dose dependent on the specific agent, dose, and time since the patient’s last dose 26

Background – ANNEXA-A and ANNEXA-R Assessed the safety and efficacy of andexanet alfa administered as a bolus or a bolus and continuous infusion vs. placebo in the reversal of apixaban or rivaroxaban, respectively, in healthy volunteers • Andexanet alfa 400 mg IV x 1, then 4 mg/min IV x 120 mins (480 mg) in those treated with apixaban • Andexanet alfa 800 mg IV x 1, then 8 mg/min IV x 120 mins (960 mg) in those treated with rivaroxaban • Significant reductions in anti-factor Xa activity following an IV bolus vs. placebo in both apixaban (94% vs. 21%; p<0. 001) and rivaroxaban (92% vs. 18%; p <0. 001) studies • Effect preserved for 1 – 2 hours following a bolus and infusion N Engl J Med. 2015; 373(25): 2413 -24. 27
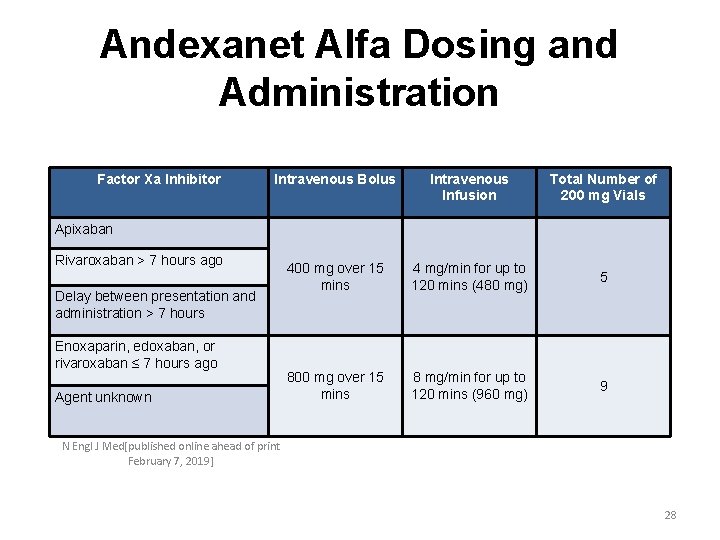
Andexanet Alfa Dosing and Administration Factor Xa Inhibitor Intravenous Bolus Intravenous Infusion Total Number of 200 mg Vials 400 mg over 15 mins 4 mg/min for up to 120 mins (480 mg) 5 800 mg over 15 mins 8 mg/min for up to 120 mins (960 mg) 9 Apixaban Rivaroxaban > 7 hours ago Delay between presentation and administration > 7 hours Enoxaparin, edoxaban, or rivaroxaban ≤ 7 hours ago Agent unknown N Engl J Med[published online ahead of print February 7, 2019] 28

Study Design Objective • Assess the safety and efficacy of andexanet alfa in patients with acute major bleeding while taking a factor Xa inhibitor Design • Multicenter, prospective, open-label, single-group study • Included patients with acute major bleeding who had received any dose of a factor Xa inhibitor within 18 hours • Key Exclusion Criteria: • Elective surgery within 12 hours • ICH with a GCS < 7 • Estimated hematoma volume > 60 cc • Anticipated survival < 30 days • Thrombotic event within prior 2 weeks N Engl J Med[published online ahead of print February 7, 2019] 29

Outcomes Primary Efficacy Endpoints • Percent change from baseline in anti-factor Xa activity • Percentage of patients with excellent or good hemostatic efficacy 12 hours post-infusion Primary Safety Endpoints • Death • Thrombotic events • Development of antibodies to andexanet alfa, factor X, or factor Xa N Engl J Med[published online ahead of print February 7, 2019] 30

Statistical Analyses • Safety analyses included all patients treated with andexanet alfa • Efficacy analyses included patients who met the following: – Baseline anti-factor Xa ≥ 75 ng/m. L (or ≥ 0. 25 IU/m. L if receiving enoxaparin) – Confirmed major bleeding • 350 patients required to provide 80% power to demonstrate > 50% of patients with hemostatic efficacy and to meet regulatory requirements for sufficient number of patients for each Xa inhibitor • Association between hemostatic efficacy and change in antifactor Xa activity examined with receiver-operating-characteristic curves N Engl J Med[published online ahead of print February 7, 2019] 31
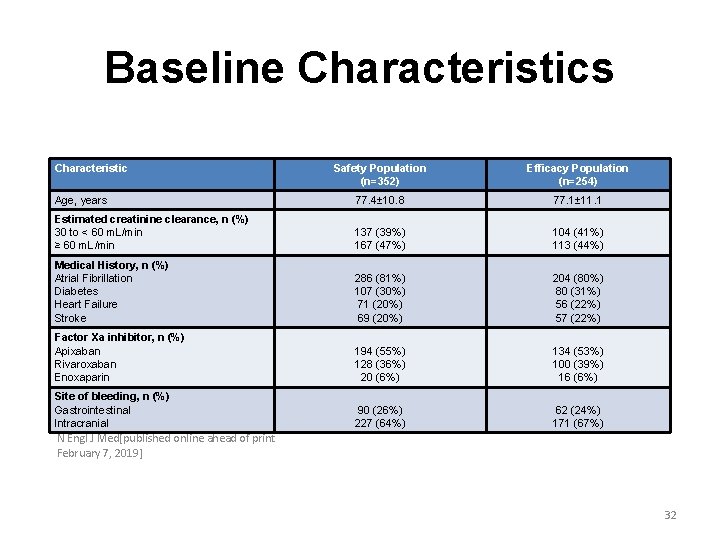
Baseline Characteristics Characteristic Safety Population (n=352) Efficacy Population (n=254) Age, years 77. 4± 10. 8 77. 1± 11. 1 Estimated creatinine clearance, n (%) 30 to < 60 m. L/min ≥ 60 m. L/min 137 (39%) 167 (47%) 104 (41%) 113 (44%) Medical History, n (%) Atrial Fibrillation Diabetes Heart Failure Stroke 286 (81%) 107 (30%) 71 (20%) 69 (20%) 204 (80%) 80 (31%) 56 (22%) 57 (22%) Factor Xa inhibitor, n (%) Apixaban Rivaroxaban Enoxaparin 194 (55%) 128 (36%) 20 (6%) 134 (53%) 100 (39%) 16 (6%) Site of bleeding, n (%) Gastrointestinal Intracranial 90 (26%) 227 (64%) 62 (24%) 171 (67%) N Engl J Med[published online ahead of print February 7, 2019] 32
![Factor Xa Activity N Engl J Med[published online ahead of print February 7, 2019] Factor Xa Activity N Engl J Med[published online ahead of print February 7, 2019]](http://slidetodoc.com/presentation_image_h2/1b8a9f2a91d2592586481192060edff6/image-33.jpg)
Factor Xa Activity N Engl J Med[published online ahead of print February 7, 2019] 33
![Factor Xa Activity N Engl J Med[published online ahead of print February 7, 2019] Factor Xa Activity N Engl J Med[published online ahead of print February 7, 2019]](http://slidetodoc.com/presentation_image_h2/1b8a9f2a91d2592586481192060edff6/image-34.jpg)
Factor Xa Activity N Engl J Med[published online ahead of print February 7, 2019] 34
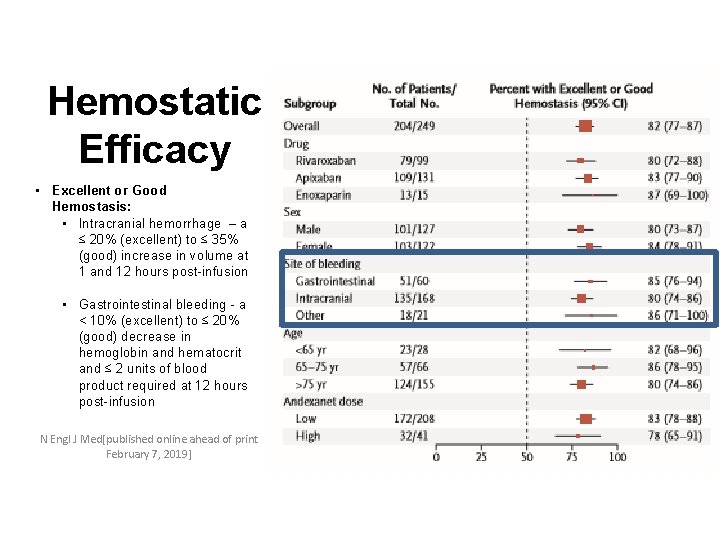
Hemostatic Efficacy • Excellent or Good Hemostasis: • Intracranial hemorrhage – a ≤ 20% (excellent) to ≤ 35% (good) increase in volume at 1 and 12 hours post-infusion • Gastrointestinal bleeding - a < 10% (excellent) to ≤ 20% (good) decrease in hemoglobin and hematocrit and ≤ 2 units of blood product required at 12 hours post-infusion N Engl J Med[published online ahead of print February 7, 2019] 35
![Biomarker-Efficacy Correlation N Engl J Med[published online ahead of print February 7, 2019] 36 Biomarker-Efficacy Correlation N Engl J Med[published online ahead of print February 7, 2019] 36](http://slidetodoc.com/presentation_image_h2/1b8a9f2a91d2592586481192060edff6/image-36.jpg)
Biomarker-Efficacy Correlation N Engl J Med[published online ahead of print February 7, 2019] 36
![Safety N Engl J Med[published online ahead of print February 7, 2019] 37 Safety N Engl J Med[published online ahead of print February 7, 2019] 37](http://slidetodoc.com/presentation_image_h2/1b8a9f2a91d2592586481192060edff6/image-37.jpg)
Safety N Engl J Med[published online ahead of print February 7, 2019] 37

Conclusions • Treatment with andexanet alfa reduces anti-factor Xa activity in patients treated with a factor Xa inhibitor – Reduction may be associated with excellent or good hemostatic efficacy at 12 hours in patients with intracranial hemorrhage – Unclear how reduction may impact clinical outcomes • Andexanet alfa should be utilized with caution in the following subgroups: – Patients treated with enoxaparin or edoxaban – Patients presenting with a gastrointestinal bleed • Unclear how this reversal agent compares to lower cost alternatives • Cost? 38

Andexanet Alfa for Bleeding Associated with Factor Xa Inhibitors Jennifer C. Cook, Pharm. D PGY 2 Cardiology Pharmacy Resident Duke University Hospital

April Journal Club • Date – Monday, April 22 nd at 2 PM EST • Presenters – Laken Warnock (St. Joseph Hospital, KY) – Jessica Wilcyznski (Thomas Jefferson Hospital, PA) • Articles – CASSINI – Rivaroxaban for thromboprophylaxis in high -risk ambulatory patients with cancer (2019 NEJM) – Outcomes of DOACs in patients with mitral stenosis (2019 JACC)
- Slides: 40