ACCP Cardiology PRN Journal Club 27 September 2019

ACCP Cardiology PRN Journal Club 27 September 2019

Presenter Bio Dr. Maxwell Ditlevson is a PGY 2 Cardiology Resident at the The Johns Hopkins Hospital. He completed his PGY 1 Pharmacy Practice Residency at The Johns Hopkins Hospital too. Max received his Pharm. D from the University of Maryland School of Pharmacy.

Mentor Bio Dr. Farzad Daneshvar received his doctor of pharmacy degree from the University of Michigan in 2013. Post graduation, he completed a clinical residency training at Henry Ford Hospital in Detroit, followed by a specialty residency training with a focus on cardiovascular pharmacotherapy at Sharp Healthcare in San Diego, CA. Upon completion of his residency training, he practiced as a cardiology professor and a cardiology clinical pharmacist in Los Angeles, CA. He then moved back to MI to pursue a position as a cardiology clinical pharmacist specialist at Beaumont Dearborn. He currently rounds with an academic Cardiology Teaching Team in a Coronary Care Unit and has additional involvement is the Cardiac Cath Lab and Cardiothoracic Surgery. He was recently promoted as a non-physician faculty member for the Cardiology Fellowship Program at Beaumont Dearborn. He is a member of the Michigan Chapter and National American College of Cardiology (ACC) and has presented at both state-wide and national conferences.

Digoxin Discontinuation and Outcomes in Patients With Heart Failure With Reduced Ejection Fraction Maxwell Ditlevson, Pharm. D PGY 2 Cardiology Pharmacy Resident The Johns Hopkins Hospital, Baltimore, MD

Disclosures • Maxwell Ditlevson and all other contributors have no relevant financial relationships to disclose

Background • Cardiac glycoside isolated from the Digitalis (foxglove) genus • Indications: • Heart failure with reduced ejection fraction (HFr. EF) • Rate control in atrial fibrillation
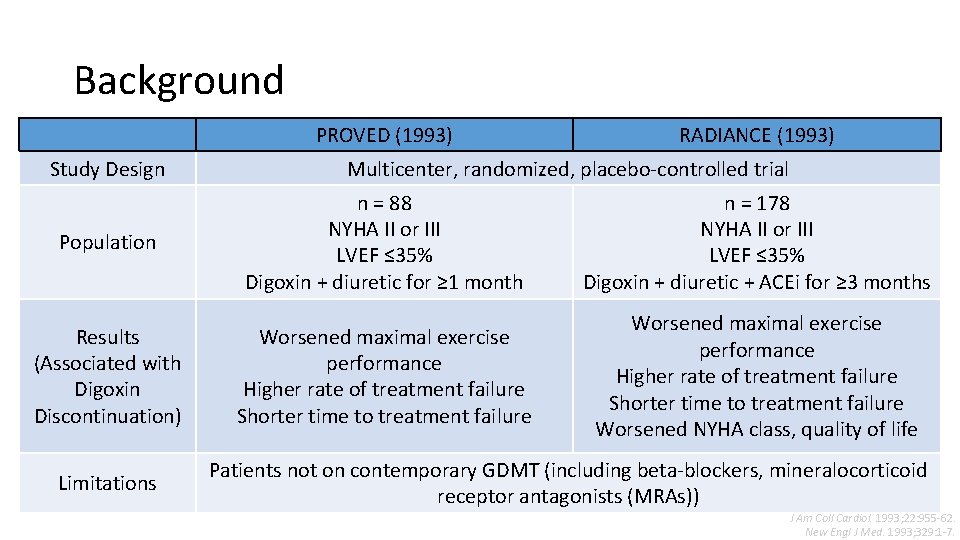
Background PROVED (1993) Study Design RADIANCE (1993) Multicenter, randomized, placebo-controlled trial Population n = 88 NYHA II or III LVEF ≤ 35% Digoxin + diuretic for ≥ 1 month n = 178 NYHA II or III LVEF ≤ 35% Digoxin + diuretic + ACEi for ≥ 3 months Results (Associated with Digoxin Discontinuation) Worsened maximal exercise performance Higher rate of treatment failure Shorter time to treatment failure Worsened NYHA class, quality of life Limitations Patients not on contemporary GDMT (including beta-blockers, mineralocorticoid receptor antagonists (MRAs)) J Am Coll Cardiol. 1993; 22: 955 -62. New Engl J Med. 1993; 329: 1 -7.
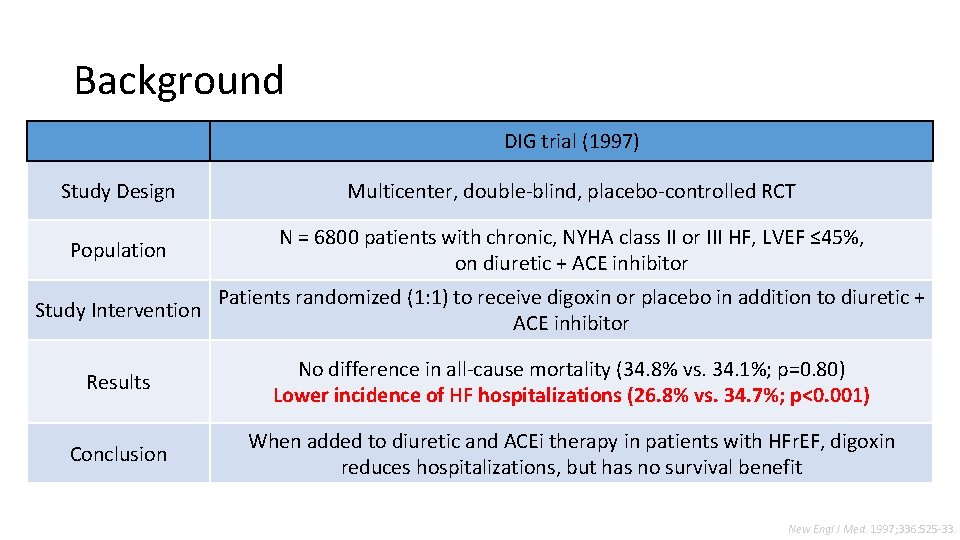
Background DIG trial (1997) Study Design Multicenter, double-blind, placebo-controlled RCT Population N = 6800 patients with chronic, NYHA class II or III HF, LVEF ≤ 45%, on diuretic + ACE inhibitor Patients randomized (1: 1) to receive digoxin or placebo in addition to diuretic + Study Intervention ACE inhibitor Results No difference in all-cause mortality (34. 8% vs. 34. 1%; p=0. 80) Lower incidence of HF hospitalizations (26. 8% vs. 34. 7%; p<0. 001) Conclusion When added to diuretic and ACEi therapy in patients with HFr. EF, digoxin reduces hospitalizations, but has no survival benefit New Engl J Med. 1997; 336: 525 -33.

Background • 2013 ACCF/AHA HF Guidelines: • “Digoxin can be beneficial in patients with HFr. EF, unless contraindicated, to decrease hospitalizations for HF” (Class IIa, LOE B) J Am Coll Cardiol. 2013; 62: 147 -239.
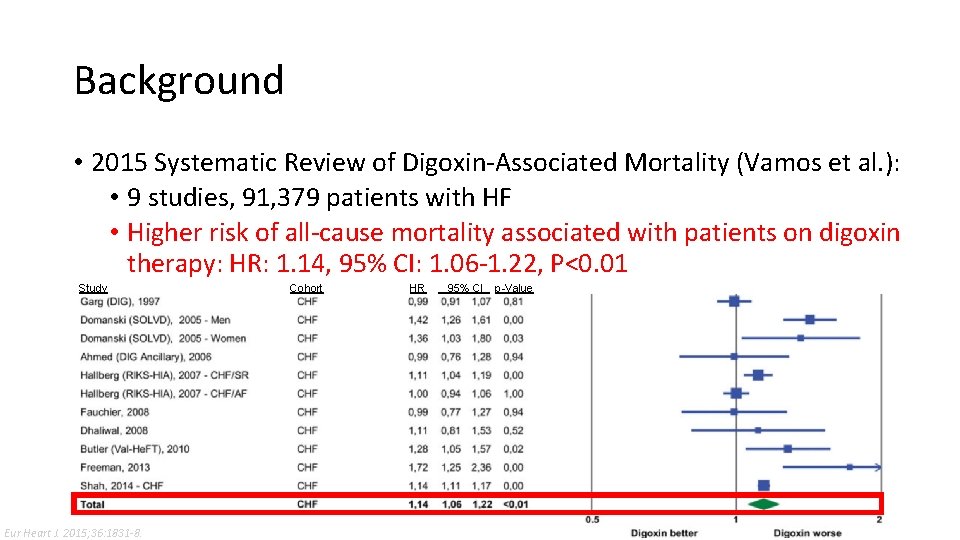
Background • 2015 Systematic Review of Digoxin-Associated Mortality (Vamos et al. ): • 9 studies, 91, 379 patients with HF • Higher risk of all-cause mortality associated with patients on digoxin therapy: HR: 1. 14, 95% CI: 1. 06 -1. 22, P<0. 01 Study Eur Heart J. 2015; 36: 1831 -8. Cohort HR 95% CI p-Value

Objective • To evaluate the relationship between discontinuation of preadmission digoxin therapy and outcomes in a cohort of hospitalized patients with HFr. EF receiving contemporary GDMT J Am Coll Cardiol. 2019; 74: 617 -27.
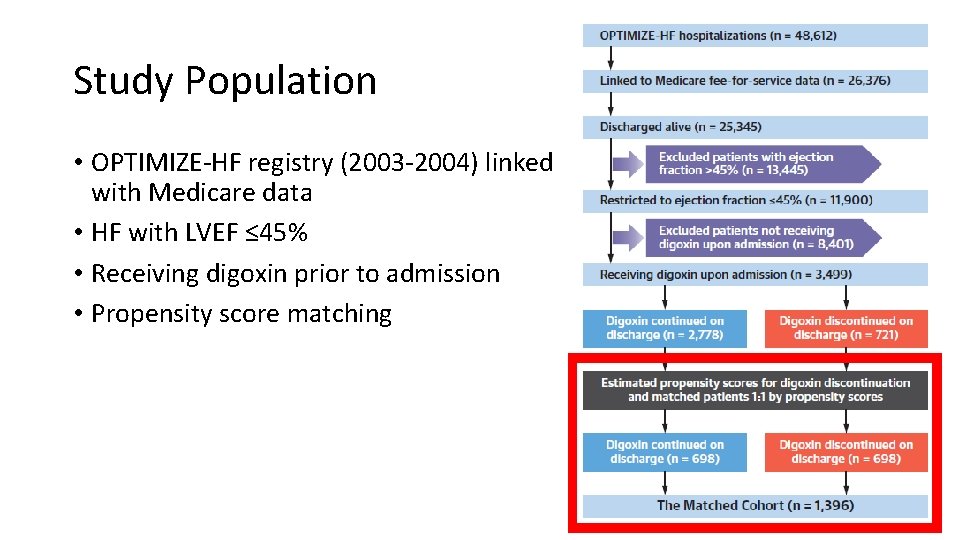
Study Population • OPTIMIZE-HF registry (2003 -2004) linked with Medicare data • HF with LVEF ≤ 45% • Receiving digoxin prior to admission • Propensity score matching

Outcomes Examined at 30 days, 6 months, 1 year, and 4 years: • Heart failure readmissions • All-cause mortality • Composite of HF readmissions or all-cause mortality J Am Coll Cardiol. 2019; 74: 617 -27.

Statistical Analysis • Propensity score matching • Sensitivity cohort to account for AKI • Sensitivity analysis to account for unmeasured confounders J Am Coll Cardiol. 2019; 74: 617 -27.
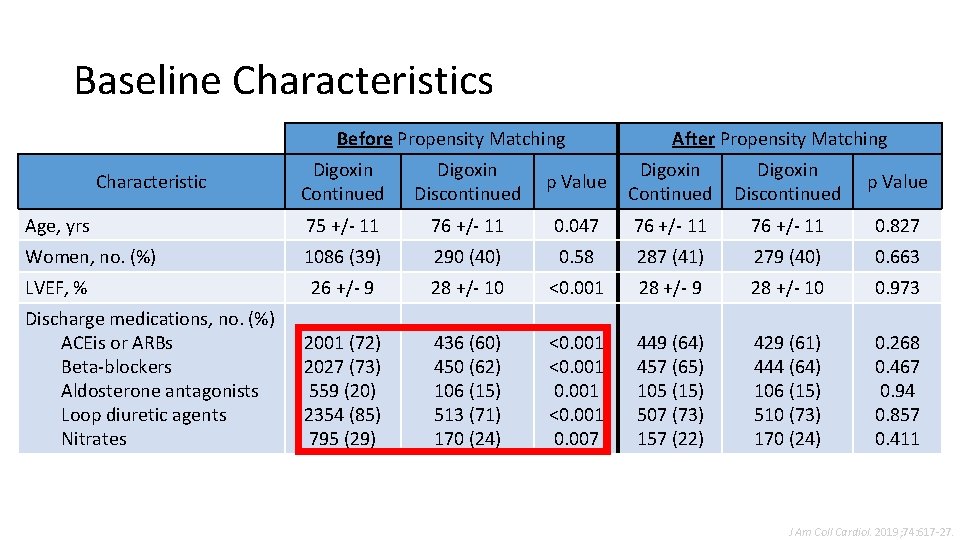
Baseline Characteristics Before Propensity Matching After Propensity Matching Digoxin Continued Digoxin Discontinued p Value Age, yrs 75 +/- 11 76 +/- 11 0. 047 76 +/- 11 0. 827 Women, no. (%) 1086 (39) 290 (40) 0. 58 287 (41) 279 (40) 0. 663 26 +/- 9 28 +/- 10 <0. 001 28 +/- 9 28 +/- 10 0. 973 2001 (72) 2027 (73) 559 (20) 2354 (85) 795 (29) 436 (60) 450 (62) 106 (15) 513 (71) 170 (24) <0. 001 <0. 001 0. 007 449 (64) 457 (65) 105 (15) 507 (73) 157 (22) 429 (61) 444 (64) 106 (15) 510 (73) 170 (24) 0. 268 0. 467 0. 94 0. 857 0. 411 Characteristic LVEF, % Discharge medications, no. (%) ACEis or ARBs Beta-blockers Aldosterone antagonists Loop diuretic agents Nitrates J Am Coll Cardiol. 2019; 74: 617 -27.
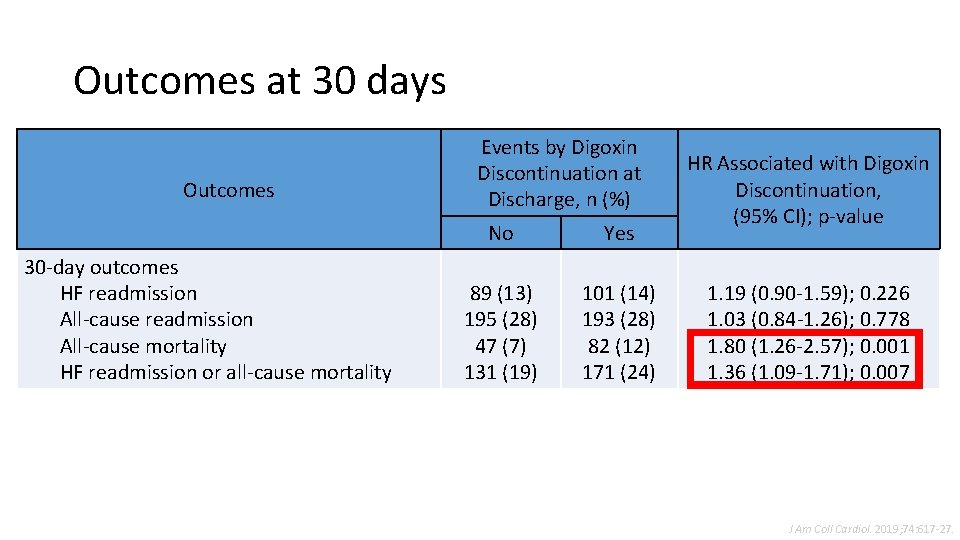
Outcomes at 30 days Outcomes 30 -day outcomes HF readmission All-cause mortality HF readmission or all-cause mortality Events by Digoxin Discontinuation at Discharge, n (%) No Yes 89 (13) 195 (28) 47 (7) 131 (19) 101 (14) 193 (28) 82 (12) 171 (24) HR Associated with Digoxin Discontinuation, (95% CI); p-value 1. 19 (0. 90 -1. 59); 0. 226 1. 03 (0. 84 -1. 26); 0. 778 1. 80 (1. 26 -2. 57); 0. 001 1. 36 (1. 09 -1. 71); 0. 007 J Am Coll Cardiol. 2019; 74: 617 -27.
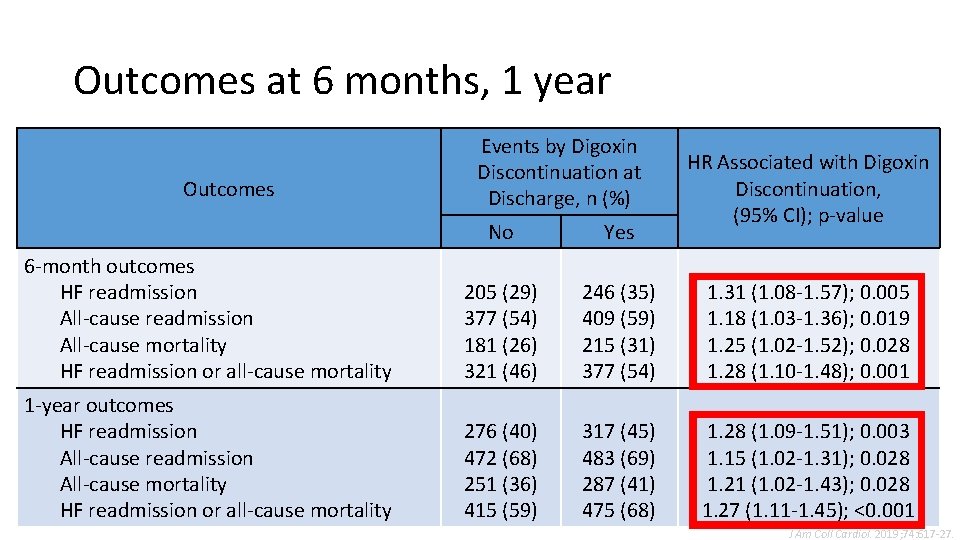
Outcomes at 6 months, 1 year Outcomes Events by Digoxin Discontinuation at Discharge, n (%) HR Associated with Digoxin Discontinuation, (95% CI); p-value No Yes 6 -month outcomes HF readmission All-cause mortality HF readmission or all-cause mortality 205 (29) 377 (54) 181 (26) 321 (46) 246 (35) 409 (59) 215 (31) 377 (54) 1. 31 (1. 08 -1. 57); 0. 005 1. 18 (1. 03 -1. 36); 0. 019 1. 25 (1. 02 -1. 52); 0. 028 1. 28 (1. 10 -1. 48); 0. 001 1 -year outcomes HF readmission All-cause mortality HF readmission or all-cause mortality 276 (40) 472 (68) 251 (36) 415 (59) 317 (45) 483 (69) 287 (41) 475 (68) 1. 28 (1. 09 -1. 51); 0. 003 1. 15 (1. 02 -1. 31); 0. 028 1. 21 (1. 02 -1. 43); 0. 028 1. 27 (1. 11 -1. 45); <0. 001 J Am Coll Cardiol. 2019; 74: 617 -27.
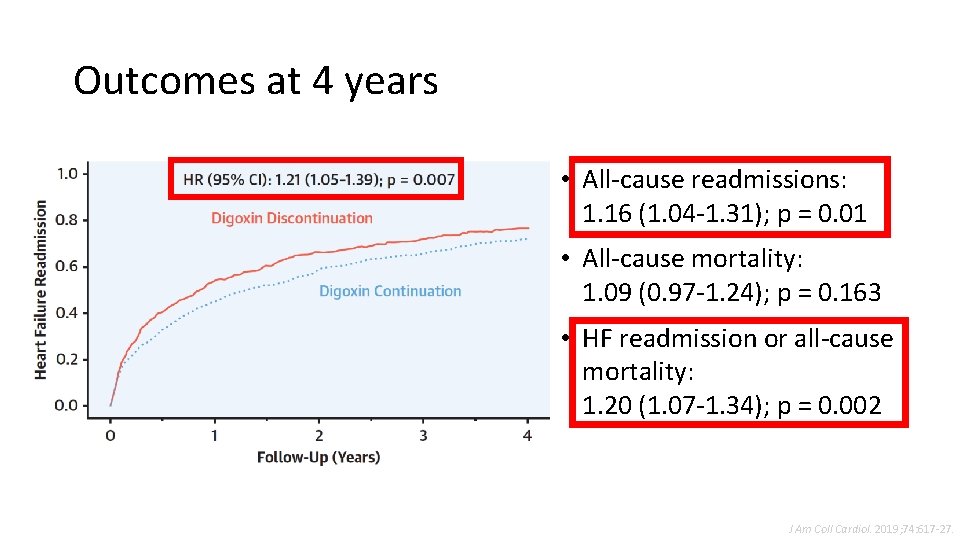
Outcomes at 4 years • All-cause readmissions: 1. 16 (1. 04 -1. 31); p = 0. 01 • All-cause mortality: 1. 09 (0. 97 -1. 24); p = 0. 163 • HF readmission or all-cause mortality: 1. 20 (1. 07 -1. 34); p = 0. 002 J Am Coll Cardiol. 2019; 74: 617 -27.
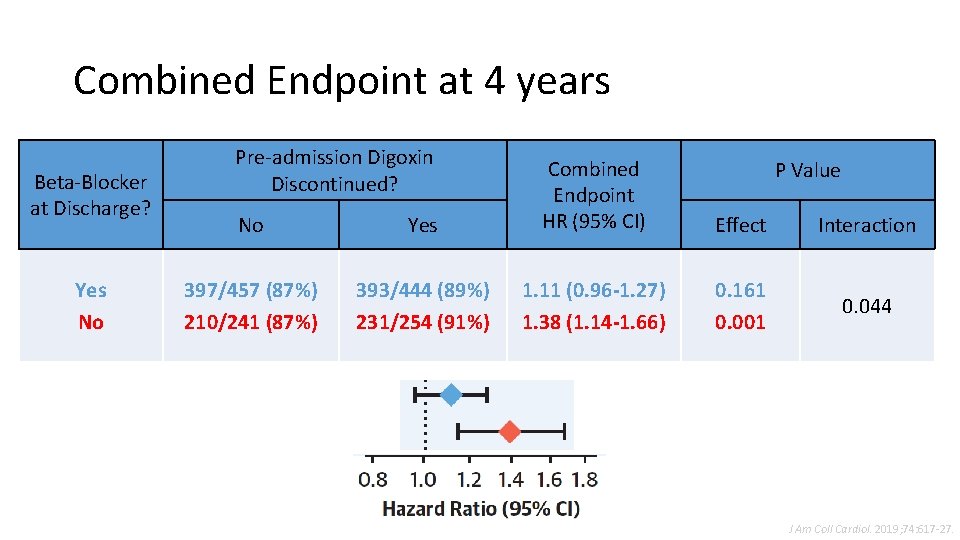
Combined Endpoint at 4 years Beta-Blocker at Discharge? Yes No Pre-admission Digoxin Discontinued? No Yes Combined Endpoint HR (95% CI) 397/457 (87%) 210/241 (87%) 393/444 (89%) 231/254 (91%) 1. 11 (0. 96 -1. 27) 1. 38 (1. 14 -1. 66) P Value Effect Interaction 0. 161 0. 001 0. 044 J Am Coll Cardiol. 2019; 74: 617 -27.
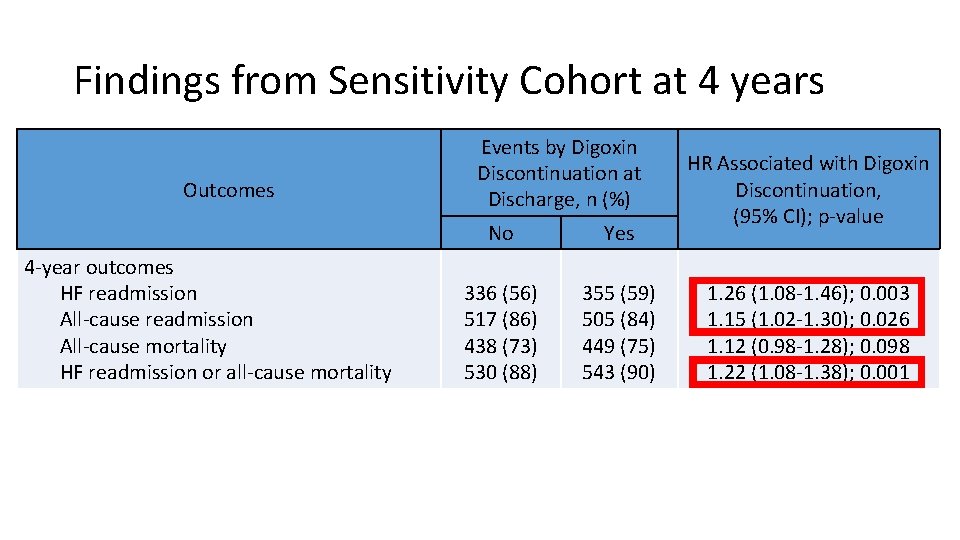
Findings from Sensitivity Cohort at 4 years Outcomes 4 -year outcomes HF readmission All-cause mortality HF readmission or all-cause mortality Events by Digoxin Discontinuation at Discharge, n (%) No Yes 336 (56) 517 (86) 438 (73) 530 (88) 355 (59) 505 (84) 449 (75) 543 (90) HR Associated with Digoxin Discontinuation, (95% CI); p-value 1. 26 (1. 08 -1. 46); 0. 003 1. 15 (1. 02 -1. 30); 0. 026 1. 12 (0. 98 -1. 28); 0. 098 1. 22 (1. 08 -1. 38); 0. 001

Findings from Sensitivity Analysis • 4 -year associations of digoxin discontinuation with all-cause readmissions and the combined endpoint were insensitive to unmeasured confounders • All-cause readmissions: • Single unmeasured baseline characteristic would need to increase the odds of digoxin discontinuation by 3% to explain away this association • HF readmissions or all-cause mortality: • Single unmeasured baseline characteristic would need to increase the odds of digoxin discontinuation by 7% to explain away this association

Authors’ Conclusions • Among hospitalized older patients with HFr. EF receiving contemporary GDMT, discontinuation of pre-admission digoxin therapy was associated with poor outcomes • It may be premature to abandon the use of digoxin in patients with HFr. EF J Am Coll Cardiol. 2019; 74: 617 -27.

Study Critique • Limited generalizability • “Contemporary” GDMT • Lack of follow-up data • Propensity score matching J Am Coll Cardiol. 2019; 74: 617 -27.
Study Critique • Limited generalizability • “Contemporary” GDMT • Lack of follow-up data • Propensity score matching J Am Coll Cardiol. 2019; 74: 617 -27.

Study Critique • Limited generalizability • “Contemporary” GDMT • Lack of follow-up data • Propensity score matching J Am Coll Cardiol. 2019; 74: 617 -27.

Study Critique • Limited generalizability • “Contemporary” GDMT • Lack of follow-up data • Propensity score matching J Am Coll Cardiol. 2019; 74: 617 -27.

Impact on Clinical Practice • Discourages the routine discontinuation of pre-admission digoxin therapy in hospitalized older patients with HFr. EF • No impact on digoxin’s place in HFr. EF therapy • No impact regarding initiation of digoxin therapy J Am Coll Cardiol. 2019; 74: 617 -27.

Digoxin Discontinuation and Outcomes in Patients With Heart Failure With Reduced Ejection Fraction Maxwell Ditlevson, Pharm. D PGY 2 Cardiology Pharmacy Resident The Johns Hopkins Hospital, Baltimore, MD

Presenter Bio Dr. Sara Ward is a PGY 2 Cardiology Resident at the University of Kentucky. She completed her PGY 1 Pharmacy Practice Residency at the Medical University of South Carolina. Sara received her Pharm. D from the University of Kentucky College of Pharmacy.

Presenter Bio Dr. Erica Sheridan is a PGY 2 Cardiology Resident at the University of Kentucky. She completed her PGY 1 Pharmacy Practice Residency at University of Pittsburgh Medical Center. Erica received her Pharm. D from the University of Toledo College of Pharmacy and Pharmaceutical Sciences.

Mentor Bio Dr. Craig Beavers is currently the Director of Cardiovascular Services at Baptist Health System in Paducah, Kentucky. He completed his Pharm. D, PGY 1 Pharmacy Practice residency, and PGY 2 Cardiology residency at the University of Kentucky.

Ticagrelor or Prasugrel in Patients with Acute Coronary Syndromes ISAR - REACT 5 Sara M. Ward, Pharm. D Erica A. Sheridan, Pharm. D, MBA PGY 2 Cardiology Pharmacy Residents University of Kentucky Health. Care

Disclosure We have no actual or potential conflicts of interest to disclose.

Background ● Dual-antiplatelet therapy is standard treatment for patients with acute coronary syndromes (ACS) ● Ticagrelor and prasugrel have a faster onset of action with more potent and consistent anti-platelet effects than clopidogrel Am Heart J 2007; 153(1): 66. e 9 -66 Circulation 2009; 120: 2577 -2585

Previous Literature TRITON-TIMI (2007) Prasugrel vs. Clopidogrel ● ● ● Significant reduction in primary endpoint, driven by reduction in nonfatal MI Significant increase in the risk of major bleeding No significant difference in overall mortality PLATO (2009) Ticagrelor vs. Clopidogrel ● ● ● Significant reduction in MACE (not observed in N. American subgroup) Significant reduction in all-cause mortality No significant increase in major bleeding NEJM 2007; 357: 2001 -2015 NEJM 2009; 361: 1045 -1057
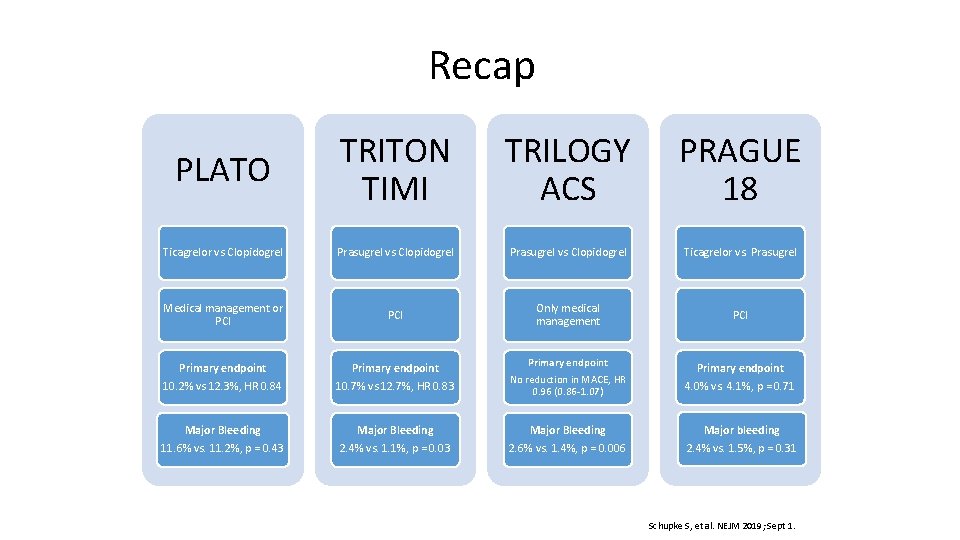
Recap PLATO TRITON TIMI TRILOGY ACS PRAGUE 18 Ticagrelor vs Clopidogrel Prasugrel vs Clopidogrel Ticagrelor vs. Prasugrel Medical management or PCI Only medical management PCI Primary endpoint 10. 2% vs 12. 3%, HR 0. 84 Primary endpoint 10. 7% vs 12. 7%, HR 0. 83 Primary endpoint No reduction in MACE, HR 0. 96 (0. 86 -1. 07 ) Primary endpoint 4. 0% vs. 4. 1%, p = 0. 71 Major Bleeding 11. 6% vs. 11. 2%, p = 0. 43 Major Bleeding 2. 4% vs. 1. 1%, p = 0. 03 Major Bleeding 2. 6% vs. 1. 4%, p = 0. 006 Major bleeding 2. 4% vs. 1. 5%, p = 0. 31 Schupke S, et al. NEJM 2019; Sept 1.
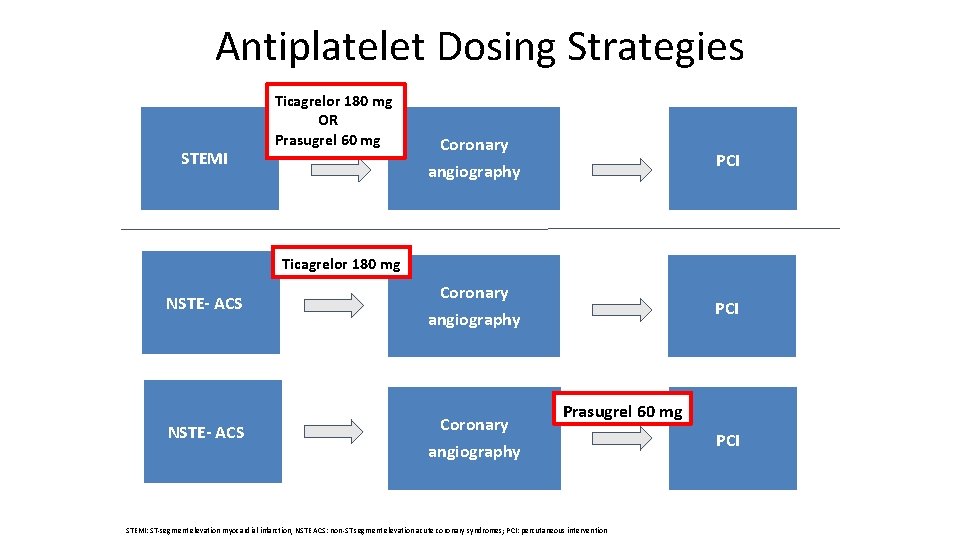
Antiplatelet Dosing Strategies STEMI Ticagrelor 180 mg OR Prasugrel 60 mg Coronary PCI angiography Ticagrelor 180 mg NSTE- ACS Coronary PCI angiography Coronary Prasugrel 60 mg angiography STEMI: ST-segment elevation myocardial infarction, NSTE ACS: non-ST segment elevation acute coronary syndromes; PCI: percutaneous intervention PCI

Methods ● Objective: Compare the efficacy and safety of two anti-platelet dosing strategies in patients with acute coronary syndromes ● Design: Investigator-initiated, phase 4, multicenter, randomized, openlabel trial Schupke S, et al. NEJM 2019; Sept 1.

Study Population Inclusion Criteria ● Adults > 18 y/o ● Hospitalization for: ○ STEMI ○ NSTEMI ○ Unstable angina ● Planned invasive evaluation (i. e. coronary angiography) Exclusion Criteria ● ● ● ● ● History of stroke or TIA History of ICH INR > 1. 5 Dialysis OAC Known intolerance to study drugs Platelets < 100, 000/μL Use of fibrinolytics Moderate or severe hepatic dysfunction ● Concomitant use of CYP 3 A inhibitors y/o: years old; STEMI: ST-segment elevation myocardial infarction, NSTEMI: non-ST segment elevation myocardial infarction; TIA: transient ischemic attack; ICH: intracranial hemorrhage; INR: international normalized ratio; OAC: oral anticoagulant Schupke S, et al. NEJM 2019; Sept 1.

Randomization Hospitalization for ACS with planned invasive evaluation Ticagrelor or Prasugrel Ticagrelor STEMI or NSTE ACS* STEMI or NSTE ACS Loading dose→ 60 mg X 1 Maintenance→ 10 mg daily + Loading dose→ 180 mg X 1 Maintenance→ 90 mg BID *LD after coronary angiography if PCI + 5 mg daily in patients > 75 y/o or < 60 kg ACS: acute coronary syndrome; STEMI: ST-segment elevation myocardial infarction, NSTEMI: non-ST segment elevation myocardial infarction, LD: loading dose Schupke S, et al. NEJM 2019; Sept 1.

Trial Endpoints Primary Outcome ● Composite of death, MI, or stroke at 1 year Secondary Outcomes ● Bleeding incidence at 1 year (BARC criteria) ● Individual components from primary endpoint ● Incidence of definite or probable stent thrombosis at 1 year MI: myocardial infarction; BARC: Bleeding Academic Research Consortium Schupke S, et al. NEJM 2019; Sept 1.

Statistical Analysis ● 1, 895 patients in each group to achieve 80% power with significance defined < 0. 05 ● Statistical analyses: ○ Student’s t-test ○ Non-parametric Wilcoxon rank-sum test ○ Cox proportional hazard models ● Pre-specified subgroup analyses ● Primary endpoint→ intention-to-treat ● Safety endpoint→ modified intention-to-treat Schupke S, et al. NEJM 2019; Sept 1.
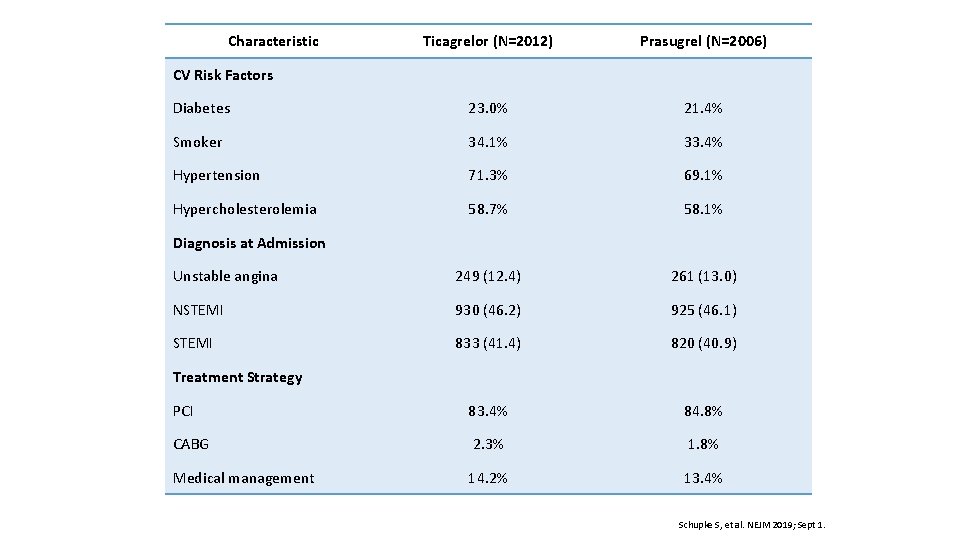
Characteristic Ticagrelor (N=2012) Prasugrel (N=2006) Diabetes 23. 0% 21. 4% Smoker 34. 1% 33. 4% Hypertension 71. 3% 69. 1% Hypercholesterolemia 58. 7% 58. 1% Unstable angina 249 (12. 4) 261 (13. 0) NSTEMI 930 (46. 2) 925 (46. 1) STEMI 833 (41. 4) 820 (40. 9) PCI 83. 4% 84. 8% CABG 2. 3% 1. 8% Medical management 14. 2% 13. 4% CV Risk Factors Diagnosis at Admission Treatment Strategy Schupke S, et al. NEJM 2019; Sept 1.
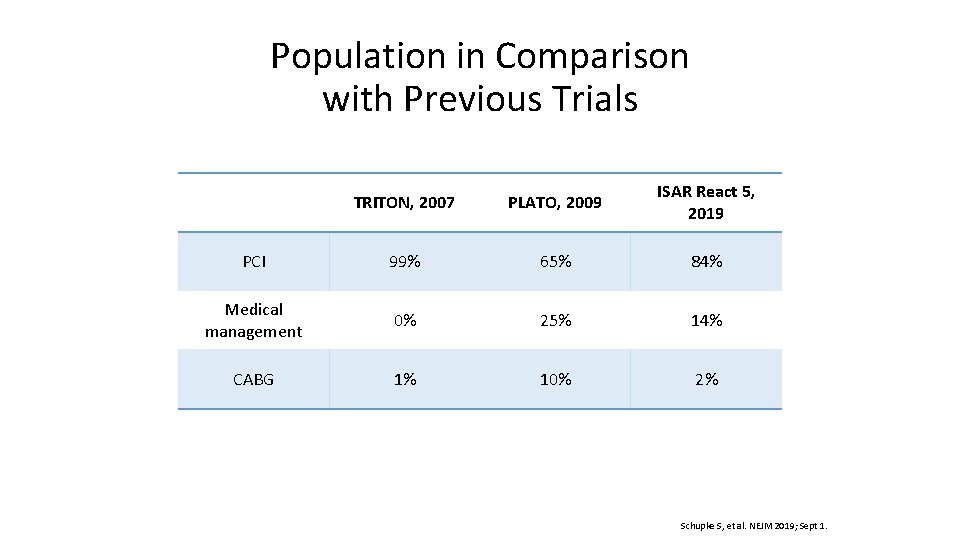
Population in Comparison with Previous Trials TRITON, 2007 PLATO, 2009 ISAR React 5, 2019 PCI 99% 65% 84% Medical management 0% 25% 14% CABG 1% 10% 2% Schupke S, et al. NEJM 2019; Sept 1.
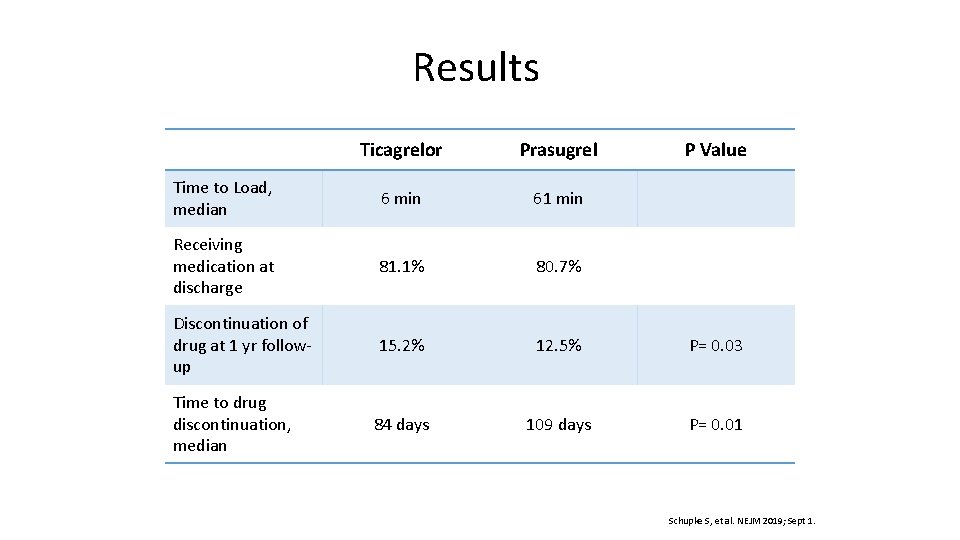
Results Ticagrelor Prasugrel Time to Load, median 6 min 61 min Receiving medication at discharge 81. 1% 80. 7% Discontinuation of drug at 1 yr followup 15. 2% 12. 5% P= 0. 03 84 days 109 days P= 0. 01 Time to drug discontinuation, median P Value Schupke S, et al. NEJM 2019; Sept 1.
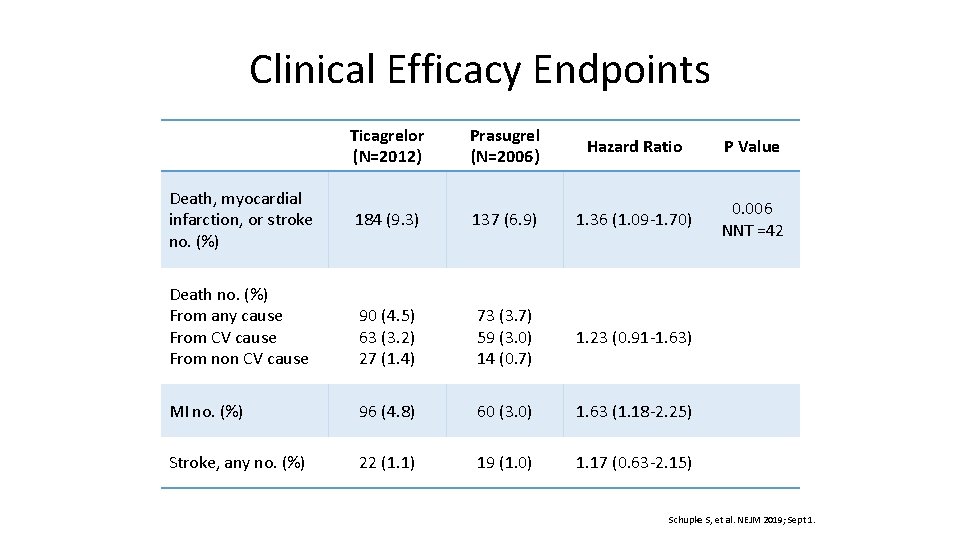
Clinical Efficacy Endpoints Ticagrelor (N=2012) Prasugrel (N=2006) Hazard Ratio P Value 184 (9. 3) 137 (6. 9) 1. 36 (1. 09 -1. 70) 0. 006 NNT =42 Death no. (%) From any cause From CV cause From non CV cause 90 (4. 5) 63 (3. 2) 27 (1. 4) 73 (3. 7) 59 (3. 0) 14 (0. 7) 1. 23 (0. 91 -1. 63) MI no. (%) 96 (4. 8) 60 (3. 0) 1. 63 (1. 18 -2. 25) Stroke, any no. (%) 22 (1. 1) 19 (1. 0) 1. 17 (0. 63 -2. 15) Death, myocardial infarction, or stroke no. (%) Schupke S, et al. NEJM 2019; Sept 1.
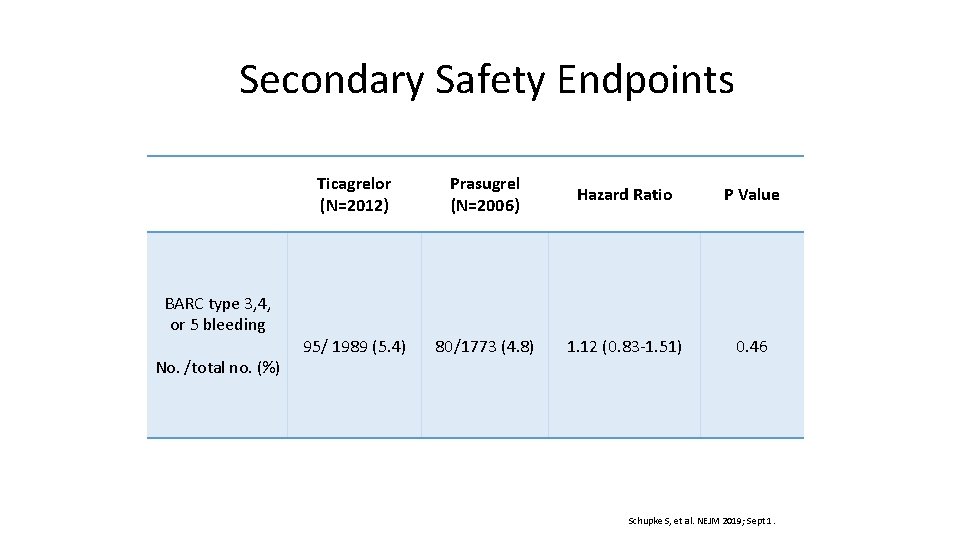
Secondary Safety Endpoints BARC type 3, 4, or 5 bleeding No. /total no. (%) Ticagrelor (N=2012) Prasugrel (N=2006) Hazard Ratio P Value 95/ 1989 (5. 4) 80/1773 (4. 8) 1. 12 (0. 83 -1. 51) 0. 46 Schupke S, et al. NEJM 2019; Sept 1.
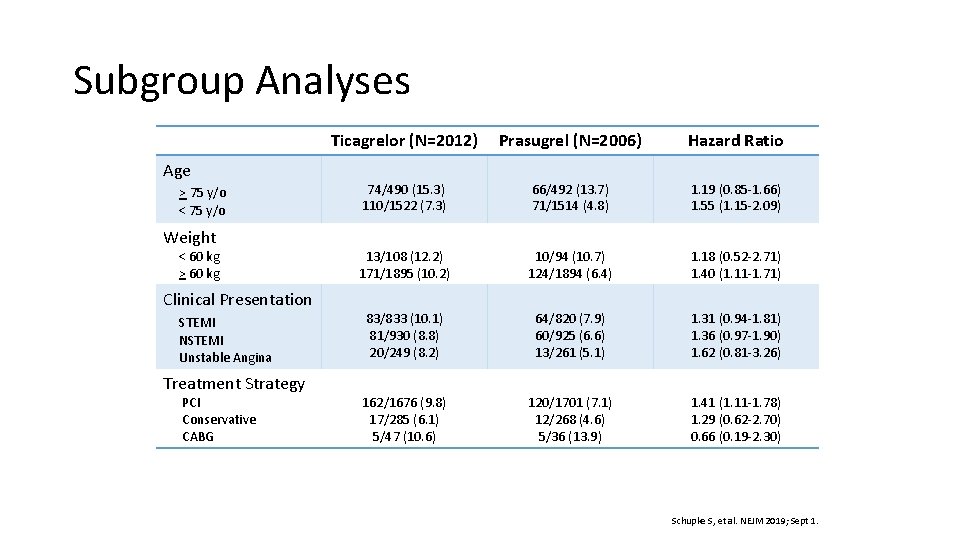
Subgroup Analyses Age > 75 y/o < 75 y/o Weight < 60 kg > 60 kg Clinical Presentation STEMI NSTEMI Unstable Angina Treatment Strategy PCI Conservative CABG Ticagrelor (N=2012) Prasugrel (N=2006) Hazard Ratio 74/490 (15. 3) 110/1522 (7. 3) 66/492 (13. 7) 71/1514 (4. 8) 1. 19 (0. 85 -1. 66) 1. 55 (1. 15 -2. 09) 13/108 (12. 2) 171/1895 (10. 2) 10/94 (10. 7) 124/1894 (6. 4) 1. 18 (0. 52 -2. 71) 1. 40 (1. 11 -1. 71) 83/833 (10. 1) 81/930 (8. 8) 20/249 (8. 2) 64/820 (7. 9) 60/925 (6. 6) 13/261 (5. 1) 1. 31 (0. 94 -1. 81) 1. 36 (0. 97 -1. 90) 1. 62 (0. 81 -3. 26) 162/1676 (9. 8) 17/285 (6. 1) 5/47 (10. 6) 120/1701 (7. 1) 12/268 (4. 6) 5/36 (13. 9) 1. 41 (1. 11 -1. 78) 1. 29 (0. 62 -2. 70) 0. 66 (0. 19 -2. 30) Schupke S, et al. NEJM 2019; Sept 1.

Author’s Conclusion ● ● Rate of death, myocardial infarction, or stroke is significantly lower in patients with ACS taking prasugrel as compared to ticagrelor There is no difference in major bleeding in patients with ACS taking prasugrel as compared to ticagrelor Schupke S, et al. NEJM 2019; Sept 1.

Strengths and Limitations Investigator initiated Open label to investigators Head to head comparison Patients did not get medication free as part of trial Generalizable 20% of both groups weren’t discharged on study drug Lower adherence to ticagrelor as compared to prasugrel Exclusion of stroke/TIA patients Schupke S, et al. NEJM 2019; Sept 1.

Considerations Is there a need for repeat study to confirm findings? Is there an unidentified pharmacodynamic benefit of prasugrel? Could be advocated for in patients with poor adherence benefitting from once daily dosing?
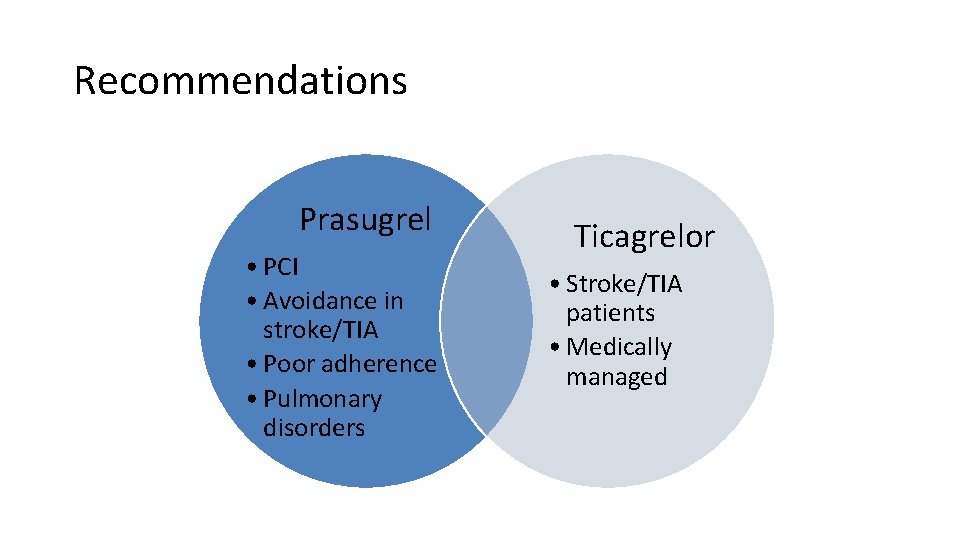
Recommendations Prasugrel • PCI • Avoidance in stroke/TIA • Poor adherence • Pulmonary disorders Ticagrelor • Stroke/TIA patients • Medically managed

Thank you! • Craig Beavers, Pharm. D, FACC, FAHA, BCCP, BCPS-AQ Cardiology, CACP • Josh Jacobs, Pharm. D • Jessie Dunne, Pharm. D, BCPS • Ashley Schenk, Pharm. D, BCCP, BCPS • Ayesha Ather, Pharm. D, BCCP, BCPS

Ticagrelor or Prasugrel in Patients with Acute Coronary Syndromes ISAR - REACT 5 Sara M. Ward, Pharm. D Erica A. Sheridan, Pharm. D, MBA PGY 2 Cardiology Pharmacy Residents University of Kentucky Health. Care
- Slides: 54