ACCP Cardiology PRN Journal Club 1302019 Dr Rob

ACCP Cardiology PRN Journal Club 1/30/2019

Dr. Rob Di. Domenico is an associate professor at the UIC College of Pharmacy. He received his Pharm. D from UIC and went on to complete his PGY 1 pharmacy residency and a cardiovascular pharmacotherapy fellowship at UIC. Dr. Di. Domenico covers the inpatient cardiology service and serves as the director of the PGY 2 cardiology pharmacy residency.

Dr. Kristen de Almeida is they PGY 2 Cardiology Pharmacy Resident at West Palm Beach VA Medical Center. She earned her Pharm. D degree from Palm Beach Atlantic University and completed her PGY 1 Pharmacy Residency at West Palm Beach VA Medical Center. Her professional interests include arrhythmias, heart failure, and valvular disease.

Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy Journal Club Kristen de Almeida, Pharm. D PGY 2 Cardiology WPB VA Medical Center Mentor: Robert J. Di. Domenico, Pharm. D, FCCP, FHFSA, FACC

Disclosure • I, Kristen de Almeida, have no relevant relationships to disclose

Background • Transthyretin amyloid cardiomyopathy (ATTR‐CM) vs Amyloid light chain • Characterized by accumulation of amyloid fibrils composed of misfolded transthyretin proteins • Transthyretin is a protein produced by the liver • Functions to transport thyroxine and retinol • Amyloid deposits in the myocardium lead to • cardiomyopathy • conduction system • bundle‐branch block, AV block, sinoatrial disease and atrial fibrillation Circulation. 2012 Sep 4; 126(10): 1286‐ 300. N Engl J Med. 2018 Aug 27.
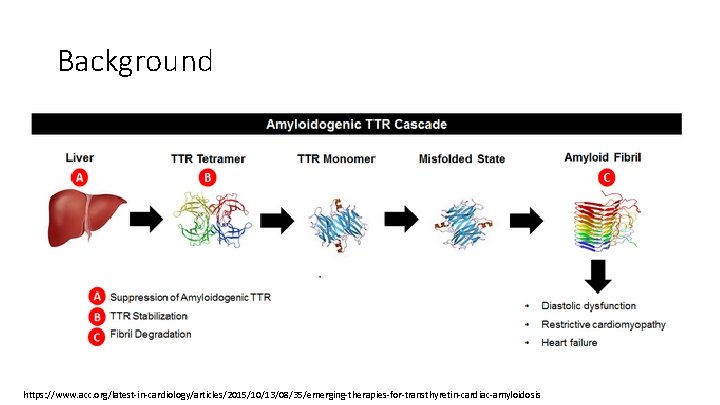
Background https: //www. acc. org/latest‐in‐cardiology/articles/2015/10/13/08/35/emerging‐therapies‐for‐transthyretin‐cardiac‐amyloidosis
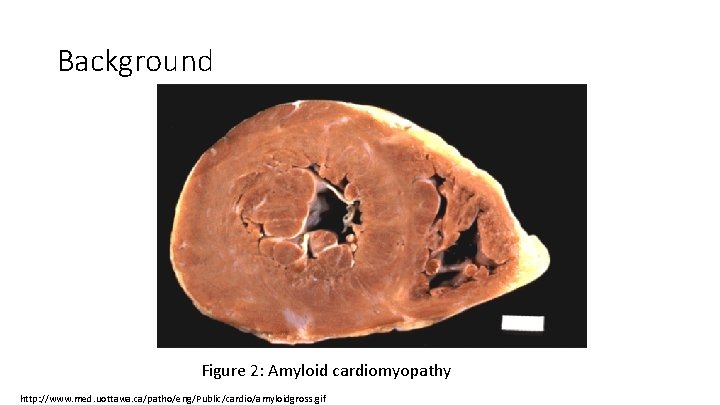
Background Figure 2: Amyloid cardiomyopathy http: //www. med. uottawa. ca/patho/eng/Public/cardio/amyloidgross. gif
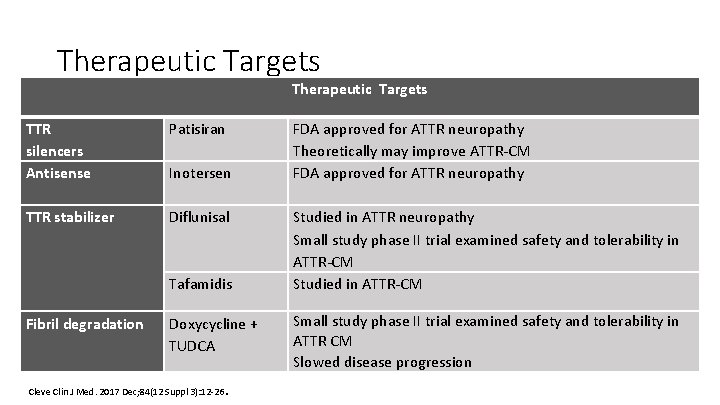
Therapeutic Targets TTR silencers Antisense Patisiran TTR stabilizer Diflunisal Inotersen Tafamidis Fibril degradation Doxycycline + TUDCA Cleve Clin J Med. 2017 Dec; 84(12 Suppl 3): 12‐ 26 . FDA approved for ATTR neuropathy Theoretically may improve ATTR‐CM FDA approved for ATTR neuropathy Studied in ATTR neuropathy Small study phase II trial examined safety and tolerability in ATTR‐CM Studied in ATTR‐CM Small study phase II trial examined safety and tolerability in ATTR CM Slowed disease progression

Background • Treatment is limited to supportive care • No guideline‐recommended treatment • Tafamidis: • Benzoxazole derivative lacking nonsteroidal anti‐inflammatory drug activity • Inhibits the dissociation of tetramers into monomers • Shown to slow progression of peripheral neurologic impairment in ATTR polyneuropathy • Prior studies showed tafamidis 20 mg daily stabilized transthyretin in ATTR‐ CM N Engl J Med. 2018 Aug 27. Orphanet J Rare Dis. 2013; 8: 31 .

Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy The New England Journal of Medicine August 27, 2018 N Engl J Med. 2018 Aug 27 .
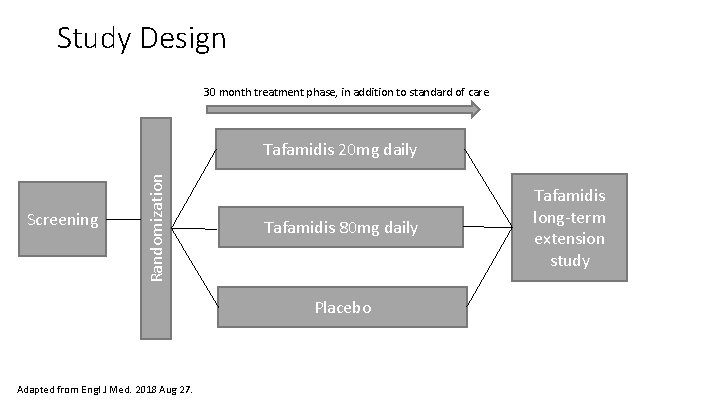
Study Design 30 month treatment phase, in addition to standard of care Screening Randomization Tafamidis 20 mg daily Tafamidis 80 mg daily Placebo Adapted from Engl J Med. 2018 Aug 27. Tafamidis long‐term extension study

Inclusion/Exclusion • Key Inclusion Criteria • Presence of amyloid deposit in biopsy tissue (cardiac or non cardiac) and TTR precursor protein identification by mass spectroscopy, immunohistochemistry or scintigraphy • Evidence of cardiac involvement by echocardiography with end‐diastolic interventricular septal wall thickness > 12 mm • Past medical history of heart failure with at least 1 prior hospitalization for HF • NT‐pro. BNP ≥ 600 pg/ml • 6‐minute walk test distance >100 meters • Key Exclusion Criteria • • NYHA class IV GFR <25 m. L/min/1. 73 m 2 Concurrent treatment with NSAID drugs Modified body mass index <600 kg/m 2 g/L N Engl J Med. 2018 Aug 27
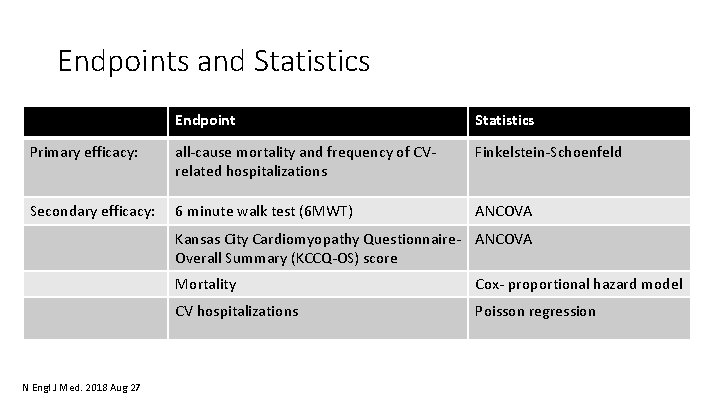
Endpoints and Statistics Endpoint Statistics Primary efficacy: all‐cause mortality and frequency of CV‐ related hospitalizations Finkelstein‐Schoenfeld Secondary efficacy: 6 minute walk test (6 MWT) ANCOVA Kansas City Cardiomyopathy Questionnaire‐ ANCOVA Overall Summary (KCCQ‐OS) score N Engl J Med. 2018 Aug 27 Mortality Cox‐ proportional hazard model CV hospitalizations Poisson regression
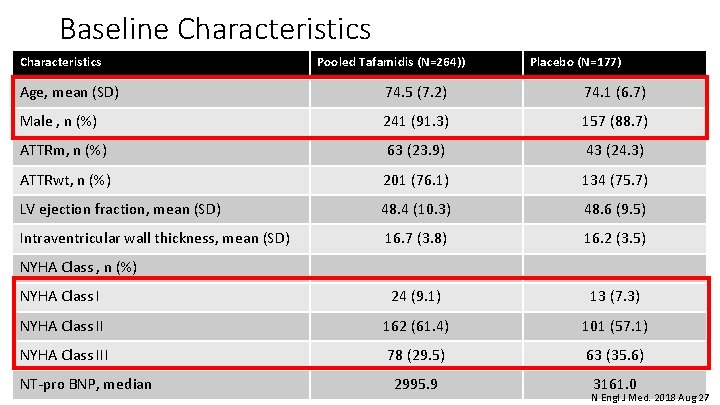
Baseline Characteristics Pooled Tafamidis (N=264)) Placebo (N=177) Age, mean (SD) 74. 5 (7. 2) 74. 1 (6. 7) Male , n (%) 241 (91. 3) 157 (88. 7) ATTRm, n (%) 63 (23. 9) 43 (24. 3) ATTRwt, n (%) 201 (76. 1) 134 (75. 7) LV ejection fraction, mean (SD) 48. 4 (10. 3) 48. 6 (9. 5) Intraventricular wall thickness, mean (SD) 16. 7 (3. 8) 16. 2 (3. 5) NYHA Class I 24 (9. 1) 13 (7. 3) NYHA Class II 162 (61. 4) 101 (57. 1) NYHA Class III 78 (29. 5) 63 (35. 6) NYHA Class , n (%) NT‐pro BNP, median 2995. 9 3161. 0 N Engl J Med. 2018 Aug 27
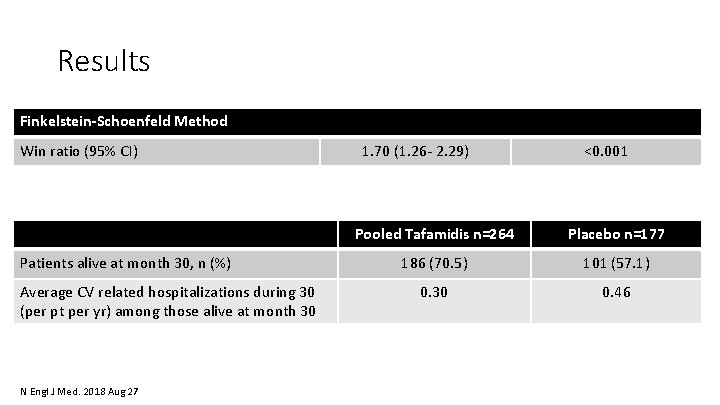
Results Finkelstein-Schoenfeld Method Win ratio (95% CI) Patients alive at month 30, n (%) Average CV related hospitalizations during 30 (per pt per yr) among those alive at month 30 N Engl J Med. 2018 Aug 27 1. 70 (1. 26‐ 2. 29) <0. 001 Pooled Tafamidis n=264 Placebo n=177 186 (70. 5) 101 (57. 1) 0. 30 0. 46
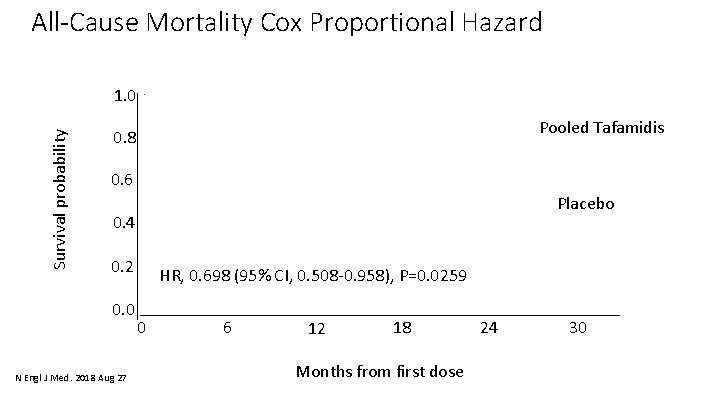
All-Cause Mortality Cox Proportional Hazard Survival probability 1. 0 Pooled Tafamidis 0. 8 0. 6 Placebo 0. 4 0. 2 0. 0 N Engl J Med. 2018 Aug 27 HR, 0. 698 (95% CI, 0. 508‐ 0. 958), P=0. 0259 0 6 12 18 Months from first dose 24 30

Results Frequency of CV related Hospitalizations Pooled Tafamidis n=264 Total (%) number of patients with CV‐related hospitalizations per yr Pooled tafamidis vs placebo treatment difference relative risk ratio (95% CI) P‐value N Engl J Med. 2018 Aug 27 Placebo n=177 138 (52. 3) 107(60. 5) 0. 48 0. 70 0. 68 (0. 56‐ 0. 81) <0. 0001
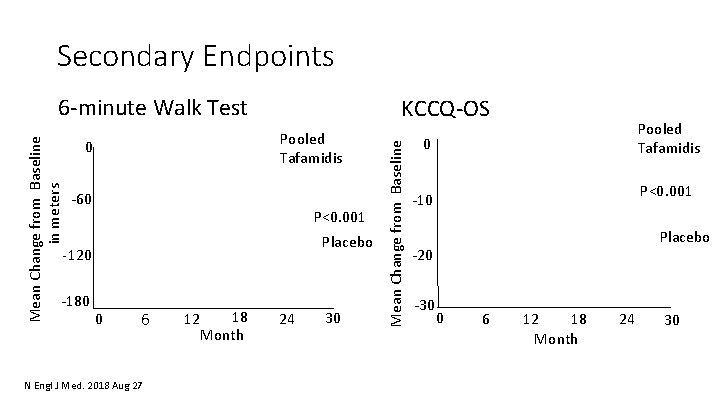
Secondary Endpoints KCCQ‐OS Pooled Tafamidis 0 ‐ 60 P<0. 001 Placebo ‐ 120 ‐ 180 0 6 N Engl J Med. 2018 Aug 27 18 12 Month 24 30 Mean Change from Baseline in meters 6‐minute Walk Test Pooled Tafamidis 0 P<0. 001 ‐ 10 Placebo ‐ 20 ‐ 30 0 6 12 18 Month 24 30

Safety • No significant difference in adverse drug reactions in tafamidis vs placebo • Previous studies with tafamidis reported higher rates of urinary tract infection, diarrhea, and abdominal pain

Critique • Strengths: • • Novel drug Appropriate statistical tests Compliance assessed 30 month duration • Weakness: • Use of NSAIDs • Cost

Upcoming trials • Tafamidis received breakthrough therapy designation from FDA • Long term safety tafamidis in ATTR‐CM • 60 months • Due 2024 • Coming therapies • Monoclonal antibody degrade TTR amyloid deposits https: //clinicaltrials. gov/ct 2/show/NCT 02319005? cond=Transthyretin+Cardiac+Amyloidosis&rank=3 https: //clinicaltrials. gov/ct 2/show/NCT 02791230? cond=Transthyretin+Cardiac+Amyloidosis&rank=10

Conclusion • Patients with heart failure due to ATTR‐ CM, treatment with tafamidis reduced all‐cause mortality and CV‐related hospitalizations • Tafamidis significantly reduced the decline in functional capacity and quality of life • Tafamidis is an effective therapy for patients with ATTR‐CM N Engl J Med. 2018 Aug 27

Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy Journal Club Kristen de Almeida, Pharm. D PGY 2 Cardiology WPB VA Medical Center Mentor: Robert J. Di. Domenico, Pharm. D, FCCP, FHFSA, FACC

Dr. Addy Schoening is the PGY 2 Cardiology Pharmacy Resident at Abbott Northwestern Hospital. She earned her Pharm. D from the University of Iowa and completed her PGY 1 Pharmacy Residency at Abbott Northwestern. Her professional interests include heart failure, pulmonary hypertension, transplant, and research.

Angiotensin‐Neprilysin Inhibition in Acute Decompensated Heart Failure Addy Schoening, Pharm. D PGY 2 Cardiology Pharmacy Resident Abbott Northwestern Hospital Minneapolis, MN Velazquez EJ, et al. N Engl J Med. 2018; [Epub ahead of print]

Disclosures • No actual or potential conflicts of interest related to the content of this presentation to disclose 27

Background • Standard of care for advanced heart failure exacerbation includes IV diuretics, IV vasodilators, IV inotropes • Few recent therapies shown to alter outcomes in HF in acute setting • Ideally, patients begin GDMT prior to discharge • Recent trials assessing sacubitril‐valsartan – PARADIGM‐HF – TRANSITION Mc. Murray JJV, et al. N Eng J Med. 2014. 371(11): 993‐ 1004.

PARADIGM‐HF • Design: – Prospective, randomized, active‐controlled – 3 phases • Screening period; single‐blind run‐in period; double‐blind treatment period • Population: – Ambulatory, adult patients with HFr. EF with NYHA class II ‐ IV already on GDMT (N=8442) • Primary endpoint: – Composite of death from CV causes or first hospitalization for HF • Results: – Sacubitril‐valsartan had significant reduction in mortality • (HR 0. 80; 95% CI, 0. 73 to 0. 87; P<0. 001) – Sacubitril‐valsartan had significant reduction in first hospitalization for HF • (HR 0. 79; 95% CI, 0. 71‐ 0. 89; P<0. 001) Mc. Murray JJV, et al. N Eng J Med. 2014. 371(11): 993‐ 1004.

TRANSITION • Design: • Multicenter, open‐label • Population: • Adult patients hospitalized for ADHF (new or pre‐existing HF) with NYHA Class II–IV and LVEF ≤ 40% • Intervention: • Initiation of sacubitril‐valsartan prior to discharge vs. post‐discharge • Primary endpoint: • Proportion of patients in the pre‐discharge and post‐discharge treatment initiation groups who achieve the target sacubitril‐valsartan dose at the end of Week 10 after randomization • Results (unpublished): • > 86% of patients were receiving sacubitril‐valsartan for 2 weeks or longer without interruption, roughly half of patients in the study achieved the primary endpoint which was a target dose of 200 mg of sacubitril‐valsartan twice daily within 10 weeks in both groups Pascual‐Figal D, et al. ESC Heart Fail. 2018. 5(2): 327‐ 336. Althoff E and Estes A. 2018. Accessed from http: //www. novartis. com.

PIONEER‐HF

Inclusion • Hospitalized for ADHF • Within 24 hours – 10 days of presentation meeting the following criteria: – SBP ≥ 100 mm. Hg within 6 hours of randomization – No intensification of IV diuretics within 6 hours – No IV inotropes within 24 hours of randomization – No IV vasodilators including nitrates within 6 hours of randomization • LVEF ≤ 40% within past 6 months • NT‐pro. BNP ≥ 1600 pg/m. L OR BNP ≥ 400 pg/m. L Exclusion • Use of sacubitril‐valsartan in previous 30 days • Known history of angioedema to ACEi or ARB therapy • e. GFR < 30 ml/min/1. 73 m 2 • Serum K > 5. 2 m. Eq/L at screening • Hepatic impairment or history of cirrhosis with evidence of portal hypertension • ACS, stroke, TIA, cardiac, carotid, other major CV surgery; PCI, carotid angioplasty with the prior month
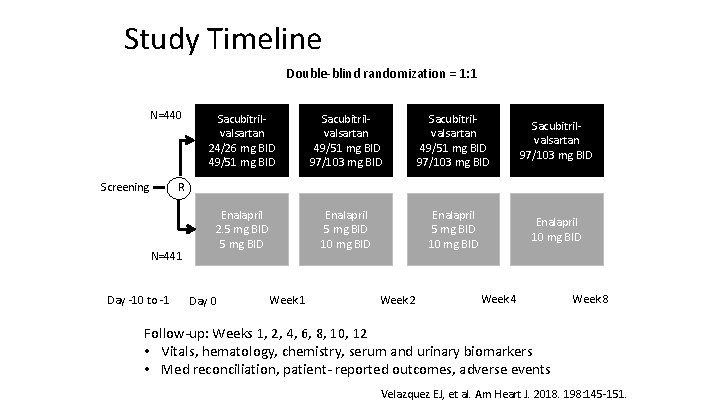
Study Timeline Double‐blind randomization = 1: 1 N=440 Screening Sacubitril‐ valsartan 24/26 mg BID 49/51 mg BID Sacubitril‐ valsartan 49/51 mg BID 97/103 mg BID Sacubitril‐ valsartan 97/103 mg BID Enalapril 2. 5 mg BID Enalapril 5 mg BID 10 mg BID Enalapril 10 mg BID R N=441 Day ‐ 10 to ‐ 1 Day 0 Week 1 Week 2 Week 4 Week 8 Follow‐up: Weeks 1, 2, 4, 6, 8, 10, 12 • Vitals, hematology, chemistry, serum and urinary biomarkers • Med reconciliation, patient‐ reported outcomes, adverse events Velazquez EJ, et al. Am Heart J. 2018. 198: 145‐ 151.

Dosing Protocol Randomization SBP ≥ 100 ‐ <120 mm. Hg • Sacubitril‐valsartan 24/26 mg BID • Enalapril 2. 5 mg BID SBP ≥ 120 mm. Hg • Sacubitril‐valsartan 49/51 mg BID • Enalapril 5 mg BID Week 1 Week 2, 4, 6 SBP < 110 mm. Hg • Sacubitril‐valsartan 24/26 mg BID • Enalapril 2. 5 mg BID SBP < 100 mm. Hg • Sacubitril‐valsartan 24/26 mg BID • Enalapril 2. 5 mg BID SBP ≥ 110 mm. Hg • Sacubitril‐valsartan 49/51 mg BID • Enalapril 5 mg BID SBP ≥ 100 mm. Hg • Sacubitril‐valsartan 49/51 mg BID • Enalapril 5 mg BID SBP < 110 mm. Hg • Sacubitril‐valsartan 49/51 mg BID • Enalapril 5 mg BID SBP < 100 mm. Hg • Sacubitril‐valsartan 49/51 mg BID • Enalapril 5 mg BID SBP ≥ 110 mm. Hg • Sacubitril‐valsartan 97/103 mg BID • Enalapril 10 mg BID SBP ≥ 100 mm. Hg • Sacubitril‐valsartan 97/103 mg BID • Enalapril 10 mg BID

Endpoints Primary • Time‐averaged proportional change in NT‐ pro. BNP concentration from baseline through weeks 4 and 8 Exploratory • Incidence of a composite of death • Re‐hospitalization for heart failure • Implantation of a LVAD • Listing for heart transplantation • Unplanned visit for acute heart failure requiring IV diuretics, increase in dose of diuretics of > 50%, or the use of an additional drug for heart failure Safety • Incidence of worsening renal function, hyperkalemia, symptomatic hypotension, and angioedema

Statistics • Assuming threshold for statistical significance of. 05 and 85% power, assuming a value of 0. 95 for enalapril group – Sample size needed to detect 18% reduction in geometric mean= 882 patients • Primary endpoint analysis: intention‐to‐treat – Least‐squared means from an analysis of covariance model • Exploratory endpoint: cumulative clinical‐event rates were calculated according to the Kaplan–Meier method – Differences in clinical outcomes were assessed with the log‐ rank test – Hazard ratios and associated 95% confidence intervals were calculated with a Cox proportional‐hazards model. Velazquez EJ, et al. Am Heart J. 2018. 198: 145‐ 151.

Results 964 screened 887 randomized 443 assigned to sacubitril‐valsartan ‐ 3 excluded for randomization error 444 assigned to enalapril ‐ 3 excluded for randomization error 91 discontinued treatment prematurely (20. 4%) ‐ 51 adverse event ‐ 4 died while receiving treatment drug ‐ 5 lost to follow‐up 96 discontinued treatment prematurely (21. 7%) ‐ 45 adverse event ‐ 6 died while receiving trial drug ‐ 7 lost to follow‐up 440 in efficacy analysis 379 had data for primary efficacy outcome at baseline and weeks 4, 8 441 in efficacy analysis 374 had data for primary efficacy outcome at baseline and weeks 4, 8 439 in safety analysis 436 in safety analysis
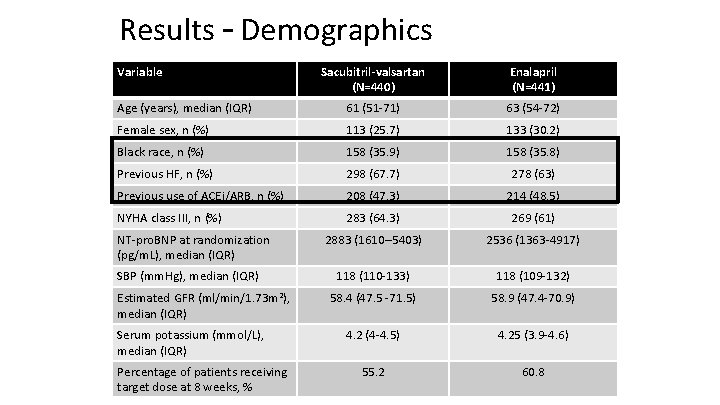
Results – Demographics Variable Sacubitril-valsartan (N=440) Enalapril (N=441) Age (years), median (IQR) 61 (51‐ 71) 63 (54‐ 72) Female sex, n (%) 113 (25. 7) 133 (30. 2) Black race, n (%) 158 (35. 9) 158 (35. 8) Previous HF, n (%) 298 (67. 7) 278 (63) Previous use of ACEi/ARB, n (%) 208 (47. 3) 214 (48. 5) NYHA class III, n (%) 283 (64. 3) 269 (61) 2883 (1610– 5403) 2536 (1363‐ 4917) 118 (110‐ 133) 118 (109‐ 132) 58. 4 (47. 5 ‐ 71. 5) 58. 9 (47. 4‐ 70. 9) 4. 2 (4‐ 4. 5) 4. 25 (3. 9‐ 4. 6) 55. 2 60. 8 NT‐pro. BNP at randomization (pg/m. L), median (IQR) SBP (mm. Hg), median (IQR) Estimated GFR (ml/min/1. 73 m 2), median (IQR) Serum potassium (mmol/L), median (IQR) Percentage of patients receiving target dose at 8 weeks, %
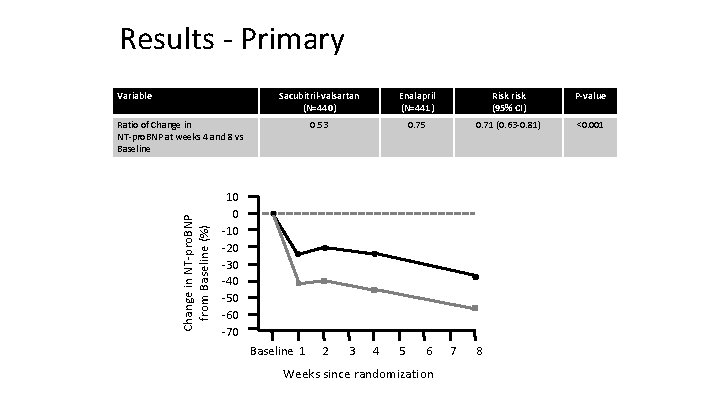
Results ‐ Primary Variable Sacubitril-valsartan (N=440) Enalapril (N=441) Risk risk (95% CI) P-value 0. 53 0. 75 0. 71 (0. 63‐ 0. 81) <0. 001 Change in NT‐pro. BNP from Baseline (%) Ratio of Change in NT‐pro. BNP at weeks 4 and 8 vs Baseline 10 0 ‐ 10 ‐ 20 ‐ 30 ‐ 40 ‐ 50 ‐ 60 ‐ 70 Baseline 1 2 3 4 5 6 Weeks since randomization 7 8
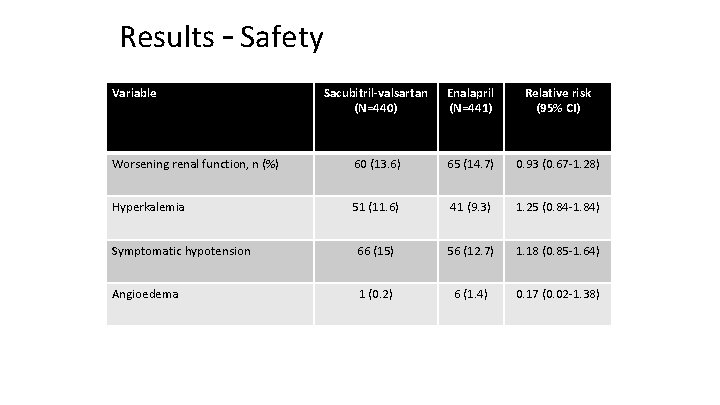
Results – Safety Variable Sacubitril-valsartan (N=440) Enalapril (N=441) Relative risk (95% CI) Worsening renal function, n (%) 60 (13. 6) 65 (14. 7) 0. 93 (0. 67‐ 1. 28) Hyperkalemia 51 (11. 6) 41 (9. 3) 1. 25 (0. 84‐ 1. 84) Symptomatic hypotension 66 (15) 56 (12. 7) 1. 18 (0. 85‐ 1. 64) Angioedema 1 (0. 2) 6 (1. 4) 0. 17 (0. 02‐ 1. 38)
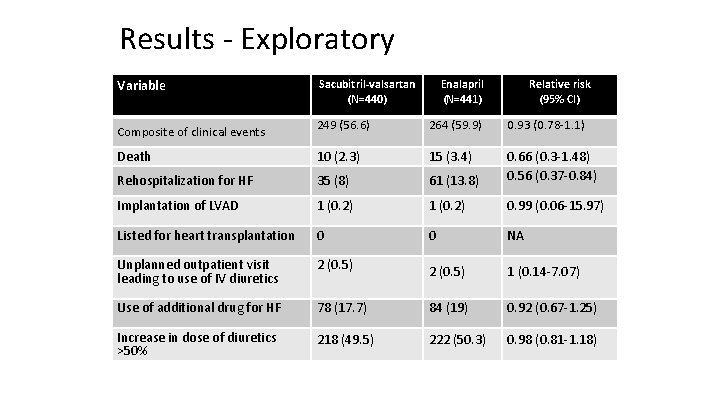
Results ‐ Exploratory Variable Sacubitril-valsartan (N=440) Enalapril (N=441) Relative risk (95% CI) Composite of clinical events 249 (56. 6) 264 (59. 9) 0. 93 (0. 78‐ 1. 1) Death 10 (2. 3) 15 (3. 4) Rehospitalization for HF 35 (8) 61 (13. 8) 0. 66 (0. 3‐ 1. 48) 0. 56 (0. 37‐ 0. 84) Implantation of LVAD 1 (0. 2) 0. 99 (0. 06‐ 15. 97) Listed for heart transplantation 0 0 NA Unplanned outpatient visit leading to use of IV diuretics 2 (0. 5) 1 (0. 14‐ 7. 07) Use of additional drug for HF 78 (17. 7) 84 (19) 0. 92 (0. 67‐ 1. 25) Increase in dose of diuretics >50% 218 (49. 5) 222 (50. 3) 0. 98 (0. 81‐ 1. 18)

Discussion • Sacubitril‐valsartan successful in lowering NT‐pro. BNP when initiated prior to discharge – Clinical outcomes? • Sacubitril‐valsartan is safe to start in hospitalized patients – Black race, new diagnosis of HF, ACEi/ARB naïve – Increase in hospital initiation • Cost‐effectiveness

Critiques Strengths • Prospective, double‐blind, randomized clinical trial • Active comparator • Relevant clinical question • Underrepresented population • Inclusive of low‐dose sacubitril‐ valsartan in dosing protocol Weaknesses • Surrogate endpoint • Exploratory analysis of clinical outcomes • Local clinician‐investigators initiating without central validation • Monitoring required could prolong hospitalization

Impact on Clinical Practice • Improvements in transitions of care – Increased utilization of GDMT – Reduction in readmissions • Increased access to sacubitril‐valsartan – More data may allow increased inclusion on insurance formularies

Acknowledgments • Robert Di. Domenico, Pharm. D, BCPS‐AQ Cardiology, FCCP, FHFSA, FACC • Matt Lillyblad, Pharm. D, BCCCP, BCPS‐AQ Cardiology

Questions? ? ? Angiotensin‐Neprilysin Inhibition in Acute Decompensated Heart Failure Addy. Schoening@allina. com PGY 2 Cardiology Resident Abbott Northwestern Hospital Minneapolis, MN Velazquez EJ, et al. N Engl J Med 2018; [Epub ahead of print]
- Slides: 46